Abstract
Rheumatoid arthritis (RA), axial spondylarthritis (axSpA) and psoriatic arthritis (PsA) significantly impact global health, causing chronic pain and inflammation. Despite effective treatments like adalimumab (ADA), high costs limit its use in India. Moreover, the absence of India-specific guidelines creates subjective biases for effective treatments. To address this lacuna, the RAPID-India steering committee, comprising experienced academic rheumatologists, used a three-phase Delphi process to create a nationwide consensus for ADA treatment for RA, axSpA and PsA. A 22-question survey was sent to 32 rheumatologists nationwide who rated statements on a 5-point Likert scale, with consensus being defined as ≥75% agreement. ADA was recommended for treatment of aggressive early RA and established RA, axSpA and PsA with axial, peripheral and extra-musculoskeletal manifestations. ADA was also recommended for co-morbid conditions like chronic kidney disease (CKD), cardiovascular diseases, fatty liver, osteoporosis and metabolic syndrome (MetS). For patients achieving six consecutive months of remission, a slow tapering of ADA was advised over instant discontinuation to avoid disease flares. ADA is also recommended during pregnancy, right up to the end of the second trimester and during lactation. Furthermore, ADA biosimilars are cheaper and affordable for a larger number of patients in India. This consensus provides evidence-based recommendations to optimise disease outcomes by guiding healthcare professionals with special emphasis and focus only on ADA use in inflammatory arthritis.
Keywords
Introduction
Rheumatic diseases like rheumatoid arthritis (RA), axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA) significantly impact global health, affecting millions with chronic pain and inflammation. 1 Optimising treatment for specific patient populations is complex despite established guidelines.2,3
India has a notable prevalence of rheumatic diseases, with 42.19 million RA cases in 2020. 4 axSpA, the second most common inflammatory rheumatic disease, is also on the rise globally, particularly in Europe and Asia. 5 The global PsA prevalence in psoriasis patients is estimated to be 17.58%. 6 In India, an 8.7% prevalence of PsA was reported among psoriasis patients in a one-year study in 2014. 3
Treatment options have improved, with conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), biologic DMARDs (bDMARDs) like tumour necrosis factor (TNF) inhibitors and targeted synthetic DMARDs (tsDMARDs) significantly enhancing patients’ quality of life.7,8 Current American College of Rheumatology (ACR) and European League Against Rheumatism (EULAR) guidelines recommend a treat-to-target approach. For RA, step-up therapy with bDMARDs like adalimumab (ADA) is recommended to achieve the goal of remission or low disease activity (LDA).9,10 In axSpA, non-steroidal anti-inflammatory drugs (NSAIDs) are first-line, with bDMARDs recommended for inadequate NSAID response. 11 For PsA, bDMARDs are suggested if csDMARDs are ineffective. 12 ADA safety and efficacy in RA, axSpA and PsA have made it a cornerstone treatment.13,14
In India, the lack of national guidelines tailored to local needs poses challenges in managing RA, axSpA and PsA. These diseases’ diverse manifestations require nuanced approaches. Despite effective treatments like ADA, high costs preclude its wider use. This consensus provides evidence-based recommendations for effective ADA use in India, akin to international guidelines.
Methodology
A steering committee comprised of six seasoned rheumatologists from the RA, axSpA and PsA Delphi in India (RAPID-India) group facilitated the creation of consensus statements to define the role of ADA in managing RA, axSpA and PsA in India (Figure 1). The committee conducted an extensive review of the literature to identify gaps in existing treatment approaches for these conditions. To maintain focus, each manuscript was restricted to a single therapeutic molecule without including comparisons with other treatments.
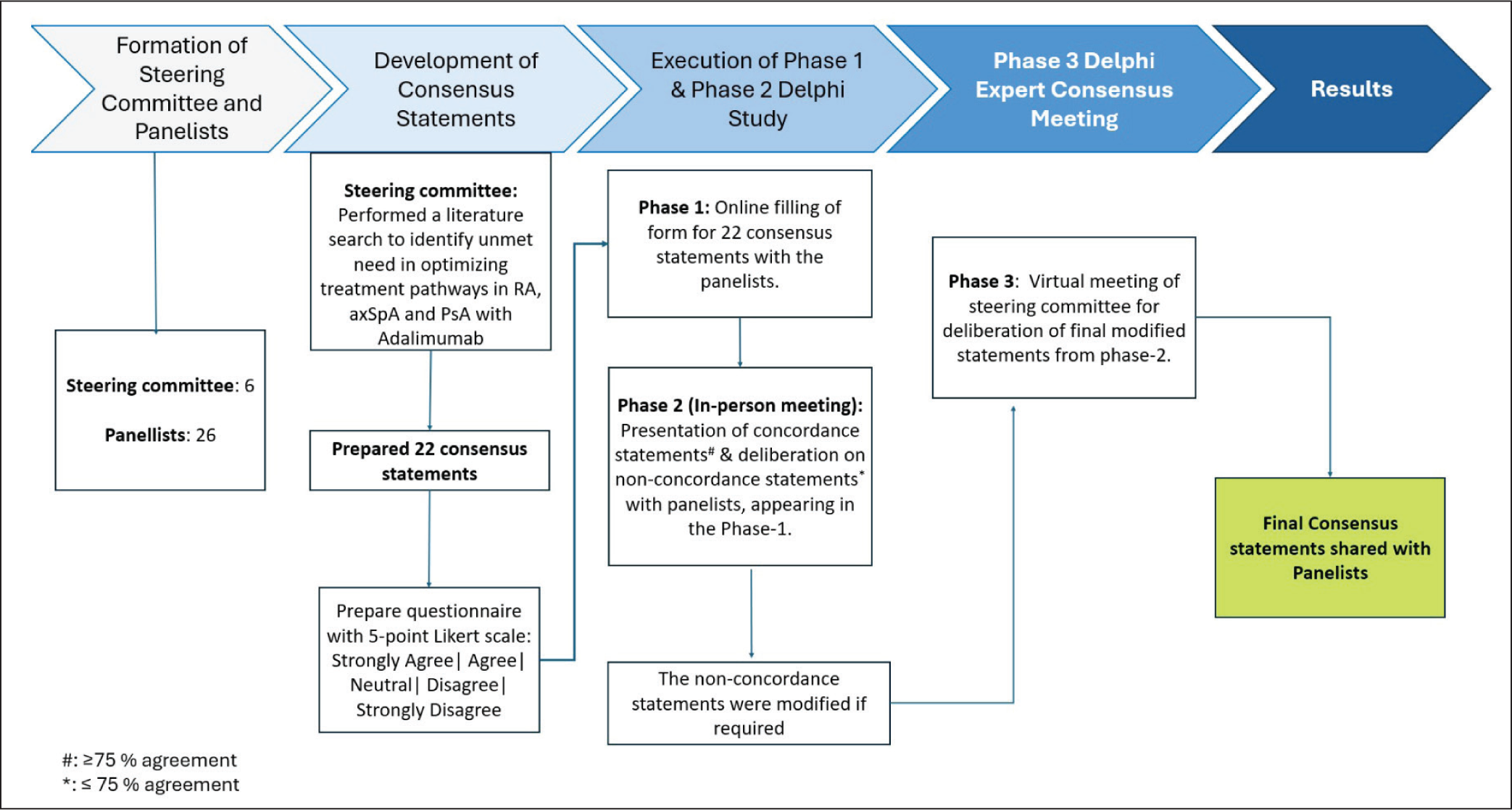
A selection of 26 senior rheumatologists was made based on factors such as geographic representation, expertise in the field, and prior publications. The panel reviewed the statements drafted by the steering committee through a structured three-phase Delphi process. During Phase 1, panelists completed an online survey (Questionnaire-Supplementary Material), rating their agreement with 22 statements on a 5-point Likert scale. Statements with more than 75% agreement were considered to have reached a consensus. Phase 2 involved an in-person meeting aimed at discussing and refining statements that did not achieve the 75% agreement in the previous phase. A descriptive analysis was conducted to finalise the consensus statements, and those that were not in agreement were revised based on feedback from the panel. Phase 3 included a virtual meeting where the steering committee reviewed and finalised the revisions from Phase 2. The completed consensus statements were then shared with the panel for confirmation. We had 22 statements in Phase 1 through an online survey, for which consensus was reached for 16. The remaining six were deliberated during Phase 2, and during Phase 3, all the statements were refined and finalised (Supplementary Table 1).
Overarching Principles.

This Delphi process facilitated a structured and evidence-driven method for formulating recommendations on the use of ADA in the context of RA, axSpA and PsA within Indian clinical practice.
Search Strategy
The literature search encompassed PubMed, Embase, Cochrane Library and Google Scholar databases, covering studies published in the previous 20 years (2003-2024). The search strategy included Medical Subject Headings (MeSH) terms and keywords such as RA, axSpA, PsA, ADA and bDMARDs, using Boolean operators for precise retrieval. Filters were applied to include human studies, English-language articles, clinical trials, systematic reviews, meta-analyses and case reports. However, studies reporting the pharmacokinetics and pharmacodynamics of ADA and those involving animal models were excluded. Following duplicate removal, 98 unique articles were retained from an initial pool of 125 articles. Titles and abstracts were screened by two independent reviewers based on predefined inclusion and exclusion criteria, and 90 full-text articles underwent detailed assessment. After rigorous evaluation, 74 studies were deemed eligible and included in the evidence synthesis to support the consensus recommendations.
Overarching Principles
The overarching principles of this study have been outlined in Table 1.
Results
Clinical Scenarios for ADA Therapy
Recommendation #1: For patients with axSpA with episodes of refractory uveitis, the panel recommends the use of anti-TNFα monoclonal antibodies like ADA over fusion proteins (etanercept) to reduce the recurrence of uveitis.
The panel recommends anti-TNFα monoclonal antibodies, like ADA, for refractory uveitis in axSpA patients, aligning with the Pan American League of Associations for Rheumatology (PANLAR) and Assessment of Spondyloarthritis International Society-European League Against Rheumatism (ASAS-EULAR) guidelines, which prefer ADA over etanercept due to its efficacy in recurrent uveitis.11,15 This was also indicated in a prospective open-label study where ADA significantly prevented anterior uveitis flares in patients with active axSpA (Supplementary Table 2). 16
Recommendation #2: For patients with axSpA with an active/history of inflammatory bowel disease (IBD), the panel recommends treatment with anti-TNF monoclonal antibodies like ADA over etanercept.
The panel agreed on ADA’s superior efficacy for treating IBD associated with managing axSpA. The ASAS-EULAR guidelines recommend TNF monoclonal antibodies like infliximab and ADA for active IBD since other anti-TNFs like etanercept and secukinumab lack efficacy for axSpA with IBD. 11 The Spanish Society of Rheumatology also advises using bDMARDs like ADA for axSpA and IBD, excluding etanercept due to its lack of efficacy (Grade of Agreement: D; Level of Evidence: 5; Grade of Recommendation: 100%). 17
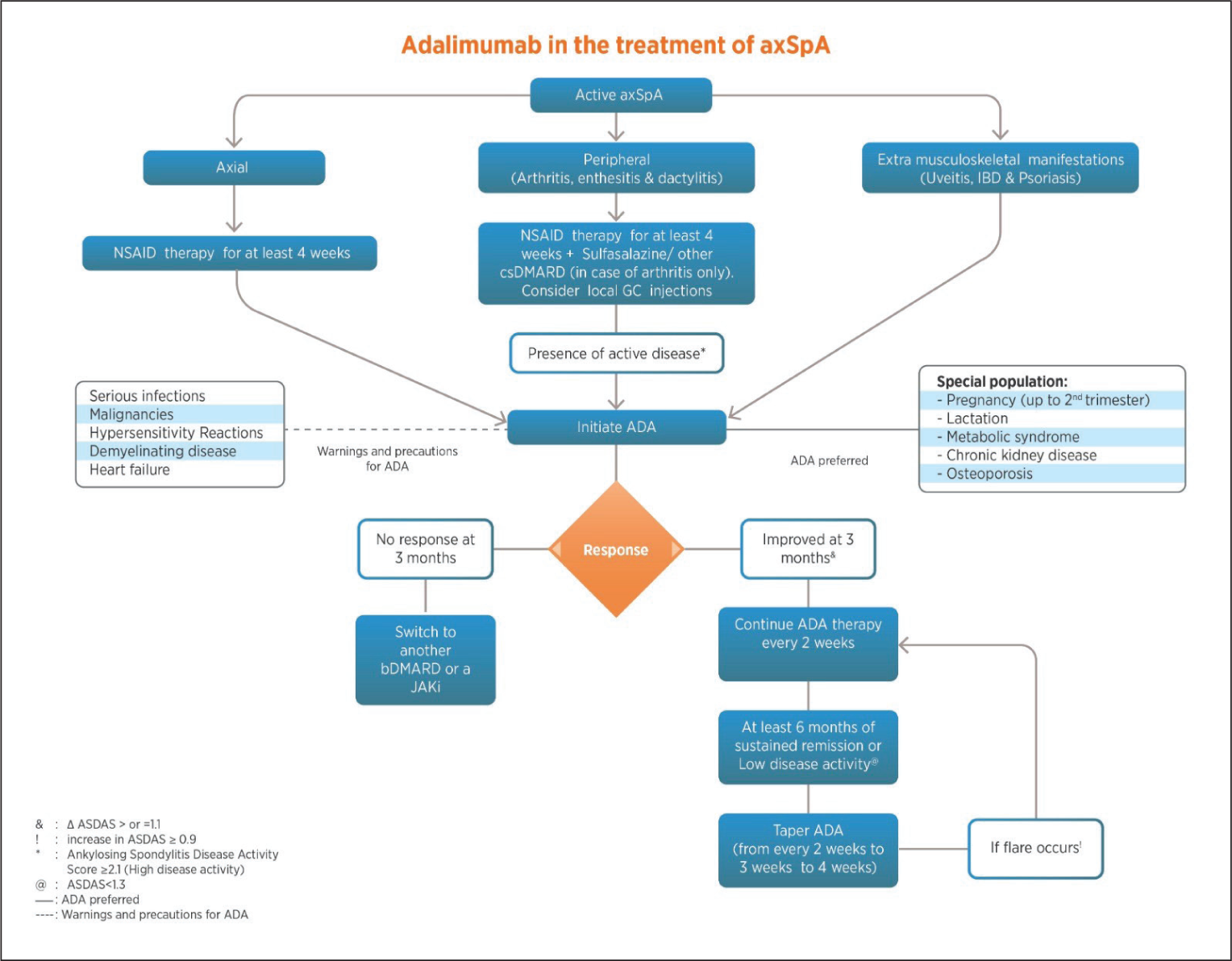
Recommendation #3: For patients with severe active non-radiographic axial spondylarthritis (axSpA), the panel recommends the use of an anti-TNFα monoclonal antibody (mAb) like ADA (monotherapy) in adults who either respond inadequately to or do not tolerate NSAIDs.
The panel members recommend ADA monotherapy for individuals with active axSpA who cannot tolerate NSAIDs. ADA also alleviates non-radiographic(nr) axSpA symptoms, even when used alone. This statement is based on data published in the Spondylitis Association of America/Spondyloarthritis Research and Treatment Network (SAA/SPARTAN). 18 In the ABILITY-1 trial, 36% of ADA-treated nr-axSpA patients achieved Assessment of SpondyloArthritis International Society (ASAS 40) response at week 12, compared to 15% on placebo (P < .001). Patients with positive MRI or elevated C-reactive protein (CRP) at baseline had a higher response to ADA (41%) versus placebo (14%). 19 Expert opinion suggests that ADA is effective and well-tolerated for treating nr-axSpA with active inflammation in patients unresponsive to standard therapy. 20
Recommendation #4: For patients with axSpA and peripheral manifestations including either enthesitis or dactylitis, or both, the panel recommends the use of an anti-TNFα mAb like ADA in adults, who either responded inadequately to, or do not tolerate NSAIDs.
In clinical practice, axSpA may present with peripheral manifestations, including dactylitis and enthesitis. When NSAIDs are ineffective or not tolerated, the panel strongly recommends anti-TNFα drugs like ADA. Another expert consensus suggests considering TNF-α, IL-17, or Janus kinase inhibitors (JAKi) for patients with refractory enthesitis. 21 A study on ADA in non-psoriatic peripheral arthritis patients demonstrated significantly higher Peripheral SpA Response Criteria (PSpARC40) response rates at week 12 compared to placebo, with improvements observed as early as week 2 (39% vs. 20%; P = .006). ADA also improved global assessments of disease activity, pain, tender joint count, swollen joint count and total enthesitis count compared to placebo. 22
Recommendation #5: For patients with moderate to severe RA and showing inadequate response to methotrexate (MTX)* with poor prognostic factors (high disease activity, failure of >2 cs DMARDs), the panel recommends the addition of a TNF inhibitor like ADA to MTX.
*A trial of 25 mg/week of MTX for three months OR the maximum tolerated dose of MTX for three months.
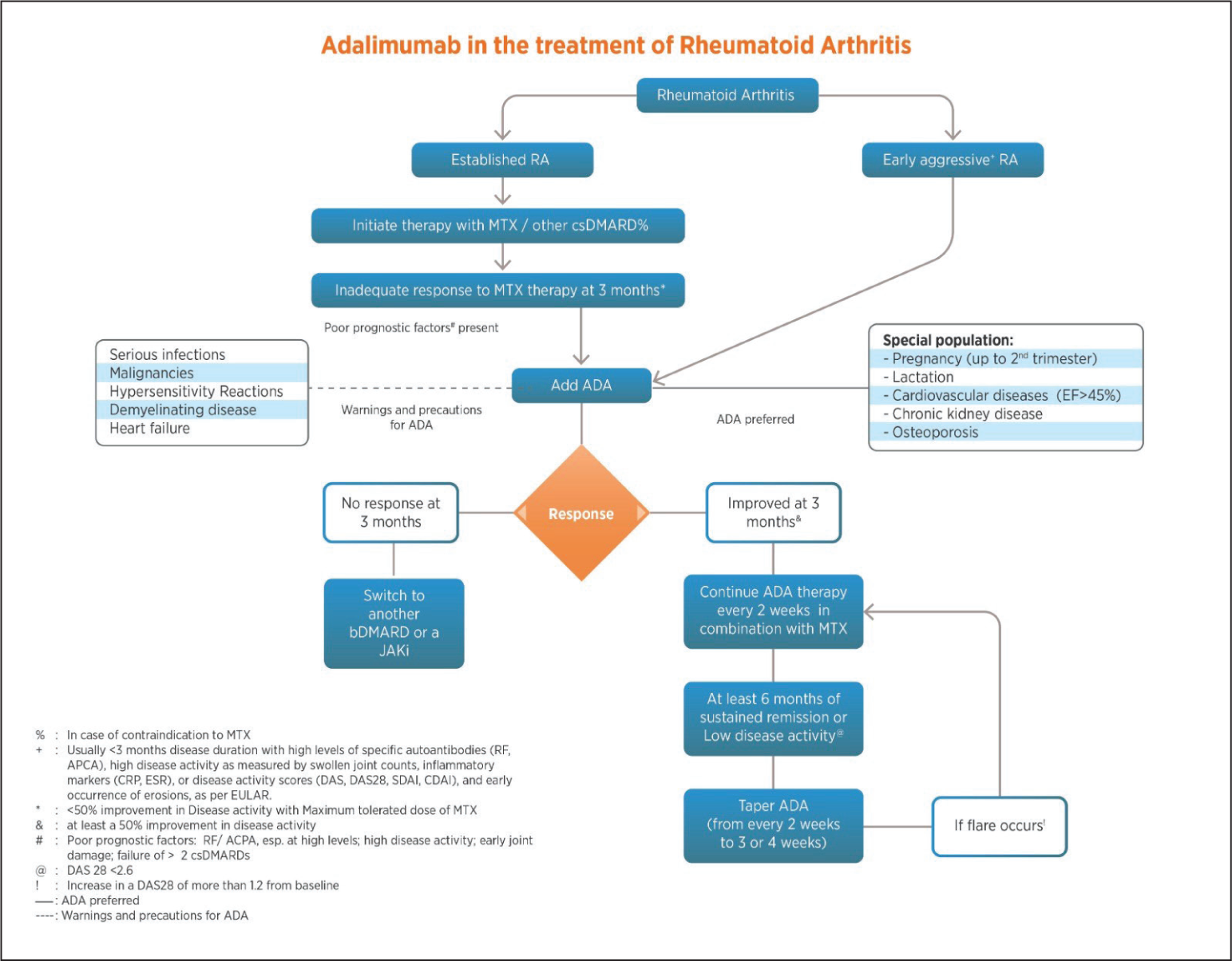
MTX monotherapy often fails due to limited bioavailability and poor gut absorption. 23 Experts recommend combining ADA with MTX for RA patients who do not reach treatment targets on MTX and have poor prognostic factors. The panel advises against concomitant corticosteroid use with MTX, instead suggesting initial treatment with bDMARDs for RA patients with high inflammatory load, poor prognostic factors or both. A more aggressive approach to managing RA is advocated in the presence of poor prognostic factors suggested by the ACR and the EULAR guidelines.9,10 In the DE020 study, after ten years of ADA treatment, 71.2% of DMARD-refractory RA patients achieved Disease Activity Score 28 with C-reactive Protein (DAS28-CRP) ≤3.2, 42.4% achieved health assessment questionnaire - disability index (HAQ-DI) <0.5, and 37.3% achieved both. DAS28-CRP or SDAI-based remission was observed in 57.2% and 29.7% of patients, respectively. 23
Recommendation #6: For patients with aggressive early* RA, the panel recommends the upfront initiation of a TNF inhibitor like ADA, in combination with methotrexate (MTX), for better outcomes, particularly in the prevention of radiographic progression.
*Criteria for aggressive early: Early—usually less than three months disease duration. 24 Aggressive—EULAR classifies aggressive disease based on three key factors: (a) high levels of specific autoantibodies (rheumatoid factor and anticitrullinated peptide antibodies), (b) high disease activity as measured by swollen joint counts, inflammatory markers (CRP, ESR), or disease activity scores (DAS, DAS28, SDAI, CDAI) and (c) early occurrence of erosions. 25
The consensus among the panel was to initiate combination therapy with ADA and MTX in early and aggressive RA for better outcomes, over and above the use of combination csDMARDs for this indication. ADA treatment for ten years is associated with fewer erosions, indicating reduced long-term deformity risk, 26 but its use is limited due to higher costs and infection (especially tuberculosis) risk. This approach is particularly beneficial in preventing radiographic progression in early RA, 27 challenging the traditional paradigm of combination-DMARD (e.g., the triple csDMARD—methotrexate, leflunomide, hydroxychloroquine) use before biologics. Numerous other clinical studies have also demonstrated the benefits of this combination (Supplementary Table 3).
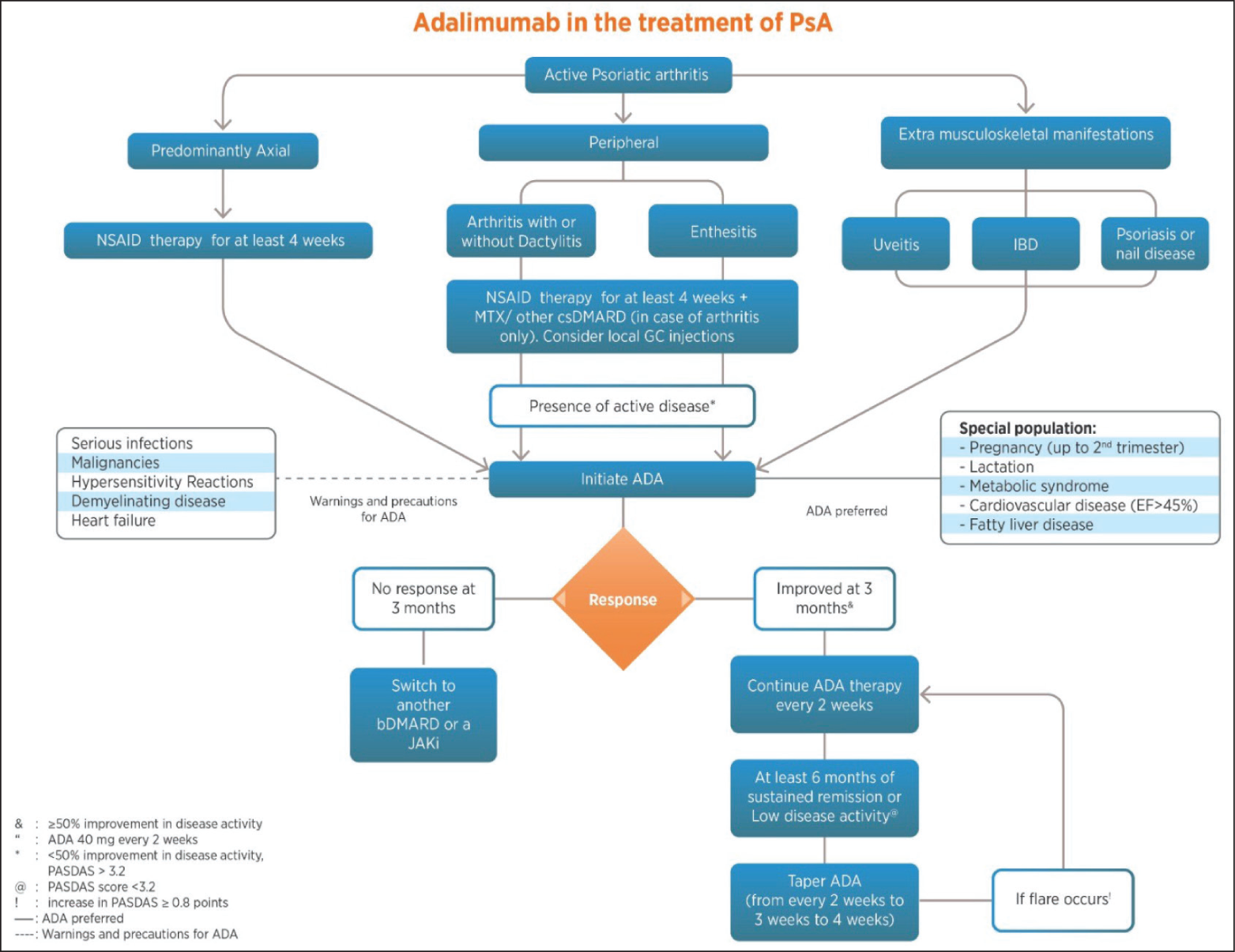
Recommendation #7: For patients with active PsA with predominantly axial disease and insufficient response to NSAIDs, the panel recommends the use of a TNF inhibitor like ADA.
For PsA patients with predominant axial involvement, NSAID treatment may be insufficient for achieving LDA. Thus, the panel recommends ADA for its sustained benefits in inhibiting radiographic progression, inducing and maintaining remission, and long-term safety.12,28 In a study by Makino et al., ADA treatment for PsA with axial involvement who had an inadequate response to NSAIDs showed improvements in spondylitis and sacroiliitis on MRI, regardless of clinical symptoms. The mean total grade of sacroiliac joint (SIJ) involvement improved significantly from 6.76 to 5.27 after 24 weeks of treatment. 29
Recommendation #8: For patients with active PsA and predominant enthesitis, dactylitis, skin and/or nail psoriasis, the panel recommends a TNF inhibitor like ADA as first-line biological therapy.
In PsA, ADA is the recommended first-line biological treatment for patients with active disease, especially associated with dactylitis and enthesitis and is also resistant to conventional treatments. The EULAR guidelines suggest NSAIDs, local glucocorticoid injections and rapid csDMARD initiation. TNF inhibitors are the recommended first-line bDMARDs for PsA with peripheral features. 30 The British Society for Rheumatology (BSR) guidelines also recommend TNFi in active psoriatic enthesitis (multi-site), dactylitis, or axial involvement with either inadequate csDMARD response or intolerance. In the presence of psoriasis with or without nail disease, all currently licensed b/tsDMARDs can be offered. However, it is recommended to prioritise therapies specifically indicated for psoriasis, such as TNFi, IL-17i, IL-12/23i, or IL-23i (GRADE 1A, SoA 93%). 31 In a real-life cohort study, D’Angelo et al. found ADA highly effective in peripheral PsA, significantly reducing tender joint count (TJC) and swollen joint count (SJC) from baseline at 24 months (TJC: 7.0 ± 5.7 vs. 2.3 ± 3.5, P < .001; SJC: 2.7 ± 2.8 vs. 0.4 ± 0.9, P < .001). The enthesitis prevalence decreased from 47.3% to 15.1%, and dactylitis decreased from 27.8% to 3.2% over the follow-up period. 32
Efficacy Data
Recommendation #9: Significant clinical response to ADA occurs as early as two weeks in adults with active ankylosing spondylitis/axSpA, with moderate to severe active RA and with active PsA. Therefore, the panel recommends ADA as an add-on therapy in emergent situations.#
#High disease activity affecting activities of daily living.
The panel agrees that ADA shows significant clinical response as early as two weeks in adults with active ankylosing spondylitis (axSpA), moderate to severe RA, and active PsA. Pain, stiffness and inflammation improve quickly, particularly in axSpA patients, who may experience relief after the first dose. RA or PsA patients typically see improvement after two to three doses, though results may vary.
Trials comparing tofacitinib and ADA in PsA showed similar times to initial HAQ-DI score response, averaging 15-16 days. 33 In the ATLAS study, significant ASAS20 response rates by week 12 were observed in axSpA patients, with responses as early as two weeks. 34 Additionally, in the ARMADA trial, the ACR20 responses at the first scheduled visit (week 1) were notable across different ADA doses compared to placebo. 13
Dosing Optimisation
Recommendation #10: For patients with at least six consecutive months of remission in axSpA, RA & PsA, tapering of ADA could be considered & the panel does not recommend an instant discontinuation because this can lead to disease flares.
The expert panel recommends gradual tapering of ADA doses for patients in prolonged remission, lasting for at least six months in axSpA, RA, or PsA. This approach maintains LDA and is economically logical in resource-limited settings. The panel advises against abrupt discontinuation to avoid disease flares. Even in the PREDICTRA phase IV study, which investigated the impact of ADA dose tapering versus withdrawal in RA patients in sustained remission. Results showed that tapering led to fewer flares than withdrawal, with a longer time to flare. 35 Similarly, evidence supports the tapering of ADA in axSpA and PsA.36-38
The PANLAR guidelines also advise against sudden bDMARD discontinuation. For axSpA patients in sustained remission on bDMARDs for 6-12 months, tapering is conditionally recommended. 29 Published data indicates that tapering anti-TNF therapy maintains remission or LDA in most axSpA patients, while instant discontinuation often leads to disease flares. 39 In a study by Tanaka Y. et al. involving 75 RA patients, 48% and 62% sustained DAS28-ESR <2.6 and <3.2, respectively, for one year in the ADA continuation group. Among patients in sustained remission (DAS28-ESR ≤1.98), these rates increased to 68% and 79%, with no significant difference between ADA discontinuation and continuation groups. Additionally, 100% of patients who sustained DAS28-ESR <3.2 during ADA discontinuation remained in structural remission, and 94% remained in functional remission. Notably, 79% of patients in deep remission stayed ADA-free for one year without flaring, showing no functional or structural damage. 40
Recommendation #11: The panel recommends continuous ADA therapy, which is associated with a higher likelihood of long-term clinical response or remission/ radiographic progression in axSpA, RA and PsA, compared to discontinuation.
According to the panel, continuous ADA therapy demonstrates superior outcomes in axSpA, RA and PsA, including higher remission rates, reduced joint damage and improved physical function compared to intermittent treatment.
In the ARMADA trial extension, RA patients on continuous ADA therapy showed improved ACR20/50/70 response rates over time, with stable remission rates increasing from 34% at year 1 to 43% at year 4. 13 Similar results were observed in the ABILITY-3 trial, where continued therapy with ADA in axSpA was associated with lesser flare rates compared to discontinuation (70% vs. 47%; P < .0001) through week 68. 41 Additionally, in the ADEPT trial extension, ACR20 response rate remained stable at about 57% at 104 weeks, along with sustained improvements in other disease parameters. 14
Recommendation #12: The panel recommends assessing the response to ADA treatment at 12 weeks after the start of treatment. If there is an inadequate response (primary treatment failure), consider switching to a different class of biologic or a JAK inhibitor.
Unanimous agreement exists on evaluating response to TNF inhibitors like ADA in axSpA after 12 weeks; if treatment is ineffective, switching to an IL-17 inhibitor or a JAK inhibitor is recommended. 42 The NICE guidelines for axSpA reinforce this, suggesting a continuation of ADA treatment when there is evidence of response at the 12-week mark. 43 The ASAS-EULAR 2022 guidelines recommend that an absence of response to treatment prompts re-evaluation of the diagnosis and consideration of differentials. 11
The PANLAR recommends switching to a bDMARD with a different mechanism or a JAK inhibitor for primary TNFi failure. For secondary failure, options include cycling to a bDMARD with the same mechanism, switching to one with a different mechanism of action, or trying a JAK inhibitor. 15 A prospective cohort study indicates that choosing a bDMARD with a different mechanism of action may be more beneficial for primary TNFi failure, while patients discontinuing the initial TNFi due to secondary failure showed better response. 44 Consequently, in a 15-year monocentric real-life study, nearly half of PsA patients switched biologics after initial bDMARD failure, with persistent effectiveness observed with second-line bDMARDs, highlighting the benefit of switching biologics. 45
Recommendation #13: In patients with axSpA who achieve a stable or inactive disease activity after treatment with ADA and NSAIDs and/or csDMARDs, the panel recommends the discontinuation of NSAIDs, tapering ADA doses, while continuing csDMARDs.
In axSpA, where remission is achieved, experts recommend considering the withdrawal of NSAIDs while maintaining the patient on a csDMARD and reducing the dosing or frequency of ADA. In India, cost considerations may influence drug sequencing, but the consensus is to stop NSAIDs; persistent remission may allow for biologic tapering. Shefali et al. found sulfasalazine effective in axial disease, with a significant Ankylosing Spondylitis Disease Activity Score (ASDAS) improvement in the treatment group (67.7%) compared to placebo (15.1%, P = .001). The difference in the Bath Ankylosing Spondylitis Disease Activity Index (ΔBASDAI) was higher in the treatment group (3.29 ± 0.97) as compared to placebo (1.47 ± 0.99, P = .00). Clinical improvement was greater in those with disease duration ≤ 4 years in the treatment group (P = .04). 46 In a couple of systematic reviews, it was shown that the tapering of TNF inhibitors was successful in maintaining LDA or remission in patients with axSpA.36,37 The panel suggests spacing ADA between one and four months instead of continuing at fortnightly intervals. Sulfasalazine should be continued for maintenance therapy in axSpA with peripheral manifestations. After remission, csDMARD dosages can be reduced and ADA tapered. The most effective drug, such as ADA, is retained in developed countries while others are discontinued.
Co-morbidities
Recommendation #14: For patients with active RA and PsA, along with cardiovascular diseases, the panel recommends TNFs like ADA, which reduces the risk of cardiovascular events (CVE), reduce corticosteroid and NSAIDs use.
The panel members opine that bDMARDs, such as TNF inhibitors (e.g., ADA) and csDMARDs like methotrexate, particularly when used in combination, reduce the risk of CVE in patients by lowering inflammation. Elevated RA disease activity and persistent inflammation increase the risk of CVE. Combining MTX with TNFi, like ADA, can reduce CVE risk and decrease reliance on corticosteroids and NSAIDs, which elevate cardiovascular risk. 47 Belgian Evidence-based Treatment Advice in Psoriasis (BETA-PSO) supports ADA for psoriasis patients with cardiovascular risk, except those with heart failure. 48 The Canadian Dermatology-Rheumatology Comorbidity Initiative advises that MTX and/or TNF inhibitors may decrease CVE risk in RA and PsA. Many other clinical reports suggest that TNFi, like ADA, are associated with a reduced risk of CVE, including MI, stroke and cardiovascular accident (CVA) 49 (See Supplementary Table 4).
Recommendation #15: For patients with active RA and chronic kidney disease (CKD), the panel recommends the use of bDMARDs like ADA over tsDMARDs. ADA does not interfere with renal function and has even been used in patients undergoing haemodialysis.
Renal impairment restricts the use of certain targeted DMARDs, such as JAK inhibitors, in patients with RA, primarily due to the risk of adverse events (AEs). In patients with compromised kidney function, JAK inhibitors can accumulate, further exacerbating renal dysfunction because of their partial or predominant renal excretion. Consequently, these targeted therapies may worsen renal function, requiring careful monitoring and dose adjustments. 50 However, anti-TNF agents are safe in patients with CKD as they are hydrolysed at lysosomes and are safe even in patients with end-stage renal disease (ESRD). Hence, they can be preferred in all stages of CKD and do not need any dose adjustment depending on the CKD stage.51-53 Also, anti-TNF agents like ADA are valid for CKD patients since their excretion is not dependent on renal functions. 54
In a retrospective study by Sumida et al. (2013), the safety of ADA in RA patients with renal insufficiency, including those on haemodialysis, was assessed over three years. Among 65 patients, including two on haemodialysis, ADA treatment did not significantly worsen renal function. 55 The majority of the panel members recommend the use of ADA in this subset of patients.
Recommendation #16: For adults with active PsA and the metabolic syndrome (MetS), the panel recommends a TNFi like ADA, which does not worsen the components of the MetS.
Experts recommend ADA for PsA due to its effectiveness in improving MetS components, mainly by reducing inflammation and enhancing lipid profiles, such as increasing HDL, compared to methotrexate. 56 Biologic therapies, supported by research, are safe for patients with psoriasis and MetS, usually preferable to conventional systemic therapies like methotrexate. 57 In a retrospective study, ADA demonstrated efficacy superior to methotrexate in improving the MetS among PsA patients. 58 BETA-PSO also recommends TNF inhibitors like ADA use as a systemic treatment for adult psoriasis patients with the MetS. 45 In PsA patients, JAK inhibitors are known to increase lipid levels similar to RA and psoriasis treated patients, and are usually avoided in the MetS patients due to dyslipidaemia and weight gain risks. 59
Special Population
Recommendation #17: In patients with active rheumatic disease, including RA, axSpA and PsA during pregnancy, the panel recommends that ADA may be continued until the end of the second trimester.
Based on several case reports and per the BSR guidelines, experts advise pregnant women with RA to continue ADA until the end of the second trimester, with caution in the third trimester. However, ADA can be used to manage rheumatic diseases throughout pregnancy; if administered after 28 weeks, live vaccinations for the newborn must be postponed until it is six months old. The Bacillus Calmette-Guérin (BCG) vaccine is often postponed, while the rotavirus vaccine can be given at three months. Overall, TNF inhibitors can be used up to 28 weeks of pregnancy if clinically indicated. Note, however, that the decision to continue beyond this time point should factor in neonatal vaccination and infection risk status. 28
Recommendation #18: In geriatric patients with active RA, the panel recommends exercising caution with ADA because of the higher incidence of infections in the elderly.
Older patients are susceptible to an increase in infectious complications, with most studies reporting a two- to three-fold higher risk of serious infections for those aged >65 years. 60 This panel agrees unanimously in exercising caution in this population, especially those with comorbidities or taking additional immunosuppressants, ADA can increase the risk of severe infections. This has also been demonstrated in several clinical studies, providing strong evidence for these recommendations (See Supplementary Table 5).
Recommendation #19: The panel recommends vaccinating all patients prior to starting ADA (preferably at least four weeks before) with inactivated vaccines (pneumococcal and influenza) and other vaccines recommended by the National Guidelines for Adult Vaccinations unless contraindicated. Live vaccines should not be administered during ADA therapy.
Long-term immunosuppressive therapies reduce T cell-dependent antibody production and increase infection risk. 61 The panel recommends vaccinating patients at least four weeks before starting ADA, which does not affect antibody production and is compatible with attenuated vaccines and should be administered when the disease is in remission or at low activity. Due to logistical challenges in India, such as inadequate cold chain facilities, transportation problems in remote or rural areas, limited healthcare facilities in the peripheries, etc., vaccines and ADA are often administered simultaneously. The panel also recommends the Herpes zoster vaccine for individuals over 50 and the Human Papillomavirus (HPV) vaccine for women aged 9-26. Various guidelines offer specific vaccination recommendations for patients on biologics. The recommendations by various guidelines have been summarised in Supplementary Table 6.
Safety Data
Recommendation #20: The panel recommends the use of citrate-free formulation, low volume & 29-gauge needle in patients who experience injection site reactions with conventional ADA* leading to drug discontinuation.
*Original ADA formulation including a citrate and phosphate containing buffering system, ADA 40 mg (within an 0.8 ml injection volume) and pre-filled syringes with 27-gauge (27G) needles.
The original ADA formulation caused injection site reactions (e.g., erythema, pruritus and pain or swelling). 62 A new formulation with improvements like citrate-free excipients and thinner needles was introduced in 2018 to reduce these reactions. 63 The panel suggests switching to enhance adherence.
According to a comprehensive review done by Pilunni D. et al., the change in ADA formulation led to fewer injection site reactions. 64 For patients with a lower pain tolerance/threshold, any factors such as reduced injection volume/frequency and the inclusion of fewer excipients, which may reduce the overall sensation of injection-site pain, may be helpful.
Recommendation #21: In the absence of innovator ADA (Humira), the panel recommends the use of biosimilars of ADA, which are functionally equivalent to the originator molecule.
According to Central Drugs Standard Control Organisation (CDSCO) and Department of Biotechnology (DBT) guidelines, a biosimilar matches an approved reference product in quality, safety and efficacy. Biosimilars meet the same standards as all biological medicines and undergo Phase 3 studies to confirm similar efficacy. Experts and panel members agree that ADA biosimilars are as effective as the original, enabling safe switching and significant cost reductions. 65 A study comparing biosimilar ADA in Indian patients with active RA on MTX found it as effective and well-tolerated as reference ADA, with both achieving ACR20 responses (96.43%) in intention-to-treat analysis at week 12. 66
Recommendation #22: The panel recommends dose interruption of ADA before surgery or if a serious infection occurs.
Immunosuppressive therapy, including ADA, can hinder wound healing, elevate infection risk and delay recovery, raising concerns for surgeons about perioperative cardiovascular risks and complications. 67 To mitigate these issues, the panel recommends pausing ADA before surgery or during severe infections to balance the risk of disease flare-ups with perioperative outcomes.
The ACR/American Association of Hip and Knee Surgeons (AAHKS) guidelines recommend stopping the ADA dosing interval before surgery and restarting it 14 days postoperatively to reduce infection and wound healing risks while maintaining disease control. Similarly, the updated guidelines from the BSR also recommend interrupting most bDMARDs for one dosing interval (or five half-lives for higher-risk surgeries). 68 Another consensus recommends withholding ADA for an entire dosing cycle preoperatively and scheduling elective surgery at least three weeks after discontinuation to enhance patient safety and reduce the risk of postoperative complications. 69
Discussion
This RAPID-India Group consensus is the first to systematically address the management of RA, axSpA and PsA with ADA in India in the absence of formal Indian guidelines. While the ACR/EULAR guidelines provide a universal template, they may not fully address the unique clinical needs and practice conditions for Indian patients. To ensure clarity, the committee decided to focus on one molecule per paper, with this consensus spotlighting ADA for the treatment of RA, axSpA and PsA in India. The committee also developed a treatment algorithm based on the consensus statements (Figures 2, 3 and 4) to provide clinicians with an evidence-based guide for the use of ADA in these conditions. This approach is intended to provide a clear understanding of its role in therapy without suggesting any bias against or preference for other effective treatments that are not covered here. ADA has established itself as a vital therapeutic in most inflammatory musculoskeletal disorders. Not only is it effective for the synovitis of RA, but it also works very effectively for axial manifestations of SpA, enthesitis and dactylitis of PsA, and IBD and uveitis of musculoskeletal disorders. Additionally, in axSpA, patients’ responses start as early as two weeks into ADA therapy. This consensus also emphasises early and aggressive ADA use to prevent irreversible joint damage and disability, as well as advocating ADA as a possible first-line biologic after csDMARD failure.
ADA is a fully human mAb, unlike infliximab, which is a chimeric mAb. The risk of anti-drug antibodies (ADAs) still exists for ADA. Several risk factors influence the formation of ADAs, which can be drug-related, patient-related, or disease-related. There are two types of ADAs: (a) neutralising and (b) non-neutralising. The first type reduces the efficacy of the molecule, while the second increases the risk of AEs such as allergic reactions. In the Indian context, the use of many biosimilars and the spacing of injections can create unique challenges regarding the assays needed to measure ADAs and the increased risk of ADA formation. Indeed, measuring ADAs and therapeutic drug monitoring is essential for managing Autoimmune rheumatic diseases.70–72
However, the panel does not recommend it routinely due to feasibility, cost issues and the complexity of standardising assays. The panel also recommends the concomitant use of anti-proliferative agents like methotrexate as they reduce the formation of ADAs.
While most guidelines do not address the long-term effects of ADA, this consensus highlights its superior long-term outcomes. The group highlights the importance of vaccination prior to initiating ADA therapy and outlines monitoring parameters to minimise adverse outcomes. Additionally, it is recommended to screen for active and latent tuberculosis through a detailed patient history, risk assessment and tests such as the tuberculin skin test (TST) or Interferon-Gamma Release Assay (IGRA) before initiating treatment, as highlighted by ACR and EULAR.73,74 The expert panel believes that in a country like India, where TB is highly endemic, it is better to conduct both TST and IGRA to enhance sensitivity. Patients with latent TB should complete at least one month of treatment, while those with active TB must undergo the full course of treatment before initiating biologic therapy. 75 According to the World Health Organisation (WHO) recommendation, for LTBI (latent tuberculous infection) in countries with a high incidence of TB, such as India, treatment with rifampicin plus isoniazid daily for three months or rifapentine and isoniazid weekly for three months is preferred over isoniazid monotherapy for six months.75–77 ADA is also the preferred choice for special populations, including pregnant women (up to the 2nd trimester), lactating mothers and individuals with cardiovascular diseases (EF > 45%), CKD and osteoporosis, underscoring ADA’s safety in this population. However, the panel recommends interrupting biologic therapies for a single dosing interval before surgery or in the presence of a serious infection. Like the ACR/EULAR guidelines, tapering the dose of ADA is recommended for patients in sustained remission, but abrupt discontinuation is discouraged. Given resource constraints in India, complete discontinuation of csDMARDs is not preferred; instead, patients are maintained on csDMARDs with extended intervals between ADA doses. In the context of healthcare resource allocation in India, biosimilar ADA offers a cost-effective alternative to the originator ADA.
The strength of this consensus statement is the diversity of the panel members in terms of their expertise, academic background and clinical experience. The main objective was to guide the clinicians in the best possible way to use ADA in their daily clinical practice. The statements chosen are based on practical relevance, emphasising the effectiveness and safety of ADA when used to manage inflammatory arthritis. A few statements (safety in CKD, use of low-volume citrate formulation, preference in MetS) are new, and some (tapering of ADA, caution in elderly) have been modified or adjusted to suit the Indian context. Our manuscript has a few limitations. We did not use any guidelines to grade the quality of the evidence synthesised. No specialists from other medical specialities or patient representatives were in the expert panel. We did not cover the individual aspects of each inflammatory arthritis and could not touch upon the comorbidities in detail.
Conclusion
In our pursuit of effective and personalised management for RA, axSpA and PsA in India, this consensus, meticulously crafted by the RAPID-India Group, provides evidence-based recommendations. It emphasises the critical role of shared decision-making between patients and healthcare professionals. ADA is an upfront choice for aggressive early RA and in established RA with poor prognostic factors after failure of conventional treatment. ADA is an upfront choice in active axSpA with extra-musculoskeletal manifestations like uveitis, IBD & psoriasis and in axSpA with purely axial or peripheral manifestations after failing NSAID treatment. TNF inhibitor like ADA is recommended as first-line biological therapy in active PsA with predominant axial disease and in active PsA with enthesitis, dactylitis, or nail psoriasis. ADA’s treatment response is typically observed within two weeks. While tapering is an option for patients in sustained remission, abrupt discontinuation is not advised. Continuous ADA therapy yields better long-term outcomes compared to discontinuation. As the intricate landscape of healthcare resource allocation is navigated, ADA biosimilars should be championed as allies in the mission to enhance patient well-being and transform lives.
Supplemental Material
Supplemental materials for this article are available online.
Footnotes
Acknowledgements
The authors express their gratitude to Dr Ajaz Kariem Khan, Dr Ajit Nalawade, Dr Alok Kalyani, Dr Ashit Syngle, Dr Atul Kakkar, Dr Bharat K. Singh, Dr Bimlesh Dhar Pandey, Dr B. Rajasekar, Dr Dhaiwat Shukla, Dr Durgesh Srivastava, Dr G. Narsimhulu, Dr Keerthi T., Dr K. Shanmuganandan, Dr N. Raja, Dr Piyush Joshi, Dr Ramesh Jois, Dr Reena Sharma, Dr Renu Saigal, Dr Samir Rajadhyaksha, Dr Sanjiv Kapoor, Dr Shailaja Sabnish, Dr U.R.K. Rao, Dr Ved Chaturvedi, Dr Vineeta Shoba, Dr Vikram Raj Jain and Dr V. Krishnamurthy for their invaluable contributions to Phase 1 and Phase 2 of the Delphi process. Dr Yogesh Sharma and the Abbott medical team were instrumental in facilitating the proceedings and helping with the writing and data collection for these articles. The authors thank IntelliMed Healthcare Solutions, Mumbai, for medical writing support.
Author Contributions
The conception and design of the study, acquisition of data, or analysis and interpretation of data: Dr Sundeep Kumar Upadhyaya, Dr Shashank M. Akerkar & Dr Sham Santhanam.
Drafting the article or revising it critically for important intellectual content: All Authors.
Final approval of the version to be submitted: All authors.
Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved: All authors.
Data Availability
Not applicable.
Declaration of Conflicting Interests
All the authors have received speaker fees and have been on the advisory board of various companies that manufacture/market adalimumab.
Ethical Approval
Ethical permission was not applicable for this article, as this is a review article drafted from various research articles and not from patients directly.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Abbott Healthcare Pvt Ltd.
Informed Consent
Consent was not applicable, as this is a review article compiled from various research articles and guidelines and not from patients directly.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
