Abstract
Background:
While patients with systemic sclerosis (SSc) are predisposed to cardiovascular events, data regarding this from India is sparse. We analysed correlates of subclinical atherosclerosis in Indian patients with SSc.
Methods:
Patients with SSc fulfilling the 2013 classification criteria (n = 61) were recruited after obtaining written informed consent. Clinical risk factors for cardiovascular disease (CVD) were assessed. A carotid ultrasound was performed to assess the mean carotid intima-media thickness (CIMT). Total and endothelial microparticles (EMP, positive for CD31 and CD146) were estimated from plasma. Serum cytokines known to play a role in atherosclerosis (interleukin-1β [IL-1β], tumour necrosis factor-alpha, IL-6 and IL-17) were assessed. Forty-one age- and sex-similar healthy controls were recruited for comparison. Clinical and serological risk factors for CVD were compared between SSc with and without carotid plaque. Linear regression analyses were conducted to identify predictors of CIMT and carotid plaque in SSc.
Results:
Patients with SSc had lower body-mass index than healthy controls, however, had higher CIMT, higher serum IL-1β, IL-6, total microparticles and EMP than control subjects. SSc with carotid plaque (n = 13) were older, and more likely to be male, but demonstrated no differences in serological markers of CVD. On multivariable-adjusted regression analyses, age was the only significant predictor of CIMT in SSc and male sex was the only significant predictor of carotid plaque in SSc.
Conclusions:
Carotid plaques were present in one-fifth of young patients with SSc. Older age and male sex predicted a higher risk of subclinical atherosclerosis in SSc.
Keywords
Introduction
Systemic sclerosis (SSc) is an autoimmune disease which more commonly affects female subjects and is characterized by excessive fibrosis in the skin, lungs and internal organs. Vasculopathy and endothelial injury are key pathophysiologic correlates of SSc, manifesting clinically as Raynaud’s phenomenon. 1 Cardiovascular disease (CVD) is a major cause of morbidity in SSc. Patients with SSc have a 2.36 times increased risk of overall CVD events (1.52 times increased risk of stroke and 2.36 times increased risk of myocardial infarction) than control subjects. 2 Comparative studies have revealed a similar risk of CVD in SSc and rheumatoid arthritis. 3
Atherosclerosis is now recognized as being driven by systemic inflammation which contributes both to plaque progression and plaque instability and is amenable to targeting by blocking inflammatory cytokines such as interleukin-1β (IL-1β) and IL-6.2,4,5 Microparticles are small extracellular particles (size 0.1–1 µm) released from multiple sources including the endothelium. Endothelial microparticles (EMP) have been associated with cardiovascular risk in diabetes mellitus and other diseases where CVD risk is increased. Total as well as EMP are increased in SSc and have been associated with interstitial lung disease and vasculopathy.6–8 Carotid intima-media thickness (CIMT) assessed using carotid ultrasound is a marker of subclinical atherosclerosis which has also been positively correlated with coronary atherosclerosis.9,10
Given that information from India regarding the risk of CVD in SSc is scarce, we evaluated classical CVD risk factors, potential novel CVD markers (total and EMP, inflammatory cytokines implicated in the pathogenesis of atherosclerosis: IL-1β, IL-6, tumour necrosis factor-alpha [TNF-α], IL-17) and carotid ultrasound findings for CIMT and carotid plaque in patients with SSc compared with healthy controls.
Methods
The study design was cross-sectional. Adult patients (age more than 18 years) with SSc fulfilling the 2013 classification criteria from the American College of Rheumatology and the European Alliance of Associations for Rheumatology 11 were consecutively recruited from the outpatient and indoor services at the Department of Clinical Immunology and Rheumatology at Sanjay Gandhi Postgraduate Institute of Medical Sciences (SGPGIMS), Lucknow, India. Patients with SSc with prior cardiovascular events, critically sick patients, pregnant women and patients older than 60 years or younger than 18 years were excluded. A detailed clinical assessment was undertaken. Demographic characteristics, disease duration (defined from the first symptom, whether Raynaud’s or non-Raynaud’s), body mass index (BMI), waist-hip ratio, the type of SSc (limited or diffuse) at diagnosis, prevalent CVD risk factors (diabetes mellitus, hypertension, tobacco use), the use of disease-modifying anti-rheumatic drugs (DMARDs) and atherogenic parameters on fasting lipid profile (total cholesterol: high-density lipoprotein-cholesterol [HDL-c]; HDL-c: low-density lipoprotein-cholesterol) were recorded. For comparison, consenting clinically healthy individuals of similar age and sex were recruited as controls. Approval was obtained from the Institute Ethics Committee at SGPGIMS (document submission number 2017-208-EMP-100, date of ethics approval 29 December 2017) before initiating the study. The study was supported by a research grant from the Indian Rheumatology Association. All the enrolled patients and controls were recruited after obtaining written informed consent between January 2018 and June 2018.
Based on a published study 12 where an effect size of 0.07 mm in CIMT between SSc and controls and standard deviation (SD) of CIMT 0.10 mm were observed, with the assumptions of α = 0.05 and study power of 80%, with a 6:4 ratio between SSc and controls, the minimum number of patients and controls required was calculated as 40 and 27, respectively, using online software. 13 In this ratio, we enrolled 61 patients with SSc and 41 healthy controls.
Carotid ultrasound using B-mode ultrasound (Esaote MyLab Xvision) was performed with a linear transducer array, utilizing electrocardiographic gating to measure the CIMT. Measurement was performed on the common carotid artery distal arterial wall 5 mm proximal to the carotid bulb. During mid-diastole, three measurements were obtained on the left and right sides, the mean of which was calculated to compute the CIMT. Standard definitions were used for carotid plaques.10,14
Plasma was collected and centrifuged at 12,000g(2 minutes), following which supernatant was collected and again centrifuged at 20,000g (90 minutes). The pellets after centrifugation were resuspended in 1 mL phosphate buffer saline (PBS) and centrifuged twice at 20,000g (90 minutes) after resuspending the pellet in PBS. Thereafter, the pellets obtained were stored in 50 µL of PBS at -80°C for future analysis. At the time of analysis, the microparticles were dissolved and resuspended in 100 µL of PBS. In two separate tubes, 5 µL of this solution was collected for test and isotype controls. After treatment with Anti-Annexin V APC, anti-human CD31 FITC, anti-human CD146 PE microparticles and 7 µm size microbeads (all obtained from BioLegend, USA) for 30 minutes, microparticles were analysed using flow cytometry. Total microparticles were identified after comparison with microbeads for Annexin V-positive particles of size 0.1–1 µm. EMP were further identified as positive for CD31 and CD146 (Supplementary Figure S1). Serum cytokines IL-1β, TNF-α, IL-6 and IL-17 were analysed as per the instructions of the manufacturer using enzyme-linked immunosorbent assay kits from R and D Biosciences, USA.
Comparisons (SSc vs controls; SSc with vs without carotid plaque) were performed between continuous variables (mean ± SD) with unpaired Student’s t-test, whereas, categorical variables were compared using the chi-square test. Linear regression models were developed for CIMT and for carotid plaque separately using clinical risk factors for CVD, serum cytokines and microparticles. p < .05 was considered as indicative of statistical significance. STATA 16.1 I/C (STATACorp, USA) was used for statistical analyses.
Results
Sixty-one patients with SSc (38 with diffuse SSc, 23 with limited SSc) and 41 age- and sex-similar healthy controls were recruited. Diabetes mellitus (1/61) and hypertension (2/61) were present in a few SSc patients, who also had lower BMI than controls. CIMT was significantly increased in patients with SSc than controls. Serum IL-1β, IL-6, total microparticles and EMP were elevated in SSc than in controls (Table 1).
Characteristics of Patients with Systemic Sclerosis and Healthy Controls.
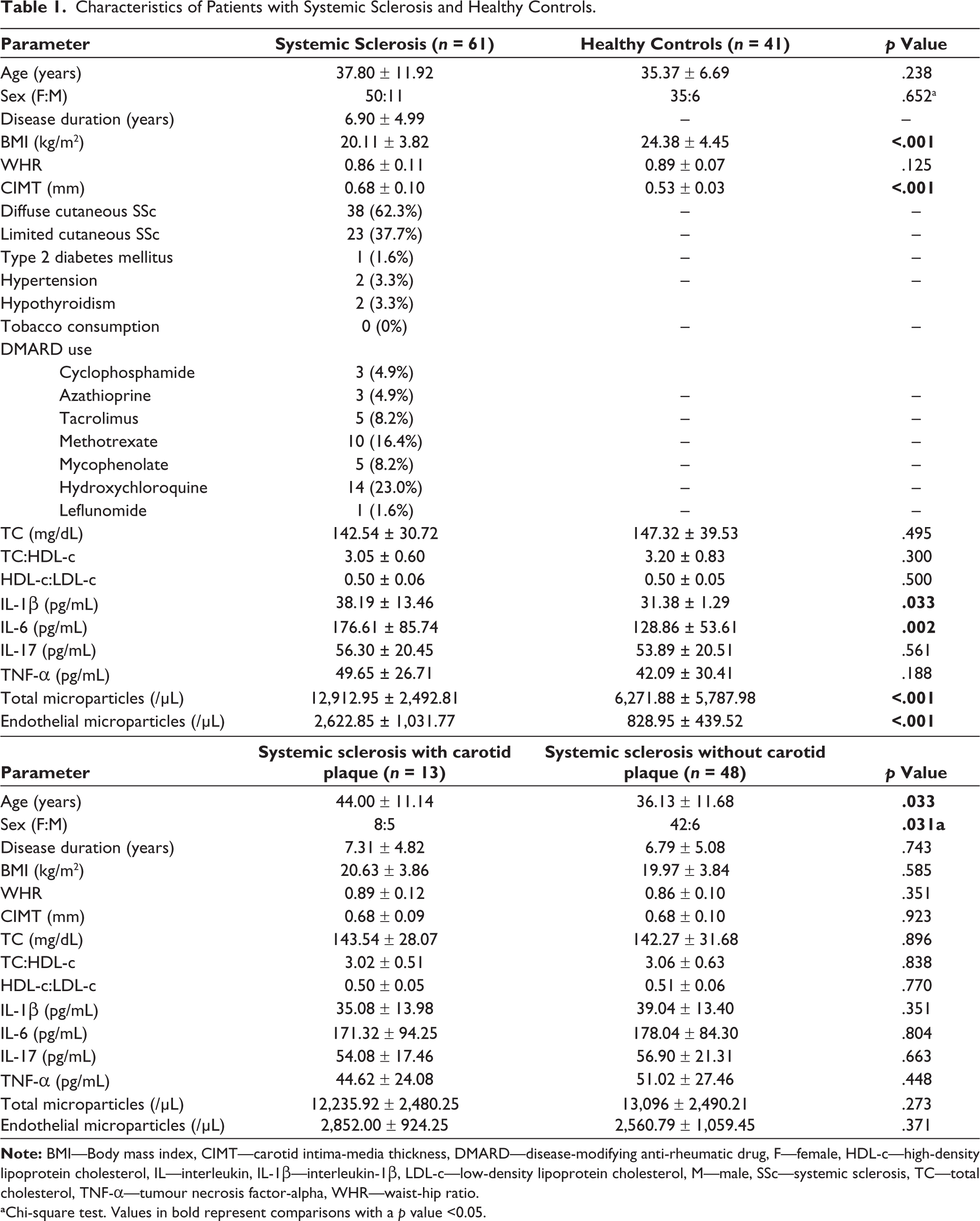
aChi-square test. Values in bold represent comparisons with a p value <0.05.
Thirteen patients with SSc (21.31%) had carotid plaques. SSc with carotid plaques were older and more likely to be male subjects, without observed differences in other traditional CVD risk factors, serum cytokines and total or EMP (Table 1).
On multivariable-adjusted linear regression analyses, only older age predicted a higher CIMT in patients with SSc. Male sex predicted carotid plaques in patients with SSc. Neither inflammatory cytokines nor total or EMP predicted CIMT or carotid plaques in patients with SSc (Supplementary Table S1).
Discussion
Patients with SSc had demonstrable subclinical atherosclerosis denoted by carotid artery plaques in 21%. Despite lower BMI, the CIMT was higher in SSc than in healthy controls. Older age predicted higher CIMT and male sex predicted the presence of carotid plaques in patients with SSc.
The high prevalence of carotid plaques in young patients with SSc was worrisome. Most patients with SSc lacked traditional risk factors for CVD such as diabetes mellitus, hypertension or tobacco use. Indian subjects per se are at a greater risk of CVD. 15 The presence of systemic inflammatory states such as SSc likely drives accelerated atherogenesis. 2 The CIMT was higher in SSc than in healthy controls. Recent literature has criticized the CIMT as possibly reflecting smooth muscle hypertrophy which can occur in the presence of hypertension rather than simply reflecting atheromatous deposits in the carotid artery. 2 However, few of our patients with SSc had hypertension. SSc per se is associated with lower blood pressures other than in the presence of scleroderma renal crisis. The higher burden of subclinical atherosclerosis indicated by higher CIMT in SSc was further corroborated by the high prevalence of carotid plaques. CIMT and carotid plaques correlated with traditional risk factors for CVD, viz., age and male sex. These findings emphasize the need to seek out and manage CVD risk factors appropriately in patients with SSc, as well as the need to advise regarding appropriate lifestyle modification to favourably modulate CVD risk such as regular exercise and a healthy diet.
Although total and EMP as well as inflammatory cytokines IL-1β and IL-6 were increased in SSc, they did not correlate with CIMT or the presence of carotid plaques. A factor influencing the levels of these serological markers could have been that some patients with SSc in the study were on immunosuppressive therapy. Evaluating these markers in newly diagnosed, treatment-naïve SSc might provide more sound insights into their potential role in driving CVD risk in this disease setting.
This study had its limitations. It assessed surrogate markers of CVD in a cross-sectional manner, thereby, causal associations cannot be inferred. We have not evaluated highly-sensitive C-reactive protein as a biomarker of CVD in SSc. The demonstration of hard outcomes such as myocardial infarction, stroke or CVD mortality was beyond the scope of this study and remains an avenue for research in the future. Other, more robust measures to demonstrate subclinical atherosclerosis such as ultrasound-based evaluation of aortic stiffness would have added corroborative evidence to that provided by carotid ultrasound. Future cohort studies of cardiovascular risk in SSc should also explore the effect of DMARDs on subclinical atherosclerosis in SSc.
To conclude, Indian patients with SSc have a considerable burden of carotid atherosclerosis. It is imperative to identify and appropriately manage CVD risk factors in patients with SSc in the clinic.
Supplemental Material
The supplemental material for this article is available online.
Footnotes
Author Contributions
The conception and design of the study—DPM, acquisition of data, analysis and interpretation of data—DPM, HG, MKR, NJ, NM, SK, VA. Drafting the article—DPM, MKR, HG; revising it critically for important intellectual content—NJ, NM, SK, VA. Final approval of the version to be submitted—DPM, HG, MKR, NJ, NM, SK, VA. Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved—DPM, HG, MKR, NJ, NM, SK, VA.
Data Availability Statement
All the analyses performed for this article have been reported in the main text or the supplementary files. Data pertaining to the article shall be shared on reasonable request to the corresponding author (Durga Prasanna Misra,
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
The study was approved by the Institute Ethics Committee of SGPGIMS, Lucknow (document submission number 2017-208-EMP-100, date of approval 29-12-2017). All the study participants provided written informed consent.
Funding
This research was funded by Indian Rheumatology Association research grant (2017–2018) to Durga Prasanna Misra dated 15.07.2017.
Prior Conference Presentations
Presented as a poster at the British Society for Rheumatology conference at Liverpool, United Kingdom in 2019. Presented as a poster at Indian Rheumatology Association Conference (IRACON) 2018 at Guwahati, India.
