Abstract
Background:
Over the decades, studies from various populations have found fibromyalgia and dysautonomia to be linked. However, recent evidence from Indian populations contradicts these findings.
Methods:
This cross-sectional study, conducted in a quaternary care centre in South India, recruited 30 women with fibromyalgia (2016 ACR criteria) and 30 age and sex-matched controls. We assessed all participants using Composite Autonomic Symptom Score (COMPASS-31) questionnaires, Sympathetic Skin Response (SSR) for sympathetic dysfunction and Heart Rate Variability battery of tests for parasympathetic dysfunction.
Results:
Median age of the women was 40 years. Fibromyalgia group reported severe levels of pain and disease impact on life. Fourteen participants (46.7%) with fibromyalgia tested positive for either sympathetic or parasympathetic dysfunction or both, while none of the control participants tested positive for either (odds ratio [OR] = 53.6, 95% confidence interval [CI] = 3–957, p = 0.0068).
Conclusion:
Dysautonomia is strongly associated with fibromyalgia syndrome. Further research is needed to determine causality.
Introduction
Fibromyalgia remains a mysterious primary chronic pain syndrome, affecting 2% of the general population globally 1 — featuring widespread musculoskeletal pain lasting more than three months, associated with disturbed sleep, poor cognition, chronic fatigue and depression. 2 Fibromyalgia has no clear etiopathogenesis but is hypothesised to be of central nervous system origin with impaired noxious inhibitory control.3,4 Experts suspect three mechanisms play key roles in pathophysiology—central Sensitisation, temporal summation (abnormal pain processing) and small fibre neuropathy.5–7
Small-fibre peripheral neuropathy (SFPN) has been implicated in about half of the patients who have fibromyalgia. 8 With the loss of the small C-type fibre innervation, these patients can present with worse pain of a burning nature with paraesthesia and allodynia.9,10 The loss of these small fibres has also been attributed to the other somatic autonomic symptoms associated with fibromyalgia – like dry eyes/mouth, erratic bowel and bladder symptoms – which severely affect the quality of the patient’s life. It is not clear if autonomic dysfunction is a causal factor or an effect of fibromyalgia. Reports from Western countries report a 45% prevalence of autonomic dysfunction in patients with fibromyalgia.11–14 The presence of these autonomic symptoms presents as a challenge for clinicians attempting symptomatic management of fibromyalgia syndrome.
However, existing published evidence—two observational studies from north Indian populations—suggests that patients with fibromyalgia are not more likely to exhibit autonomic dysfunction compared to age and sex-matched controls.15,16 The reasons for this discrepancy between western and Indian populations are unclear. In this study, we aim to determine the association of autonomic dysfunction in patient with fibromyalgia, compared to age- and sex-matched controls, recruited from a south Indian population.
Materials and Methods
We obtained institutional ethical committee clearance before prospectively recruiting participants to the current cross-sectional study. We screened patients, aged 18- to 70-year-old, attending the general medicine and rheumatology outpatient clinics of a quaternary care hospital—between December 2019 and August 2021. We excluded patients suffering from other rheumatological conditions that might lead to secondary fibromyalgia, chronic infections or other chronic non-rheumatological inflammatory conditions, other chronic pain syndromes, major depressive disorder, uncontrolled diabetes (HbA1c ≥ 9%), uncontrolled hypertension, untreated hypo or hyperthyroidism, respiratory or neurological illness, chronic kidney or liver disease, substance abuse problems in the last two years, cancer within five years, current chemotherapy eating disorder within the previous five years. We recruited consecutive, eligible participants who provided written informed consent. An internal medicine specialist (RAH) and a rheumatologist (SS) independently assessed and recruited new patients diagnosed with fibromyalgia using the 2016 revised American College of Rheumatology (ACR) criteria. 2 Similarly, we evaluated and recruited age- and sex-matched asymptomatic controls in a 1:1 ratio from bystanders of outpatient clinic patients. da Cunha Ribeiro et al. (2011) detected cardiac autonomic dysfunction in 57.1% of the fibromyalgia case group and none in the control group. 17 Assuming a 0.1% odds of autonomic dysfunction in the control group, with 80% power and 95% confidence level (CI), we calculated the minimum sample size of 12 participants in each group (total of 24) using OpenEpi. 18
For all participants, we collected data on demographic information, blood specimens for routine investigations (complete blood counts, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), liver and renal function tests) as well as administered the Composite Autonomic Symptom Score (COMPASS)-31 questionnaire and autonomic function tests. We further administered the Revised Fibromyalgia Impact Questionnaire (FIQR) and Brief Pain Inventory (BPI) questionnaire to the participants with fibromyalgia. The COMPASS-31 is a refined and abbreviated instrument to assess autonomic symptoms. 14 It measures neurodegenerative system symptoms through 31 patient-reported questions across six weighted domains: orthostatic intolerance, vasomotor, secretomotor, gastrointestinal, bladder and pupillomotor – and calculated on a weighted score of 0 to 100. The FIQR is an updated version of the original FIQ, developed to assess the impact of fibromyalgia on patients’ lives. 19 It includes three domains, function, overall impact and symptoms, with additional questions on memory, tenderness, balance and environmental sensitivity, all graded on a 0-10 numeric scale. The BPI is a self-reported questionnaire developed to rapidly assess the severity of pain and its impact on daily functioning. 20
We asked the participants to temporarily stop any α or β blocker medications (and prescribed a substitute if needed) for a period of 72 hours before testing. Then, a team of neurotechnologists, blinded to the participant’s disease status, administered the autonomic function tests under the supervision of a senior consultant neurologist who interpreted the results to discern either sympathetic or parasympathetic dysfunction. They administered the Sympathetic Skin Response (SSR) tests as per the technique previously described by Shahani et al. 21 The team maintained the participant’s skin temperature at 30°C. In the supine position, they performed the test using a Nicolet VikingQuest (Natus Medical Inc., WI, USA) electro-myogram machine (Version 21). They used a low-frequency filter setting of 0.1 Hz and a high-frequency filter setting of 100 Hz with the sweep duration set at 1s /division and the sensitivity between 100 and 500 μV/division. They chose a limb side randomly and conducted the test with two electrodes attached to the dorsoventral surfaces of the feet (for lower limb test) and the palms (for upper limb test), a third ‘earth’ electrode attached 7 cm proximally on the same limb. They then delivered an electrical stimulus (10–30 mA for 0.1–0.5 ms) on a peripheral nerve (median or common peroneal nerve) of the participant’s contralateral limb. They measured the onset latency, peak latency and amplitude of skin conductance, representing the sweat gland activity controlled by the sympathetic nervous system. They trialled the stimulus every 1–2 minutes to avoid habituation. After ten trials, the absence of a waveform in either the lower or upper limbs signifies sympathetic autonomic dysfunction, and they further confirm the waveform absence on the contralateral limb. However, researchers have judged SSR to have poor reproducibility due to habituation/fatigue of the C-type nerve fibres and poor diagnostic accuracy due to the nociceptive stimuli activating an emotional rather than thermoregulatory response. 22
The neurotechnologist team also tested the cardiac autonomic dysfunction, assessing the Heart Rate Variability (HRV) using the Expiratory-to-Inspiratory (E/I) ratio (in the supine position) and the 30:15 ratio (supine to standing position) of the R-R interval—as described in the Ewing’s battery. 23 Initially, the team first places two unipolar electrocardiogram electrodes to the chest wall and ensures a normal baseline reading. Then, the teams ask the participant to breathe deeply at a rate of six breaths/minute (five seconds of inhalation or exhalation per breath) for three cycles. The team records the maximum and minimum R-R intervals in three respiratory cycles. Then, they calculate the mean of the longest R-R interval during expiration and shortest R-R intervals during inspiration from three readings. E/I ratio is the ratio between the respective mean R-R intervals. For the 30:15 ratio, the team repeats the initial steps as before and then asks the participant to stand upright for one minute. The 30:15 ratio is the ratio between the R-R interval at the 30th beat to the 15th beat, when standing upright. The team trials the test for eight times every three minutes and the mean of these 30:15 ratios is considered for interpretation. We classified ratios below the normal for age threshold to be suggestive of parasympathetic dysfunction. 24 If a participant tested positive for either sympathetic or parasympathetic dysfunction (or both), the neurologist diagnosed the participant with autonomic dysfunction, after clinically correlating with patient symptoms and signs.
For descriptive analysis, we reported categorical data summary estimates as frequencies (percentages) and continuous data summary estimates as median (interquartile range) and compared using Fischer’s exact/Pearson’s χ 2 and Mann–Whitney U tests, respectively. We will report the odds ratio (OR), its 95% CI and the P-value. We performed all statistical analyses on SPSS Statistics Version 26 (IBM Corp, Armonk, NY, USA).
Results
For the primary analysis, we included 30 women with fibromyalgia as cases and 30 age-sex matched women as controls—a total of 60 women, who provided informed consent and completed all components of the study (Figure 1). There were no missing data. The median participant age was 40 (21) years. The median COMPASS-31 score of the cohort was 27 (31). On average, the fibromyalgia group reported high pain levels and disease severity with a median visual analogue scale (VAS) of 8 (3), a median BPI score of 800 (391) and a median FIQR score of 57.5 (48). We depict the participant characteristics, stratified by cases and controls, in Table 1.
Participant Characteristics, Stratified by Case Versus Control (N = 60).
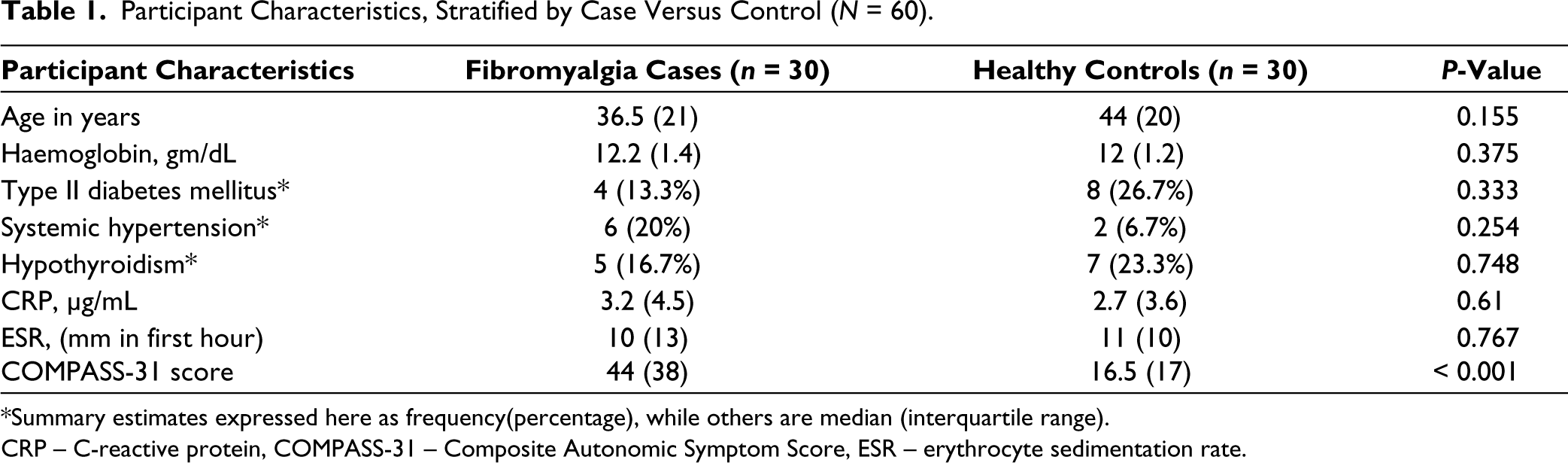
*Summary estimates expressed here as frequency(percentage), while others are median (interquartile range).
CRP – C-reactive protein, COMPASS-31 – Composite Autonomic Symptom Score, ESR – erythrocyte sedimentation rate.
Age, proportions of type II diabetes mellitus, systemic hypertension, hypothyroidism, levels of haemoglobin, CRP, ESR were similar between cases and controls. However, strong evidence that suggested that COMPASS-31 scores were higher in cases, compared to controls.
We summarised the SSR and HRV test parameters in Table 2. While there was no evidence that SSR parameters differed between cases and controls in the upper limb, there was very weak evidence that SSR onset and latency were lower in cases compared to the control in the lower limb—with six patients reporting absent waves on both sides. There was no evidence that E/I ratio of the R-R interval differed between groups, while there was strong evidence that the 30:15 ratio was on average lower in cases than controls.
Summary Estimates of Sympathetic Skin Response (SSR) and Heart Rate Variability (HRV) Tests (N = 60).
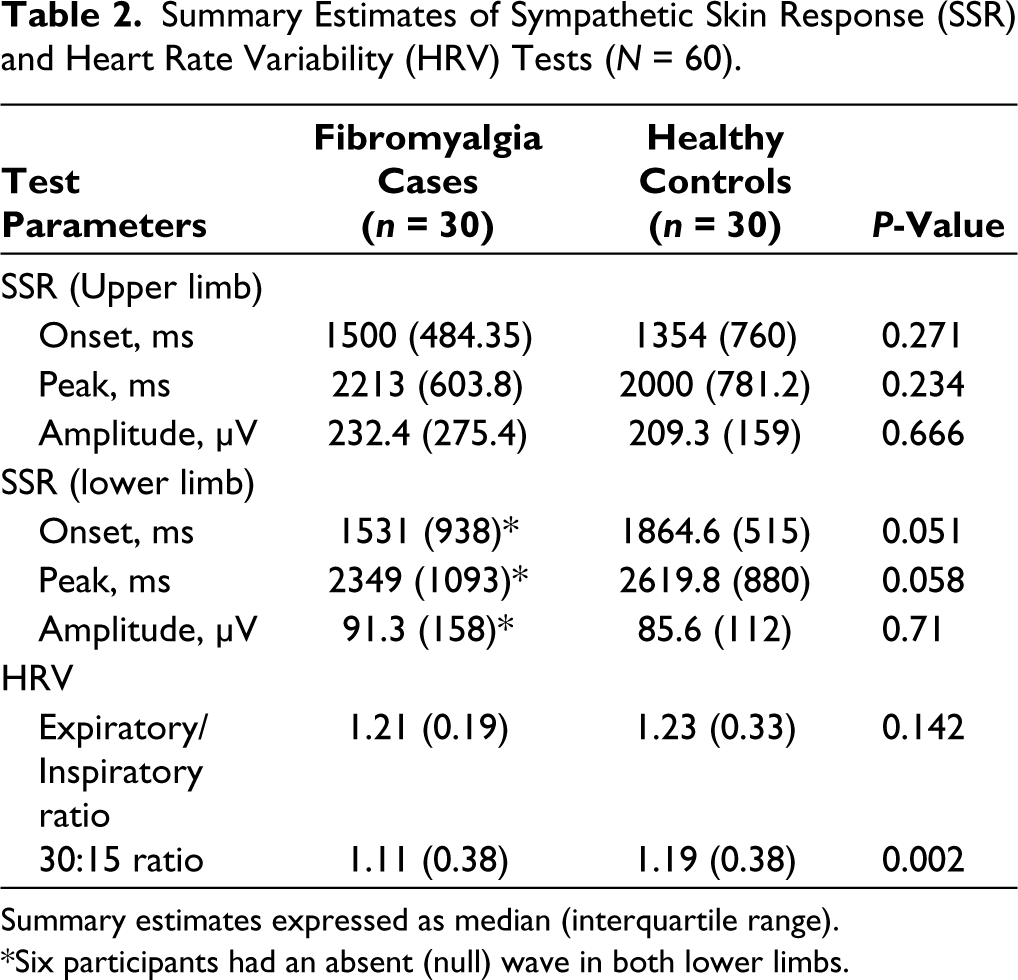
Summary estimates expressed as median (interquartile range).
*Six participants had an absent (null) wave in both lower limbs.
We have tabulated the results of the primary interpretations in Table 3. We found that autonomic dysfunction existed exclusively in cases (n = 14, 46.7%), while none of the control participants tested positive for sympathetic or parasympathetic dysfunction (OR = 53.6, 95% CI = 3–957, p = 0.0068). We detected that autonomic dysfunction strongly correlated with COMPASS-31 scores (Pearson’s r = 0.437, P < 0.001).
Primary Analysis of Autonomic Dysfunction (N = 60).
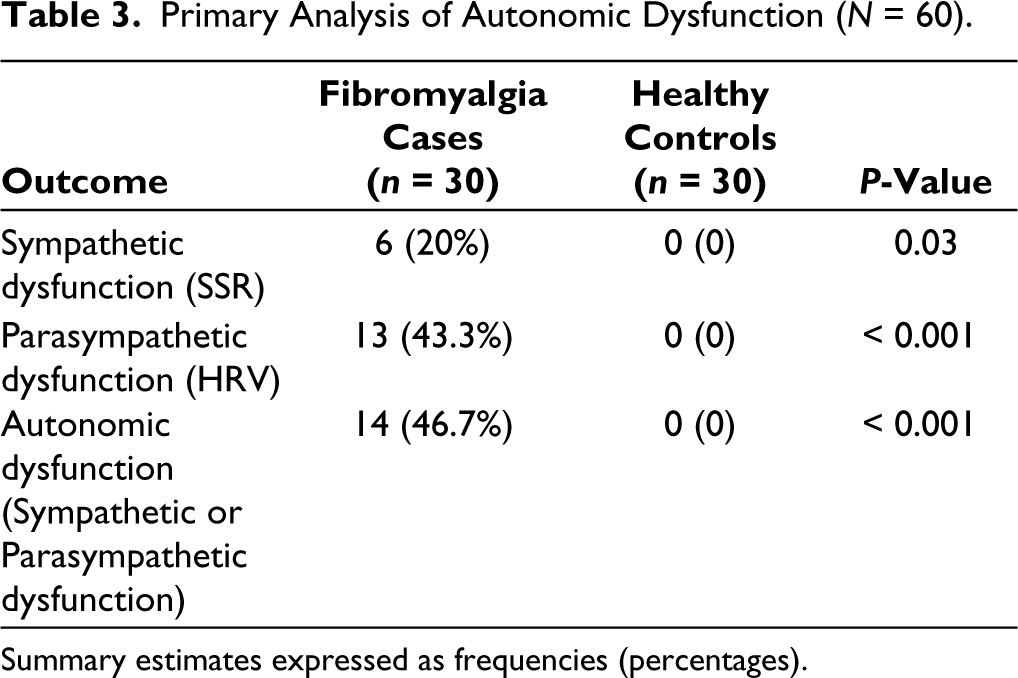
Summary estimates expressed as frequencies (percentages).
Discussion
In our cross-sectional study of 60 women (30 women with fibromyalgia), we detected autonomic dysfunction exclusively in fibromyalgia participants, while age- and sex-matched control participants seemed unaffected. Parasympathetic dysfunction appears to be the predominant form of autonomic dysfunction in our South Indian cohort of women with fibromyalgia. However, both sympathetic and parasympathetic dysfunctions can co-exist too.
The prevalence of autonomic dysfunction, exclusively in participants with fibromyalgia, is most likely a true representation of the general population with fibromyalgia. As we excluded all participants with other possible causes of autonomic dysfunction or small fibre neuropathy, it is conceivable that none of the healthy controls tested positive for autonomic dysfunction. Thus, despite a wide confidence interval, we believe that there is very strong evidence for an association between autonomic dysfunction and fibromyalgia disease. However, although most of the studies across the decades concur with our findings, there is heterogeneity between study populations.12,25–30 As per studies performed on Indian populations, Kulshreshtha et al. (2012) reported conflicting evidence, who performed a more comprehensive battery of autonomic function tests on 42 patients with fibromyalgia and age-matched controls. 16 They concluded that the sympathetic and parasympathetic functions are maintained in fibromyalgia patients. However, they also noted that both systolic and diastolic blood pressure were higher in fibromyalgia cases than controls. They attribute this elevated blood pressure and any other autonomic symptoms to be mediated by muscle deconditioning, a consequence of ‘pain-related physical inactivity’ of patients with fibromyalgia, and suspect that the autonomic reflex arc is maintained in fibromyalgia.
In another study from India, Hazra et al. (2020) (N = 100) could not find a difference in autonomic activity between fibromyalgia and control groups. 15 However, they did find variable levels of sympathetic activity within the fibromyalgia group and a blunted response to stress, possibly mediated by the central sensitisation—detected in these fibromyalgia patients. It is unclear why our results contradict both Hazra et al. and Kulshreshtha et al., whose hypotheses do not align, even though they conclude that there is no inherent autonomic dysfunction associated with fibromyalgia. One possible explanation for the disparity is that autonomic dysfunction may be related to disease severity—which may be higher in our cohort if fibromyalgia is a causative factor. Our cohort reported a higher pain at rest (VAS = 8 ± 3) than Hazra et al. (VAS = 6.3 ± 1.3), with Kulshreshtha et al. not reporting any baseline disease severity. Another cause for the disparity may also be the differing ethnicity in the South Indian population, which we sampled, as opposed to the North Indian populations studied by the other researchers.
It is unlikely that our study is reporting a false positive estimate, as similar findings have been replicated in populations across the globe, all of which suggest autonomic dysfunction and small fibre neuropathy are prevalent in about half of all patients who have fibromyalgia. 31 We also believe that our battery of autonomic function tests is validated by its correlation with COMPASS-31 scores in the same patients. We have tried to exclude possible confounders that could be biased towards a positive association, like severe anxiety or depressive disorders. Since they were newly diagnosed fibromyalgia patients, they were also not on any medication that would alter the autonomic function test results.
However, due to the nature of this study, it is not clear if the association between fibromyalgia and autonomic dysfunction is causal or even speculate about the direction of causality. However, researchers and clinicians concur that many of the symptoms or syndromes with fibromyalgia, like anxiety disorders, irritable bowel syndrome and sleep disturbances, may be a consequence of the autonomic dysfunction, facilitated by chronic hyperstimulation of β adrenergic receptors, leading to receptor desensitisation and down-regulation. 32 From a therapeutic standpoint, it is not yet clear if medications licensed for fibromyalgia are particularly effective for autonomic symptoms or small-fibre mediated pain—which may be an avenue for future research.
To determine the causality between fibromyalgia and dysautonomia, researchers may consider using large real-world time series data of patient hospital visits to track a patient’s natural history of symptoms before a formal diagnosis of fibromyalgia and following up the symptomatology after the diagnosis. It remains difficult to formally test the hypothesis as a longitudinal study due to the uncertainty of the onset of symptoms in patients. A large real-world dataset may also inform about the effect of the duration and the severity of fibromyalgia on autonomic dysfunction, which may support the argument for a causal relationship.
Strength and Limitations
The results of the present study may be robust, as we exercised a strict inclusion criterion with the data collected prospectively. However, as a single-centre study with a small sample size and only women included, the results may not be generalisable to all Indian populations. While autonomic dysfunction has been reported in men with fibromyalgia, too, 33 we were unable to recruit any men during the study period, thus incidentally recruiting only women. In a clinical setting, considering feasibility and time consumption, we could not perform a comprehensive battery of autonomic function tests. For similar reasons, our results may not be comparable directly to other studies but offer a differing insight into the status of autonomic function in Indian patients with fibromyalgia. Since SSR’s diagnostic accuracy is low, we may have underestimated the proportion of participants with sympathetic dysfunction. In contrast, the proportion of parasympathetic dysfunction is likely more accurate despite the age-based threshold for R-R interval ratios being based on non-Indian populations.
Conclusion
Fibromyalgia patients are more likely to present with autonomic dysfunction, compared to healthy age- and sex-matched controls in our Indian population. Further research is needed to confirm our results in this population, and determine causality using disease duration and severity.
Footnotes
Acknowledgements
We would like to thank Dr MGK Pillai, Professor and Head Department of General Medicine for his clinical support and Ms Renjitha, Department of Biostatistics at Amrita Institute of Medical Sciences, Kochi, India for aiding in data analysis.
Credit Author Statement
BLC, RAH and SS designed the study and collected data.
MP performed data analysis.
BLC and MP wrote the first draft of this article, while others contributed significant revisions to the final draft.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
We obtained ethical committee clearance from the Institutional Review Board of Amrita Institute of Medical Sciences, Kochi, India (IRB-AIMS-2020-227).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The first author undertook this study per the MD residency requirements at Amrita Institute of Medical Sciences, Amrita Vishwa Vidyapeetham, Kochi, India, and received monetary support for the conduct of the study from Amrita Vishwa Vidyapeetham University.
Informed Consent
Written informed consent were obtained from all participants before inclusion.
