Abstract
Background:
Patients with spondyloarthropathy (SpA) have a higher risk of subclinical atherosclerosis (SCA). We assessed clinical and serological determinants of SCA in Indian SpA patients.
Methods:
Patients with SpA (fulfilling ASAS 2010 criteria; n = 104) attending our hospital were recruited; mean carotid intima-media thickness (CIMT) was performed by carotid ultrasonography, along with clinical assessment and traditional risk factor evaluation. Microparticles were extracted from plasma and total, as well as endothelial microparticles (EMP), platelet microparticles, T lymphocyte microparticles and B lymphocyte microparticles, were analysed by flow cytometry. Serum samples were analysed for inflammatory cytokines previously implicated in atherosclerosis, namely interleukin 1β (IL-1β), IL-6, IL-17, IL-27, IL-33 and tumour necrosis factor-alpha. Thirty-eight healthy controls were used for comparison. Subgroup analyses compared parameters between SpA with SCA (i.e., with carotid plaque or more than 75th percentile of CIMT for that age and sex in the Indian population) versus those without SCA. Ethical approval and written, informed consent were obtained.
Results:
Despite significantly younger age, lower body mass index and higher total cholesterol in controls compared to SpA, those with SpA had higher CIMT. Traditional cardiovascular risk factors (older age, higher waist-hip ratio) and novel markers of inflammation (serum IL-1β, IL-6) were associated with SCA. While total microparticles, EMP, T lymphocyte and B lymphocyte microparticles were increased in SpA than in healthy controls, they were not associated with SCA.
Conclusions:
Traditional risk factors and serum inflammatory cytokines IL-1β and IL-6 are associated with higher SCA in Indian SpA patients.
Keywords
Introduction
Spondyloarthropathy (SpA) refers to a group of inflammatory joint diseases predominantly affecting young adult males, characterised by an inflammatory type of low backache and involvement of large joints, predominantly of the lower limbs, with enthesitis. 1 The prevalence of SpA amongst Indians is estimated to be 0.23%–0.36%. 2 Such patients are treated with non-steroidal anti-inflammatory drugs (NSAIDs) as the first line, which may be continued for many years as there is some evidence to suggest that NSAIDs have a disease-modifying role in this disease. Ideally, patients who fail NSAIDs are candidates for biologic therapies such as those targeting tumour necrosis factor-alpha (TNF-α), the use of which potentially reduces NSAID use. However, most patients in an Indian scenario are unable to afford such costly biologic agents and require continuous NSAID therapy for many years for adequate relief of symptoms. 3
Current understanding of atherosclerosis suggests that it has a primary inflammatory basis. 4 Use of NSAIDs has been found to predispose to increased risk of cardiovascular events. 5 Hence, it is logical to assume that in the systemic inflammatory state present in patients with SpA, the use of NSAIDs contributes to an additional risk of atherosclerosis. The pathogenesis of atherosclerosis involves endothelial dysfunction and activation of platelets. Microparticles derived from endothelial cells (endothelial microparticles—EMP) and platelets (platelet microparticles—PMP) are emerging as novel biomarkers of atherosclerosis based on this understanding, and contribute towards increased risk of plaque progression and cardiovascular risk. 6 Only one study has assessed circulating EMP and PMP in patients with ankylosing spondylitis (AS) in a Turkish population. 7 This study failed to detect any difference in EMP and PMP between 82 patients with AS and 53 healthy controls. T lymphocyte populations play a role in the pathogenesis of SpA. 8 Therefore, lymphocyte microparticles might also play a role in driving disease processes and cardiovascular risk in spondyloarthropathies. Cytokines secreted by immune cells such as interleukin (IL) 1β, IL-6, TNF-α and IL-17A have been demonstrated to play a role in promoting atherosclerotic plaque development. Other anti-inflammatory cytokines such as IL-27 and IL-33 are thought to have a protective role in atherosclerosis. 9 Traditional risk factors for atherosclerosis identified in different studies include the ratio of high-density lipoprotein cholesterol (HDL-c) to low-density lipoprotein cholesterol (LDL-c), with lower values of HDL-c and higher LDL-c indicating a higher risk of cardiovascular events. Diabetes mellitus which results in higher levels of fasting plasma glucose is also an important risk factor for cardiovascular disease. 10
Studies on atherosclerosis assessed clinically by incident cardiovascular events and subclinically by imaging of the carotid arteries to look for the carotid intima-media complex are sparse in Indian patients with SpA. However, a meta-analysis of different studies from other populations (predominantly Caucasian) suggests a small but definite risk of increased cardiovascular events in patients with SpA. 11 Hence, there is an unmet need to assess subclinical atherosclerosis (SCA) in Indian patients with SpA. In this study, we evaluated traditional and novel cardiovascular risk factors (total microparticles, endothelial, platelet, T lymphocyte and B lymphocyte microparticles, IL-1β, IL-6, TNF-α, IL-17, IL-27, IL-33) in patients with SpA and healthy controls and their relationship with SCA assessed using carotid intima-media thickness (CIMT) using colour Doppler ultrasonography.
Methods
This was a cross-sectional study. Consecutive adult patients (>18 years of age) with SpA fulfilling ASAS 2010 criteria 1 attending the OPD/Ward of the Department of Clinical Immunology and Rheumatology and healthy controls from the attendants of patients in the OPD were included in the study after seeking written informed consent. Detailed history-taking and clinical examination were undertaken and standard proformas were used to assess disease activity (BASDAI) and functional limitation (BASFI). 1 Traditional cardiovascular risk factors (body mass index [BMI] and waist-hip ratio). Fasting blood sugar and fasting lipid profile were analysed. Patients belonging to vulnerable groups such as pregnant or lactating women, patients younger than 18 years or older than 60 years, and critically sick or terminally handicapped patients were excluded. The study was approved by the Institute Ethics Committee of SGPGIMS, Lucknow (document submission number 2017-76-IMP-97, date of approval 22 June 2017) and funded by Intramural funding from the Institute. Patients were recruited between January 2018 and June 2019.
The sample size was calculated based on a published study 12 with an α value of 0.05 and β value of 0.20, the effect size of 0.05 mm difference in CIMT between patients and controls (ratio of patients to controls 3:1), and a standard deviation (SD) of 0.09 mm using an online tool. 13 the minimum calculated number of patients and controls were 102 and 34, respectively. Therefore, we have enrolled 104 patients with SpA and 38 healthy controls.
CIMT measurement was performed on the distal wall of the common carotid artery coinciding with diastole timed through electrocardiographic gating approximately 5 mm before the carotid bulb using a linear transducer array with B-mode ultrasound (Esaote MyLab Xvision). 14 The mean of three readings on each side carotid artery was taken as the CIMT. Carotid plaques were identified using standard definitions. 15 SCA was defined as CIMT more than the 75th percentile of CIMT for that age and sex in the Indian population 16 or the presence of carotid plaques. The risk of developing a heart attack or stroke over the next 10 years was calculated using QRISK3. 17 Since this tool provides estimates of cardiovascular risk for those between 25 and 84 years, those patients with SpA in our cohort who were younger than 25 years were assumed to have this lower limit of age for QRISK3 calculation.
An amount of 6 mL of plasma was collected from patients with SpA and healthy controls. After spinning at 12,000 g for 2 minutes, the supernatant was collected and spun again at 20,000 g for 90 minutes. Thereafter, the pellets were collected and 1 mL of phosphate buffer saline (PBS) was added and spun at 20,000 g for 90 minutes. Again, pellets were collected and 1 mL PBS was added, spun at 20,000 g for 90 minutes and the pellet was collected and stored in 50 µL of chilled PBS at –80°C.
The stored microparticles were thereafter dissolved in 100 µL PBS. An amount 5 µL were collected for testing and isotype control. Incubation was performed for 30 minutes with Anti-Annexin V APC and microbeads of 7 µm size for total microparticles, Anti Human CD31 Alexa fluor488 and Anti-Human CD146 PE for EMP, Anti Human CD42a BV421 and Anti Human CD62L PE/Cyanine7 for PMP (separate tube), and Anti-Human CD19 PE (B lymphocyte microparticles) and Anti-Human CD3 FITC (T lymphocyte microparticles) in another separate tube. Microparticles were identified as a size between 0.1 and 1 µm on flow cytometry. All flow cytometry antibodies were requisitioned from BioLegend, USA. IL-1β, IL-6, IL-17, IL-27, IL-33 and TNF-α were estimated using ELISA kits procured from R and D Biosciences, USA as per manufacturer instructions.
Continuous variables were represented using mean ± SD and compared using unpaired Student’s t-test. Categorical variables were compared using the chi-square test or Fisher’s exact test (if any of the four cells had a value less than five). Comparisons were performed between patients with SpA and healthy controls, between SpA with or without SCA/carotid plaques. QRISK3 scores were compared between SpA with or without SCA. Differences in CIMT between SpA and controls were established after adjustment for any differences in traditional cardiovascular risk factors between patients and controls. Thereafter, linear regression models were constructed to establish significant factors associated with CIMT, SCA or carotid plaque with separate models for conventional cardiovascular risk and disease-related factors, serum cytokines and microparticle populations. The relationship between CIMT and QRISK3 scores was calculated using Pearson’s correlation coefficient. A P value <.05 was indicative of statistical significance. Statistical analyses were performed using STATA 16.1 I/C.
Results
One hundred and four SpA (103 axial SpA, 76 peripheral SpA) and 38 healthy controls were recruited. Among SpA, 3.85% had type 2 diabetes mellitus, 11.54% hypertension and 23.08% consumed tobacco. Nearly a third had on-demand use of NSAIDs, whereas about two-thirds continuously used NSAIDs. About one-half were on conventional disease-modifying anti-rheumatic drugs (DMARDs). Biologic use was scarce in our patients. Patients with SpA had a mean (±SD) 2.39 (±3.92)% risk of developing a heart attack or stroke over the next 10 years as per the QRISK3 score. Patients with SpA were slightly older and had lower BMI and total cholesterol levels than the 38 controls. CIMT was significantly higher in SpA than in healthy controls even after adjustment for differences in age, BMI and total cholesterol levels. QRISK3 had a moderate correlation with CIMT (Pearson’s r = 0.63) in patients with SpA. SCA was present in 40.38% SpA (Figure 1), whereas 14.42% had carotid plaques. Patients with SpA had higher levels of IL-1β, IL-6, TNF-α, IL-27 and lower levels of IL-17 than controls. Total microparticles, EMP, T lymphocyte microparticles and B lymphocyte microparticles were higher in SpA than in controls (Table 1). SpA with SCA had a lower HDL-c:LDL-c ratio and higher QRISK-3 scores, however, no other differences in cardiovascular risk factors, inflammatory cytokines or microparticle populations were observed (Table 2). SpA with carotid plaques were older, had longer disease duration, had higher waist-hip ratio, higher QRISK-3 scores and higher serum levels of IL-1β than those without carotid plaques (Table 3).
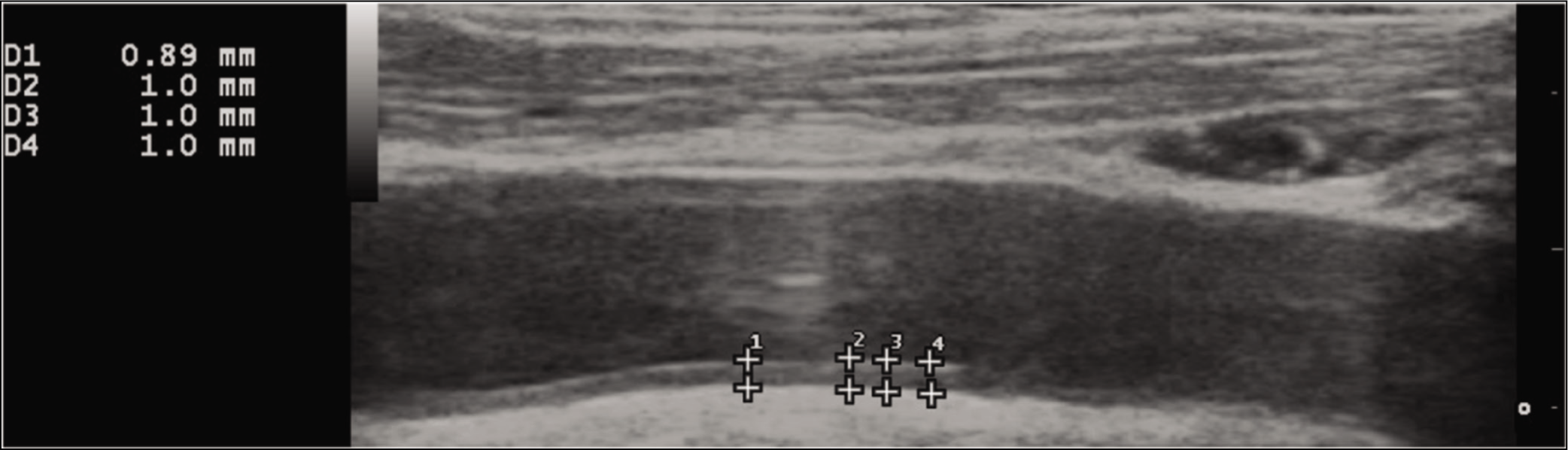
Thickened Carotid Intima-media Complex in a Patient with Spondyloarthropathy.
Characteristics of Patients with Spondyloarthropathy and Healthy Controls.
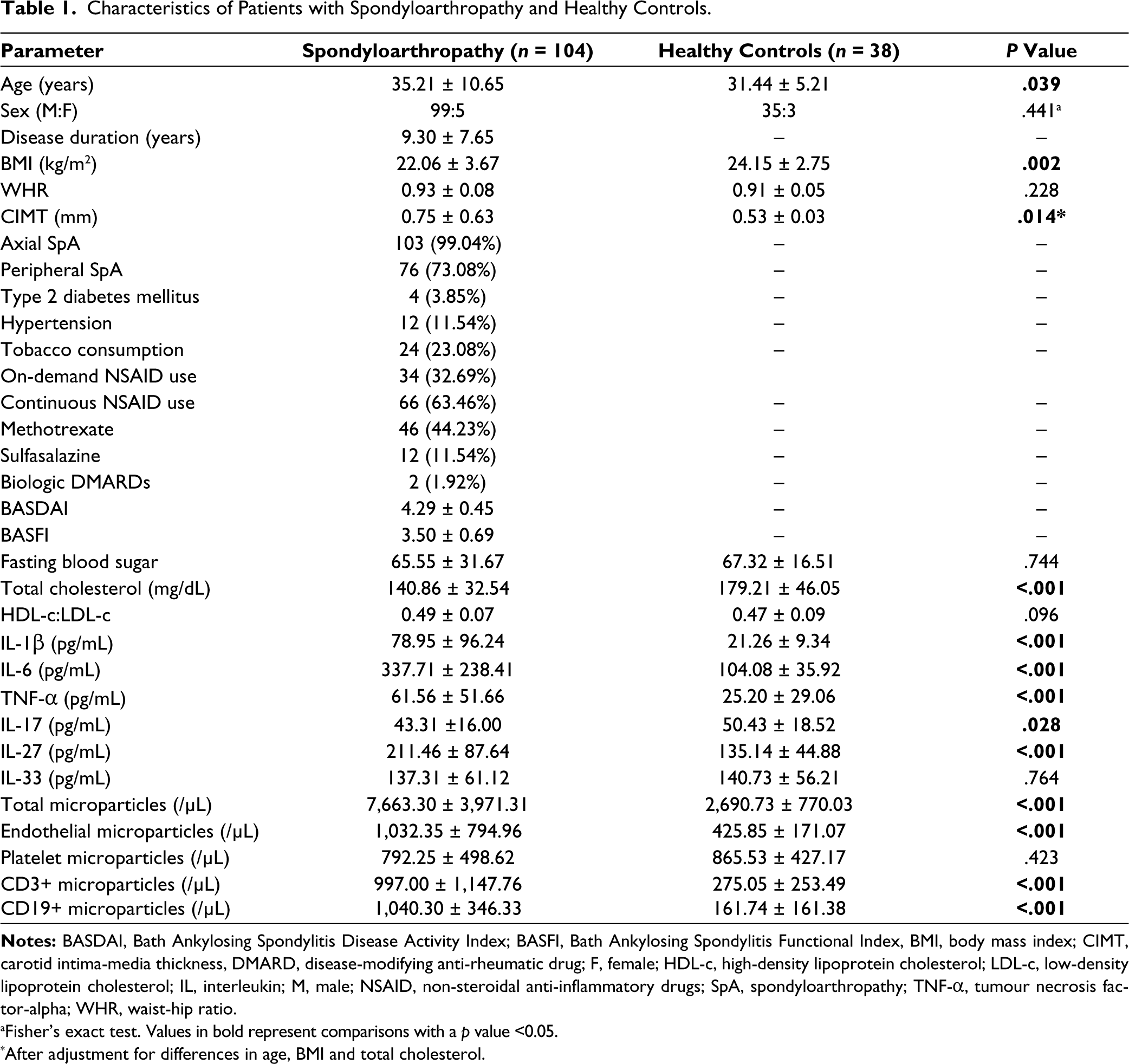
aFisher’s exact test. Values in bold represent comparisons with a p value <0.05.
*After adjustment for differences in age, BMI and total cholesterol.
Comparison Between Patients With Spondyloarthropathy With or Without Subclinical Atherosclerosis.
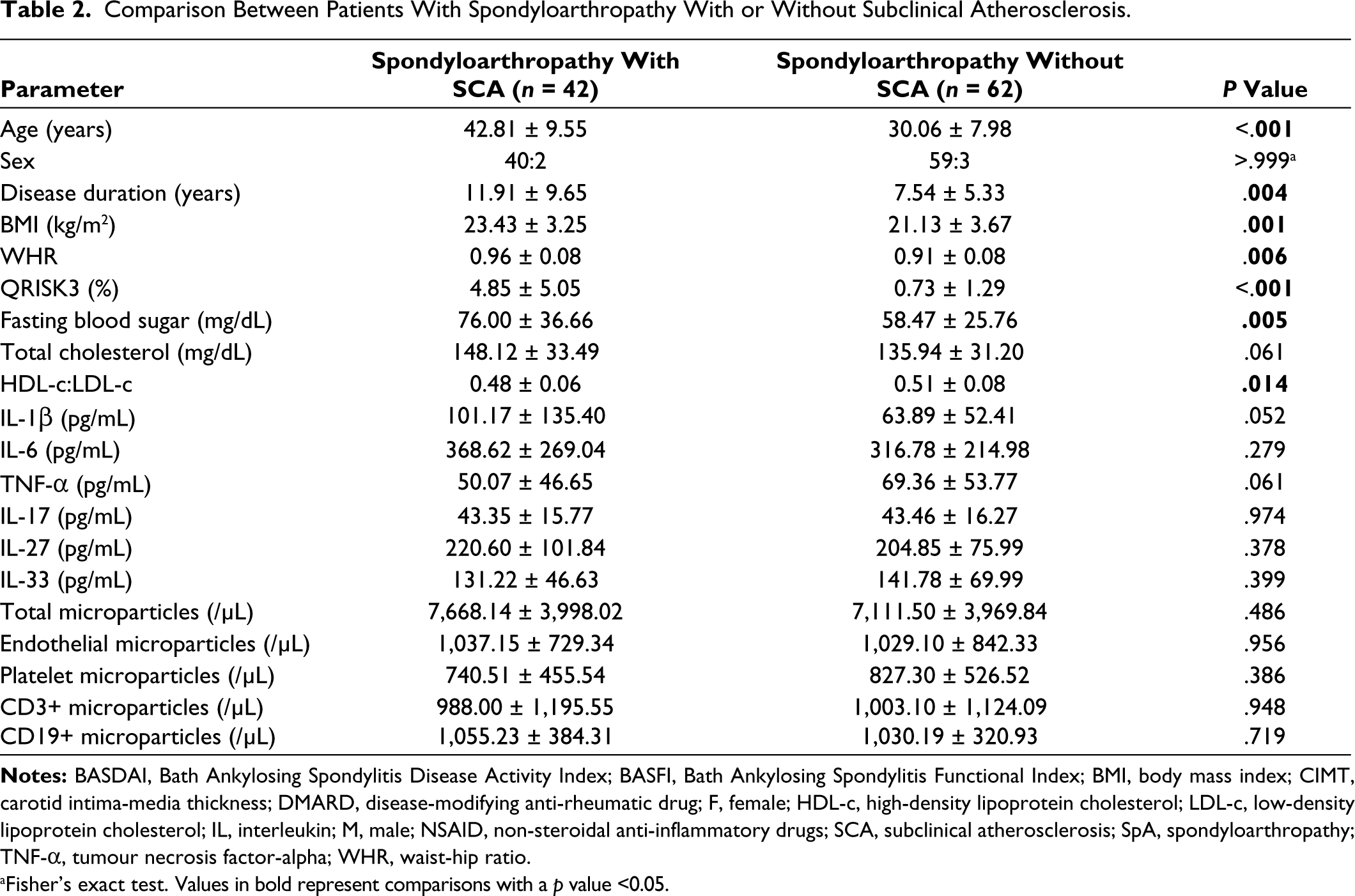
aFisher’s exact test. Values in bold represent comparisons with a p value <0.05.
Comparison Between Patients with Spondyloarthropathy With or Without Carotid Plaque.
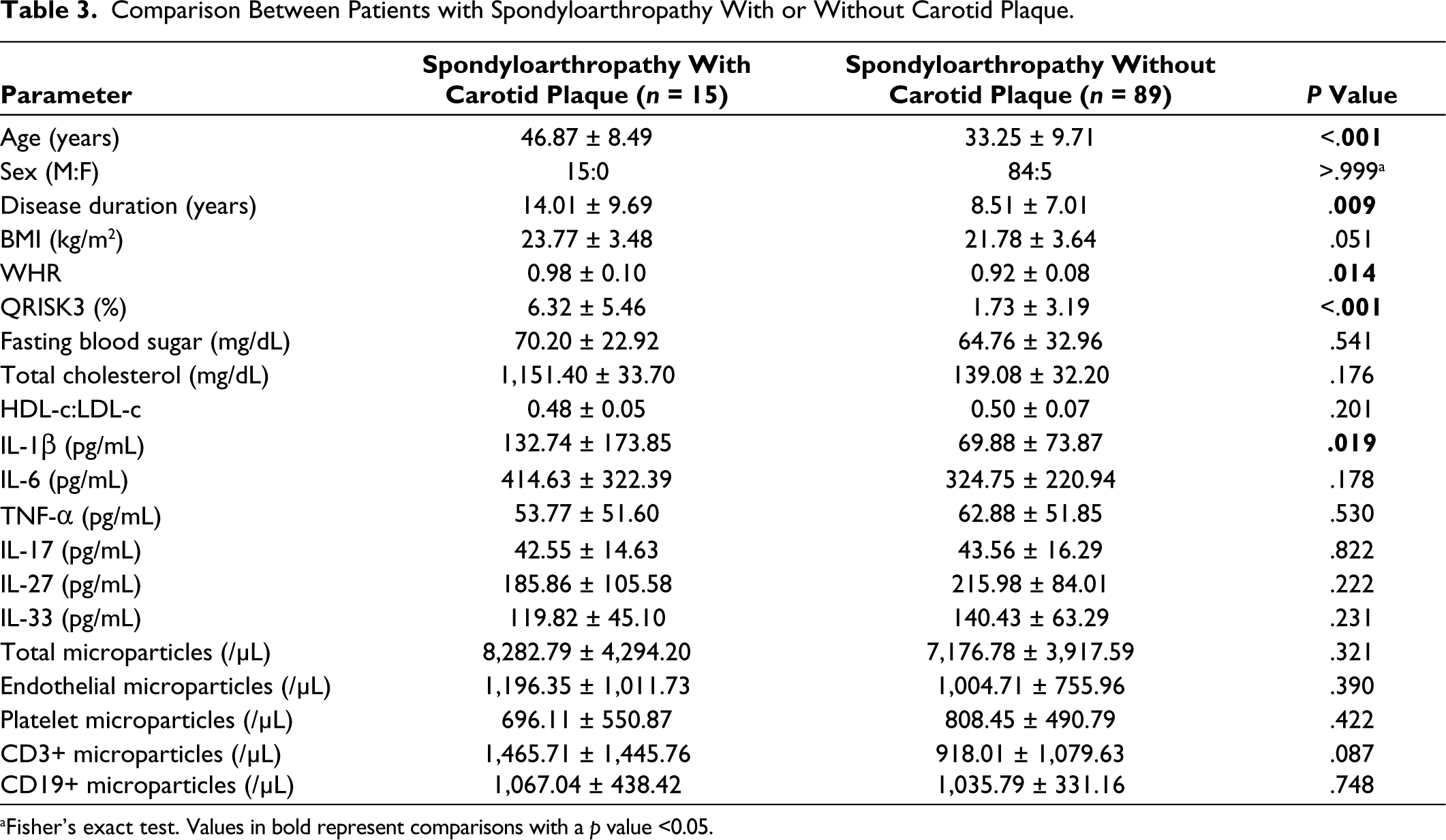
aFisher’s exact test. Values in bold represent comparisons with a p value <0.05.
Multivariable-adjusted linear regression models did not identify any significant predictors of CIMT in patients with SpA (Table 4). Older age, higher BASDAI, higher body-mass index, increased levels of IL-1β and IL-6 and decreased levels of TNF-α were associated with SCA in SpA (Table 5). Older age, increased levels of IL-1β, and decreased levels of TNF-α were associated with carotid plaque (Table 6).
Linear Regression Models for Predictors of Carotid Intima-media Thickness in Spondyloarthropathy.
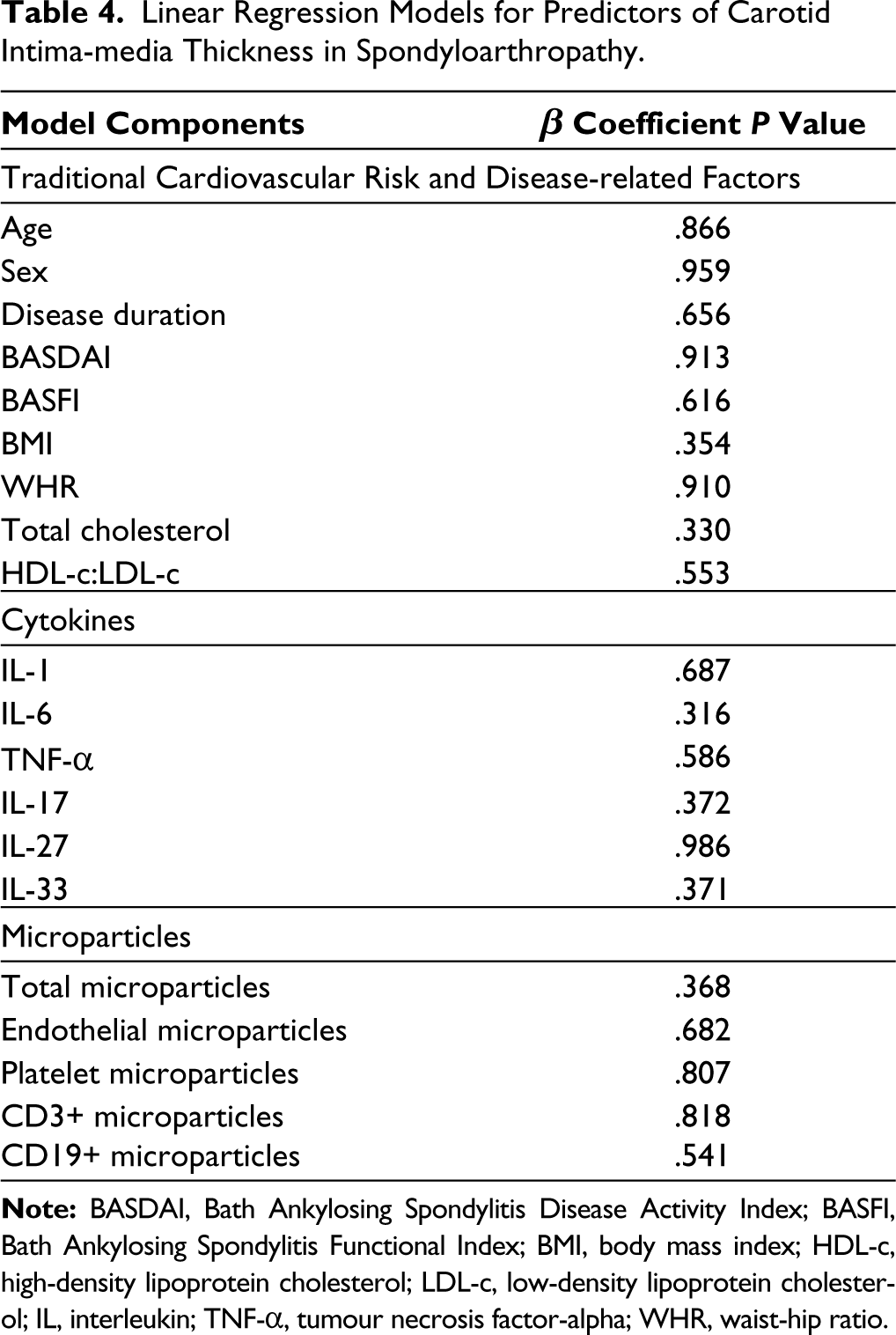
Linear Regression Models for Predictors of Subclinical Atherosclerosis in Spondyloarthropathy.
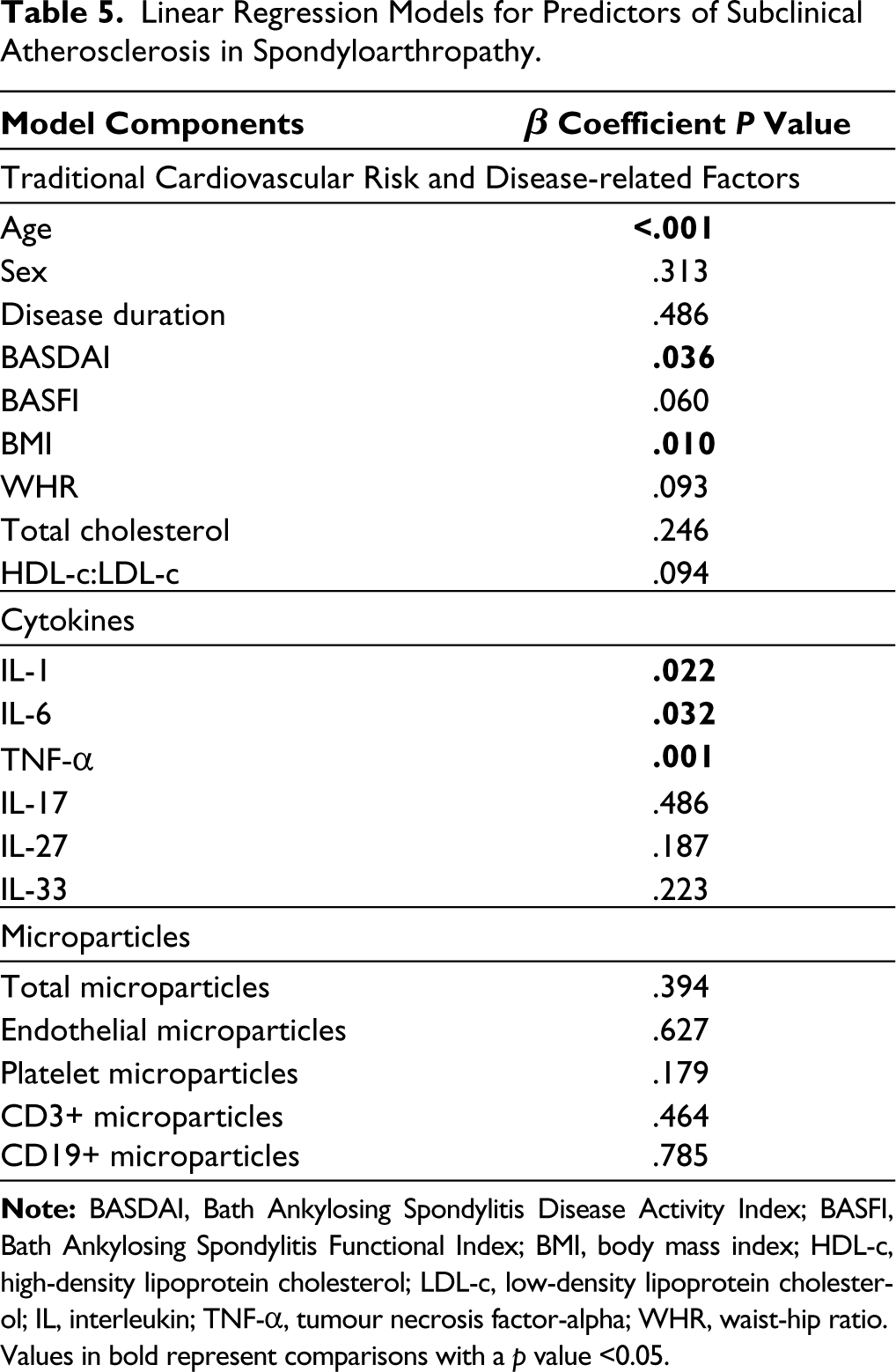
Values in bold represent comparisons with a p value <0.05.
Linear Regression Models for Predictors of Carotid Plaque in Spondyloarthropathy.
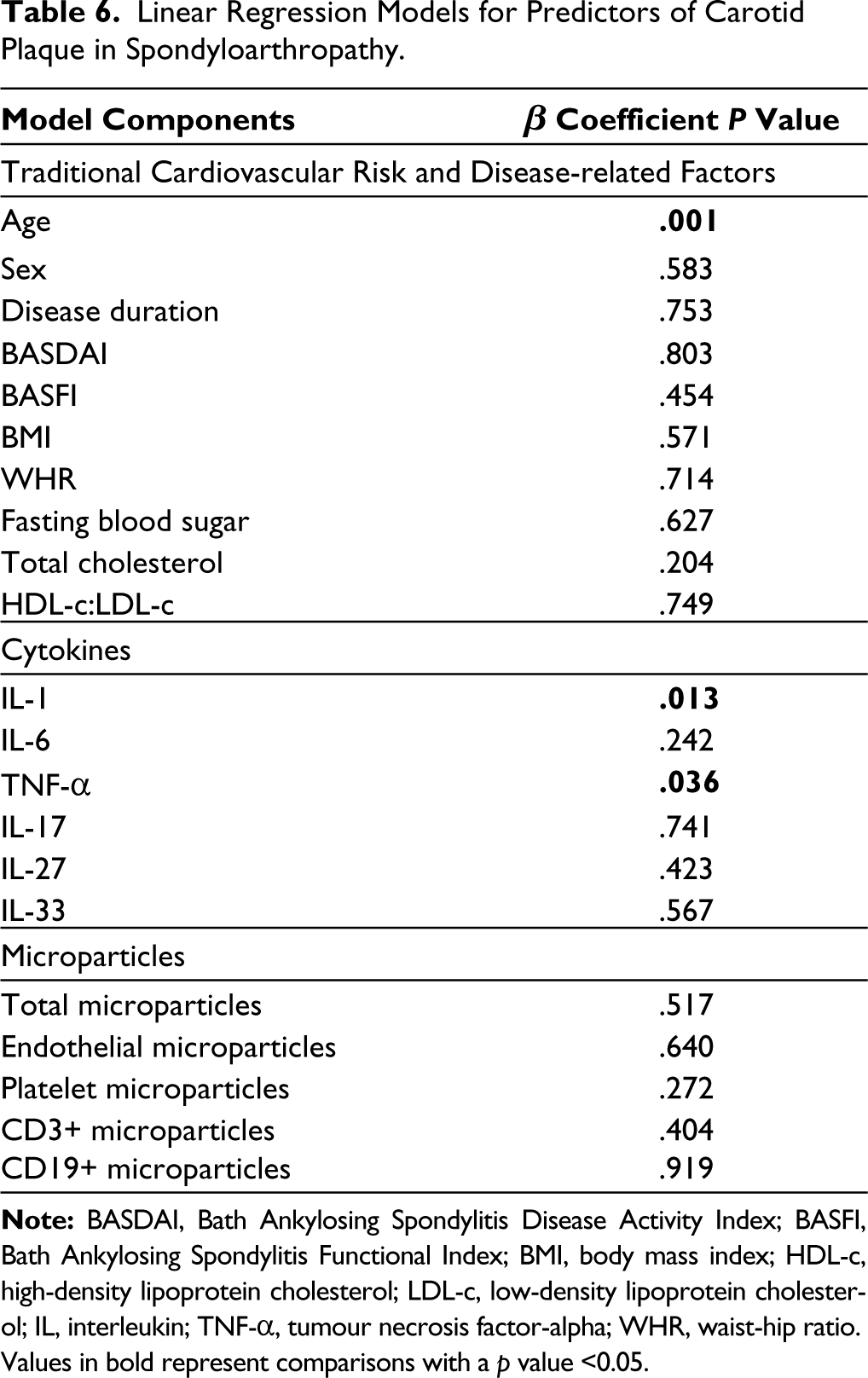
Values in bold represent comparisons with a p value <0.05.
Discussion
We identified a high burden of SCA (40.38%) and carotid plaques (14.42%) and increased CIMT in Indian patients with SpA. Traditional cardiovascular risk factors (older age, higher waist-hip ratio) and novel markers of inflammation (serum IL-1β, IL-6) were associated with SCA. While total microparticles, EMP, T lymphocyte and B lymphocyte microparticles were increased in SpA than in healthy controls, they were not associated with SCA.
We observed SCA in more than a third of patients with SpA, a disease that commonly affects young persons. Also, carotid plaques were present in nearly one-seventh of patients with SpA. Such a high burden of SCA is a matter of concern. Carotid plaques are thought to be surrogates of cardiac atherosclerotic burden.18,19 Studies from India have identified SCA in a similar proportion (14%) of patients with rheumatoid arthritis. 15 Indians per se are at a greater risk of cardiovascular disease which also has a younger age of onset than their Western counterparts. 20 The higher burden of SCA in Indian patients with SpA is a matter of concern, particularly because cardiovascular risk factor management is often sidelined in busy outpatient clinics. 21 Rheumatologists should be aware of the heightened risk of cardiovascular disease in Indian patients with SpA and manage cardiovascular risk factors better.
The higher burden of SCA in our patients with SpA was driven predominantly by traditional cardiovascular disease risk factors such as older age, higher waist-hip ratio and more atherogenic lipid profile (lower HDL-c, higher LDL-c). This was observed despite a lower BMI in patients with SpA than in healthy controls. These observations reiterate the need to assess and manage risk factors for cardiovascular disease even in young individuals with SpA.
Some novel markers of cardiovascular risk also were associated with SCA in our patients with SpA. Higher levels of inflammatory cytokines IL-1β and IL-6 were associated with SCA and carotid plaques in SpA. The inflammatory origins of atherosclerosis are increasingly being understood. The landmark CANTOS trial demonstrated that the IL-1 inhibitor canakinumab resulted in a reduction in subsequent cardiovascular events in patients with prior cardiovascular events who were already on optimal doses of statins and anti-platelet agents. 22 IL-6 is another key cytokine driving atherosclerosis. While the IL-6 receptor antagonist tocilizumab increases cholesterol and triglyceride levels, this does not translate into an increase in the risk of cardiovascular events.23,24 A trial of IL-6 inhibition in patients with myocardial infarction had to be prematurely terminated due to poor enrolment. 25 The landmark ENTRACTE study showed comparable cardiovascular safety with tocilizumab or etanercept. 26 A novel IL-6 antagonist ziltivekimab has demonstrated potential efficacy for cardiovascular risk reduction in a phase II trial. 27 While IL-1 and IL-6 antagonists are not of clinical use in SpA, whether their use might reduce cardiovascular risk in SpA might be an avenue for future exploration. Surprisingly, lower levels of TNF-α were associated with SCA in our study. This might relate to the limitation of the study that a majority of SpA had long-standing disease and were on DMARDs.
The cross-sectional design of the study was a limitation. It would be useful to follow up on these individuals over a period to assess evolution in CIMT and the burden of SCA, however, it was beyond the remit of this study. SCA was assessed using carotid ultrasound. The CIMT was significantly higher in our patients with SpA even after statistical adjustment for differences with healthy controls concerning age, BMI and total cholesterol levels. While the CIMT has traditionally been used as a marker of SCA, it is now recognised that smooth muscle hypertrophy might also drive an increase in CIMT, particularly in individuals with hypertension. 28 About one-ninth of our patients with SpA had hypertension. To counter the limitations of assessing SCA with CIMT alone, we also separately analysed the factors associated with carotid plaques, which are a harder end-point of SCA. Multimodality assessment of SCA using coronary computed tomography calcium score, ultrasound assessment of aortic stiffness using aortic augmentation index or aortic pulse wave velocity, or assessment of endothelial dysfunction using brachial flow-mediated dilatation would have added value to the study. 10 The patients with SpA included in the study had a relatively long disease duration and were on treatment with DMARDs, which might have influenced the assessment of inflammatory markers and microparticle populations in the peripheral blood. The use of hard end-points of cardiovascular events by following up patients with SpA after baseline assessment of the traditional and inflammatory markers of cardiovascular disease risk would lend greater credence to the observed increased burden of SCA in these patients and is a work in progress. Our patients with SpA were mostly male, however, this represents the demographics of the patient population seen in our clinic. Of late, increased cardiovascular risk has been recognised with tofacitinib, which is also being used in patients with SpA.29,30 However, none of our patients were on Janus kinase inhibitors. Few of our patients were on biologic DMARDs. This relates to the limited accessibility to costly therapies such as biologic DMARDs in a government hospital such as ours. Therefore, this limits the generalisability of the findings of our study to settings such as in Europe or North America where biologic DMARDs are much more commonly used in patients with SpA.3,30
To conclude, we observed a considerable burden of SCA in our patients with SpA, driven both by traditional, disease-related and novel cardiovascular risk factors. The findings of our study reiterate the need to screen for and manage cardiovascular risk appropriately in Indian patients with SpA despite the younger age of this group of patients.
Footnotes
Author Contributions
The conception and design of the study—DPM, acquisition of data, analysis and interpretation of data—DPM, MKR, NJ, NM, SK, VA. Drafting the article—DPM, MKR; revising it critically for important intellectual content—NJ, NM, SK, VA. Final approval of the version to be submitted—DPM, MKR, NJ, NM, SK, VA. Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved—DPM, MKR, NJ, NM, SK, VA.
Data Availability Statement
All the analyses performed for this article have been reported in the main text or the supplementary files. Data pertaining to the article shall be shared on reasonable request to the corresponding author (Durga Prasanna Misra,
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
The study was approved by the Institute Ethics Committee of SGPGIMS, Lucknow (document submission number 2017-76-IMP-97, date of approval 22 June 2017). All the study participants provided written informed consent.
Funding
This research was funded by SGPGIMS intramural research fund to Durga Prasanna Misra (Ref. No. PGI/DIR/RC/974/2017) dated 14 October 2017.
Prior Conference Presentations
Presented as a poster at the British Society for Rheumatology conference in Liverpool, United Kingdom, in 2019. Presented as an oral paper at the Indian Rheumatology Association Conference (IRACON) 2019 in Puducherry, India.
