Abstract
Rheumatoid arthritis (RA), axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA) pose major global health challenges. These conditions are characterised by chronic widespread inflammation, leading to considerable morbidity and excess cardiovascular mortality. Despite advancements in therapy, very little in the form of consensus exists in India among musculoskeletal physicians and rheumatologists for the management of these conditions. The recommendations presented in this paper were developed by a steering committee, the RAPID-India, comprising academic rheumatologists who are key opinion leaders with diverse interests in rheumatology. With the help of the Delphi technique, they convened 3 advisory board meetings to discuss optimising to facitinib therapy for RA, axSpA and PsA. The paper particularly highlights aspects of pre-treatment screening, toxicity monitoring and specifics of use in the elderly. Additionally, long-term therapy with tofacitinib is advocated to prevent radiographic progression and maintain disease control; tapering strategies are also discussed. This consensus provides evidence-based recommendations to optimise disease outcomes by guiding healthcare professionals with special emphasis and focus only on tofacitinib use in inflammatory arthritis.
Keywords
Introduction
Inflammatory joint diseases (IJDs), including rheumatoid arthritis (RA), axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA), pose global health and socio-economic challenges. These conditions are characterised by persistent inflammation, resulting in debilitating symptoms such as pain, stiffness and functional impairment. Despite shared pathology, each disease exhibits distinct clinical, immunological and genetic features.1,2
RA, an autoimmune disorder which disproportionately affects women, comprised 70% of the diagnosed cases globally in 2019, according to the World Health Organization (WHO). 3 The prevalence of RA increases with age, with over half of diagnosed individuals being over 55 years of age. 4 The prevalence rate of axSpA in Asia is reported to be 167 per 10,000, while in India, it ranges between 0.03% and 0.2%. 5 PsA affects 8.7% of people with psoriasis in India. 6 These conditions significantly affected work productivity, with median presenteeism at 20% and overall productivity dropping by 20.34%, thus contributing to the financial burden of the illness. Additionally, 8% of patients had to quit their jobs, and daily activities were impaired by up to 40%. 5 The Community Oriented Program for Control Of Rheumatic Diseases (COPCORD) study in India reports prevalence rates of IJDs as RA(0.34%), axSpA(0.01%) and PsA(0.01%), highlighting their significant presence in the population. 7
The European League Against Rheumatism (EULAR) and the American College of Rheumatology (ACR) guidelines advocate a ‘treat-to-target’ approach for RA, aiming for remission or low disease activity (LDA). Initial therapy typically includes methotrexate (MTX) and glucocorticoids (GC), alone or in combination with other conventional disease-modifying anti-rheumatic drugs (csDMARDs), targeting LDA or clinical response (CR) results within 6 months. If LDA or remission is not achieved in 6 months and adverse prognostic indicators are present, the EULAR recommends biologic DMARDs (bDMARDs) or targeted synthetic DMARDs (tsDMARDs) such as tofacitinib.8,9 Although non-steroidal anti-inflammatory drugs (NSAIDs) are considered the first-line treatment option for axSpA, Janus kinase inhibitors (JAKi) or biologics may be added in patients with persistently high disease activity despite treatment with conventional therapies. 10 For PsA, the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) 2021 guidelines recommend that tsDMARDs be used as first-line therapy in musculoskeletal (MSK) domains other than peripheral arthritis. 11
Tofacitinib, as per the United States Food & Drug Administration (USFDA) recommendation, is indicated for adult patients with moderately to severely active RA, PsA and axSpA who have had an inadequate response (IR) or intolerance to one or more tumour necrosis factor (TNF) blockers. 12 The European Medicine Agency (EMA) recommends tofacitinib, in combination with MTX, for treating moderate to severe active RA in adult patients who have responded inadequately or are intolerant to one or more DMARDs. Tofacitinib can also be used as monotherapy if MTX is contraindicated or there is intolerance to MTX. For active PsA in adults, tofacitinib combined with MTX is indicated after an IR or intolerance to prior DMARD therapy. Additionally, tofacitinib is indicated for the treatment of active axSpA in adults who have responded inadequately to conventional therapies. 13
Despite notable therapeutic advances, significant gaps persist in delivering optimal care for IJDs, such as RA, PsA and axSpA. Research methodologies, particularly randomised controlled trials (RCTs), often lack real-world applicability due to short-term follow-ups and reliance on placebo comparators. Furthermore, the inherent heterogeneity of IJDs complicates treatment decisions, especially in regions like India, where specific treatment recommendations are lacking. Addressing these challenges is imperative for enhancing patient outcomes and optimising rheumatology practice. The evolving landscape of IJD treatments underscores the need for collaborations to establish optimal management strategies. This consensus document, formulated by a consortium of Indian rheumatology experts, aims to provide treatment recommendations for tofacitinib for the management of RA, PsA and axSpA.
Methodology
A steering committee of 6 experienced rheumatologists from the RA, axSpA and Psoriatic Arthritis Delphi in India (RAPID-India) group led the consensus development process to define the role of tofacitinib in managing RA, axSpA and PsA in India (Figure 1). The steering committee conducted a comprehensive literature review to identify unmet needs in treatment pathways for these conditions. To ensure clarity, the focus was limited to one molecule per paper without bias towards other treatments.
Flow Chart of Delphi Process in Framing Recommendations for Tofacitinib Usage in Inflammatory Arthritis in the Indian Context.
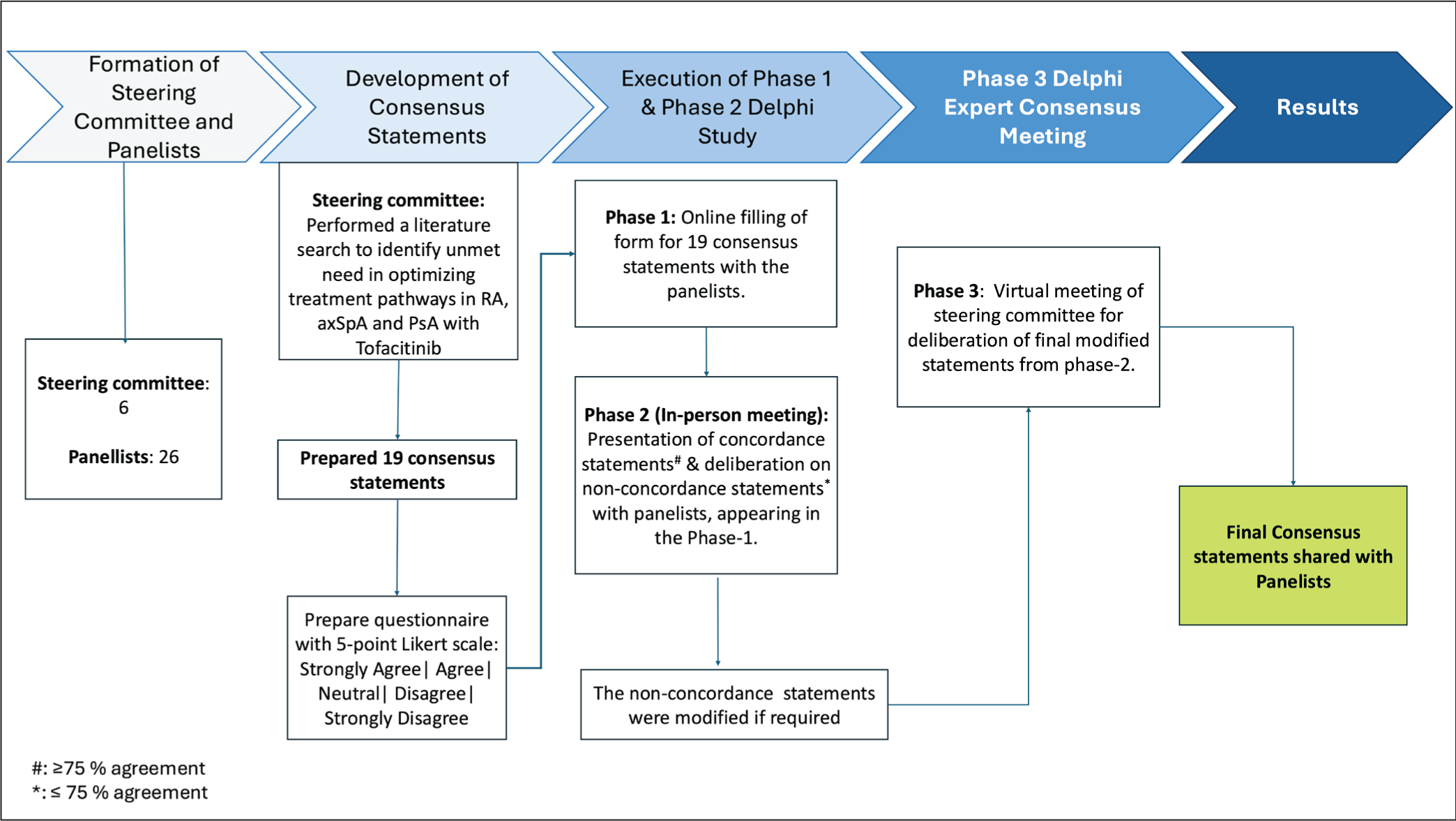
axSpA, axial spondyloarthritis; PsA, psoriatic arthritis; RA, rheumatoid arthritis.
A panel of 26 senior rheumatologists was selected based on geographic diversity, their therapy area experience and publications. These panellists were tasked with evaluating the statements developed by the steering committee. The process involved a 3-phase Delphi study. In Phase 1, panellists filled out an online questionnaire (see Supplementary Material) to rate their agreement with 19 key statements using a 5-point Likert scale, with consensus set at over 75% agreement. In Phase 2, an in-person meeting was held to review and discuss statements that didn’t reach 75% agreement in Phase 1. A descriptive analysis was conducted, and statements with 75% or more agreement were finalised, while non-concordant statements, that is, <75% agreement were modified based on feedback.
In Phase 3, a virtual meeting of the steering committee was conducted to review and finalise the modified statements from Phase 2. The finalised consensus statements were then shared with the panellists. This Delphi process ensured a rigorous and transparent approach, leading to evidence-based recommendations for using tofacitinib in the Indian clinical context.
Overarching Principles
Adhering to these overarching principles, tofacitinib can be utilised to optimally manage inflammatory arthritis in the Indian context. The overarching principles of this study have been outlined in Table 1.
Overarching Principles.

Expert Opinion
Clinical Profiles
A consensus by the panel recommends that patients diagnosed with RA and identified as having poor prognostic markers, like high disease activity, high titre autoantibody positivity (rheumatoid factor and/or anti-cyclic citrullinated peptide), or dual antibody positivity, presence of joint damage early in the disease or failure of >2 csDMARDs, should receive tofacitinib in combination with MTX in contrast to the addition of another csDMARDs. This recommendation aligns with the updated EULAR guidelines 2022 for RA that suggests the addition of bDMARD or JAKi, like tofacitinib, when csDMARDs fail to achieve treatment targets, particularly in patients with poor prognostic factors (Level of Evidence: Efficacy 1a; Safety: 1b, Strength of Recommendation: Efficacy A; Safety B). 14 Supplementary Table 1 mentions clinical trials evaluating the efficacy and safety of tofacitinib for RA.15,16 The panel noted that in developing countries like India, where biologics are unaffordable for most patients, tofacitinib offers a convenient alternative. Additionally, Indian MSK physicians often use triple therapy methotrexate-sulfasalazine-hydroxychloroquine (MTX-SSZ-HCQ), either initially or as a rescue therapy for MTX failure, achieving, in many instances, efficacy similar to anti-TNF-α agents combined with MTX. 17 However, the latest EULAR recommendations have not advocated that these combinations did not add to clinical efficacy but instead were associated with more adverse events and led to higher discontinuation rates. 14
The expert panel unanimously recommends combining MTX and tofacitinib for patients with difficult-to-treat RA who have failed at least two bDMARDs. Tesser et al. analysed Phase II and III trial data, focusing on the effectiveness in bDMARD-IR patients. They highlighted the percentage of patients achieving specific outcomes based on the number of prior failed biologic treatments, as mentioned in Supplementary Table 2. The study concluded that in patients who had previously failed bDMARDs, the success rates (ACR20/50/70 response and Disease Activity Score-28 remission) for tofacitinib were similar regardless of how many prior bDMARDs they had failed (1 or 2 or more). 18 This recommendation aligns with the USFDA, which suggests using JAKi only after at least one TNFi has failed. However, EULAR places JAKi at the same level as bDMARDs, that is, MTX-refractory RA. 14
The panel recommends tofacitinib as a monotherapy for RA patients who cannot tolerate or have contraindications to MTX and other csDMARDs. Tofacitinib is FDA-approved for treating adults with moderate to severe RA who have not responded adequately or are intolerant to MTX. It can be used alone or with MTX or other non-biologic DMARDs. 12 The EMA recommends tofacitinib as a monotherapy for patients intolerant to or unsuitable for MTX use. Many studies show high MTX intolerance, ranging from 11% to 50.5%, with symptoms reported in 16.5% to 88.7% of cases.19-23 Pooled trial data for tofacitinib (5 or 10 mg twice daily) versus placebo, both as monotherapy and with csDMARDs, showed improved efficacy at month 3 across all groups: non-MTX csDMARD-IR (n=537), MTX-IR (n=3113) and bDMARD-IR (n=782). Tofacitinib demonstrated clinical benefits in RA patients, with greater efficacy and a better benefit/risk ratio in those with fewer prior treatments. 18
The panel unanimously agreed to initiate tsDMARDs like tofacitinib for patients with active PsA and axial disease or PsA with dactylitis/enthesitis. 24 Experts noted the low evidence for using csDMARDs in treatment-naive axial disease patients. 25 Despite this, the panel opined that csDMARDs can be used as first-line therapy, especially in patients who also have peripheral arthritis. According to the 2021 GRAPPA guidelines, psoriasis treatment typically starts with topical therapies, while arthritis management often begins with csDMARDs. However, bDMARDs or tsDMARDs may be used as first-line therapy if appropriate. GRAPPA guidelines state that MTX is strongly recommended for DMARD naïve peripheral arthritis or conditionally recommended for peripheral arthritis with inadequate DMARD response. 11 For PsA patients with axial involvement, radiographic damage or severe skin/joint symptoms, early initiation of targeted therapy is recommended over MTX. 17 The EULAR 2024 PsA guideline states that in patients with active PsA and predominant enthesitis, axial, dactylitis or nail disease and who are not responding to NSAIDs and local GC injections, bDMARD should be initiated. Alternatively, JAKi-like tofacitinib should be considered. 26 A Cochrane review evaluated low-dose MTX (≤15mg/week) versus placebo for PsA. The MTX group showed higher disease response compared to placebo. However, the study could not assess side effects or long-term benefits. 27 Therefore, the panel considers that Indian data needs to be generated for this setting.
The panel recommends tofacitinib for axSpA patients not responding to NSAIDs and when bDMARDs are contraindicated or unavailable, which is in line with the Pan American League of Associations for Rheumatology (PANLAR-2023). 28 A real-world clinical setting evaluated the csDMARD and NSAID-sparing capability of generic tofacitinib for active axSpA at 6 months. Over 92% of patients significantly reduced or stopped NSAIDs, and around 28% needed fewer csDMARDs. Additionally, 50.6% reduced or discontinued both csDMARDs and NSAIDs. Tofacitinib was well-tolerated, and 57.9% of patients achieved a clinically significant improvement in disease activity score. 29 Another recent study from India compared tofacitinib and adalimumab for treating 266 patients with axSpA. Both therapies showed similar improvements in disease activity scores, such as Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) and Ankylosing Spondylitis Disease Activity Score (ASDAS), at 6 months. 30 The panel opined that for a low-resource country like India, tofacitinib offers a low-cost alternative to the more expensive biologic/biosimilar.
Therapeutic Assessment
The panel reached a consensus on the analgesic properties of tofacitinib, with the study showing rapid pain relief within 2 weeks of treatment initiation. 31 Significant pain reduction is observed within a month for RA, PsA and axSpA patients. 32 A post hoc analysis revealed significant pain relief by week 2 in RA and PsA patients treated with tofacitinib 5 and 10 mg twice daily (BID), compared to placebo, measured by the least square (LS) mean change from baseline in the Patient’s Assessment of Arthritis Pain (PAAP) scores, and the Short-Form Health Survey 36 version 2 (SF-36v2) Question (Q)7 Bodily Pain domain score. 33 A prospective study on RA patients investigated the effects of 10 mg of tofacitinib daily for 6 months. Tofacitinib rapidly reduces pain and central sensitisation, with significant decreases in pain intensity and central sensitisation index by Day 28. 32 Additionally, clinical trials have demonstrated that the analgesic effects of JAK inhibitors like tofacitinib can occur as early as 1 to 2 weeks, even before the control of inflammation. 34
The expert panel unanimously agreed on data showing tofacitinib’s efficacy in preventing radiographic progression, a key marker of severe disease. Three studies investigated tofacitinib’s effectiveness in limiting joint damage in RA. In a Phase 3 study, tofacitinib with MTX showed better results than placebo in preventing structural damage at 6 months. 35 A separate RCT with over 950 patients confirmed tofacitinib’s advantage over MTX in reducing joint damage on X-rays after 1 and 2 years. 36 Finally, a long-term study with over 1200 patients showed that tofacitinib limited radiological progression over 3 years, regardless of whether it was used alone or with other medications. These findings suggest tofacitinib’s potential for preventing joint damage in RA even at 5-year follow-ups. 37
For seronegative RA patients, the panel recommends using tofacitinib over rituximab. In seronegative patients, rituximab treatment had a less pronounced effect on radiographic progression and no effect on CR. The REFLEX trial indicated that while some seronegative patients responded to rituximab, joint protection was primarily observed in seropositive patients. 38 Conversely, a Phase 3 study evaluated the response to tofacitinib in patients with seropositive and seronegative RA. Results showed that tofacitinib significantly improved ACR20/50/70 response rates, disease remission (DAS28-4[ESR]) and physical functioning (SF-36) across all subgroups, with stronger responses in seropositive (anti-CCP+/RF+) patients compared to seronegative (anti-CCP-/RF-) patients. Safety profiles were similar across all groups. Overall, tofacitinib was effective in improving clinical outcomes for both seropositive and seronegative RA patients. 16
Dose Optimisation Approaches
The expert panel recommends a patient-centred management strategy for RA. Following initial steroid control, tapering and potential discontinuation of NSAIDs are advocated while maintaining targeted therapies and csDMARDs, particularly MTX in older patients. The dosing of JAK inhibitors, such as tofacitinib, may be individualised based on disease activity and patient preference. Optimising outcomes necessitates considering patient preferences, the heterogeneity of clinical presentations, and adherence to medication regimens. Additionally, the panel highlights the importance of achieving rapid control during flare-ups for RA patients in remission. In a prospective study, patients who restarted tofacitinib after a flare regained remission within a month, allowing dose reduction or withdrawal. 39 A pooled data on tofacitinib in RA patients assessed Clinical Disease Activity Index (CDAI) responses with or without MTX and GC. Discontinuation of MTX or GC was defined as not using them within 30 days before the year-3 visit, while initiation was defined as starting MTX or GC on or before the year-3 visit. Results showed that a significant proportion of patients achieved and maintained CDAI remission or LDA over 3 years, regardless of MTX/GC discontinuation. Patients who did not respond well to tofacitinib alone showed notable improvement when MTX was added, whereas adding GC led to only minor improvement. 40 Future research is required in this direction. The panel suggests that tapering will depend on these patients’ multimorbidity and baseline characteristics.
In the context of maintaining remission in RA, a majority of the panel members proposed transitioning patients from tofacitinib 5mg (BID) to the once-daily (OD) extended-release formulation of tofacitinib 11mg (XR). This shift has the potential to enhance treatment adherence. Bioavailability measurements showed equivalent total and peak exposures when the XR and immediate-release formulations were compared. Tofacitinib XR 11mg reached a steady state within 48 hours, with minimal accumulation upon repeat dosing. Tofacitinib XR 11mg was well-tolerated in healthy volunteers, offering a convenient OD dosing option. 41 In the ORAL Shift study, tofacitinib modified-release (MR) 11mg OD plus MTX improved disease activity measures, with 84.5% of patients achieving CDAI-defined LDA after 24 weeks. 42 Two noninterventional cohort studies showed improved adherence and comparable effectiveness with tofacitinib MR compared to immediate-release formulation over 12 months. The claims database study highlighted better adherence, especially in patients without prior advanced therapy (bDMARDs or JAK inhibitors other than tofacitinib). 43
The panel strongly advocates dose optimisation for patients with at least 6 months of remission or LDA in axSpA, RA and PsA. Concerns were raised about the short 6-month follow-up for axSpA, stressing the need for sustained remission assessment using sonographic and immunologic parameters. The EULAR 2023 guidelines for PsA management recommend tapering DMARDs in sustained remission cases. Tapering refers to dosage reduction, not discontinuation, to avoid flares. More long-term data on the tapering of bDMARDs and tsDMARDs is being published. Despite stronger recommendations, shared decision-making between patients and doctors remains crucial. (LoE: 2b, GoR: B) 26 EULAR 2022 treatment algorithm recommends that if bDMARDs or tsDMARDs demonstrate improved outcomes at 3 months and achieve treatment targets at 6 months, reducing the dose or increasing the interval between doses with minimal risk of flares is safe. However, immediate discontinuation is linked to a high risk of flare-ups. 14 Experts suggest that dose reduction or increased dosing intervals may be considered potential tapering strategies for JAKi therapy in appropriate patients. The panel unanimously discourages abrupt cessation of tofacitinib therapy. A Phase 4 study examined the effects of tofacitinib withdrawal or dose reduction in RA patients with stable disease control. Patients continuing their current dosage had the longest flare-free duration (5.8 months), followed by those with dose reduction (4.7 months) and those discontinuing tofacitinib (2.4 months). 44
The expert panel recommends reducing tofacitinib dosage for patients with compromised liver or kidney function. For severe renal impairment (creatinine clearance < 30 mL/min) or moderate hepatic impairment (Child-Pugh B classification), a lowered dosage of 5 mg OD is suggested to maintain treatment efficacy while minimising risks. Regular creatinine and transaminase level assessments are advised. Tofacitinib is contraindicated in severe hepatic impairment (Child-Pugh C) but can be used without dose adjustment in mild hepatic impairment (Child-Pugh A). For moderate hepatic impairment (Child-Pugh B), the dose should be reduced to 5 mg OD. 45
Precautions Before Initiation
The panel recommends a thorough pre-treatment evaluation, including comprehensive laboratory tests, before initiating tofacitinib therapy (Table 2).
Evaluating and testing for latent and active tuberculosis according to guidelines is crucial. For latent tuberculosis (LTBI) screening, standard guidelines 46 recommend either interferon gamma release assay (IGRA) or tuberculin skin testing (TST), alongside a chest X-ray (PA view). Each test has its own strengths and limitations. The expert panel suggests that while either test can be chosen based on resource availability, performing both tests is preferred in a high-burden country like India to enhance sensitivity.9,47-49 Patients testing positive should be treated for LTBI. In line with 2022 EULAR, US FDA and Australian guidelines, close monitoring for tuberculosis signs and symptoms throughout treatment is also essential.12,50,51 The panel recommends that a chest X-ray be performed within the last 3 months before initiating tofacitinib.
The panel discussed the association between inflammation and lipid abnormalities. The more severe the underlying inflammatory disease, the more consistently abnormalities in lipids and lipoproteins are observed. Table 2 shows the monitoring of tofacitinib therapy as per the FDA label.
Studies indicate that tofacitinib is an effective immunosuppressive agent for renal transplantation. However, its use in current protocols is linked to higher risks of Cytomegalovirus (CMV), BK and Epstein-Barr virus (EBV) viral infections, as well as anaemia, leukopenia and post-transplant lymphoproliferative disorder. 52
The panel recommends using the recombinant VZV vaccine 2 to 4 weeks before starting tofacitinib and advises against live vaccines during therapy. Ideally, both doses of the recombinant vaccine (at 0 and 2 months) should be administered before initiating JAKi. However, in patients with autoimmune rheumatic diseases (AIRDs), vaccination is best timed during disease quiescence, which may not align with the need to start JAKi during active disease.46,53-55 As a recombinant vaccine, it can still be safely administered after JAKi initiation. The FDA advises avoiding live vaccines with tofacitinib and following guidelines for immunosuppressive agents. The effectiveness of the recombinant VZV vaccine may be reduced if given after starting tofacitinib. 56 Supplementary Table 3 shows different guidelines for administering the VZV vaccine in patients receiving tsDMARDs.46,57
Safety Considerations
The panel endorsed the use of tofacitinib even in elderly patients >60 years, who have low or negligible CV risk factors and patients with active disease unresponsive to multiple bDMARDs. Retrospective analysis reveals that strict adherence to EMA recommendations in clinical practice may lead to the exclusion of a significant number of RA patients from treatment with JAKi. To better customise RA management, it is recommended to accurately assess the risk of major adverse cardiovascular events (MACE) using dedicated tools like Expanded Risk Score in RA (ERS-RA). 58 It is recommended that tofacitinib be used with caution in the elderly, depending on the risk of MACE, until more real-world data is available. Figure 2 shows a simple prescribing guidance tool that stratifies patients by risk. 59 Supplementary Table 4 shows data from pooled Phase 3 and long-term extension studies showing lower incidence of CV events with tofacitinib. 60 Analysis from the ORAL Surveillance trial revealed that both the 5mg and 10mg BID doses of tofacitinib were associated with a heightened risk of malignancies and MACE. Risk of lung cancer is higher in tofacitinib (1.0% non-small cell lung cancer) excluding non-melanoma skin cancers (HR: 1.47, 95% CI: 1.00, 2.18 and HR: 1.48, 95% CI: 1.00, 2.19, respectively). Continued vigilance is warranted in monitoring potential risks, especially in specific patient demographics or prolonged treatment durations. 61 A cohort study found no heightened malignancy risk with tofacitinib compared to TNFi in RA patients (pooled weighted HR: 1.01, 95% CI: 0.83, 1.22). However, prolonged treatment may slightly increase the risk. 62 The panel concurred with the FDA’s black box warning based on the available evidence. They emphasise ongoing monitoring and benefit-risk assessment, especially for older patients or those on long-term therapy.
While discussing the management of ILD, leflunomide was noted to potentially worsen ILD, while MTX may offer some protective benefits. Tofacitinib, however, emerged as a potentially promising option, with some evidence suggesting it might even reduce existing ILD and be suitable for use in the early stages of the disease. 63 A cohort study noted a lower incidence of ILD among RA patients receiving tofacitinib compared to those treated with all classes of bDMARDs. Furthermore, patients treated with tofacitinib had a 69% reduced risk of ILD compared to those treated with bDMARDs. 64 Similarly, post-hoc analysis from 21 clinical trials shows that incidence rates for ILD with both doses of tofacitinib were 0.18 per 100 patient-years, and these rates generally remained consistent over time. Among the ILD events, 17 out of 42 (40.5%) were classified as SAE, while 35 out of 42 events (83.3%) were characterised as mild to moderate in severity. 65 While data is limited, tofacitinib’s safety profile appears favourable for RA-ILD patients. Further long-term studies are needed to confirm these promising observations. 63
Discussion
The recommendations provided by the RAPID-India group for using tofacitinib in RA, PsA and axSpA distinguish themselves from the ACR/EULAR guidelines in several significant aspects, even while offering India-specific treatment recommendations. The RAPID-India group recommends tofacitinib as monotherapy for RA when MTX and other csDMARDs are not tolerated or contraindicated. Furthermore, for PsA with predominant axial disease, dactylitis or enthesitis refractory to NSAIDs, the RAPID-India group recommends tofacitinib use as an alternative to the more traditional use of a bDMARD in this setting. In axSpA, tofacitinib is advised when NSAIDs fail, and bDMARDs are contraindicated or considered expensive, reflecting a pragmatic approach tailored to resource-limited settings. The group also highlights the rapid analgesic effects of tofacitinib, often within 2 weeks, which is a crucial consideration for improving patient-related outcomes (PROs).
Additionally, the long-term use of tofacitinib to prevent radiographic progression in RA and its efficacy and safety profile comparable to bDMARDs highlights a shift towards its wider acceptance. The recommendation for tapering corticosteroids and switching to tofacitinib XR to enhance compliance, dosing adjustments for renal and hepatic impairments and a well-defined, comprehensive pre- and post-treatment monitoring reflects the broad coverage of tofacitinib’s indications in this consensus paper. To provide clarity, the committee chose to focus on one specific molecule per paper, leading this consensus to place particular emphasis on tofacitinib for managing RA, axSpA and PsA in India. This focus is intended for simplicity and does not imply bias or preference over other effective therapies that are not discussed. In summary, the RAPID-India group’s recommendations, specific to the unique Indian setting and tailored to the average Indian patient’s requirements, are important in caring for patients with musculoskeletal disorders in India. Figures 3, 4 and 5 highlight the positioning of tofacitinib in RA, axSpA and PsA.
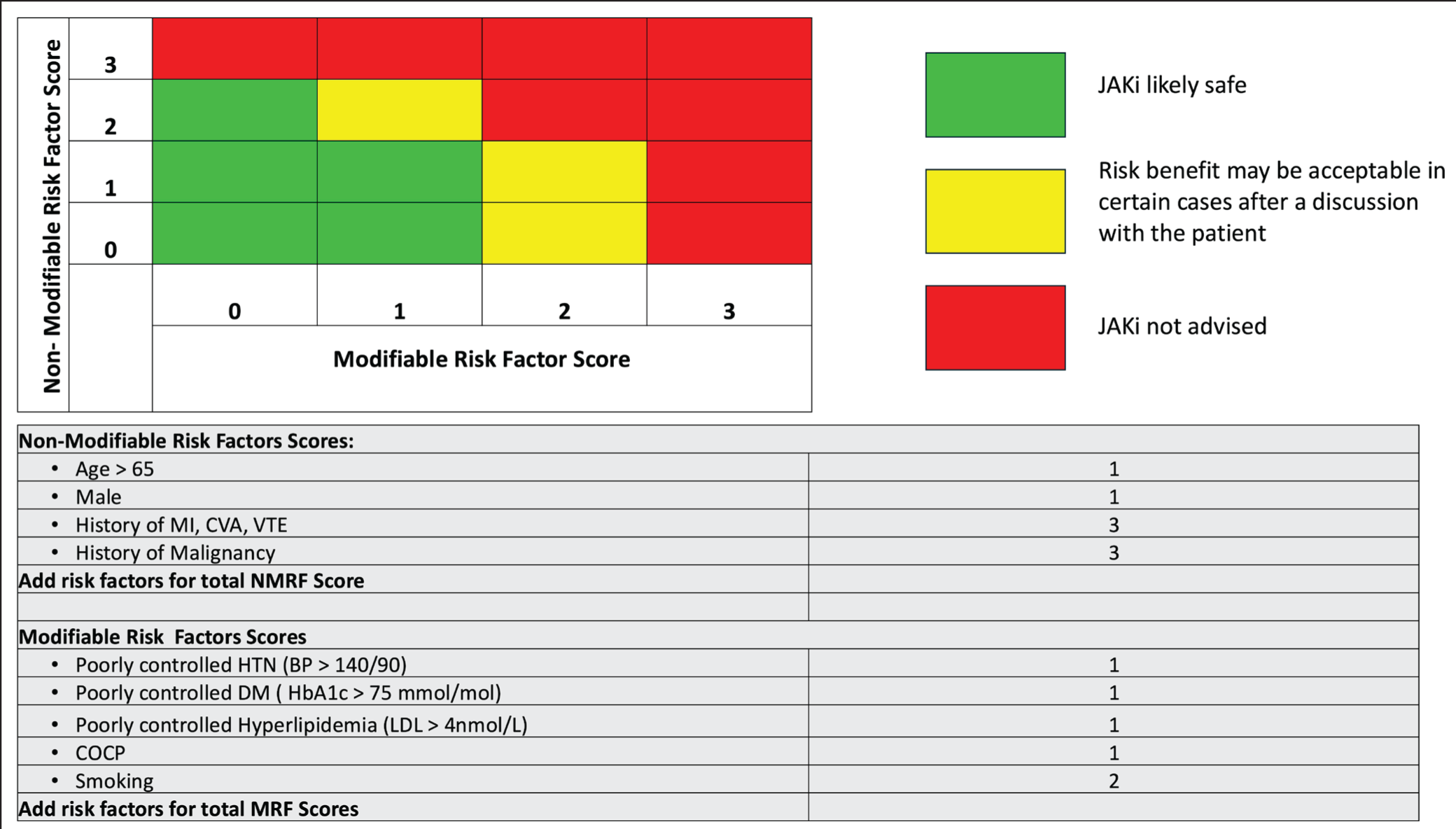
BP: blood pressure, COCP: combined oral contraceptive pill, CVA: cerebrovascular accident, DM: diabetes mellitus, HbA1c: glycated haemoglobin, HTN: hypertension, JAKi: Janus kinase inhibitors, LDL: low-density lipoprotein, MI: myocardial infarction, VTE: venous thromboembolism.
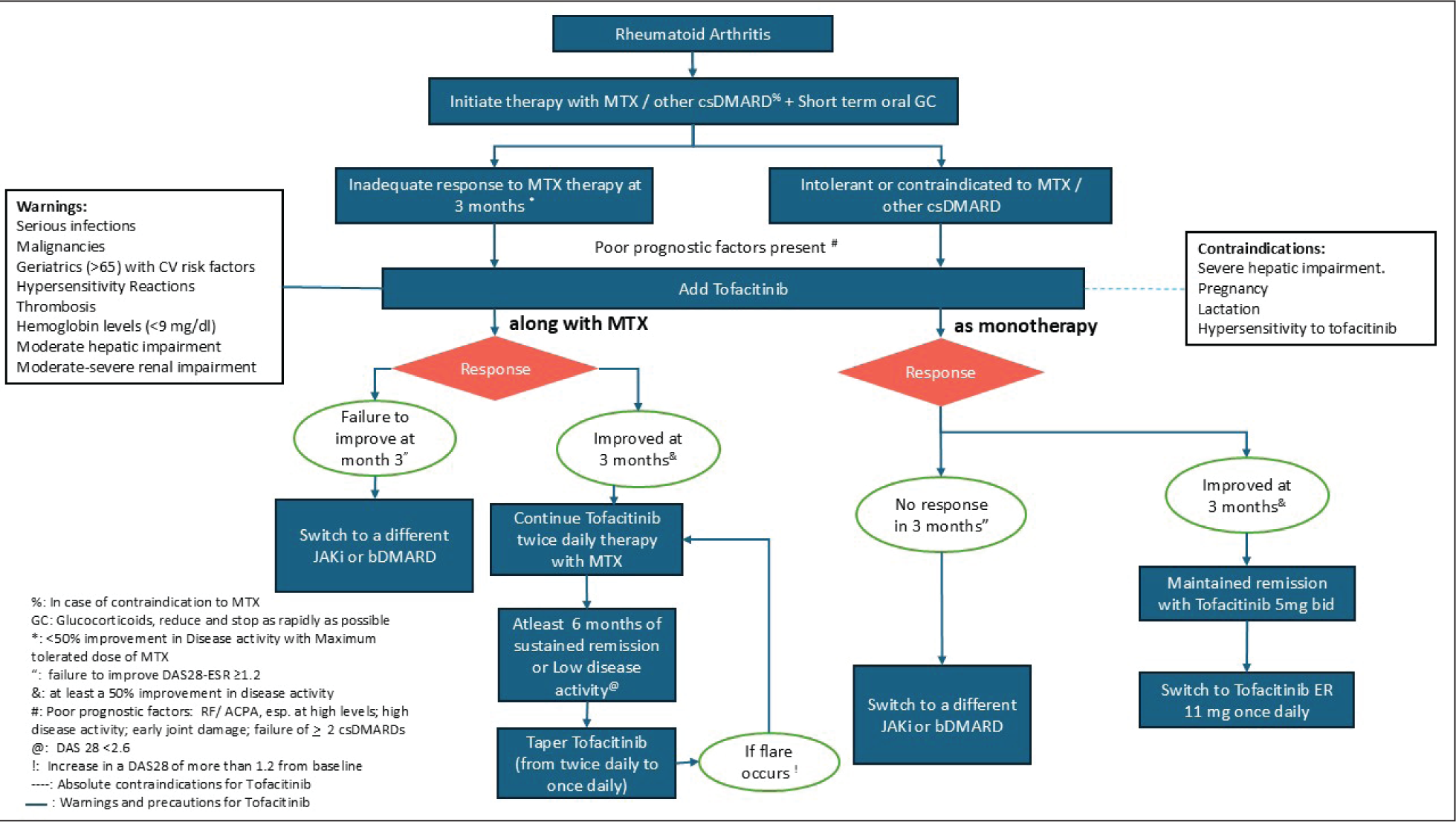
ACPA: anti-cyclic citrullinated peptide, BID: twice daily, bDMARD: biological disease-modifying anti-rheumatic drug, csDMARD: conventional disease-modifying anti-rheumatic drug, CV: cardiovascular, DAS28-ESR: Disease Activity Score in 28 joints (erythrocyte sedimentation rate), ER: extended-release, GC: glucocorticoids, JAKi: Janus kinase inhibitors, MTX: methotrexate, RF: rheumatoid factor
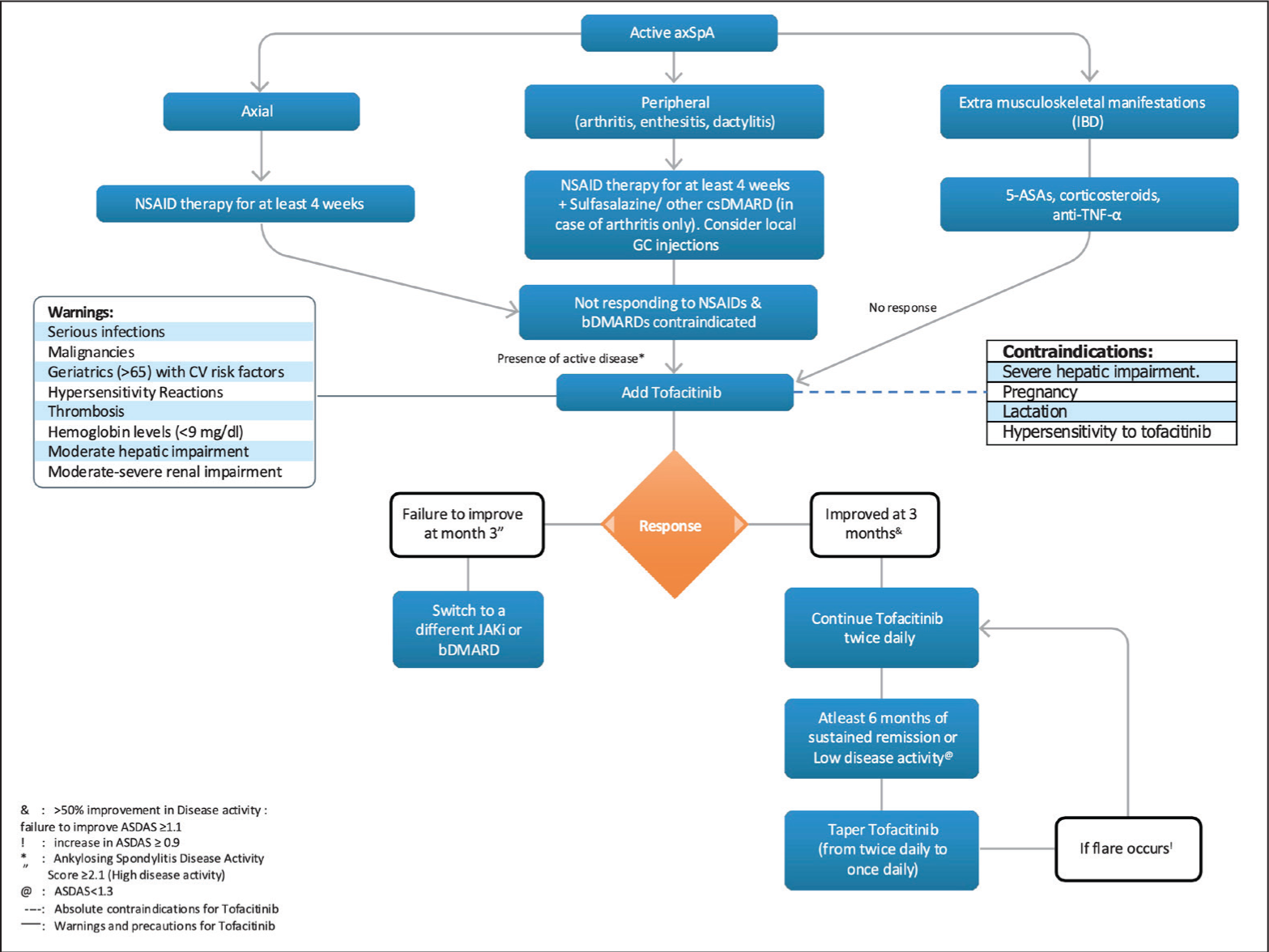
ASA: aminosalicylic acid, ASDAS: Ankylosing Spondylitis Disease Activity Score, axSpA: axial spondyloarthritis, BID: twice daily, bDMARD: biological disease-modifying anti-rheumatic drug, csDMARD: conventional disease-modifying anti-rheumatic drug, CV: cardiovascular, ER: extended release, GC: glucocorticoids, IBD: inflammatory bowel disease, JAKi: Janus kinase inhibitors, MTX: methotrexate, NSAID: non-steroidal anti-inflammatory drug, TNF: tumour necrosis factor.
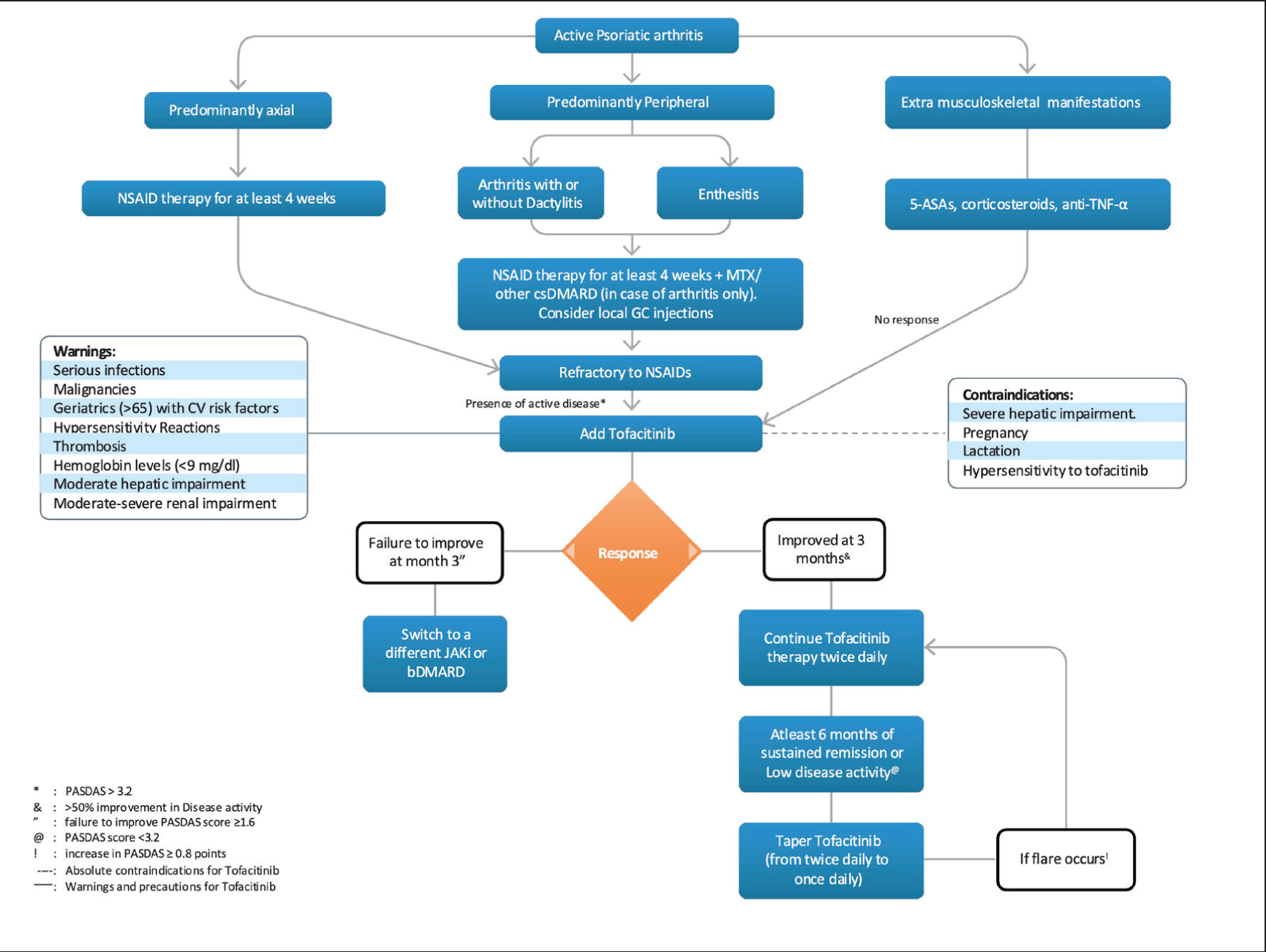
ASA: aminosalicylic acid, ASDAS: Ankylosing Spondylitis Disease Activity Score, axSpA: axial spondyloarthritis, BID: twice daily, bDMARD: biological disease-modifying anti-rheumatic drug, csDMARD: conventional disease-modifying anti-rheumatic drug, CV: cardiovascular, ER: extended release, GC: glucocorticoids, IBD: inflammatory bowel disease, JAKi: Janus kinase inhibitors, MTX: methotrexate, NSAID: non-steroidal anti-inflammatory drug, PASDAS: psoriatic arthritis disease activity score, TNF: tumour necrosis factor
Conclusion
The RAPID-India provides recommendations for the use of tofacitinib in RA, PsA and axSpA for the Indian setting. Tofacitinib is recommended for patients with IR to MTX or other csDMARDs, difficult-to-treat RA and as monotherapy where csDMARDs are not tolerated. It shows rapid analgesic effects and efficacy comparable to biologic DMARDs across various demographics. Key recommendations include comprehensive pre-treatment screening, regular monitoring and careful use in elderly patients due to elevated malignancy risks. Long-term therapy with tofacitinib is advocated to prevent radiographic progression and maintain disease control alongside tapering strategies, even while sustaining remission and minimising the chances of flares. Tofacitinib thus emerges as a useful addition in optimising treatment outcomes and enhancing the quality of life for patients with RA, PsA and axSpA.
Supplement Material
Supplement material for this article is available online.
Footnotes
Acknowledgements
We acknowledge Dr Ajaz Kariem Khan, Dr Ajit Nalawade, Dr Alok Kalyani, Dr Ashit Syngle, Dr Atul Kakkar, Dr B. Rajasekar, Dr Bharat K Singh, Dr Bimlesh Dhar Pandey, Dr Dhaiwat Shukla, Dr Durgesh Srivastava, Dr G Narsimhulu, Dr K Shanmuganandan, Dr Keerthi. T, Dr N Raja, Dr Piyush Joshi, Dr Ramesh. Jois, Dr Reena Sharma, Dr Renu Saigal, Dr Samir Rajadhyaksha, Dr Sanjiv Kapoor, Dr Shailaja Sabnish, Dr URK Rao, Dr V Krishnamurthy, Dr Ved Chaturvedi, Dr Vikram Raj Jain and Dr Vineeta Shoba for their contribution in Phase 1 and 2 of Delphi. Dr Yogesh Sharma and the Abbott medical team were instrumental in facilitating the proceedings and helping with the writing and data collection for these papers. The authors also thank IntelliMed Healthcare Solutions, Mumbai, for medical writing support.
Author Contributions
The conception and design of the study, acquisition of data or analysis and interpretation of data: Dr Upadhyaya Sundeep Kumar, Dr Akerkar Shashank M and Dr Santhanam Sham Drafting the article or revising it critically for important intellectual content: All Authors Final approval of the version to be submitted: All authors Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved: All authors
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: All the authors have received speaker fees and have been on the advisory board of various companies that manufacture/market adalimumab.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Abbott Healthcare Pvt Ltd.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

