Abstract
Primary Sjögren’s disease (pSjD) is a complex autoimmune disorder with a broad spectrum of presentations, often requiring invasive procedures such as lip biopsy for diagnosis. Recent advances in salivary gland ultrasonography (SGUS) have highlighted its value as a noninvasive, real-time imaging modality capable of detecting glandular abnormalities in patients with suspected pSjD. This review evaluates the diagnostic utility of SGUS, with a focus on the standardised Outcome Measures in Rheumatology Clinical Trials (OMERACT) scoring system, and its correlation with histological and serological findings. SGUS demonstrates high specificity and reproducibility, and when integrated with clinical and laboratory features, it may reduce reliance on invasive procedures. Furthermore, the article explores comparative advantages over traditional tests, challenges in operator variability and future directions including the integration of artificial intelligence to enhance diagnostic accuracy and clinical decision-making. With increasing evidence and improved standardisation, SGUS is positioned to play a central role in the diagnostic pathway for pSjD, particularly in early, seronegative, or equivocal cases.
Keywords
Introduction
Primary Sjögren’s disease (pSjD) is a chronic, systemic autoimmune disorder that primarily affects exocrine glands, leading to symptoms such as xerostomia (dry mouth) and keratoconjunctivitis sicca (dry eyes). It can also involve multiple organ systems, including the lungs, kidneys, skin and nervous system. The pathogenesis involves lymphocytic infiltration and destruction of salivary and lacrimal glands and an increased risk of lymphoma in longstanding disease.1,2 Due to the heterogeneous presentation of pSjD, diagnosis remains complex and multifactorial.
According to the 2016 ACR/EULAR classification criteria, the SjD diagnosis is based on a combination of clinical symptoms, serological markers (anti-SSA/Ro and anti-SSB/La), Schirmer’s test and minor salivary gland biopsy (mSGB), with each assigned significant weight in the diagnostic score. 3 Labial salivary gland biopsy remains a cornerstone in this framework, as supported by both historical and recent evidence.4,5 Table 1 mentions the 2016 ACR/EULAR classification criteria for SjD diagnosis. However, lip biopsy, while being a cornerstone of classification criteria, 3 is invasive, can result in discomfort or complications and may sometimes be inconclusive as it may yield variable results depending on sample quality and histopathologist expertise.4,5
2016 ACR-EULAR Classification Criteria for Primary Sjögren’s Disease (pSjD). 3

A total score greater than 4 indicates a positive diagnosis of pSjD.
Inclusion criteria: Any patient presenting a positive response with at least one of the following symptoms: persistent dry eyes or mouth for more than 3 months, recurrent sand or gravel sensation in the eyes, using tear substitutes more than three times a day or frequently drinking liquids.
In recent years, ultrasound (US) of salivary glands has gained prominence as a potential noninvasive alternative or adjunct to histological examination. Salivary gland ultrasonography (SGUS) is increasingly recognised as a valuable, noninvasive diagnostic tool. It enables real-time visualisation of glandular changes and is particularly useful in patients with early, seronegative, or equivocal disease presentations.
This review provides a comprehensive evaluation of SGUS in pSjD, focusing on its diagnostic performance, standardisation via Outcome Measures in Rheumatology Clinical Trials (OMERACT) scoring, comparison with earlier Hocevar scoring systems and evaluation of whether it can reliably reduce or replace the need for lip biopsy.
Methodology
This article is a narrative review that synthesises current evidence on the role of SGUS in diagnosing pSjD, with a focus on the OMERACT scoring system, comparative diagnostic utility and technological innovations. A comprehensive and purposeful literature search was conducted using three electronic databases, yielding the following number of articles during the initial search: PubMed (n = 86 articles), Google Scholar (n = 428 results screened) and ScienceDirect (n = 375 articles), covering publications from 2000 to 2024.
Search terms included combinations of the following keywords: ‘salivary gland ultrasound’, ‘primary Sjögren’s syndrome’, ‘OMERACT scoring’, ‘ultrasound vs lip biopsy’, ‘noninvasive diagnosis’ and ‘artificial intelligence in Sjögren’s syndrome’. Additional sources were identified through manual reference screening of key review articles, clinical guidelines and landmark studies. As this was not a systematic review, no formal inclusion/exclusion criteria or Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) flowchart were applied. Instead, studies were selectively included based on their relevance to the following core themes:
Diagnostic performance of SGUS and standardisation efforts (e.g., OMERACT, Hocevar scoring) Comparative studies involving lip biopsy and serological markers Technical advancements such as elastography, Doppler and artificial intelligence (AI) integration Reproducibility and interobserver agreement data Clinical applicability in early, seronegative, or equivocal pSjD cases
The review also incorporates authorial clinical perspectives derived from real-world use of SGUS and OMERACT scoring in rheumatology practice. The aim was to offer a comprehensive, practical and forward-looking synthesis to inform clinicians and researchers of SGUS’s emerging diagnostic value in pSjD.
Ultrasound as a Diagnostic Tool in pSjD
SGUS is a nonionising, noninvasive and less expensive technique that can detect structural gland abnormalities in the early stages of the disease, and is gaining traction in the diagnostic aids of SjD.6,7 SGUS enables assessment of the parotid and submandibular glands using greyscale and Doppler US. Standardised SGUS protocol recommendations emphasise the use of high-frequency linear probes, typically ranging from 8 to 15 MHz in mode B, which provide optimal resolution for superficial structures such as the salivary glands. The patients must be in seated or supine position with the neck extended and chin turned away from the examined side.8-10 Longitudinal and transverse views of each gland should be obtained, and greyscale imaging is essential for structural evaluation. Colour Doppler or Power Doppler may be used to assess vascular flow, which is particularly useful in detecting advanced or fibrotic disease. These standardised imaging conditions improve reproducibility and facilitate the application of scoring systems such as Hocevar or OMERACT.
The SGUS is highly indicative in the diagnosis of SjD, because of the proven specificity of 98% in a study performed by Baldini et al.
11
on a group of suspected pSjD patients. The key sonographic features indicative of pSjD include:
Heterogeneous echotexture with multiple hypoechoic areas suggestive of lymphocytic infiltration and acinar destruction.6,11 Hyperechoic bands representing fibrotic changes or fatty infiltration.
12
Loss of glandular borders or architecture, often seen as a lobulated or irregular gland shape.
13
Reduced vascular flow, particularly noted in advanced disease with gland atrophy.
14
These features mirror histopathological findings such as periductal lymphocytic infiltrates and acinar atrophy. US has been shown to correlate well with both disease activity and glandular pathology, and can even predict positive lip biopsy results.15,16
Ultrasound Grading Scale: A Walkthrough on Previous Methods
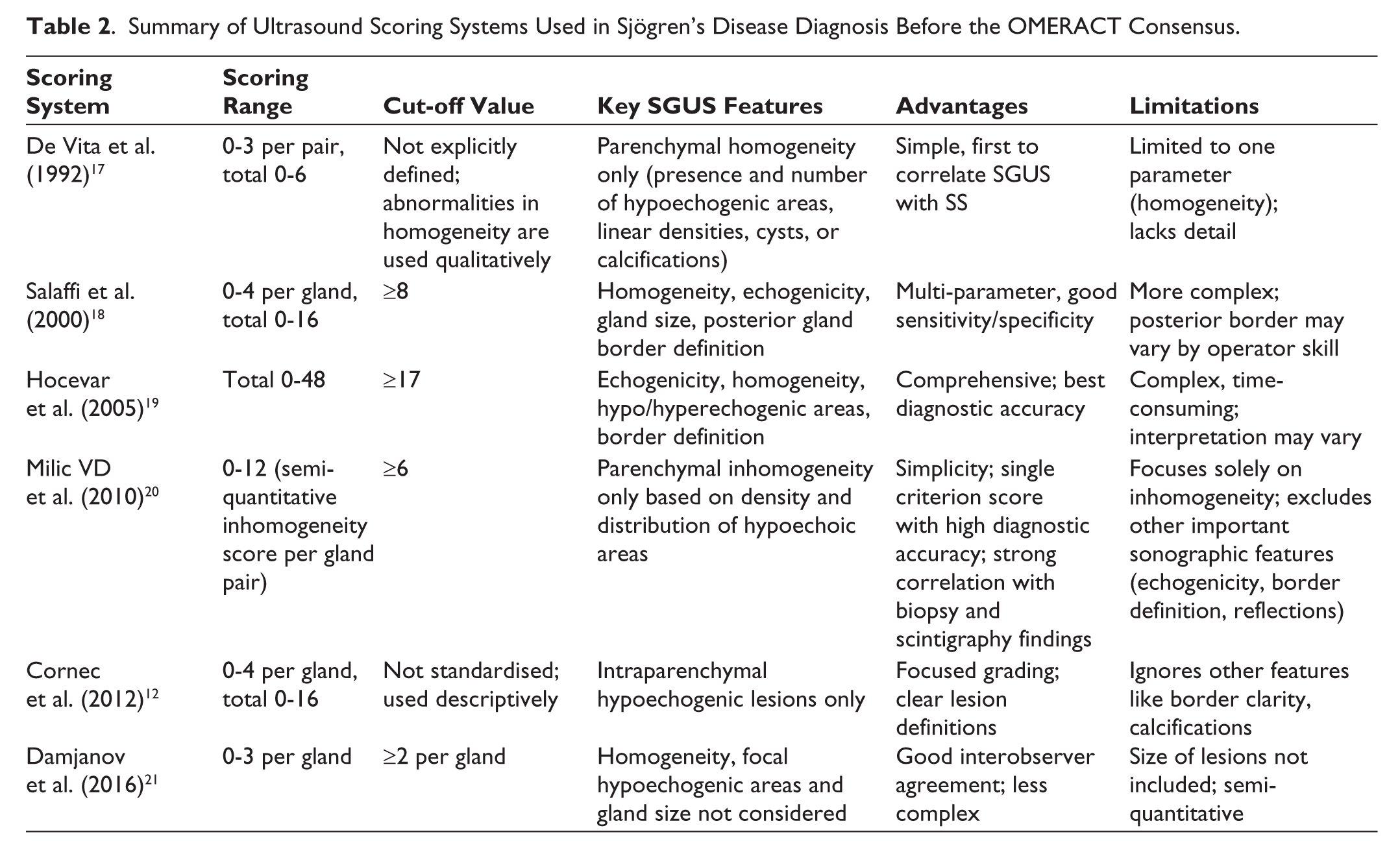
Over the years, multiple US grading systems have been developed to evaluate salivary gland involvement in SjD, each with unique criteria, number of glands assessed and levels of complexity. These scales have aimed to quantify the extent of glandular damage, especially parenchymal inhomogeneity, hypoechogenic areas and gland border irregularities. While early models provided simple visual assessments, later versions incorporated more granular features and scoring thresholds to enhance diagnostic accuracy. Table 2 summarises key US scoring systems developed before the OMERACT consensus.
Summary of Ultrasound Scoring Systems Used in Sjögren’s Disease Diagnosis Before the OMERACT Consensus.
Standardisation: The OMERACT Scoring System
To standardise US in pSjD, the OMERACT US working group developed a novel, semiquantitative grading system, consensus definitions and an image atlas based on greyscale changes observed in the major salivary glands in patients with known or suspected pSjD. 22
Table 3 presents the OMEACT scoring and definitions for Salivary Gland Ultrasound. The OMERACT scoring system defines a positive US finding for pSjD as the presence of a score ≥2 in at least one salivary gland. 23 In a study by Fana et al., this cut-off value demonstrated a sensitivity of 0.72 and a specificity of 0.91 when applied to 43 patients with pSjD who met the 2016 ACR/EULAR classification criteria. The study also found that patients with a score ≥2 in any gland frequently exhibited positive autoantibodies, abnormal sialometry and Schirmer’s test results, as well as positive labial gland biopsies. 23
In a study of 419 patients with suspected SjD, incorporating the SGUS OMERACT score (cut-off value ≥2) into the 2016 ACR/EULAR classification criteria maintained excellent diagnostic accuracy (AUC 0.974), with high sensitivity (96.4%) and specificity (86.5%). The SGUS OMERACT score alone demonstrated strong predictive value (area under the curve [AUC] 0.868), comparable to the total OMERACT score and the Hocevar score (AUC 0.864). The sensitivity and specificity of the Hocevar score of ≥15 in predicting clinical diagnosis were 59.9% and 97.2% respectively, whereas the highest SGUS OMERACT scoring showed 74.9% and 85.7% respectively. These results support SGUS as a valuable addition to standard criteria, improving diagnostic precision in clinical practice. 25
In a single-centre cohort, a global SGUS OMERACT score >6 showed strong correlation with key objective sicca measures, including positive labial gland biopsy, Antinuclear antibodies (ANA) and Ro-SSA antibody positivity and salivary gland fibrosis, despite moderate sensitivity (32.4%) and high specificity (96.8%). 26 In a larger multicentre study with 246 pSjD and 167 non-pSjD patients, SGUS with the OMERACT scoring achieved excellent diagnostic accuracy (AUC ~0.91), with optimal per-gland cut-offs yielding 75%-77% sensitivity and >91% specificity. SGUS positivity was also associated with clinical indicators of disease severity such as prolonged disease duration, parotid enlargement, dental loss and elevated serological markers. 27 These findings support SGUS with the OMERACT scoring as an effective, noninvasive imaging tool that correlates well with traditional diagnostic measures and may serve as a reliable alternative, especially when biopsy or serological tests are inconclusive or unavailable.
Recent studies have validated the reliability and reproducibility of the OMERACT system across different operators and clinical settings.24,28 Ultrasonographic evaluation of minor salivary glands using the OMERACT scoring system by Izzetti et al. demonstrated good inter-reader reliability, with intraclass correlation coefficient (ICC) values exceeding 0.9 for scores 0 and 1, and acceptable agreement for scores 2 and 3 (ICC = 0.873 and 0.785 respectively). These findings support the reproducibility of US interpretation among independent examiners in 170 patients with suspected pSjD. 24 In another study, the consensually agreed ultrasonographic scoring system for assessing major salivary gland lesions in SjD demonstrated substantial intra- and inter-reader reliability, with inter-reader kappa (κ) values of 0.62 for both parotid and submandibular gland grading. Intrareader weighted κ values ranged from 0.44 to 1 for parotid glands and 0.59 to 1 for submandibular glands. In contrast, the inter-reader agreement for detecting Sjögren’s signature was lower, with κ values of 0.36 for parotid glands and 0.03 for submandibular glands. 28 In cohorts with suspected pSjD, US findings in major salivary glands strongly correlated with positive lip biopsy findings and anti-SSA and/or Anti SSB positivity. 29
Ultrasound Versus Biopsy: Comparative Performance and Advantages
Salivary gland biopsy is considered to be the gold standard in the diagnosis of SjD.
10
Yet, SGUS offers several practical and clinical advantages over biopsy:
Noninvasiveness: US does not require tissue sampling, avoiding the risks of pain, infection, scarring and sensory nerve damage.
30
Real-time visualisation: Allows immediate assessment of gland morphology and can be repeated to monitor progression or therapeutic response. Reproducibility: With the OMERACT scoring, interobserver agreement has improved significantly.24,28 Jousse-Joulin et al. reported that intraobserver reliability for detecting and scoring ultrasonographic abnormalities was excellent (Cohen’s kappa 0.81) and interobserver reliability was good (Light’s kappa 0.66).
22
Patient acceptability: Higher tolerability, especially in elderly or co-morbid patients. Cost-effective and accessible: Particularly valuable in low-resource settings where histopathology services may be limited.
The literature on the SGUS versus biopsy yields inconclusive results on the performance of each diagnostic mode. For instance, a systematic review and meta-analysis by Martins et al. assessed how accurately major SGUS can diagnose SjD compared to mSGB. In the first meta-analysis, SGUS demonstrated strong diagnostic performance, with pooled sensitivity and specificity of 86% and 87% respectively, when compared to biopsy outcomes. However, in the second meta-analysis, direct comparisons showed that mSGB slightly outperformed SGUS, with higher sensitivity (80% vs. 71%) and specificity (94% vs. 89%). These findings suggest that SGUS offers high diagnostic value and may help reduce the need for invasive biopsy in selected cases. 7 Baldini et al. compared the performance of SGUS with mSGB in a study performed on 50 pSjD and 57 control patients. The study demonstrated that SGUS is equally specific (98%) and less sensitive (66%) compared to mSGB. 11 Based on multiple studies results, biopsy remains essential, especially when serological and clinical findings are inconclusive. SGUS should be viewed not as a replacement, but as a frontline, noninvasive imaging tool to guide diagnostic decisions and potentially reduce the need for biopsy in clearly positive or negative cases.
Comparative Advantages Over Traditional Diagnostic Tests
Traditional diagnostic tools for pSjD, such as Schirmer’s test, unstimulated salivary flow measurement, serological markers and lip biopsy, each have their limitations in terms of accuracy, patient burden and time consumption. For example, Schirmer’s test, while widely used, has low specificity and can yield false positives due to factors unrelated to autoimmune disease. 31 Similarly, salivary flow measurements are influenced by hydration status, medications and anxiety, and require multiple timed measurements to be reliable. 32 Anti-SSA/Ro antibodies, while specific, may be absent in a significant number of pSjD patients, leading to delayed diagnosis. 33 Lip biopsy, although definitive, poses risks and logistical burdens. In this context, Salaffi et al. performed a comparative study to evaluate the performance of SGUS with contrast salivary sialography and scintigraphy. The receiver operating characteristic (ROC) curve analysis showed SGUS (AUC = 0.863) to be the best performer followed by sialography (AUC = 0.804) and scintigraphy (AUC = 0.783). 34
US offers a major advantage as a quick, point-of-care imaging tool, often taking less than 10 minutes with no patient prior preparation.8,23 It can be performed at bedside or in outpatient settings, and unlike Schirmer’s test or salivary measurement, it is objective, reproducible and not subject to patient cooperation or external variables. When combined with the OMERACT grading, it allows standardised and operator-independent scoring of disease severity. Moreover, SGUS can identify candidates who are likely to have a positive lip biopsy, potentially reducing the need for invasive procedures.
In many clinical scenarios, US may outperform clinical tests by directly visualising glandular pathology. For example, a patient with dry mouth, borderline anti-SSA levels and equivocal Schirmer’s test may benefit from a targeted US to rapidly assess for pSjD without waiting for lab results or arranging a biopsy. Given its speed, accessibility and reliability, US may emerge as the first-line diagnostic modality, especially in early or seronegative disease.
Limitations and Future Directions
Despite its promise, SGUS is not without limitations. Operator expertise and equipment quality significantly affect image acquisition and interpretation. Differentiating pSjD from conditions such as immunoglobulin G4-related (IgG4)-related disease, amyloidosis or sarcoidosis remains challenging due to overlapping sonographic features.35-37 While pSjD often presents with marked glandular inhomogeneity and multiple hypoechoic foci, conditions like sarcoidosis and amyloidosis may exhibit nodular hypoechoic changes with preserved architecture.35,36 IgG4-RD may mimic pSjD but usually shows more diffuse hypoechogenicity and increased vascularity. 37 Thus, SGUS findings should be interpreted alongside clinical, serological and histopathological data to avoid misclassification.
Future directions include:
Broader training of rheumatologists in SGUS techniques. Integration of SGUS into international classification and diagnostic criteria. A study performed by Cornec et al. in a cohort of 101 patients with suspected SjD, which added SGUS to the 2012 ACR criteria, improved diagnostic sensitivity from 64.4% to 84.4%, with a minor reduction in specificity from 91.1% to 89.3%.
38
These findings support the integration of SGUS into future classification criteria to enhance diagnostic accuracy. Following the standard imaging protocol to enhance the reproducibility of the test results. Large-scale prospective trials validating AI-driven SGUS models.
Clinical Integration and Algorithmic Use
Emerging data suggests that when combined with serology (anti-SSA) and clinical features, SGUS can yield diagnostic sensitivity and specificity comparable to full ACR/EULAR criteria.3,33 The studies26,27 findings are highly proved correlation and importance of incorporating serological markers to improve the diagnostic accuracy of SjD. Astouri et al. have proposed triaging patients for biopsy based on US scores and serology to reduce unnecessary biopsies. The retrospective study was performed on 85 patients, and US demonstrated 85% positive predictive value (PPV) and 96% negative predictive value (NPV). 39 Additionally, Mo et al. demonstrated that combining SGUS with serological data in a risk matrix could predict biopsy positivity with over 85% accuracy. 15 This supports a triage model where high SGUS scores may obviate the need for biopsy, while low scores in seronegative patients may still necessitate histopathological confirmation.
Emerging Technologies and AI Integration
Technological advancements continue to refine US’s role in pSjD. Contrast-enhanced ultrasound sialendoscopy (CEUSS) and elastography are being explored for better tissue characterisation.40,41 AI and machine learning tools are being developed to automate image analysis, reduce inter-observer variability and enhance diagnostic precision. 42 Longitudinal studies are assessing US as a tool for monitoring disease activity and treatment response.13,43
The combination of standardisation (OMERACT scoring), AI integration and improved training could help make US a central, reliable and noninvasive tool in the pSjD diagnostic algorithm.
Conclusion
SGUS represents a paradigm shift in the diagnostic landscape of pSjD. Its noninvasive nature, correlation with histological and serological markers and rapid bedside applicability make it a valuable tool in clinical rheumatology. While not a complete substitute for lip biopsy in all scenarios, SGUS can substantially reduce the need for invasive procedures, particularly in patients with positive serology or overt glandular abnormalities. With continued standardisation and validation, SGUS has the potential to not just complement but redefine diagnostic algorithms in pSjD.
Author’s Viewpoint
We have incorporated bedside SGUS into our clinical workflow for patients with suspected pSjD. The application of OMERACT scoring allows objective assessment of glandular changes, especially in early or seronegative cases. When used alongside serology and clinical evaluation, US has significantly reduced our reliance on lip biopsy. As training and AI tools improve accessibility and accuracy, US has the potential to become a frontline tool for rheumatologists managing pSjD.
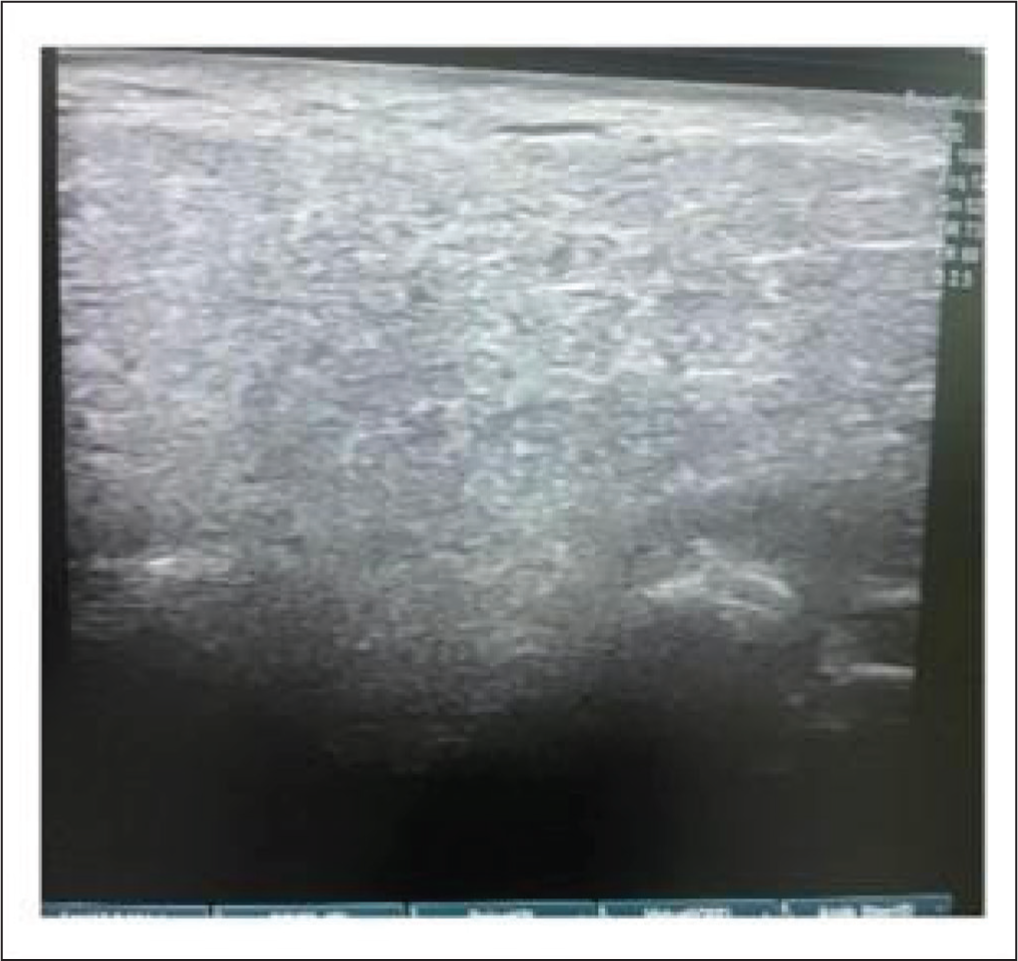
Normal Parotid gland-Grade 0
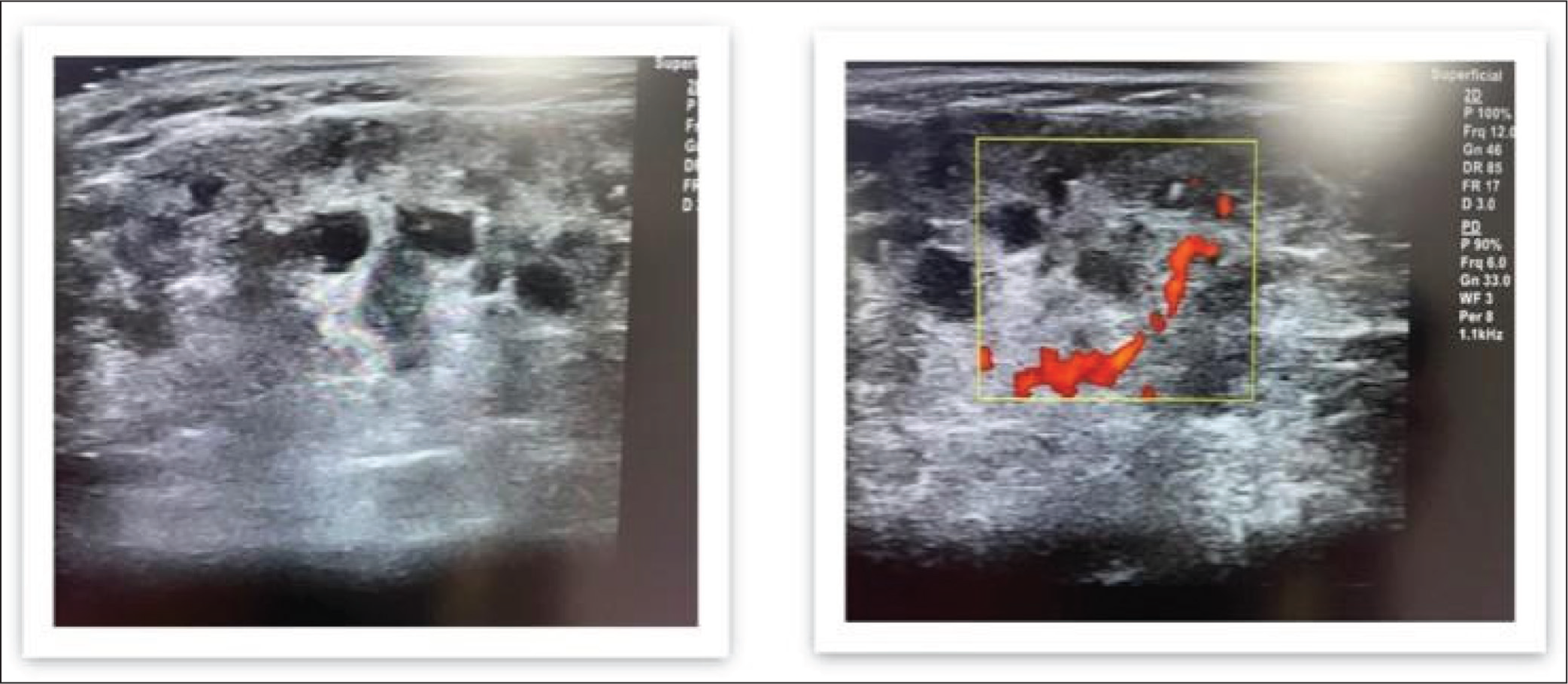
Parotid USG: Grade-3 changes; Parotid USG: Grade-3 changes with Doppler
Images of bedside parotid gland US showing normal parotid gland and USG with grade 3 changes with and without Doppler.
Footnotes
Data Availability
This review article is based on previously published literature. No new datasets were generated or analysed during the current study.
Declaration of Conflicting Interests
The authors declare no conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval and Informed Consent
This is a narrative review article. No human participants or animals were involved, and therefore, ethical approval and informed consent were not required.
Funding
No specific grant from any funding agency in the public, commercial or not-for-profit sectors was received for the preparation of this article.

