Abstract
Background:
The economic burden of rheumatoid arthritis (RA) and ankylosing spondylitis (AS) in India is significant.
Aim:
To estimate the cost of illness (COI) and out-of-pocket expenditure of AS and RA among patients on conventional disease-modifying antirheumatic drugs.
Methodology:
A retrospective, cross-sectional study on 87 participants using a prevalence-based, bottom-up approach and the human capital method was adopted to calculate the direct and indirect costs of RA and AS.
Results:
The average COI for patients with RA was estimated to be ₹51,741 person per year, whereas the COI for patients with AS was ₹41,379 per person per year (Consumer Price Index 2021: 134.44). Direct cost was the major contributor to COI constituting 65.21% [₹33,743] and 58.84% [₹24,348] of the total COI of RA and AS, respectively. Increasing disease severity, longer duration of illness and increasing impairment of daily activities were significantly associated with increasing COI of both RA and AS. The main expense towards annual direct costs per year in patients with RA was medication expenses, whereas for patients with AS, it was investigation expenses. Out-of-pocket spending was significant as nearly half (54.95%) of the direct expenditure on health for both RA [₹18,544] and AS (39.2%) [₹9,544] was borne by patients themselves. RA and AS caused catastrophic health expenditure in 14.92% and 6.67% of households, respectively. Indirect costs of RA and AS were ₹17,998 and ₹17,030, respectively. Indirect costs of RA and AS increased as productivity at work decreased. Average overall work productivity impairment and overall activity impairment reached close to 50% and was comparable for RA and AS.
Conclusion:
The economic burden of RA and AS is significant. There is an urgent need to prioritise policies and interventions for management of rheumatological conditions.
Introduction
Musculoskeletal disorders are the leading cause of disability in the world accounting for almost half (48.06%) of all years lived with disability (YLD) owing to this disease. Lower back pain and rheumatoid arthritis (RA) alone account for 20.64% and 0.7% of YLD globally with the average disability scores of 0.11 and 0.18, respectively. 1
RA is a chronic, inflammatory arthritis with a nationwide prevalence of approximately 0.28% to 0.75%. It predominantly affects middle-aged women across the socioeconomic spectrum with prevalence in rural areas matching that of urban and peri-urban areas. Patients with RA commonly present with joint pain and systemic symptoms and later with joint deformity.2,3
The prevalence of HLA-B27 has been estimated to be approximately 6% in the Indian population, and thus ankylosing spondylitis (AS) is another common chronic inflammatory rheumatological disorder that affects nearly 7 to 9 persons per 10,000 population. 4 Most patients are young males aged 20 to 40 years presenting with lower back pain, joint stiffness and extra-articular manifestations.
The costs of RA and AS are not well documented from the Indian perspective. The debilitating pain, musculoskeletal deformities and systemic symptoms associated with these rheumatological conditions lead to a considerable limitation of physical functioning, impaired quality of life, substantial consumption of health resources and decreased economic productivity. More significantly, most of these patients belong to the economically productive age group which has important ramifications for the patient’s family, the healthcare system and society from the economic standpoint. Traditionally, non-steroidal anti-inflammatory drugs (NSAIDs) and disease-modifying antirheumatic drugs (DMARDs) have formed the mainstay of treatment of AS and RA. However, in recent years, the use of the more efficacious but more expensive biologics such as TNF-α inhibitors, IL-1 inhibitors and IL-6 inhibitors has increased. 5 Treatment of RA and AS also involves significant expenditure on physical therapy and rehabilitation. The measurement of loss of work productivity and impaired daily functioning is an important measure of economic burden of these chronic rheumatological conditions on vulnerable groups such as the urban poor and the rural population as manual labour is a significant contributor to income of this population.
Out-of-pocket-expenditure (OOPE) is the cost that is borne directly by the patient where public or private health insurance does not cover the healthcare expenditure. At least 30% of the population in India is devoid of any financial protection for health. 6 Overburdened public hospitals divert individuals to seek treatment in the costlier private sector. This along with the relatively low health insurance coverage leads to a high OOPE. High OOPE is associated with catastrophic health expenditure (CHE) and reduced access to health services, increased morbidity and reduced utilisation of essential healthcare services, leading to long-term impoverishment. 7
The COVID-19 pandemic has severely impacted the provision of healthcare services and their accessibility to the general public. Outpatient, inpatient and pharmacy services were greatly restricted in several government hospitals including ours. As a result, in the last 2 years, many patients may have been forced to avail private healthcare services. Estimating the costs of illness (COI) under these circumstances, with special focus on a vulnerable group, that is, people living with a chronic health condition, may also serve as an indirect measure of the economic impact of the COVID-19.
Few studies have been conducted in India to estimate the COI of RA in India.8–10 A study conducted in 2013 in a tertiary care hospital in 2013 found that the average COI of RA was estimated as ₹27,600 per year (Consumer Price Index [CPI] 2013: 88.81) which when adjusted for inflation translates to ₹54,519 per patient per year (CPI 2022: 144.35). 11 The direct cost for the current treatment of RA was found to be ₹23,148 per year (₹47,170 per patient per year in 2022, adjusted for inflation), whereas the indirect cost was estimated to be ₹3,600 per year (₹9,397 per patient per year in 2022, adjusted for inflation). However, while calculating the indirect cost, only wage losses caused by absenteeism and premature retirement were taken into account. Presenteeism, that is, percentage impairment while working because of the illness was not considered, and thus the indirect cost may actually be higher. A 2019 study 10 compared the COI of RA in 2 tertiary care centres in South India. The monthly direct cost was estimated at ₹14,577 per month (₹18,241 per month in 2022, adjusted for inflation) and ₹8600 per month (₹10,761 per month in 2022, adjusted for inflation), whereas the monthly indirect cost was estimated at ₹1397 per month (₹1,748 per month in 2022, adjusted for inflation) and ₹398 per month (₹498 per month in 2022, adjusted for inflation). A study 9 conducted in 2003 in a government teaching hospital in North India found that the average total cost of drug treatment was ₹999 ± ₹76 per month (₹3,562 per month in 2022). The average monthly direct cost of RA was estimated to be ₹623 ± ₹31 per month (2,222 per month in 2022). The average indirect cost was found to be ₹368 ± ₹62 per month (1,312 per month in 2022). To the best of our knowledge, no studies have been conducted to estimate the COI of AS in India. In Asia, studies on cost estimates of AS have been conducted in Hong Kong, 12 South Korea,13 Singapore 14 and China. 15 Many Indian patients with AS are deprived of the benefits of biologic agents because of their high costs. 16 Thus, studies that included the cost of biologic and biosimilar-based therapy in patients with AS may not be applicable to the Indian context. Except for the South Korean study, most of these studies considered the cost of biologics while estimating COI. Since the healthcare models of the United States 17 (healthcare funded chiefly by private insurance) and the United Kingdom 18 (healthcare funded fully by the state) are very different from the Indian model (a hybrid public-private model), studies conducted in those settings may not be applicable to Indian patients. Studies have shown that COI of RA is higher than that for AS.19,20 Indirect costs contribute significantly to the COI of AS and RA.19,20 The current study will reflect the societal cost of RA and AS in terms of direct costs, OOPE, catastrophic spending on healthcare, indirect costs and work productivity losses using the human capital method. This study can serve as a benchmark for other countries with similar healthcare models and may also allow us to compare how costs vary across different rheumatological conditions. By identifying the incidence and trends affecting CHE, COI studies will guide decision-makers in appropriate resource allocation for tackling rheumatological illnesses such as RA and AS.
In this study, we aim to estimate the COI and OOPE of AS and RA. By measuring the economic burden of AS and RA, we aim to highlight the urgent need to prioritise policies and interventions for management of rheumatological conditions.
Materials and Methods
A retrospective, cross-sectional study was conducted using data collected over a 2-month period from June to July 2022 from patients attending the weekly rheumatology OPD at a tertiary care centre in North India. The study was conducted in accordance with the ethical standards of the institutional ethics committee and with the 1964 Helsinki Declaration and its later amendments. A prevalence-based, bottom-up approach was used to calculate the direct and indirect costs associated with RA and AS. A total of 82 patients, 67 patients each with RA and 15 patients each with AS were consecutively enrolled for the study after taking informed consent.
All patients included in the analyses were at least 18 years old, had a clinical diagnosis of RA or AS for at least 1 year and fulfilled the American College of Rheumatology/European League Against Rheumatism 2010 classification criteria and the 1984 modified New York criteria for RA and AS, respectively. An expert’s discretion was used to overlook taking into account the expenses incurred directly because of comorbid illness to avoid the overestimation of costs. Only patients receiving conventional DMARDs were included in the study. Patients who were receiving biologics or had received them in the last 1 year were excluded.
Patients were interviewed during a single visit to the outpatient department in the study period. Clinicodemographic data and information on the direct cost, OOPE and the indirect cost of healthcare incurred over the last 1 year were collected using a bilingual, validated questionnaire (see the supplementary material available online).
Information on sociodemographic characteristics including age, gender, education level, average monthly personal income, average monthly household income and average monthly household expenditure was collected. Clinical characteristics such as disease duration, Clinical Disease Activity Index (CDAI)21,22 score and Bath Ankylosing Spondylitis Disease Activity Index score (BASDAI) 23 for RA were recorded. Information regarding medications prescribed was also collected from OPD records.
Total direct medical costs were calculated by adding up the costs of consultation fees, inpatient hospitalisation, medication including over-the-counter and traditional medicine, investigations, physiotherapy and rehabilitative therapy availed over the past 6 months. Total direct non-medical cost was calculated as the summation of expenditure on travelling, food, lodging, assistive devices, home assistance, religious rituals and relocation expenses. Patients’ self-reported fees were recorded when private facilities were used. Cost of public health services was derived by review of medical records.
Total indirect costs were calculated using the human capital method (HCM). The Work Productivity and Activity Impairment questionnaire in Specific Health Problem (WPAI:SHP V 2.0) 24 was used to assess the impairment of work productivity and activity. WPAI:SHP V 2.0 is a self-administered questionnaire for assessing the impact of disease on productivity during the previous 7 days. Four scores were derived including the percentage of absenteeism (percentage work time missed because of the problem), the percentage of presenteeism (percentage impairment while working because of the problem), an overall work impairment score (percentage overall work impairment because of the problem) and the percentage of impairment in activities performed outside of work. Higher scores indicated poorer work productivity and greater activity impairment because of the health problem. Loss of productivity caused by absenteeism was also calculated for the caregiver. For calculating work loss, the average income range was taken into account. As a significant proportion of our subjects were not gainfully used, such as homemakers suffering from RA and students suffering from AS, for such patients, the latest minimum wage rates for their highest qualification level (for students) and for unskilled labour (for homemakers) as per the Government of National Capital Territory of Delhi were used. 25 Wage loss because of the change of employment/loss of work was also accounted for.
The out-of-pocket amount spent on OPD care over the last 3 months and hospitalisation expenditure over the last 1 year was converted to monthly OOPE by dividing OPD care OOPE by a factor of 3 and by dividing hospitalisation OOPE by 12. OOPE was calculated as
OOPE (%) = (Household’s Monthly Health Care Expenditure/Household’s Monthly Consumption Expenditure) × 100.
Any reimbursement in the form of pension/health insurance schemes was deducted from total personal expenditure on healthcare.
CHE was defined as OOPE equalling or exceeding 10% of household expenditure. 26 A household was said to be ‘distress financing’ if it financed healthcare by borrowing and selling assets.
All costs were expressed as annual expenditure in the INR rounded off to the nearest rupee. Costs were adjusted for inflation for comparison with previous studies.
A standard method was used for the adaptation of the original English version of all questionnaires into Hindi language. A bilingual translator, adept at both Hindi and English and whose mother tongue was Hindi, performed an independent forward translation. An expert reviewed the translated questionnaire. Two back translations of the Hindi version were performed for comparison with the original questionnaire. The questionnaire was initially administered to 5 patients each with RA and AS to identify any difficulty experienced while filling out the questionnaire as a part of pilot testing. Data collected from patients in the pilot run were not included in the analysis.
Statistical Analysis
Univariate statistics for demographic and clinical variables were generated for descriptive analysis. Quantitative data were summarised by providing the number of observations, mean and standard deviation (SD). Frequency and percentage were used for summarising categorical variables. Unpaired t-test was used to evaluate the difference of COI between AS and RA. Regression analysis including univariate, multiple and multivariate linear regressions was used to test the effect of different factors on total expenditure, OOPE and catastrophic spending. Results were reported as regression coefficients with 95% confidence interval (CI). Stata version 15.0 was used to perform statistical analyses.
Results
In total, 82 patients (67 with RA and 15 with AS) were consecutively enrolled in this study over a 2-month period (June-July 2022). RA cases were predominantly female (89.5%, n = 60), with a mean age of 40.53 years, (SD = 13.48, range = 18–67). AS cases were predominantly male (73.3%, n = 11), with a mean age of 26.4 years, (SD = 5.76, range = 18–39) (Table 1).
Baseline Sociodemographic and Clinical Characteristics of RA and AS.
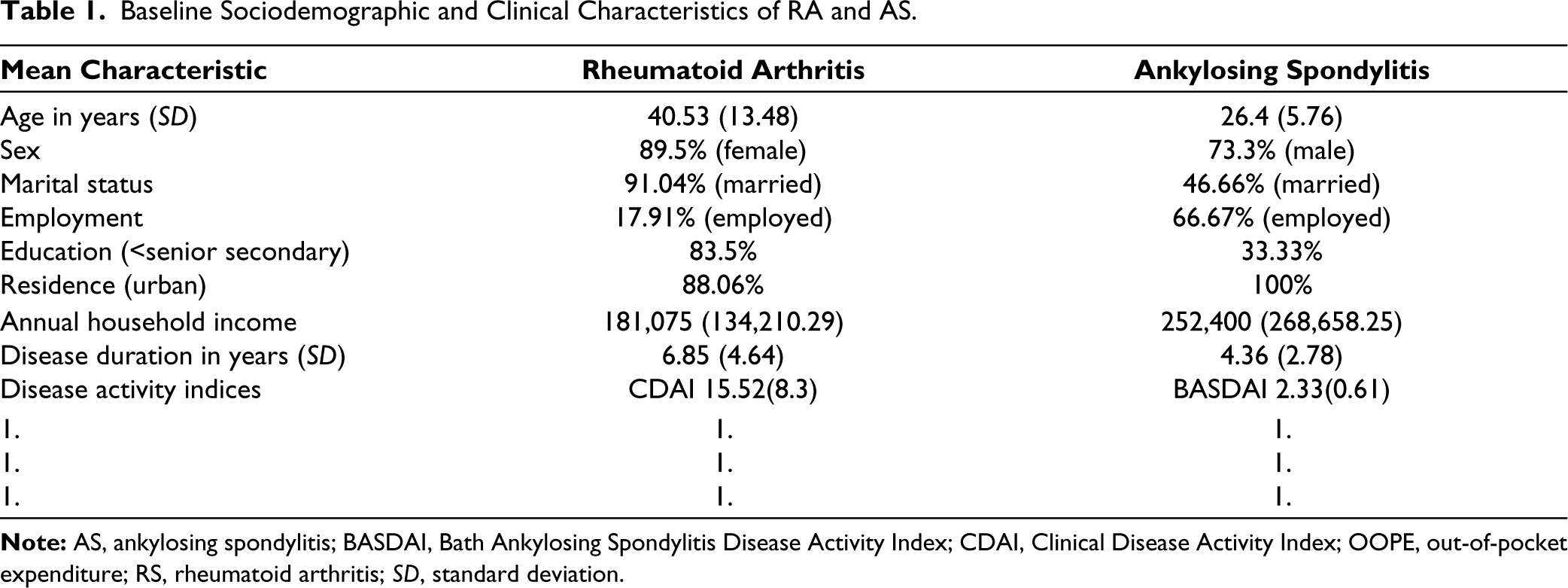
The mean disease duration (SD) of patients with RA and AS was 6.85 (SD = 4.64) years and 4.36 (SD = 2.78) years, respectively. Among RA patients, methotrexate was the most frequently prescribed DMARD (100%), followed by sulfasalazine (77.6%, n = 52) and hydroxychloroquine (73.13%, n = 50). Adjuvant drugs included: folic acid (100%), calcium (100%), NSAID (100%), pantoprazole (98.5%) (n = 66) and vitamin D (92.53%) (n = 62). Steroids were prescribed to 40.29% patients (n = 27). Biologic agents were not prescribed owing to unavailability at our centre. A total 53.74% (n = 36) patients were on DMARDs combined with 3 drugs (MTX + HCQ + SAAZ), 23.8% (n = 16) on MTX + SAAZ and 20.8% (n = 14) on MTX + HCQ.
Among patients with AS, NSAIDs (naproxen/ibuprofen) were the most frequently prescribed drug (100%). Adjuvant drugs included: pantoprazole (93.33%), calcium (80%) and sulfasalazine (42.85%). Biologic agents were not prescribed because of the unavailability of the same at our centre.
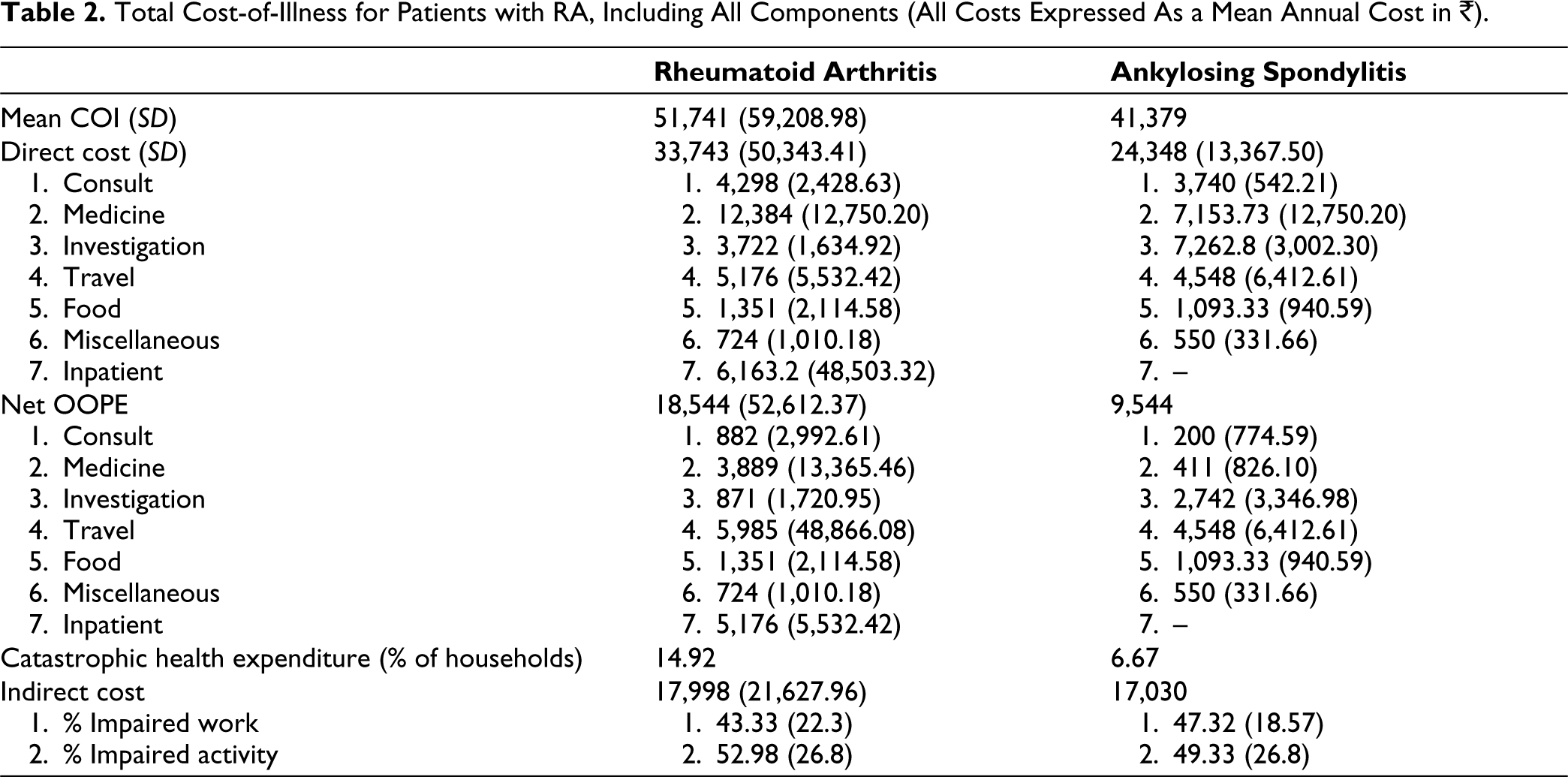
For patients with RA, the average annual household income was ₹181,075, whereas the average total COI was estimated to be ₹51,741 per person per year. Of this, direct costs amounted to ₹33,743 (65.21%), whereas indirect costs amounted to ₹17,998 (34.78%). For patients with AS, the average annual household income was ₹252,400, whereas the average total COI was estimated to be ₹41,379 per person per year. Of the total, direct costs amounted to ₹24,348 (58.84%), whereas indirect costs amounted to ₹17,030 (41.16%) (Table 2).
For patients with RA and AS, the components of direct health expenditure (Table 2) have been depicted in Table 2.
The net OOPE for RA was ₹18,544 constituting 54.95% of the direct cost. The net OOPE for AS was ₹9,544 constituting 39.2% of the direct cost (Table 2). Component-wise breakdown of OOPE has been depicted in Table 2.
Total Cost-of-Illness for Patients with RA, Including All Components (All Costs Expressed As a Mean Annual Cost in ₹).
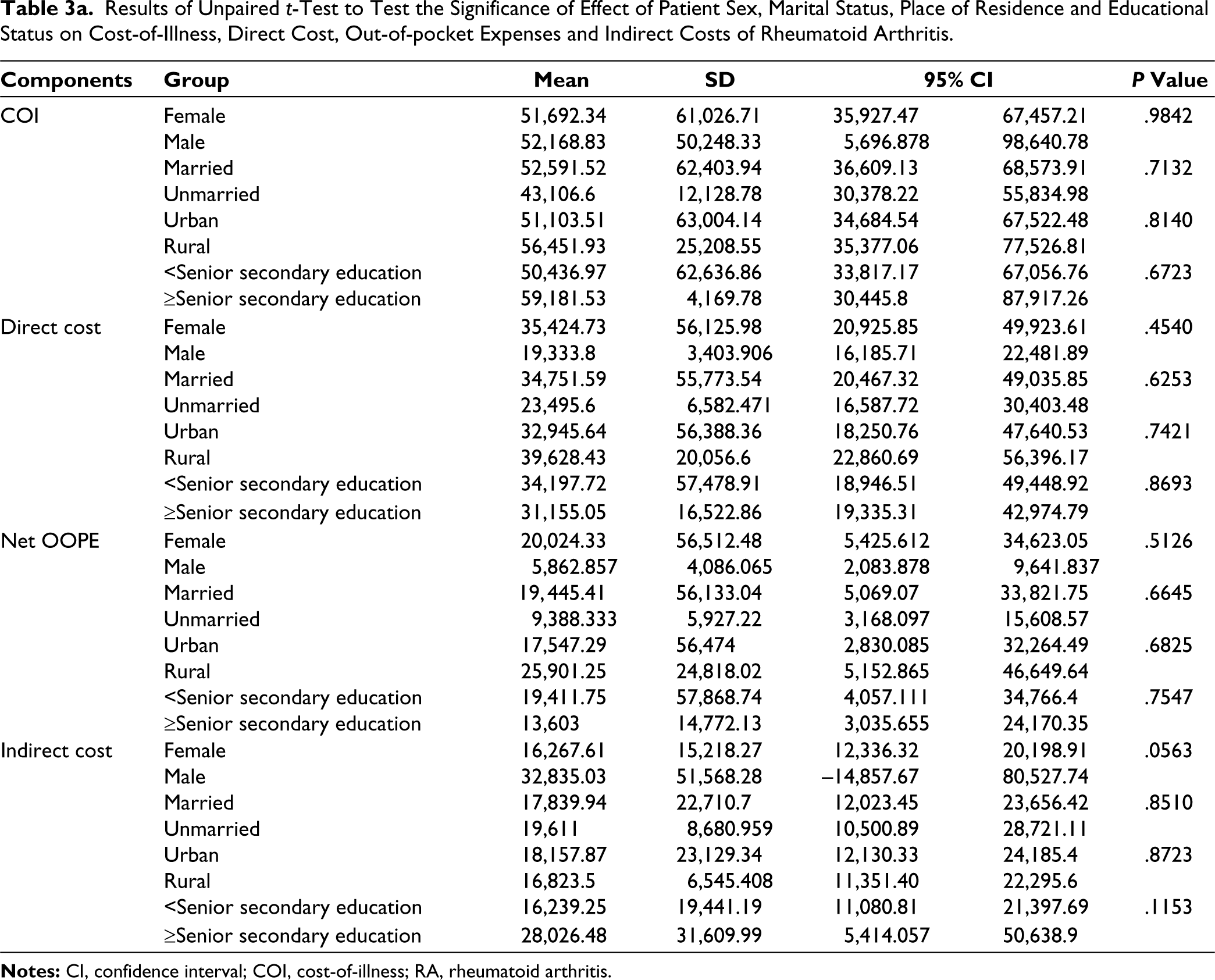
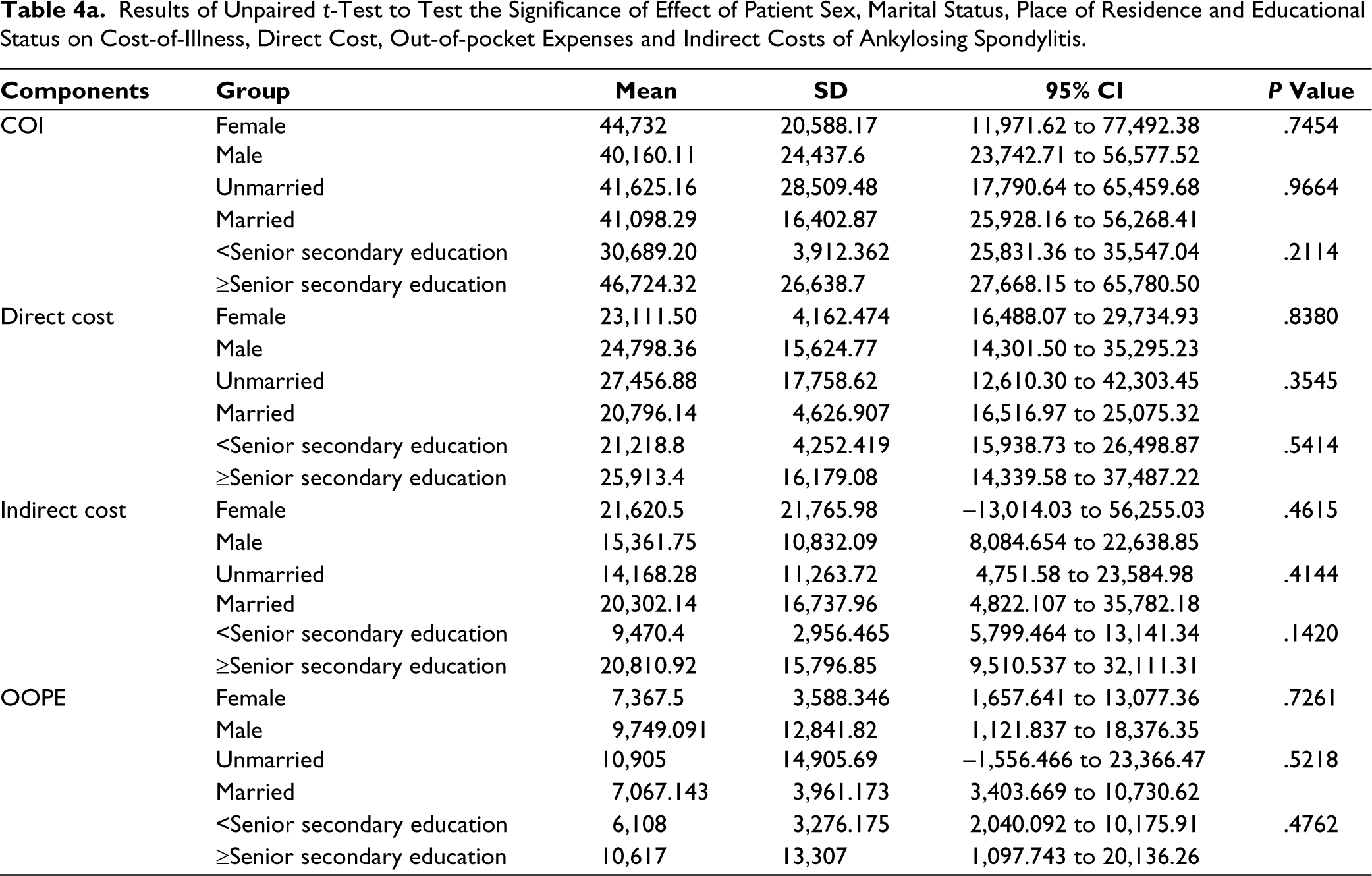
The results of the unpaired t-test revealed that sex, marital status and educational status did not have a statistically significant association with COI, direct cost, OOPE or indirect cost in the cases of both RA (Table 3a) and AS (Table 4a).
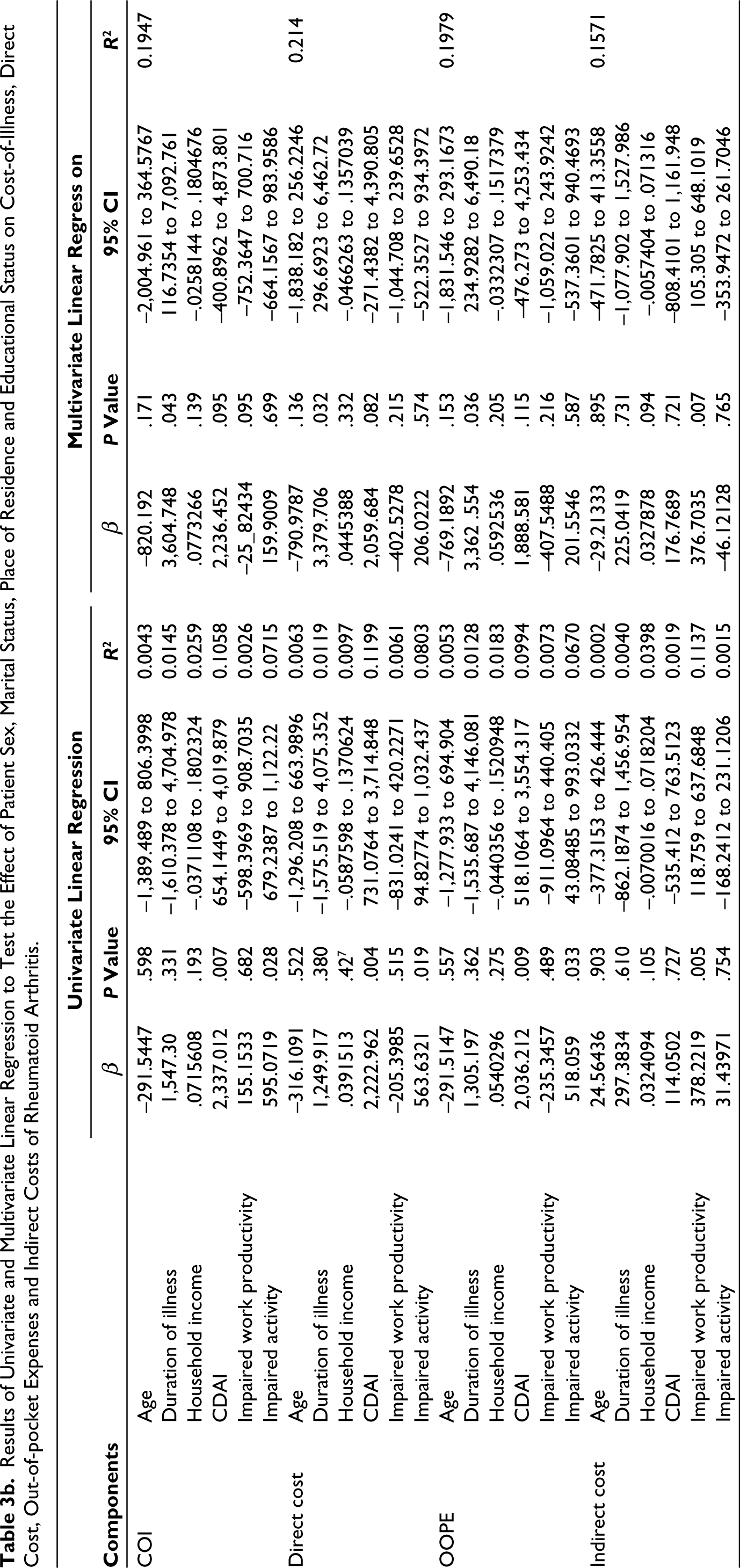
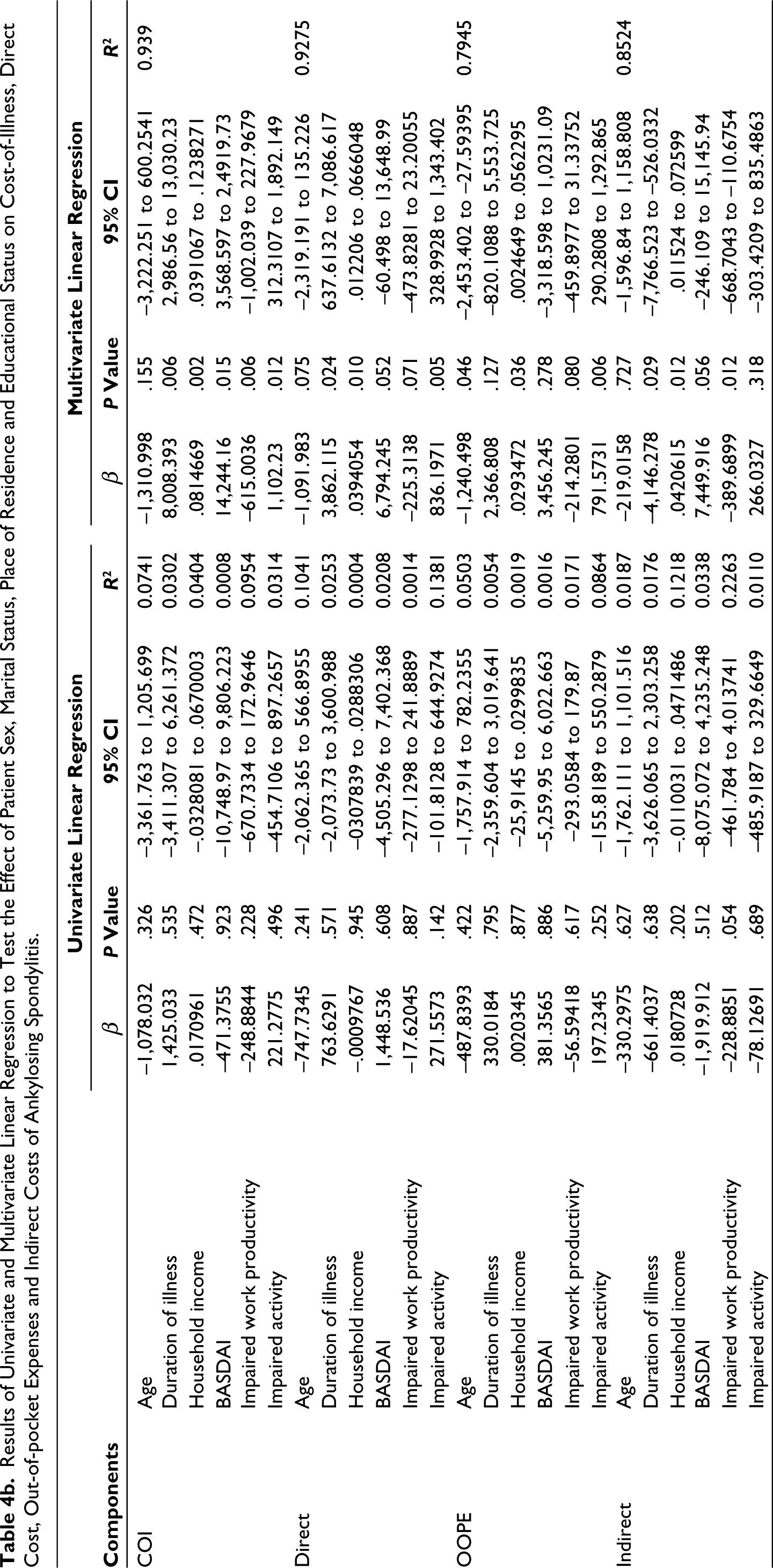
Among patients with RA, the results of univariate regression revealed that the disease activity (CDAI) and activity impairment were significant determinants of the total COI (R2,= 0.1058, β = 2,337.012, P = .007; R2 = 0.0715, β = 595.0719, P = .028), direct costs (R2 = 0.1199, β = 222.962, P = .004; R2 = 0.0803, β = 565.6321, P = .019) and OOPE (R2 = 0.0994, β = 2,036.212, P = .009; R2 = 0.0670, β = 518.059, P = .033). Higher disease severity and more impaired activity were associated with higher costs. Multiple and multivariate regression analysis showed that the duration of illness was a significant predictor of COI (R2 = 0.1940, β = 3604.748, P = .043), direct cost (R2 = 0.2114, β = 3379.706, P = .032) and OOPE (R2 = 0.1979, β = 3362.554, P = .036). As the duration of illness increased, associated costs increased too (Tables 3a and 3b).
Results of Unpaired t-Test to Test the Significance of Effect of Patient Sex, Marital Status, Place of Residence and Educational Status on Cost-of-Illness, Direct Cost, Out-of-pocket Expenses and Indirect Costs of Rheumatoid Arthritis.
Results of Univariate and Multivariate Linear Regression to Test the Effect of Patient Sex, Marital Status, Place of Residence and Educational Status on Cost-of-Illness, Direct Cost, Out-of-pocket Expenses and Indirect Costs of Rheumatoid Arthritis.
Among patients with AS, on multivariate regression analysis, it was observed that the total COI was significantly affected by the duration of illness (R2 = 0.939, β = 8,008.393, P = .006), household income (R2 = 0.939, β = 0.814, P = .002), disease activity (BASDAI score) (R2 = 0.939, β = 142b44.16, P = .015), impaired work productivity (R2 = 0.939, β = –615.0036, P = .006) and activity reduction (R2 = 0.939, β = 1,102.23, P = .012). The direct cost was significantly affected by the duration of illness (R2 = 0.9275, β = 3,862.115, P = .024), household income (R2 = 0.9275, β = 0.039, P = .01) and activity reduction (R2 = 0.9275, β = 836.1971, P = .005). Net OOPE was significantly affected by age (R2 = 0.7945, β = 2,337.012, P = .046), household income (R2 = 0.7945, β = 0.029, P = .036) and activity reduction (R2 = 0.7945, β = 791.5731, P = .006). The indirect cost was significantly affected by the household income (R2 = 0.8524, β = 0.04, P = .012), duration of illness (R2 = 0.8524, β = –4,146.278, P = .029) and impaired work productivity (R2 = 0.8524, β = –389.6899, P = .012). Increase in income, the duration of illness and disease activity resulted in an increase in respective costs, whereas older age and more impaired work productivity were associated with a decrease in respective costs (Tables 4a and 4b).
Results of Unpaired t-Test to Test the Significance of Effect of Patient Sex, Marital Status, Place of Residence and Educational Status on Cost-of-Illness, Direct Cost, Out-of-pocket Expenses and Indirect Costs of Ankylosing Spondylitis.
Results of Univariate and Multivariate Linear Regression to Test the Effect of Patient Sex, Marital Status, Place of Residence and Educational Status on Cost-of-Illness, Direct Cost, Out-of-pocket Expenses and Indirect Costs of Ankylosing Spondylitis.
RA caused catastrophic expenditure in 14.92% of households (n = 10), whereas AS resulted in catastrophic spending in 1 household (6.67%, n = 1). Results of univariate regression showed that CHE increased with worsening disease severity of RA (CDAI) (P = .016, R2 = 0.0857). As impairment of daily activity increased, the likelihood of the occurrence of CHE increased for both RA and AS (RA: P = .034, R² = 0.0673; AS: P = .017, R2 = 0.5782).
In order to meet the high cost of healthcare, patients resorted to ‘distress financing’ in the form of borrowings and sales of physical assets, such as land and jewellery. Among the households with CHE, 4 patients with RA were forced to take loans. In all 4 cases, the loans were taken from relatives and friends with the loan amount ranging between ₹20,000 and ₹300,000 with interest rates varying from 5% to 15% p.a. One of these patients was also forced to sell their ancestral property, whereas another patient had to pawn exchange jewellery for cash. Distress financing was a source of psychological stress for the family. One patient relocated to Delhi from her native state of Madhya Pradesh so that they had improved access to healthcare. All 100% subjects reported that healthcare costs were not covered by any health insurance scheme.
RA and AS severely limit an individual’s physical functioning, and thus their treatment also entails expenditure on physical therapy and rehabilitation. All (100%) patients reported that they used equipment such as heating pads, splints, crepe bandage, walking stick and wheelchair. Nearly 80.4% (n = 70) had purchased these items on their own. At least 3 patients with RA had switched to the Western toilet seat (costing them up to ₹5000) because it was difficult for them to squat on the Indian toilet seat owing to knee pain. Two patients with AS who were also students had to invest in a study table (costing up to ₹1200) on account of back pain and stiffness. Only 14.6 % (n = 12) patients had ever undergone a physiotherapy session.
Among patients with RA, 82% (n = 55) of the patients were unemployed. Average overall work impairment owing to RA among those gainfully employed was 43.33% (SD 22.3%), whereas the overall activity impairment of daily activities was 52.98% (26.8%) as measured by the overall work impairment score. Two patients were forced to quit their jobs because of their illness, whereas 2 patients reported a loss of income because of the change of occupation. Another patient had to hire a helper to assist him in his work a plumber. In contrast, 1 patient who was also a college student was forced to take up a job at a tuition centre to meet the rising cost of healthcare. About 66.6% (n = 8) of those employed reported that they had been forced to take sick leave within the last 1 year on account of their illness. The average duration of sick leave was 2.3 days (SD = 2.7). The indirect cost of RA was significantly affected by the impairment of work productivity at the 5% significance level (P = .006).
Among patients with AS, 33.33% (n = 5) of the patients were unemployed. Average overall work impairment because of AS among those gainfully employed was 47.32% (18.57%), whereas the overall activity impairment of daily activities was 49.33% (26.8%). One patient was forced to quit his job, whereas another patient had to change his occupation. A patient was forced to take up a job to meet healthcare expenses. About 80% (n = 8) of those employed reported that they had been forced to take sick leave within the last 1 year on account of their illness. The average duration of sick leave was 4.2 days (SD = 3.29).
Students (n = 5) reported that their illness had affected their academic performance and also impacted their peer group interactions. The negative psychological impact of RA and AS on the patient’s mood and psyche was also noted during interactions with subjects with several respondents expressing their dismay at the lack of a permanent cure for their health conditions. Patients used different strategies to cope with these negative emotions. At least 4 subjects reported that they had participated in religious rituals specifically for their health conditions over the last 1 year, spending an average of ₹4,500 per year. It was also noted that none of the 82 subjects sought help from a mental health professional despite 92.7% (n = 76) of them affirming that their illness was a source of psychological stress for them.
The COVID-19 pandemic resulted in the disruption of care and follow-up patients with rheumatological diseases. As the centre where the current study was performed was a COVID-19 exclusive centre from April 2020 to March 2022, all enrolled subjects were lost to follow-up during this time period. Out of the 58 patients (RA: n = 50; AS: n = 8) whose prior health records were available, 60% of patients with RA (n = 30) and 37.5% of patients with AS (n = 3) progressed to a higher disease activity category on comparing pre-pandemic and post-pandemic disease activity indices.
Non-adherence to medication (including partial adherence) during the pandemic was reported by 88% patients with RA (n = 59) and 53.33% patients with AS (n = 8). Almost all patients experienced limitation in access to healthcare services during the pandemic. Out of 82 subjects, 87.8% (15 patients with AS and 57 patients with RA [n = 72]) switched to private healthcare providers at least once, whereas 7.3% (all patients with RA [n = 6]) sought care from practitioners of alternative medicine. Among patients with RA, 4.8% (n = 4) took only a portion of the prescribed dose, whereas 1 patient (RA) stopped her prescription because of her inability to afford the medications.
Discussion
The average COI for patients with RA was estimated to be ₹51,741.12 per person per year, while that for patients with AS was ₹41,379.28 per person per year (2022). These results, when adjusted for inflation, are similar to a study on COI analysis among patients with RA in a tertiary care hospital in South India in 2013. 8 Direct costs constituted 65.21% and 58.84% of the total COI for RA and AS, respectively. This is similar to other studies on COI of RA,27–29 with indirect costs making up 22% to 37% of the total cost of RA. In comparison to AS, a lower contribution of indirect cost to patients with RA may be because a greater proportion of patients with RA were not gainfully employed. Use of minimum wage criteria for such subjects might have led to a more conservative estimate of the indirect cost.
In our study, the contribution of the indirect cost (nearly 42%) to patients with AS is nearly 2 to 4 times of the estimates reported by similar studies in New Zealand30 (22%) and Singapore14 (10%), whereas the absolute indirect cost is a fraction of the values reported by these studies. This may suggest that a large proportion of the direct cost is paid for the government in our setting, which increases the percentage contribution of the indirect cost to COI. Additionally, the lower share of the direct cost to COI in this study can be explained by the difference in prescribing patterns at our centre (biologics not prescribed) versus aforementioned studies (biologics were prescribed). Several studies31,32 have shown that average total costs were higher for patients receiving biologic therapy compared to those receiving non–biologic-based therapy.
The main expense for the mean annual direct costs per year in patients with RA was medications consistent with other studies. 27 This may be a reflection of the prescription practices in RA. The prevalence of use of combination DMARD therapy was similar to results from other studies.33,34 MTX monotherapy is the mainstay of treatment for patients with DMARD-naïve RA. Combination of DMARDs is used when disease activity remains high despite adequate monotherapy. 35 The higher medication cost might also be caused by the higher severity of disease in our setting, especially as many of our patients were lost to follow up during the pandemic.
In patients with AS, expenditure on investigations was the largest individual contributor to the direct cost closely followed by medication expenses in contrast to other recent studies.14,30 MRI has a central role in the diagnosis and management of patients with AS. 36 In most government hospitals, the timely availability of this modality is a problem because of high patient loads and massive backlog of patients. This forces patients to pay higher costs of MRI in private centres, which may contribute to high expenditure on investigations. The use of non–biologic-based treatment in our hospital may have led to decreased expenditure on medication. Notably, the results of our study are similar to a 2008 study from Hong Kong, 12 in which no participant had used biologic drugs during the study period. It was seen that the cost of technical examinations represented the largest proportion of total costs.
OOPE constituted nearly 50% of the direct expenditure on health for both RA and AS. This is in line with the national estimate of 48.2% of the OOPE as the percentage of total health expenditure as per the National Health Accounts (NHA) Estimates for India for 2018 to 2019. 37
RA caused catastrophic expenditure in 14.92% of households, whereas AS resulted in catastrophic spending in 6.67%. These results are slightly lower than the national average of approximately 17% as per a 2022 WHO report. 38 Lower CHE may be because this study was performed in a tertiary-level hospital in a metropolitan city with better quality, availability and accessibility of public health infrastructure than primary- and secondary-level hospitals in rural and semi-urban areas. However, an OOPE that is nearly equal to and a CHE that is slightly less than the national estimates suggest that the average income of subjects enrolled in this study is higher than the national average. As a result, a subject is able to spend nearly the same amount for healthcare from their own pocket with a lesser likelihood of this expense being catastrophic for the household than the average Indian.
The net OOPE and the net CHE for were higher for the patients with RA. Studies have shown that the lower household income,39,40 higher hospitalisation rates and chronicity of illness 41 are the major drivers of catastrophic and out-of-pocket health expenditure. In our study, a patient with RA was more likely to fulfil these conditions, and this may explain the difference in OOPE and CHE between the RA and AS.
In patients with RA, mean inpatient expenses had the single largest contribution to OOPE, which is similar to findings of other studies in which a major share of total costs was attributed to inpatient services.27,42,43 The pandemic resulted in the disruption of non-emergency healthcare and follow-up of these patients in our hospital. Lack of access to affordable care, poor availability and adherence to medication, and lack of timely medical supervision and intervention may have led to worsening of the disease for which the patients were forced to seek care from private healthcare providers. In study 44 conducted in 2017, the pharmacy cost was the largest contributor to OOPE. In our setting, medicines were an important component of the OOPE in RA, accounting for up to 20% of OOPE. In contrast, medicines (4%) constituted only a small part of the net OOPE in AS. This may be attributed to the relatively cheaper cost of medications for AS (chiefly NSAIDs) than RA. Moreover, as biologics were not prescribed in our centre, the percentage of OOPE because of medicines is lower for both RA and AS than figures reported in other studies.44,45 Patients with AS spend up to 25% of their OOPEs on investigations, whereas patients with RA spend only 5%. Increased OOP spending on investigation by AS patients may be attributed to the high costs and scarce availability of MRI.
The contribution of travel expenses to OOPE ranged from 28% in RA to 47% in AS. In patients with AS, travel costs were the single largest contributor to OOPE. In this study, travel costs might be higher than average because hospital policy in our centre allows the distribution of a 2-week course of medications only. Thus, travel becomes a recurring cost for our patients. Non-medical costs including food, travel and lodging are a significant burden on the patient’s pocket.11,46,47 In general, poor health infrastructure and unavailability of advanced investigations are important drivers for medical travel. Nonmedical costs can be minimised by providing measures such as travel concessions in publicly-funded transport and cheap lodging services near major hospitals. Improving access to healthcare by strengthening the primary health care system can help to reduce travel expenses.
Regression analysis revealed that as the severity of symptoms increased in both RA (measured by CDAI) and AS (measured by BASDAI), the total COI increased, which is consistent with other studies.14,48 Disease activity indices guide management of disease. As disease severity increases, there is an escalation in costs because of an increase in the dose/number of medications for the primary condition and associated complications, more frequent monitoring and increased consultation charges as well as inpatient expenses. The mean duration of illness was also a significant predictor of costs for both RA and AS with total and direct costs increasing with increasing illness duration, which is similar to findings of other studies.14,27,49 This may be attributed to factors such as the development of comorbidities over the course of disease, poor medication adherence because of chronic medication intake 50 and complications secondary to prolonged drug use.
As impairment of daily activities increased, the total COI, OOPE and CHE increased for both patients with RA and AS. A likely explanation for this could be that as patients were unable to perform even day-to-day activities, their quality of life decreased. This might have prompted patients to seek medical care more often, including OOP spending on private consults, medications and investigations, instead of waiting for several days for their scheduled OPD visit at a government hospital, to spend more money out of pocket on rehabilitation equipment, home modifications and religious rituals. It was also likely that patients would be assisted by caregivers at home which would result in increased indirect cost, including wage losses for caretakers owing to missed work. In the case of AS, as productivity at work increased, COI decreased. A possible explanation for this could be that losses of productivity led to the change/loss of occupation resulting in wage losses which affected the COI. In comparison to the workplace, an inability to perform day-to-day activities at home was more distressing or more noticeable for the patient, prompting them to seek medical care. This might have led to an increased overall cost. Additionally, as a large number of patients were not gainfully employed, a patient was more likely to relate better to activity impairment than work unproductivity.
In patients with AS, age and household income determined the net OOPE, with younger and richer patients spending more money from their own income on healthcare. These variables, however, did not significantly affect OOPE in RA. This difference can be explained by the variation in sociodemographic characteristics of the 2 diseases. A typical patient with AS tended to be a younger, employed, financially independent male, while a patient with RA was typically an older, unemployed, financially dependent female. Because AS was more likely to affect the health of a breadwinner of the family, households were more likely to spend money OOP to achieve faster and more effective remission, whereas older, unemployed women with RA were more likely to limit OOPE by seeking care in public hospitals. Unemployed patients enjoy a lesser degree of financial independence which may have led to restricted access to household income and decreased OOPE. This finding may also reflect a gender disparity in healthcare expenditures highlighted by other studies, with average health expenditure and mean OOPE being higher for men compared to women. 51 In our study, sex alone was not a significant determinant of health expenditure.
Average overall work productivity impairment and overall activity impairment were comparable for RA and AS (work impairment: RA 43.33% vs AS 47.32%; activity impairment: 52.98% vs 49.33%) which is similar to the results of previous studies.52,53
The household income affected the indirect cost of AS. With increasing income, the indirect COI increased. A 2014 study 54 on AS patients had a similar result. Higher significance of household income was a predictor of indirect costs in AS may be because a patient with AS was more likely to be gainfully employed, thus contributing directly to the household income.
Loss of work productivity would affect the patient’s personal and thus the change was more likely to reflect directly on the income of the whole household. Indirect cost of RA and AS increased as productivity at work decreased which is similar to results reported by another study on RA.27
It was also seen that as duration of illness increased, indirect cost of AS decreased. A possible explanation for this phenomenon could be a gender-based difference in coping mechanisms. In our study, most patients with AS were males. Chronic disease threatens men’s sense of masculinity and their perceived ability to fulfil expected social roles. 55 Male patients with chronic illness value self-reliance. 56 To avoid dependence on caregivers, with time patients started managing health related activities such as visits to the OPD and activities at home on their own, which might have brought down the indirect cost due to loss of caregiver income. Since AS is a chronic illness, caregiver fatigue may have had an influence. Also, it is possible that with time, the patient’s ability to cope with illness improved which might help to limit losses in work productivity.
The social and psychological costs of chronic rheumatological illnesses are difficult to quantify because of the highly subjective nature of an individual’s experiences of the world around them and a difference in their ability to cope with psychological stressors associated with their illness. Studies show that arthritis including RA and AS are associated with chronic pain and psychological distress.57–59 Self-reported health status was more strongly affected by differences in personality traits than level of disability in patients with AS. 60 RA and AS are both independently associated with a higher rate and risk of acute ED presentations and hospitalisations for mental health conditions. 61 In our study, nine out of every ten respondents stated that their illness was a source of psychological distress for them. For example, a young patient with AS stated that he could not find a suitable match for marriage due to his illness. Despite this, utilisation of mental health services was virtually non-existent among our subjects even though these services are available at our center free of cost. This may reflect the prevalent societal stigma associated with mental healthcare.62,63 In India there is a need to generate evidence base for context specific interventions that will address negative attitudes towards people with mental disorders and ensure implementation of these interventions by involving users, care-givers, community health workers and mental health service providers.
While the current study did not evaluate risk of acquiring COVID among patients of RA and AS, previous studies have shown that such a risk is low and inconsistently demonstrated.64,65 Further, a study found that AS and RA patients were more vulnerable to psychological effects of the pandemic in comparison to healthy controls. 66 Since our centre was a COVID exclusive hospital, all other health services including OPD and pharmacy services and rheumatology clinics were disrupted for nearly 2 years. Consequently, all patients were lost to follow up. Self-reported adherence to medication, number of consultations and access to healthcare decreased while disease activity and visits to private practitioners of modern medicine and alternative forms of medicine increased. These findings are similar to results of other studies.67–69
Limitations
There are some limitations to our study. First is the small number of patients of AS included in the study. Since our hospital was a COVID exclusive centre from April 2020 to March 2022, all non-essential services including the rheumatology clinic where the current study was conducted was closed to public. Once services were resumed in March 2022, the number of patients attending the clinic was substantially decreased from pre-COVID times since patients were lost to follow up for a period of nearly 2 years. Many patients switched to other healthcare providers. As a result, the desired sample size for AS could not be achieved during the 2-month period of June-July 2022 in which this study was performed. Other limitations of the present study were its cross-sectional design and inclusion of only patients being followed-up in tertiary health care centres. Since a retrospective approach was applied to determine out-of-pocket spending, this data may be a potential source of recall bias. In our study, many patients were lost to follow up during the pandemic since our hospital was a COVID-only hospital and thus routine public healthcare services were not accessible at our centre. This may have contributed to higher costs. Additionally, our centre’s policy of providing medications for 2 weeks only may have led to higher consultation and travel costs for patients. This reflects the impact of local policies on health expenditure.
Conclusion
The economic impact of RA and AS is considerable. Direct costs, driven by pharmacy and investigation expenses, are substantial. Patients face high OOPEs, constituting almost half of the direct health expenses, leading to catastrophic spending in some households. Indirect costs, including productivity losses and activity impairment, are significant. The pandemic has negatively influenced health-seeking behaviour. Hitherto expensive biologic agents, central in RA and AS management, show promise with costs decreasing because of the use of biosimilars. Future studies need to evaluate their cost-effectiveness in the Indian context. High-quality accessible healthcare and public-funded insurance schemes are crucial to reduce out-of-pocket expenses. The study emphasises the psychosocial costs of RA and AS, urging future research on psychological factors’ impact on costs, productivity and treatment adherence in the Indian context. Additionally, promoting adjunctive treatments and studying health financing sources for chronic rheumatological illnesses are essential.
Supplementary Material
The supplementary material for this article is available online.
Footnotes
Acknowledgements
This study was approved by the Indian Council of Medical Research as a part of the Short-Term Studentship 2022 programme (ICMR-STS 2022, Reference ID: 2022-00644) and has received a financial grant for the same. We are grateful to the ICMR for their support. We also thank Dr Prakhar Varshney for assistance in data collection.
Author Contributions
OB was responsible for the concepts, design, definition of intellectual content, literature search, manuscript preparation, manuscript editing and manuscript review. SS was involved in the supervision of study and the critical revision of manuscript and acted as a guarantor.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
This study was approved by the Institutional Ethics Committee of Maulana Azad Medical College and Associated Hospitals. (F.1/IEC/MAMC/90/02/2022/No/09, dated 31/05/2022).
Funding
This study was approved by the Indian Council of Medical Research as a part of the Short-Term Studentship 2022 programme (ICMR-STS 2022, Reference ID: 2022-00644) and has received a financial grant for the same.
