Abstract
Systemic lupus erythematosus (SLE) is a complex autoimmune disease characterised by multi-system involvement and diverse clinical manifestations. Among the systems affected, the gastrointestinal and hepatobiliary systems are notable taragets of the autoimmune response. Although gastrointestinal and hepatic syndromes commonly reported, gallbladder dysfunction is rare and not included in the diagnostic criteria for the disease. Biliary complications in SLE can present as a spectrum of conditions, ranging from acalculous cholecystitis and Salmonella infection to more uncommon manifestations such as primary biliary cirrhosis, autoimmune cholangiopathy, and primary sclerosing cholangitis. These complications frequently occur in the context of heightened lupus disease activity, with vasculitis identified as a key underlying mechanism, and are associated with significant morbidity. Thus, biliary involvement should be considered in the differential diagnosis of abdominal pain and related symptoms in patients with SLE. This review article explores the spectrum of gall bladder and the biliary manifestations in SLE and discusses the corresponding diagnostic and treatment strategies.
Keywords
Introduction
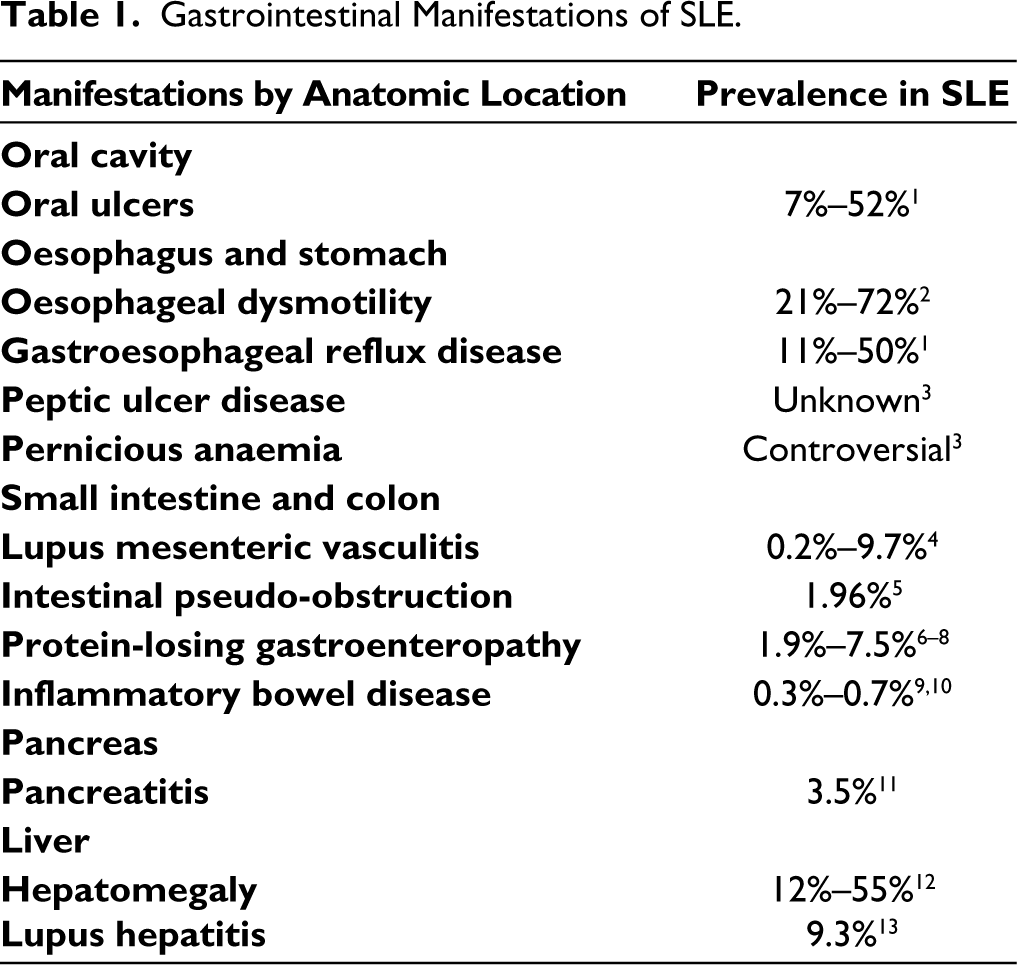
Systemic lupus erythematosus (SLE) is a complex autoimmune disease that can affect multiple organ systems, including cardiovascular, pulmonary, gastrointestinal (GI), hepatic, renal, musculoskeletal and central nervous systems. 1 GI and liver-related issues are observed in up to 40% of patients with SLE over their lifetime. These patients often present with nonspecific abdominal symptoms that require thorough and timely evaluation due to potential life-threatening enteral organ involvement. Commonly reported GI manifestations (Table 1) include protein-losing enteropathy, intestinal pseudo-obstruction, lupus-related hepatitis and acute and recurrent pancreatitis. 2
Gastrointestinal Manifestations of SLE.
In contrast, gallbladder and biliary tree involvement remains an under-researched and underreported aspect of SLE, despite extensive research on other SLE complications. A review of past publications revealed sporadic case reports of cholecystitis, Salmonella infections, primary biliary cirrhosis (PBC), and primary sclerosing cholangitis (PSC) in association with SLE.3–6 However, there is no comprehensive article that summarises lupus-related gallbladder and biliary manifestations or deals with this topic systematically. Moreover, these complications are associated with high mortality, morbidity and healthcare burden in SLE. 3 Early identification and intervention are therefore pivotal in optimising long-term outcomes.
This review aims to address this gap and provide an overview of SLE-related manifestations of the gallbladder and biliary system. The goal is to enhance understanding of gallbladder disease so that it is considered and monitored alongside other known complications of SLE. Additionally, this review will provide a framework for the diagnostic approach and treatment strategies for these pathologies in the context of SLE.
Methods
An extensive review of the literature was conducted to identify articles describing gall bladder and biliary diseases associated with SLE from inception to July 2024. We were able to narrow down to the four most reported pathologies based on their prevalence in case reports and studies, as well as their documented impact on patients with SLE: acute acalculous cholecystitis (AAC), Salmonellosis, PBC, and PSC (Table 2). To gather relevant information, a semi-structured review was performed, focusing on case reports, case series and review articles. Abstracts and non-English results were excluded from this search. The specific methodology for each pathology is outlined below.
Overview of Biliary Diseases Associated With SLE.

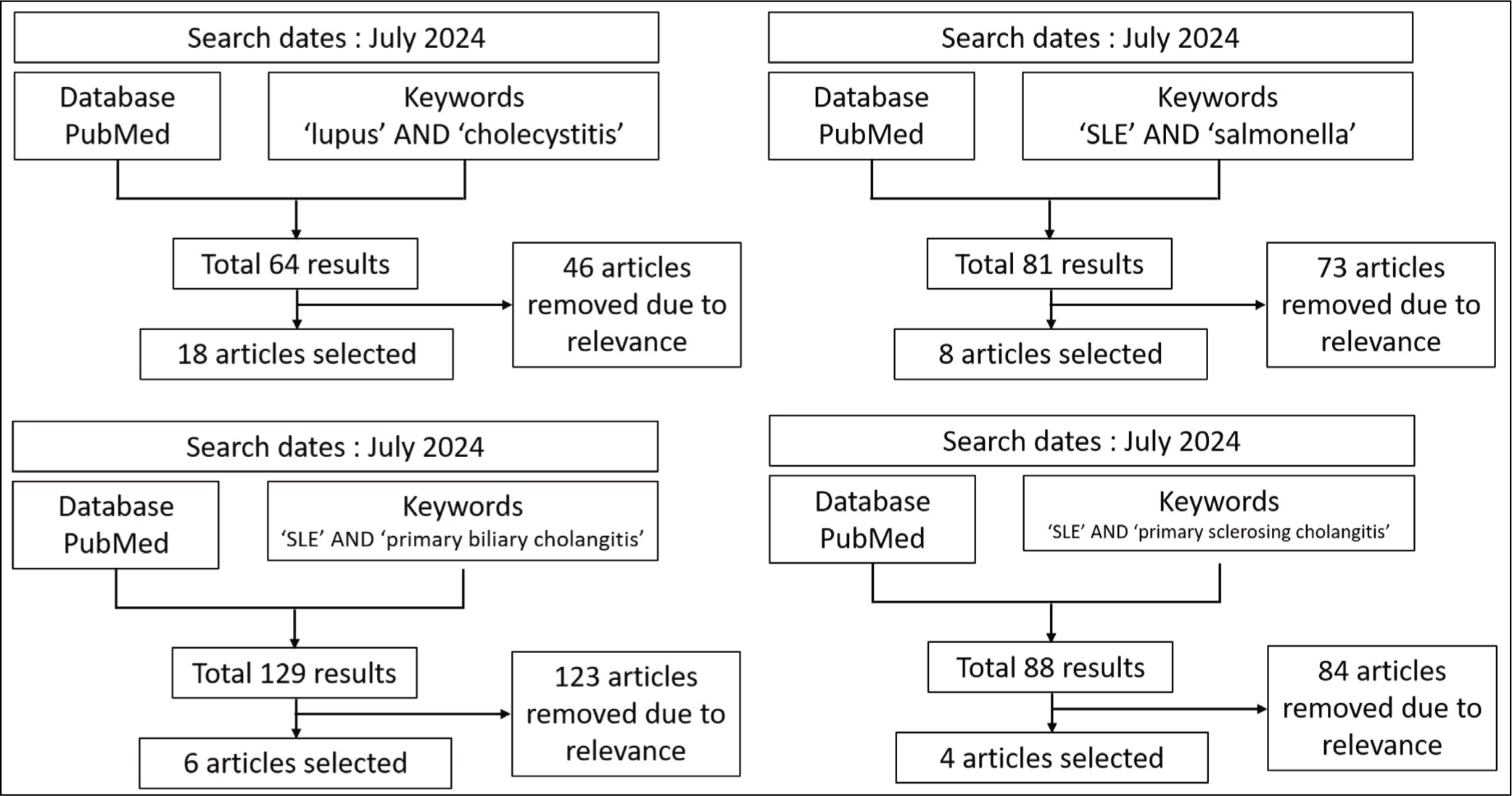
For AAC, PubMed search was performed using the keywords ‘lupus’ and ‘cholecystitis’ in conjunction with the ‘AND’ function. Of the total 64 results, 18 articles described AAC in a total of 31 patients with SLE.3,7–23 For Salmonella infections, the keywords ‘salmonella’ and ‘SLE’ were used for the search. This resulted in 81 hits, from which eight pertinent case reports, narrative reviews, and retrospective studies were selected.4,24–30 For PBC, a similar approach was used with the keywords ‘primary biliary cholangitis’ and ‘SLE’. This search identified six relevant retrospective studies and case series.6,31–35 For PSC, the literature search employed the keywords ‘lupus’ and ‘primary sclerosing cholangitis’ using the ‘AND’ function. The search revealed 88 results, of which only four case reports could be identified.36–39 These search strategies are summarised in Figure 1.
Flow Diagram Illustrating the Search Strategy for Gallbladder Disease in Patients with SLE.
Discussion
SLE-associated Acute Acalculous Cholecystitis (SLE-AAC)
Epidemiology and Clinical Features
AAC is a form of cholecystitis characterised by inflammation of gallbladder in the absence of cystic duct obstruction by gallstones. 40 It represents 5%-10% of acute cholecystitis and 0.19% of surgical intensive care unit admissions.41,42
It is the most commonly reported SLE-related biliary manifestation with an incidence as low as 0.15% in patients with SLE. 10 The review of existing case reports is presented in Table 3, all cases were from hospitalised patients. Most patients who were reported to have SLE-associated acute acalculous cholecystitis (SLE-AAC) had an established diagnosis of SLE (71%, 22 of 31). Most (93.5%, 29 of 31) of patients presented with other signs of active disease such as serositis, nephritis, arthritis or discoid rashes. From this data, SLE-AAC seems to present as part of SLE flare and rarely as a standalone presentation.
Pathophysiology
The main pathophysiology underlying SLE-AAC is not yet fully understood. Proposed mechanisms include vascular inflammation (arteritis and venulitis) of the gall bladder, ischaemia, thrombosis, mesenteric inflammatory veno-occlusive disease and serositis. In the 10 cases that underwent cholecystectomy, histologic evidence demonstrated vasculitis involving mostly the medium-sized arteries of the gallbladder wall. Only one case reported venulitis. 18 Other cases reported acute arteritis, including occlusion of arteries due to perivascular inflammation or fibrinoid changes. Two cases presented with haemobilia and calculous cholecystitis. However, both cases demonstrated mononuclear/lymphocytic infiltration of the arteries, showing histological concordance with other acalculous cholecystitis cases.
Diagnostic Approach
Diagnosing SLE-AAC requires a thorough history, physical examination, and correlation with imaging and laboratory findings. Acute calculous cholecystitis (ACC) is a more prevalent disease than AAC, and their management might differ in patients with SLE. They both share similar clinical presentations including abdominal pain, right upper quadrant tenderness and fever. However, AAC is often are accompanied by SLE flare. Thus, screening for other signs and symptoms of SLE activity might be helpful in differentiating ACC and AAC clinically. These would include thorough physical examinations to look for alopecia, skin lesions and arthritis.
Radiological imaging is the mainstay of diagnosis for AAC as it has characteristic findings. Diffuse gallbladder wall thickening of 3.0 mm or more, pericholecystic fluid accumulation and/or signs of gallbladder wall gas or oedema on abdominal computed tomography (CT) scan or abdominal ultrasound has been widely used for diagnosis. 43 Hepatobiliary Iminodiacetic Acid scans are widely used in diagnosing ACC but may have limited utility in SLE-AAC. 16
Other radiological imaging techniques including chest X-rays and echocardiograms may also be helpful as Table 3 shows SLE patients with AAC often present with serositis of the pleura and pericardium.
Case Reports of Acute Acalculous Cholecystitis (AAC) in Patients With SLE.
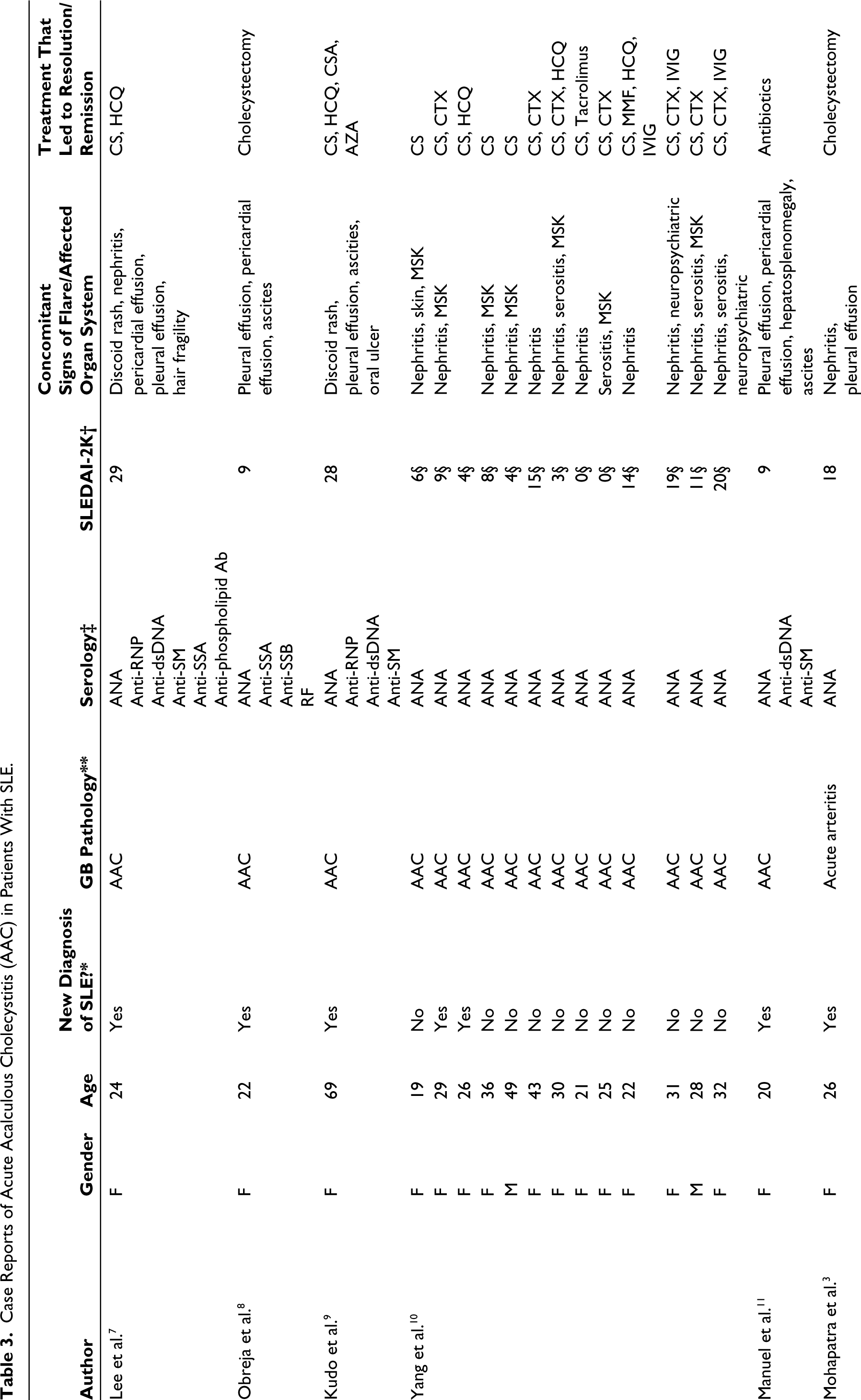
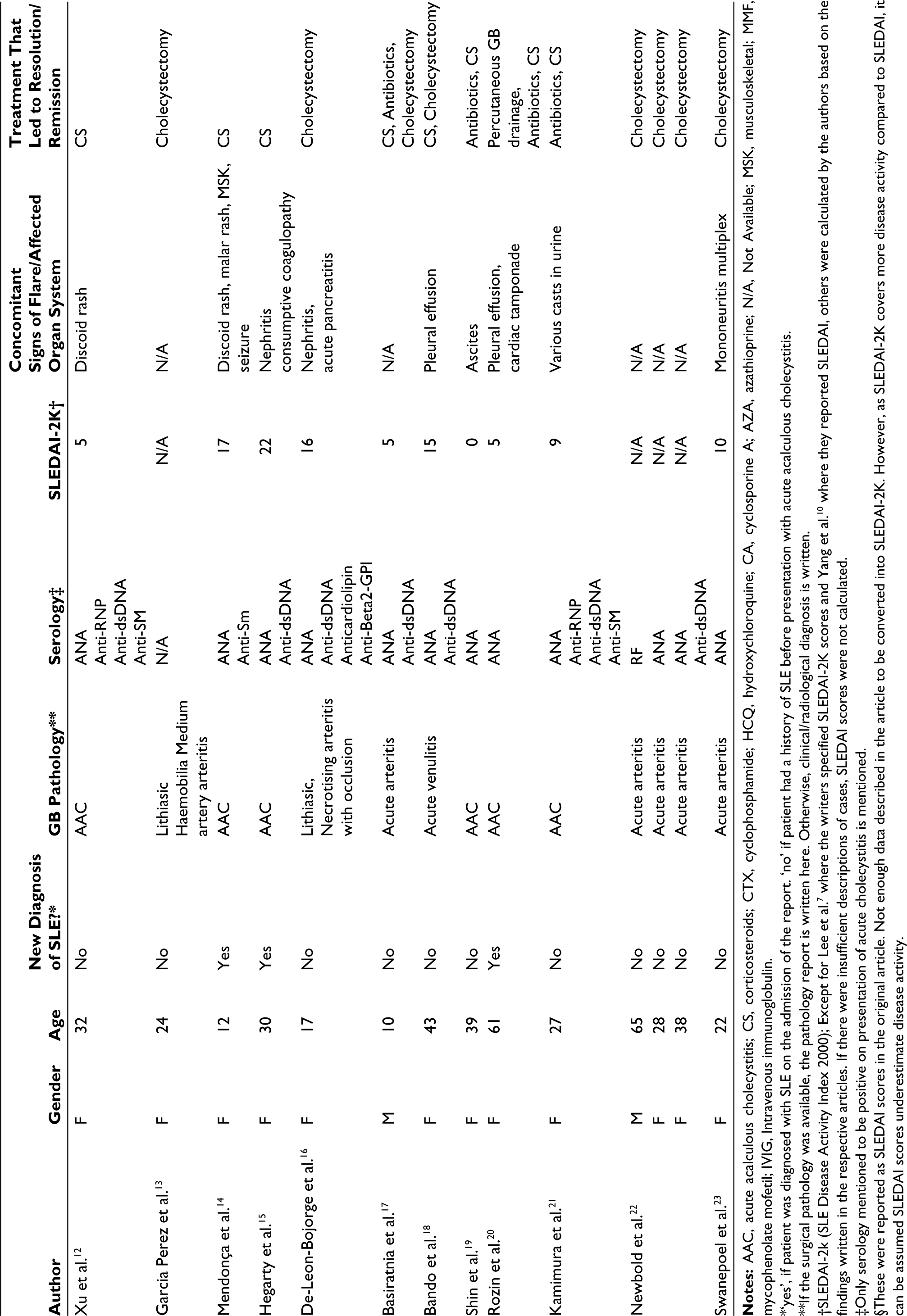
*‘yes’, if patient was diagnosed with SLE on the admission of the report. ‘no’ if patient had a history of SLE before presentation with acute acalculous cholecystitis.
**If the surgical pathology was available, the pathology report is written here. Otherwise, clinical/radiological diagnosis is written.
†SLEDAI-2k (SLE Disease Activity Index 2000); Except for Lee et al. 7 where the writers specified SLEDAI-2K scores and Yang et al. 10 where they reported SLEDAI, others were calculated by the authors based on the findings written in the respective articles. If there were insufficient descriptions of cases, SLEDAI scores were not calculated.
‡Only serology mentioned to be positive on presentation of acute cholecystitis is mentioned.
§These were reported as SLEDAI scores in the original article. Not enough data described in the article to be converted into SLEDAI-2K. However, as SLEDAI-2K covers more disease activity compared to SLEDAI, it can be assumed SLEDAI scores underestimate disease activity.
Laboratory studies will help with monitoring SLE disease activity and assessing antiphospholipid syndrome. Elevated anti-ds-DNA antibodies, low complement 3, complement 4 levels usually indicate active SLE disease. 44 Renal function tests may be helpful in screening and monitoring renal involvement of SLE.
Management
Management of SLE-AAC patients has not been standardized. There are no formal guidelines or consensus to prefer one treatment strategy over the other. However, review of the literature indicates that managing it as a lupus flare seems to be sufficient in most cases. Approximately 71% (22 of 31) of cases achieved clinical remission through medical management alone with the use of high-dose corticosteroids, immunosuppressive therapy with cyclophosphamide, intravenous gamma globulin, azathioprine, mycophenolate and hydroxychloroquine, as well as empirical antibiotics. Two cases reported by Basiratnia et al. 17 and Bando et al. 18 were the only cases where medical management failed and needed cholecystectomy. No other cases that achieved remission through medical management reported elective cholecystectomy in future follow-ups. There was one case by Rozin et al. 20 where patient required percutaneous gallbladder drainage as cholecystectomy was deemed to be too high risk. Subsequently, that case achieved remission with medical management without further need for surgical interventions.
Overall, AAC-SLE is considered a serious SLE-related syndrome and has been successfully managed as part of the medical management of SLE flare with adequate response to immunosuppressive therapy. In patients indicated for acute surgical intervention, cholecystectomy or percutaneous drainage has been pursued. For critically ill patients who cannot undergo surgery, endoscopic nasobiliary drainage has demonstrated success.3,12,45
Salmonellosis and Gall Bladder Involvement
Epidemiology and Clinical Features
In patients with SLE, Salmonella infection, particularly bacteraemia, is reported to have a higher incidence than the general population.24,26 Salmonella genus is comprised of various subtypes, each exhibiting different clinical manifestations. Typhoidal Salmonella is prevalent in immigrant population from endemic areas and primarily causes enteric fever. 46 In contrast, nontyphoidal Salmonella causes a zoonotic disease that commonly presents as acute, self-limiting gastroenteritis. 26 Invasive disease tendencies are more pronounced in extreme age groups and individuals with underlying immunocompromised conditions. 26 In patients with SLE, Salmonella bacteraemia tends to persist longer, increasing the risk of gallbladder involvement. 4
Pathophysiology
SLE is associated with impaired mononuclear phagocytic system function, decreased serum opsonising activity, functional hyposplenism, haemolysis, and the use of immunosuppressive medications such as corticosteroids, all of which increase the susceptibility to this infection. 4 Conversely, there is a bidirectional relationship where prolonged Salmonella infection increases the risk of developing SLE by stimulating the immune response and augmenting oxidative stress. 30
Diagnostic Approach
Typhoidal Salmonella infection should be suspected in patients presenting with enteric fever-like symptoms such as persistent high-grade fever, fatigue, headache, nausea, abdominal pain, diarrhoea, or constipation. 3 Salmonellosis typically manifests as self-limiting gastroenteritis. 26 Therefore, obtaining blood, stool, and urine cultures is recommended to rule out Salmonella infection in lupus patients who are febrile or presenting with gallstones.4,24 Stool cultures are also beneficial in identifying chronic carriers. 27
Management
Salmonella bacteraemia responds well to fluoroquinolones and ceftriaxone. In gallbladder carrier states, prolonged courses of fluoroquinolones, trimethoprim-sulfamethoxazole, or amoxicillin may be necessary. Cholecystectomy is recommended for these carriers to decrease the risk of recurrence. 25 As patients with SLE are likely to have chronic colonisation of Salmonella in the gallbladder, early detection and prompt initiation of antibiotic therapy is recommended. Whether prophylactic cholecystectomy or antibiotics should be warranted would need to be further studied.
Primary Biliary Cholangitis
Epidemiology and Clinical Features
PBC is an autoimmune disease driven by the interplay of genetic and environmental factors, leading to the loss of self-tolerance and immune-mediated damage to small intrahepatic bile ducts. 6
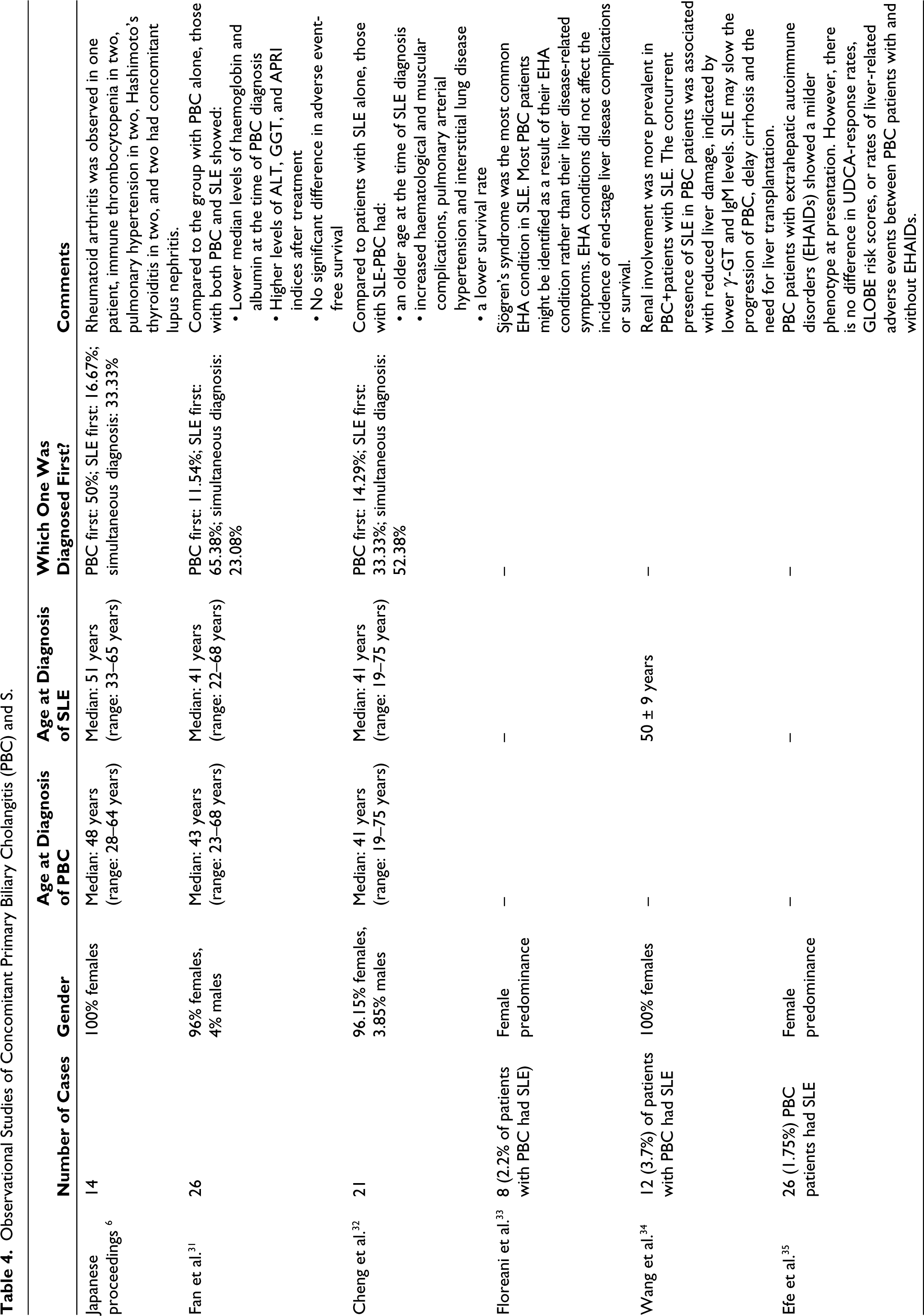
The incidence of PBC in the context of SLE is reported to range from 0% to 2.7%. 6 As many as 120 cases of combined PBC-SLE are reported mostly in the form of case reports. Data from two large centre-based studies in China and combined Japanese proceedings are summarised in Table 4.6,31,33 This included a total of 61 patients, predominantly females (97.5%). Median age at PBC diagnosis was 45, whereas that at SLE diagnosis was 41 years (age range: 19-65). The timeline of PBC diagnosis relative to SLE varied: PBC preceded SLE in 20% of cases, followed SLE diagnosis in 44%, and occurred concurrently in 36%. Histopathological examination of the liver reveals chronic, nonsuppurative, and destructive cholangitis, occasionally with epithelioid granuloma formation. Additional findings may include portal inflammation, chronic cholestasis, interface hepatitis or lobular hepatitis, and bile duct loss.6,47
Observational Studies of Concomitant Primary Biliary Cholangitis (PBC) and S.
Pathophysiology
The association between these two disorders may be attributed to shared genetic susceptibilities. Transcriptome overlap analyses have identified hub genes common to both disorders, suggesting a genetic connection. 48 In fact, each of these disorders could serve as a high-risk factor for the development of the other. 48 Surprisingly, there seems to be no correlation between lupus disease activity and the development of PBC. 6 Patients with concurrent PBC and SLE often present with multiple extrahepatic autoimmune conditions (EHA), such as Sjögren’s syndrome, immune thrombocytopenia, rheumatoid arthritis, Hashimoto’s thyroiditis, and pulmonary hypertension. 6
Diagnostic Approach
PBC commonly affects middle-aged women, often with a familial and smoking history. Common symptoms include fatigue, loss of appetite, ascites, jaundice, and hypersplenism. However, around 60% are asymptomatic.6,49 Establishing a diagnosis requires a comprehensive metabolic panel (CMP), antimitochondrial antibody (AMA) titre and/or liver biopsy. Patients meeting two of three criteria are diagnosed to have PBC: elevated serum alkaline phosphatase, positive AMA, and liver histology showing chronic, nonsuppurative, and destructive cholangitis. 47
Management
The standard treatment involves the use of ursodeoxycholic acid (UDCA) to slow the disease progression. 6 Close monitoring of haematological, cardiopulmonary, and muscular involvement is essential. 31 Tailored SLE-related immune suppressive therapy may be considered, given the lower survival rate and the presence of comorbidities in these patients. Caution is needed, as some immunosuppressants could potentially harm liver function. Therefore, agents with lower liver toxicity, such as calcineurin inhibitors, might be preferable. 31
Primary Sclerosing Cholangitis
Epidemiology and Clinical Features
PSC is an inflammatory disease of the intrahepatic and extrahepatic bile ducts, causing fibrosis and characteristic segmental dilation of the biliary tree. 32 It is commonly seen in association with inflammatory bowel disease and ulcerative colitis and is rarely described in the setting of SLE. 32 To the best of our knowledge, only four case reports describe PSC in the SLE population (Table 5). Conversely, one study reported that only 2 of 119 patients (1.7%) with PSC had SLE. 50
Case Reports of Primary Sclerosing Cholangitis (PSC) in Patients With SLE.
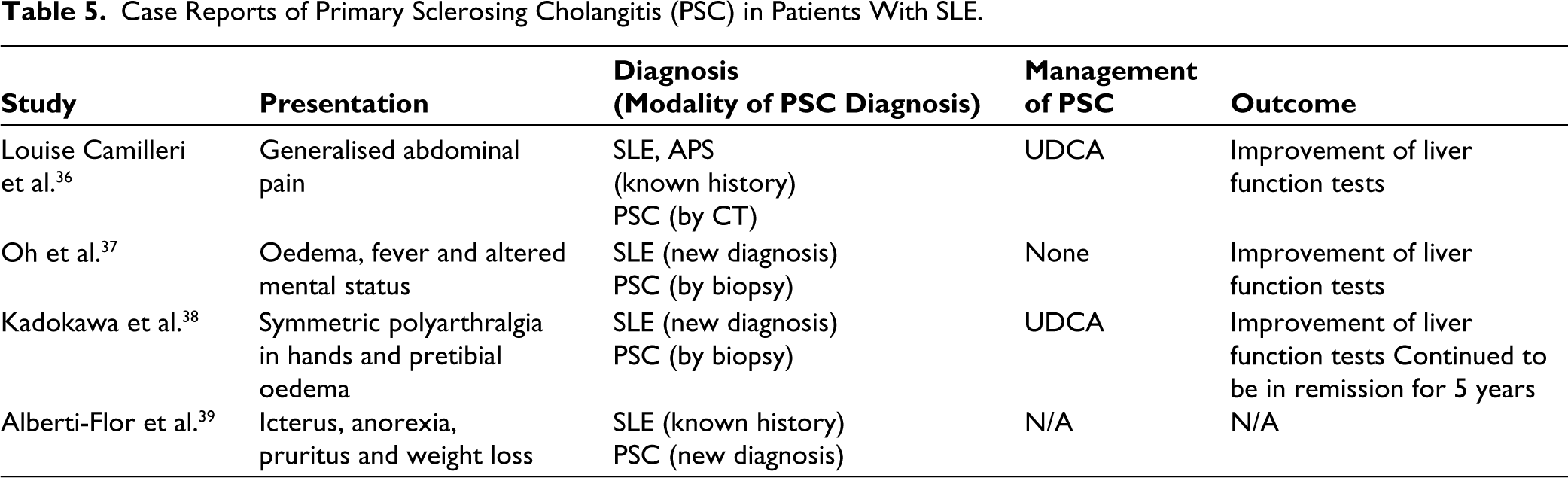
Patients who were found to have PSC had features of active lupus, such as confusion, malar rash, and polyarthritis. It is uncertain whether PSC is a result of active SLE activity due to lack of evidence in the current literature.
Pathophysiology
Biopsy of intrahepatic or extrahepatic bile ducts was once essential in diagnosing PSC and still remains the gold standard. However, with advancements in imaging modalities such as CT or Magnetic Resonance Cholangiopancreatography (MRCP), it is now more common to radiologically diagnose the disease in correlation conjunction with the clinical presentation. 51 PSC patients demonstrate characteristic multiple segmental strictures and intervening dilations (‘beading’) on biliary tree imaging. 51
It has been speculated that PSC in patients with SLE stems from T cell infiltration and circulating autoantibodies leading to arterial injury and subsequent ischaemia, as suggested by Batts and Kirby et al.52–54 However, PSC continues to be a very rare occurrence in SLE that remains to be studied further.
Diagnostic Approach
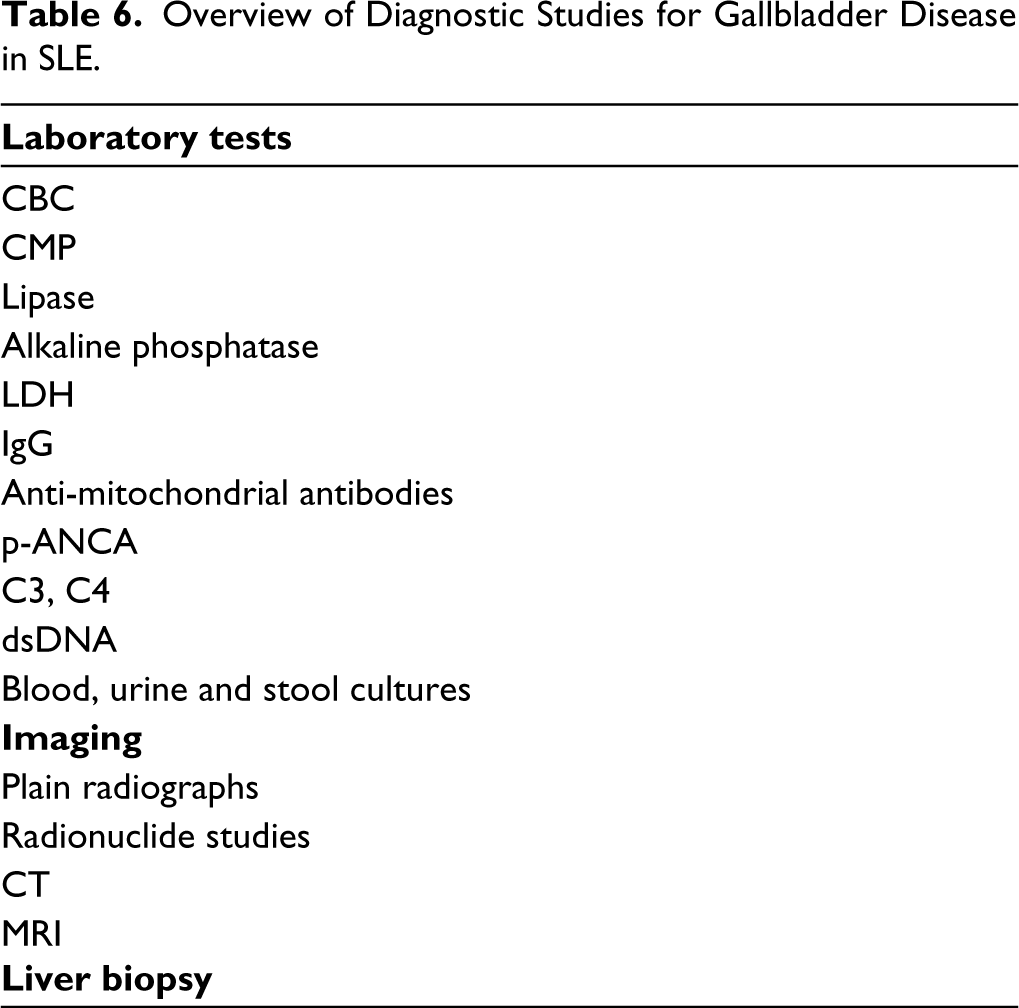
Diagnosis of PSC is difficult as half of patients can be asymptomatic. Common symptoms, when present, include fatigue, pruritis, mostly due to cholestasis. In patients with SLE, diagnosis might be incidental to laboratory work-ups during a lupus flare. Cholestatic liver function panels are commonly seen. Although liver biopsy remains the gold standard for diagnosis, abdominal CT and MRCP are now more commonly used. Characteristic multiple segmental strictures and intervening dilations (‘beading’) in combination with clinical presentation and liver function tests are often sufficient to establish the diagnosis. 55 A summary of diagnostic studies is provided in Table 6.
Overview of Diagnostic Studies for Gallbladder Disease in SLE.
Management
The standard treatment involves UDCA. This medication has been shown to improve signs and symptoms, but does not halt disease progression. 56 Very few cases of PSC associated with SLE were found to be present as part of lupus flare and subsequently resolved with immunosuppressive therapy. UDCA can also be used concomitantly. Whether UDCA use with immunosuppressive therapy results in faster resolution, is something to be studied further.
Conclusion
Gallbladder complications in SLE, although rare, are clinically significant and require a high degree of suspicion due to their vague symptoms. It is crucial to rule out infections and medication side effects during the diagnostic process. An effective diagnosis often requires a combination of laboratory tests and imaging studies. Treatment should be tailored to the specific complication and generally involves the use of immunosuppressive drugs, antibiotics or UDCA. In cases that do not respond to medical treatment, a surgical approach may be necessary.
Footnotes
Author Contributions
The review was conceptualised by M.R.S. and J.M. The initial article draft was prepared by M.R.S. and H.J. Critical revisions for important intellectual content were made by N.A.S. and J.M. J.M provided overall supervision. All authors have reviewed and approved the final manuscript version for publication. All authors agree to be accountable for all aspects of the work.
Data Availability
Data availability is not applicable, as this is a review article based on publicly available research studies and does not involve original data collection.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Ethical permission was not applicable for this article, as this is a review article drafted from various research articles and not from patients directly.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Informed consent was not applicable, as this is a review article and does not involve any direct patient data.
