Abstract
Background and Aims:
Rheumatoid arthritis (RA) is a complex autoimmune inflammatory disease-causing disability. The immunopathogenic difference between elderly onset RA (EORA) and younger-onset RA (YORA) and the factors responsible for their clinical characteristics are yet to be explored completely. The study was done to correlate inflammatory biomarkers in EORA patients and compare with YORA patients.
Methods:
A cross-sectional study comprising 30 patients each for YORA and EORA was done. Serum levels of interleukin IL-1β, IL-6, IL-8, tumour necrosis factor-alpha (TNF-α), interferon-gamma (IFN-γ) and anticyclic citrullinated peptide were determined by Enzyme-Linked Immunosorbent Assay (ELISA). These were correlated with disease activity using the DAS28, and the modified Larson score was used to assess bone erosion.
Results:
Significantly higher levels of serum IL-6 (25.1 vs. 12.8 pg/mL) and IL-8 (83.5 vs. 65.15 pg/mL) were found in EORA patients, while significantly higher levels of serum TNF-α (360.8 vs. 86.3 pg/mL) were found in patients with YORA. IL-1β, IL-8 and TNF-α have a significantly positive correlation with DAS28 in YORA, while IL-1β, TNF-α and IFN-γ are significantly associated with disease activity in EORA. Increased bone erosion was linked to EORA.
Conclusion:
Apart from clinical symptoms, serological profile, EORA, and YORA have a distinct cytokines profile. This provides a valuable insight for selecting targeted therapies, especially in managing naive and refractory RA cases of EORA. Given the higher risk of joint damage in EORA, early and aggressive management may result in early remission and improve patient’s quality of life.
Introduction
Rheumatoid arthritis (RA) is a complex, multisystem autoimmune inflammatory disorder primarily affecting the synovial linings of joints and tendons. 1 Elder onset RA (EORA) refers to cases where the disease begins at the age of ≥ 60 years and accounts for 10%–33% of total RA cases. 2 It affects up to 2.2% of individuals over 60 years of age. 3 Compared with young onset RA (YORA), EORA tends to have a more acute course, with more commonly experienced constitutional symptoms such as myalgia, weight loss, rheumatoid nodules, lymphadenopathy and neuropathy. 4 EORA patients tend to experience longer morning stiffness and higher disease activity compared with YORA. 5 EORA patients are also more likely to present with atypical forms such as remitting seronegative symmetrical synovitis with pitting oedema (RS3PE) and polymyalgia rheumatica-like forms. 6 As compared with men, women are ~3 times more likely to be affected by RA, although this gender difference diminishes in older populations. 4 The shoulder joint and other large joints are more frequently involved in EORA, and lung involvement, such as interstitial lung disease, is significantly less common compared with YORA patients.
The exact mechanism of RA is still to be deciphered; however, multiple mechanisms contribute to its development. One of the key elements in RA is the production of high amounts of inflammatory cytokines by synovial fibroblasts that activate immune cells and play a crucial part in the initiation and progression of the disease. The hallmark inflammation seen in RA is primarily driven by the abundance of these inflammatory cytokines. 7 TNF-α is considered the main pro-inflammatory cytokine and plays a main role in regulating the production of many other inflammatory mediators within the synovial tissue. 8 It also contributes to the destruction of bone and cartilage by activating chondrocytes and osteoclasts. 9 Additionally, IL-1 β, IL-6, IL-17A and IL-17F, the signature cytokines of Th17 cells, are involved in the pathogenesis of RA. 10 In addition to increasing inflammation, pro-inflammatory cytokines also promote the release of chemokines, the activation of matrix metalloproteinases, the differentiation of osteoclasts, and the production of inducible nitric oxide synthase, as well as the expression of cell adhesion molecules. 9 In response to inflammation, various anti-inflammatory cytokines such as IL-4 and IL-10 play a role as a compensatory response in the synovium. 11
Disease Activity Score based on 28 joints with Erythrocyte Sedimentation Rate (DAS-28-ESR) is widely utilised in both daily clinical practice and clinical trials for assessing disease activity and response to treatment in RA. Its validity in evaluating treatment outcomes has been well-established.12,13 Rau and Herborn introduced a modified Larsen score, which incorporates a semi-quantitative assessment of joint surface area loss. This method includes standardised reference films for multiple anatomical sites, such as metacarpophalangeal joints, proximal interphalangeal joints, wrists and metatarsophalangeal joints. The modification enhances Larsen’s original scoring system, providing a reliable measure for evaluating baseline status and tracking radiological progression in RA patients. 14
This study was done to correlate the clinical characteristics, disease activity (DAS-28 Score), Erosion score (Modified Larsen Score) and inflammatory cytokines expression profile (IL-1 β, IL-6, IL-8, TNF-α, IFN-γ) in patients with EORA and YORA.
Materials and Methods
Study Type
A cross-sectional study was conducted at a tertiary care hospital in eastern Uttar Pradesh, India.
Study Subjects
Sixty treatment-naive patients with RA diagnosed according to the 2010 American College of Rheumatology/European League Against Rheumatism criteria for RA were recruited consecutively from the outpatient department in this study. 15 No formal sample size calculation was performed for this pilot study. The number of participants was determined based on logistical feasibility and was intended to provide preliminary data for future studies.
The patients were divided into two groups—YORA (30 patients) and EORA (30 patients), based on age. Patients having any other comorbidity, such as diabetes, hypertension, coronary artery disease, or any other chronic illness, were excluded.
YORA and EORA patients were compared, based on tender joint count, swollen joint count, C-reactive protein (CRP), ESR, disease activity score (DAS28) with ESR, anti cyclic citrullinated peptide, rheumatoid factor (RF), pro-inflammatory markers including Interleukin (IL) 1β, 6, 8, tumour necrosis factor-alpha, interferon gamma (IFN-γ), and X-ray of joints for modified Larsen score. Serum levels of IL-1β, IL-6, IL-8, TNF-α, IFN-γ and Anti-Citrullinated Peptide Antibodies (ACPA) were measured by an ELISA reader (BioTek reader). Disease activity of RA was assessed using the 28-joint disease activity score. Erosion score was calculated with modified Larson scores by acquiring joint X-rays of RA patients.
Statistical Analysis
The data were collected and compiled using Microsoft Excel 2020 and subsequently analysed with IBM Statistical Package for the Social Sciences (SPSS) Statistics version 20.0 (SPSS Inc., Chicago, IL, USA). Statistical analyses were performed using SPSS version 20. Continuous variables exhibiting a normal distribution were summarised as mean and standard deviation (SD), while categorical variables and continuous variables not following a normal distribution were described using frequencies, medians and interquartile ranges. Group comparisons for continuous variables were conducted using Student’s t-test, and categorical variables were compared using the Chi-square (χ²) test. Multiple linear regression analyses were performed separately for EORA and YORA groups to identify independent predictors of disease activity (DAS28), with cytokine levels included as explanatory variables. All statistical tests were two-sided, and a P value of <.05 was considered statistically significant.
Results
Demographic Data and Clinical Characteristics
The baseline characteristics and involvement of various joints in YORA and EORA are shown in Table 1. There was no statistically significant difference in baseline characteristics between the groups.
Baseline Characteristics-demographic Data and Clinical Characteristics.
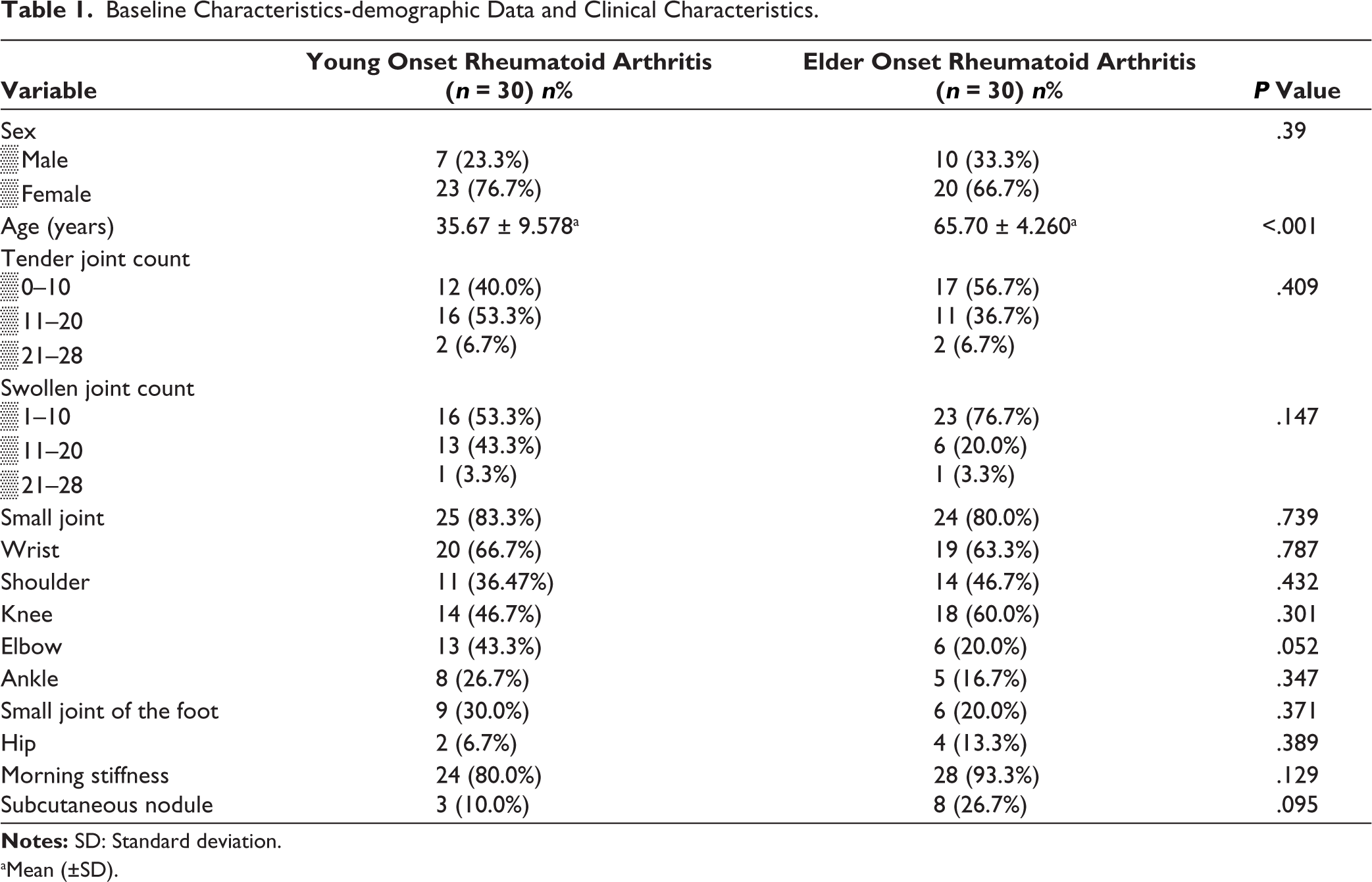
a Mean (±SD).
Radiological, Clinical Disease Activity and Metabolic Profile
Table 2 shows that the mean Larsen score was significantly higher in EORA with respect to YORA. Anti-CCP positivity was higher in YORA than EORA patients, however, not statistically significant. CBC, ESR and CRP parameters showed no significant difference between EORA and YORA patients.
Radiological, Clinical Disease Activity and Metabolic Profile.
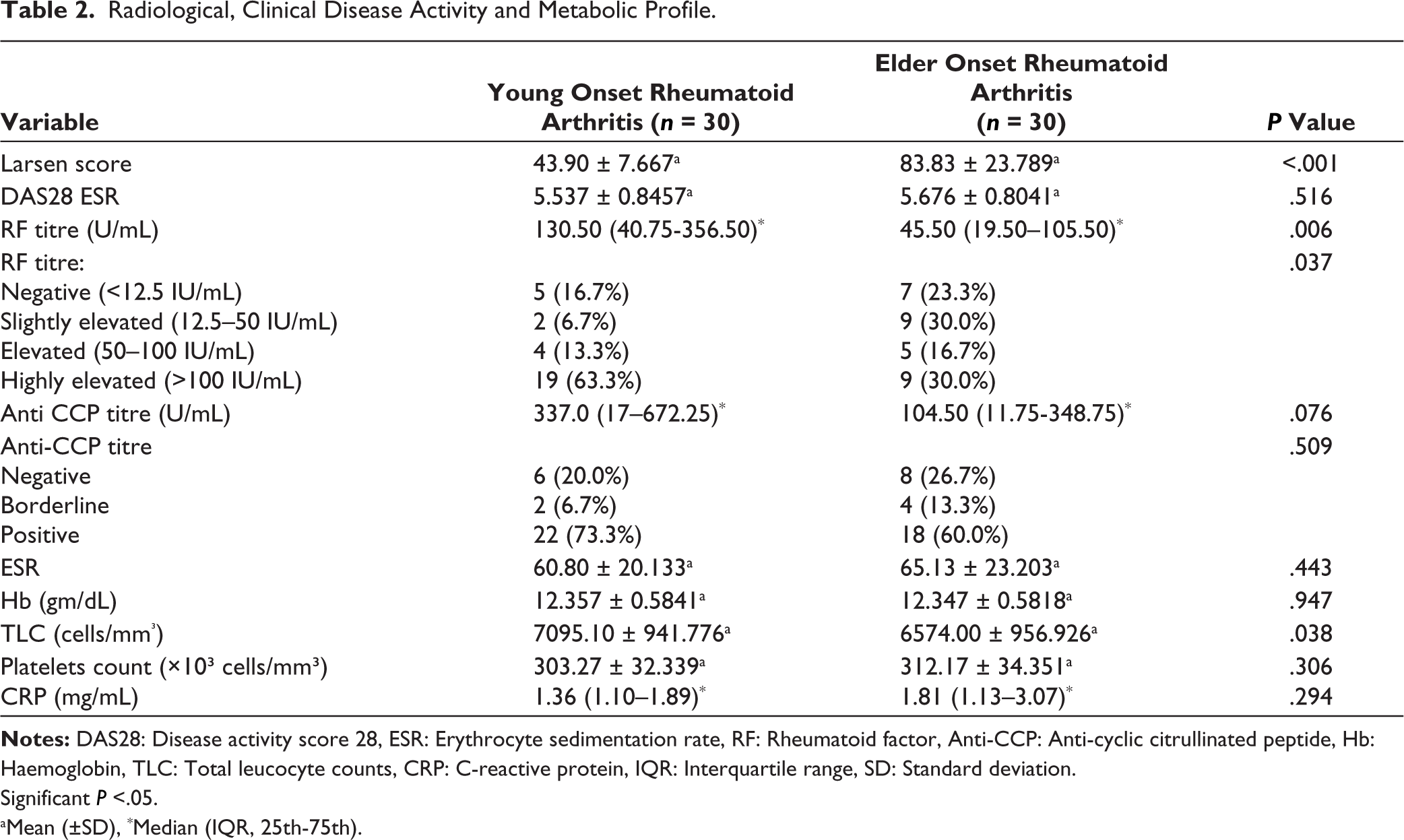
Significant P <.05.
aMean (±SD), *Median (IQR, 25th-75th).
RF titre distribution showed a significant difference between YORA and EORA (P = .037). A higher proportion of YORA patients had highly elevated RF titres, whereas in EORA, this proportion was lower, with more cases falling into the slightly elevated category. This suggests that RF titre levels tend to be higher in younger-onset cases compared with elder-onset cases.
Serum Levels of Cytokines
As shown in Table 3, higher levels of serum IL-1β, TNF-α and IFN-γ were found in YORA patients, while IL-6 and IL-8 were significantly higher in EORA.
Levels of Proinflammatory Cytokines in EORA and YORA Patients.
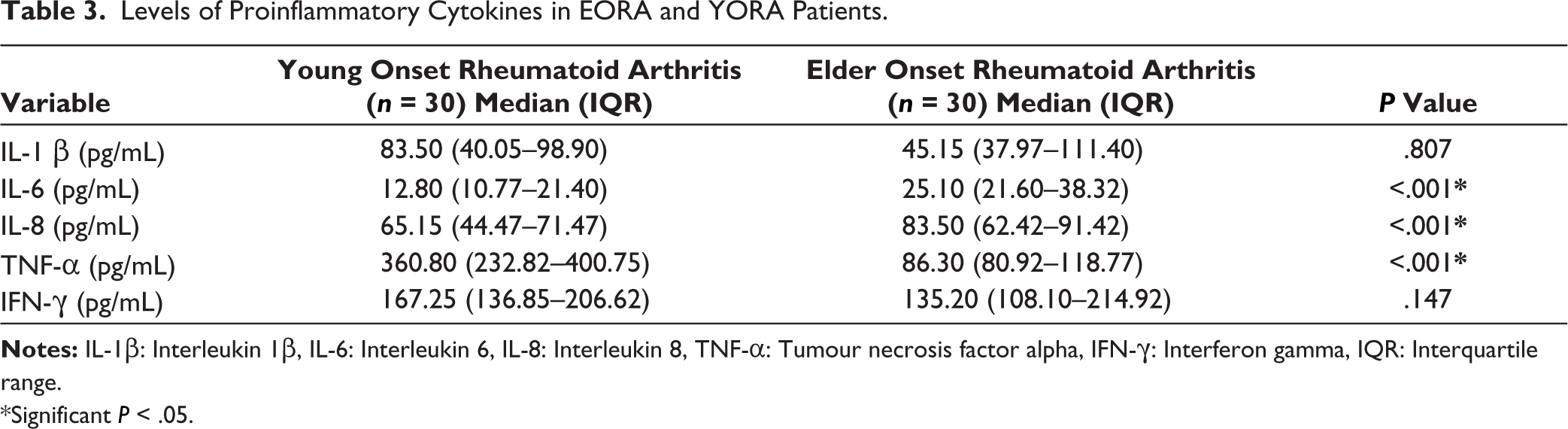
*Significant P < .05.
Correlations of Serum Cytokine Levels with DAS28 in YORA Patients
As illustrated in Table 4, IL-1β (P = .01), IL-8 (P < .001) and TNF-α (P < .001) were significantly associated with DAS28, thereby being strong predictors of disease.
Correlations of Serum Cytokine Levels With DAS28 in EORA Patients
As depicted in Table 5, IL-1β (P < .001), TNF-α (P = .046) and IFN-γ (P = .029) were significantly associated with DAS28, suggesting that their major role in disease activity and IL-1β was the strongest predictor of disease activity. IL-6 and IL-8 showed no significant correlation with DAS28.
Discussion
Individuals with RA frequently experience premature immunosenescence, an accelerated ageing of their immune system. As individuals advance in age, the innate immune system tends to exhibit heightened activity, potentially fostering a state of chronic inflammation and elevating the risk of comorbidities. Simultaneously, the adaptive immune arm of the immune system may experience a decline in efficiency, leading to a disruption of immune tolerance and an increased susceptibility to autoimmune disorders. These age-related immunological shifts likely exert a critical influence on the divergent ways in which RA manifests and progresses in individuals with EORA compared with their YORA counterparts. 16
Multiple Linear Regression Analysis of DAS28 ESR with Serum Cytokines Levels of YORA.
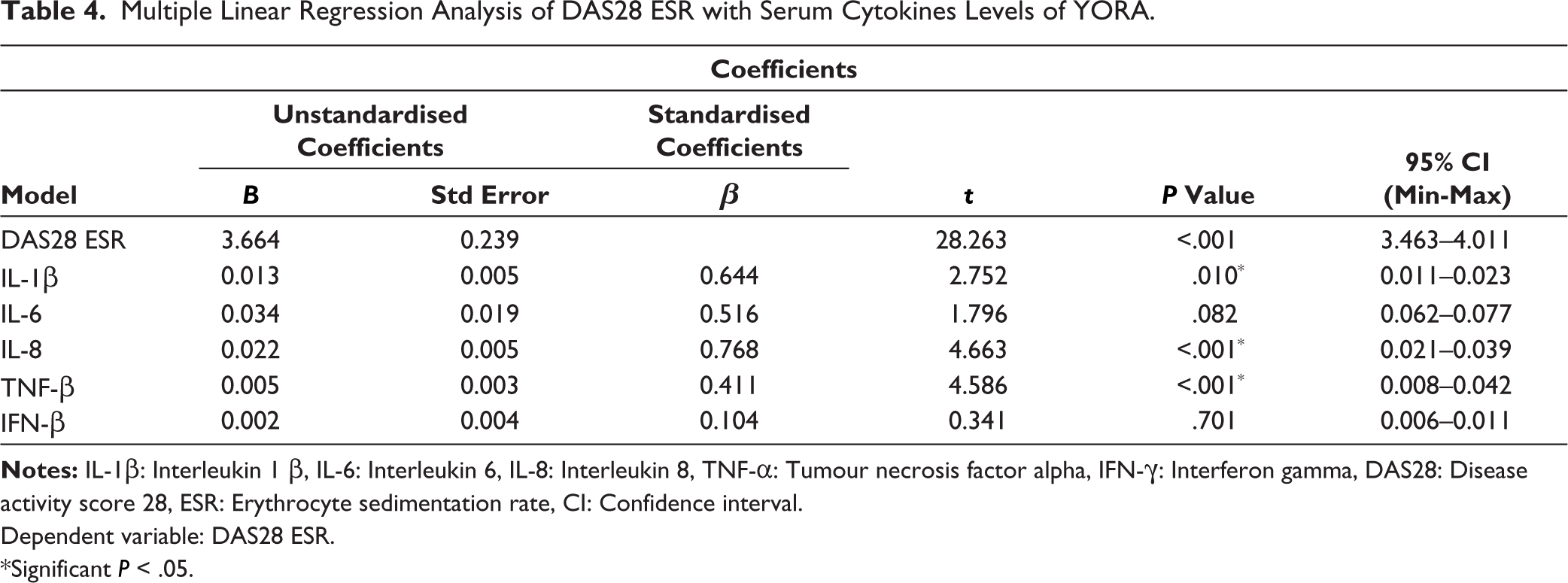
Dependent variable: DAS28 ESR.
*Significant P < .05.
Multiple Linear Regression Analysis of DAS28 ESR with Serum Cytokines Levels of EORA.
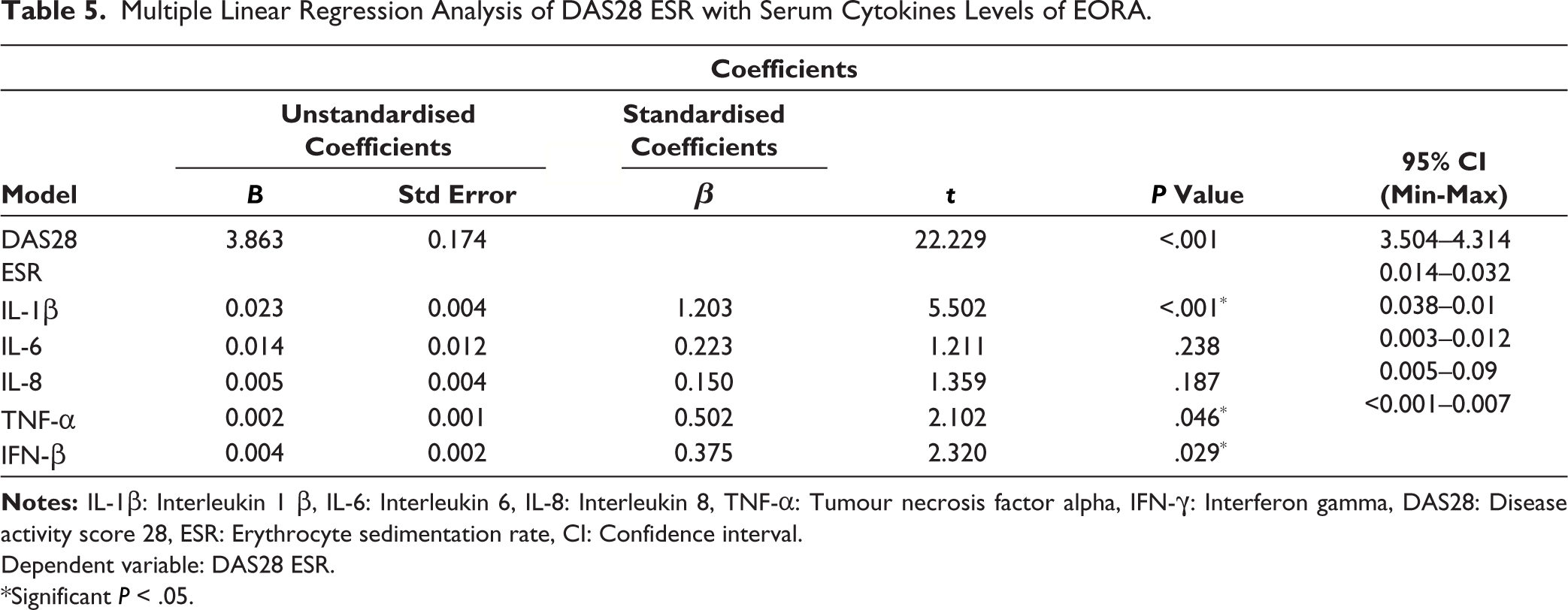
Dependent variable: DAS28 ESR.
*Significant P < .05.
The percentage of males preponderance in EORA was more than YORA; however, it was not significant, which has been previously reported by Deal et al. 17 This gender variation may be linked to the complex interplay between hormone levels (oestrogen, progesterone and androgen) and their immunomodulatory effects. Specifically, the incidence of RA in women tends to decrease following the decline in oestrogen and progesterone levels post-menopause, while older men experiencing a reduction in androgen levels face an elevated risk of developing RA. 4
The pattern of joint involvement between EORA and YORA also exhibited notable differences. Larger joints, such as the shoulder, hip and knee, along with the presence of subcutaneous nodules, were more commonly observed in the EORA group. Conversely, the YORA group showed a greater propensity for the involvement of small joints, namely the hands, wrists, elbows, ankles and feet. This distinct pattern of joint involvement is consistent with findings from several previous studies, including those by Chen, Der-Yuan et al. and Ke Yini et al., reinforcing the concept that the clinical presentation of EORA can differ substantially from that of YORA.18,19
The non-specific inflammatory markers, namely ESR and CRP, were comparatively higher in the EORA group than in the YORA group. The finding was consistent with other studies.20,21 This suggests a potentially greater overall inflammatory burden in EORA.
Various studies have shown that the presence of ACPA is generally linked with increased inflammatory activity, greater disease severity and the progression to erosions and deformities, its clinical significance in EORA remains less well-defined.22,23 In our study, ACPA positivity was more frequent in YORA patients as compared with EORA patients, similar to the study by Krams et al., who also reported a high positivity rate in YORA patients. 24 Another study found that EORA patients who presented with a polymyalgia rheumatica-like onset exhibited a higher rate of ACPA positivity, suggesting a potential subgroup within EORA with distinct serological characteristics. 24 Our study found a higher prevalence of RF positivity in the YORA group compared with the EORA group, similar to the findings of a study done by Tutuncu et al. 25
Evidence from longitudinal studies has demonstrated that, after 2 years of follow-up, there was no significant difference in disease activity, remission rates and joint damage progression between seropositive RA and seronegative RA patients, reflecting that seronegative RA is not a milder form of RA, and aggressive treatment strategies should be initiated at the earliest. 26
In addition to our previous findings, among the seropositive patients, we found that elevated RF was more prevalent among individuals in the YORA group, whereas the EORA group demonstrated a more uniform distribution of this factor. Currently, there are limited data on why RF distribution is different in EORA and YORA and how it correlates to disease prognosis, for which further investigations with larger sample sizes are required.
Our study demonstrated a significantly elevated radiographic score for bony erosion in the EORA cohort compared with their YORA counterparts, highlighting a potentially more destructive articular process in EORA. 27 Specifically, the Larsen score, a quantitative measure of radiographic joint damage, was significantly higher in EORA in our study. This increased bony erosion in EORA could be attributed to the heightened alterations in immune activity associated with ageing, potentially leading to a more aggressive pattern of joint destruction. The duration of RA before diagnosis may also play a role in the extent of bony erosion in patients. However, it was not recorded in our study.
The cytokine, IL-1β, is a potent pro-inflammatory mediator primarily produced by monocytes and macrophages. It plays a crucial role in the inflammatory cascade by stimulating chondrocytes and synovial fibroblasts to release other inflammatory molecules such as prostaglandins, chemokines, and TNF, and by regulating overall inflammatory and acute phase responses.28,29 Our study revealed that Interleukin-1 beta (IL-1β) levels, although not statistically significant, were comparatively higher in the YORA group than in the EORA group. Moreover, we observed a statistically significant positive association between IL-1β levels and disease activity in both YORA and EORA. Similar findings were seen in a study done by Chen, Der-Yuan et al. 19 The relatively higher levels of IL-1β in YORA signify that it may play a more prominent role in the inflammatory processes of younger-onset disease, while its significant correlation with disease activity in both groups underscores its general importance in RA pathogenesis irrespective of the age of onset.
Our study also showed a significant association between elevated levels of Interferon-gamma (IFN-γ) and increased bone erosion in the EORA group compared with the YORA group. IFN-γ is a key cytokine derived from T helper 1 (Th1) cells and plays a crucial role in the underlying mechanisms of RA. 30 The observed link between higher IFN-γ and greater bone erosion in EORA may be related to age-associated immune changes, as there is evidence of increased CD4 T cell and M1 macrophage activation with age, potentially exacerbating tissue damage and chronic inflammation. 31 Furthermore, activated T lymphocytes are one of the main sources of TNF-α, another cytokine which has been implicated in bone destruction in RA. 32
TNF-α was found to be lower in patients with EORA, which might indicate a potentially less aggressive disease trajectory in older individuals with RA, possibly conferring some protection against severe joint damage. This observation is particularly noteworthy considering the established clinical effectiveness of TNF-α antagonists in slowing the radiographic progression of bone erosions and joint deterioration in many RA patients. 33 However, despite these lower overall levels, we also observed a significant positive correlation between TNF-α levels and disease activity within our EORA cohort. This suggests that even at lower concentrations, TNF-α still plays a significant role in driving inflammation and disease activity in EORA. Consequently, the efficacy of TNF-α inhibitors in treating RA across different age groups, including those with EORA, is further supported, even though the baseline levels of TNF-α may be lower in EORA compared with YORA. Therefore, while lower TNF-α levels might contribute to some of the observed differences in disease severity and progression between EORA and YORA, TNF-α remains a relevant therapeutic target in both groups. Lower level of TNF-α in EORA was also found in a study by Deal et al. 17
Additionally, our study revealed significantly higher levels of Interleukin-6 (IL-6) and Interleukin-8 (IL-8) in patients with EORA. These elevated levels may lead to the distinct disease characteristics observed in this group. IL-6 is an important mediator of acute-phase responses and systemic inflammation, while IL-8 have a crucial role in promoting angiogenesis within the inflamed joint in RA.34,35 Notably, the levels of IL-6 are known to increase with age as cellular stress responses become more pronounced, potentially contributing to the chronic inflammatory state often associated with ageing. 36 This age-related increase in IL-6 provides a plausible explanation for the higher IL-6 levels we observed in the EORA group. A similar finding was found in a study done by Punzi et al., where elevated IL-6 concentrations were found in the synovial fluid of EORA patients. 37 Therefore, the increased IL-6 and IL-8 in EORA may contribute to a more pronounced systemic inflammatory burden and altered patterns of joint inflammation compared with YORA. But there was a lack of statistical significance, positive correlation of IL-6 and IL-8 with DAS28 ESR in our study, thus warranting further research with a larger sample size.
Furthermore, in our study, YORA patients demonstrated a strong positive correlation between disease activity and the levels of both IL-8, as found in the previous studies. 17 This observation implies that YORA patients exhibiting higher concentrations of these inflammatory markers are more likely to experience severe disease manifestations, including oedema and bone erosion.
Limitations
Limitation of the study includes the sample size, which limits the generalisability and statistical power of the findings. Further potential confounding factors such as medication use, disease duration, comorbidities and other lifestyle variables, all of which may influence both cytokine levels and disease activity scores. Furthermore, DAS28-ESR was employed in our study for disease activity, which could be influenced by age-dependent changes in ESR and other factors. Also, the study did not include longitudinal follow-up, which limits insights into disease progression and the temporal dynamics of inflammatory markers.
Conclusion
This study demonstrates distinct differences between EORA and YORA in clinical presentation, serological markers and cytokine profiles, emphasising the influence of immunosenescence. Elevated IL-6 and IL-8 levels in EORA suggest the presence of an age-related inflammatory pathway. These findings highlight the necessity for age-specific therapeutic management in RA, with cytokine-targeted therapies presenting promising opportunities. Further longitudinal studies are warranted to tailored treatment options based on age of onset and immunopathological mechanisms.
Footnotes
Author Contribution
Data collection, data analysis, literature review, discussion: Shivam Sharma, Sourabh Pathania, Diparani Takhelmayum. Concept, data collection, data analysis, literature review, discussion: Kailash Kumar and Anup Singh. All co-authors of the study take full responsibility for the integrity of the final version of the manuscript.
Data Availability
Data will be produced on request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee (Institute of Medical Sciences, Banaras Hindu University, Varanasi, Ethics Committee) and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Ethical approval for the study was obtained from the Institutional Ethics Committee, Institute of Medical Sciences, Banaras Hindu University, Varanasi, India (Ref No.: Dean/2019/EC/1705) on 18 November 2019.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: We thank the National Program for Healthcare of the Elderly, MoHFWs and the IOE Incentive grant BHU for support.
Patient Consent
Written informed consent was obtained from all participants before inclusion into the study, and their privacy was protected through anonymisation of data.
