Abstract
Systemic lupus erythematosus (SLE) is a complex heterogeneous autoimmune disease with protean clinical manifestations and phenotypes. As a result, any candidate molecule aiming to modulate a particular pathobiological pathway would likely fail to demonstrate efficacy in patients with mixed phenotypes. The success of the belimumab trial and the recent anifrolumab trial in SLE have provided evidence that stratifying patients based on their underlying pathobiological mechanism (e.g., endotypes) can improve the chances of success in drug trials. Various approaches to endotyping have been proposed to stratify SLE patients, such as biomarker profiling, gene expression signature fingerprinting, utilising transcriptomics and other ‘omics’ techniques for patient stratification, and molecular characterisation in both human subjects and animal models of SLE. Besides stratification of SLE patients based upon endotyping, incorporating ‘theratyping’ (which refers to outliers in any failed drug trial who exhibit a positive response) would further ‘fine-tune’ the subgrouping with uniform underlying pathobiology. Using a 2-pronged approach of defining theratypes of pre-endotyped patients could fast-track drug discovery of drugs for SLE treatment. Considering the success of recent trials, there is compelling evidence that this approach can significantly increase the likelihood of success in drug trials and pave the way for more effective treatments for SLE.
Introduction
Systemic lupus erythematosus (SLE) is a prototype systemic autoimmune rheumatic disease (SARD) with protean manifestations. The clinical heterogeneity with multiple phenotypes translates into pathobiological diversity that challenges its recognition, diagnosis, classification, disease-activity evaluation, and prognostication. 1 These features also present serious challenges for the drug development programmes for appropriate trial design and the selection of appropriate endpoints, 2 and could be one of the main reasons for drug trial failures (vide infra). 3 For a long time, the only United States Federal Drug Administration (FDA) approved drug for the treatment of SLE was aspirin (1948) followed by the approval of corticosteroids (e.g., prednisone). 4 In 1955, FDA approved the antimalarial drug hydroxychloroquine, which was shown to relieve some lupus symptoms, such as fatigue, rashes, joint pain, or mouth sores. 5 However, since then, there has been limited success in drug development. It was not until 2011 when belimumab was approved by FDA, 6 after failing to meet the primary endpoint in the initial results. This was possible only after reclassifying patients into antinuclear antibodies (ANA) positive and negative subgroups and reanalysing the results. The ANA-positive group showed a statistically significant improvement in disease activity, leading to belimumab’s approval by FDA and the European Medicines Agency (EMA) in 2011.7–11 This was the first evidence that even within the clinically diagnosed patients with SLE there is considerable heterogeneity with multiple phenotypes possibly each with different underlying molecular mechanisms involved in a variable pattern of tissue damage. Therefore, for drug trials to be successful, there was a need to include only the ‘right patient population’ within the heterogenous group of clinically diagnosed patient population. 12 The concept of ‘right patient population’ within a clinically diagnosed patient with SLE is, however, not new. Pisetsky and colleagues had already alluded to ‘new perspective in rheumatology’ namely, ‘biomarkers as entry criteria for clinical trials of new therapies for systemic lupus erythematosus’. 13 This concept was therefore factored in the new updated (2019) ‘classification criteria’ for SLE and made ‘antinuclear antibody positivity’ as the ‘entry criteria’. 14 The disease heterogeneity has received increasing importance that could also explain the frustrating experience of the failure of multiple phase-III trials of drugs in SLE that showed promising results in Phase-II. 15 In this scenario, the importance of alirocumab’s approval cannot be overemphasised. 16 It marks a breakthrough in the treatment of SLE. It is so significant that ‘The Lancet’ published an editorial with the title ‘The year 2022—a banner year for lupus’. 17
Drugs that Failed Phase III Trials in SLE After Promising Results of Phase II
Some of the drugs with promise that have failed in final stages of randomised controlled trials in SLE include rituximab, eptratuzumab, ixekizumab, regerimod, tabalumab, telitacicept, and at least 18 other molecules, mostly due to issues associated with disease heterogeneity and trial design. 18 In this regard, however, there have been two major disappointing failures namely that of baricitinib (BRAVE trials) and atacicept. In phase-II trial of baricitinib for treating SLE (called ‘BRAVE-1’) were encouraging. 19 In this trial, the primary endpoint along with exploratory secondary outcome measures in musculoskeletal and mucocutaneous domain scores were met in patients taking 4 mg baricitinib. Also, no new safety signals were observed in the patients receiving baricitinib. However, in BRAVE-1 trial, some of the formal secondary endpoints were not met and that was worrisome. With this background, a phase-III (BRAVE-II) trial of baricitinib in SLE was conducted. 20 It included 775 patients of whom 258 patients received baricitinib 4 mg, 261 patients received 2 mg baricitinib, and 256 patients received placebo. Most disappointingly, the trial did not show any differences in the percentage of patients from each group achieving the primary as well as the secondary endpoint. In addition, no secondary endpoints were met. Another and, possibly a much more perturbing drug trial failure. has been that of a theoretically promising drug, atacicept. 21 The reasons for these failures have been discussed in depth by Isenberg and colleagues from London 22 as also by the researchers in the US.23,24 These investigators have again highlighted that not selecting ‘the right patient population’ could be the possible reason for failed trials in SLE. Regarding atacicept, therefore, a retrial on an appropriately stratified patient population has been suggested. 21 These results could indicate potential confounding effect of heterogeneity that has been insufficiently addressed in the design of drug trials in SLE. However, it needs to be reminded that, in general, it is not uncommon for drugs to fail in Phase III clinical trials after showing promising results in Phase II. This can be due to a variety of additional factors other than those mentioned above, including small sample size, flawed trial design, poor choice of primary outcome measure, poor implementation of the trial, poor choice of concomitant medications, over-interpretation of the results of phase-II trials, safety issues, high placebo response rates, and specifically in the case of SLE, mandated use of steroids and its tapering, and inadequate domains of SLE assessment. The story of the approval of belimumab after failing to meet the primary endpoint has already been mentioned vide supra. In the case of belimumab, ‘serology-based response’ (ANA positive patients showed response) appears to be a clue that the patients with SLE need to be appropriately stratified to achieve homogenous groups of patient phenotypes, presumably with same/similar underlying pathobiology, for drug trials to provide relevant results. Unfortunately, the presently available ‘classification criteria’ for SLE drug trials, 14 appears to be too insensitive for such stratification of patients with SLE.
Success of Anifrolumab—The ‘Interferon-1’ Story
Obviously, finding a single treatment for a disease like SLE that has multiple pathogenic pathways in causing cell-tissue-organ damage in different patients at different ages and different stages of the disease, would be challenging. In this regard, an important observation by Moutsopoulos and his group in 1979 was that of elevated type-1 interferon (IFN-I) levels in lupus with possible implicating in its pathogenesis. 25 Several decades later, in 2017, Paul Emery’s group re-examined the IFN-1 molecular pathway’s possible role in SLE pathogenesis’ 26 Their studies pointed out abnormal levels of IFN-1 in a subset of SLE but, additionally in subsets of several other SARDs as well. Based upon these observations Emery and colleagues coined the term ‘interferonopathies’ for such overlapping groups of patients with SARDs. Over the next several years enough evidence accumulated supporting the involvement of IFN-1 pathway in the pathogenesis of SLE. 27 The obvious next step was to conduct trials with IFN-I inhibitors to test their efficacy in patients with SLE. Anifrolumab, a fully human, IgG1 monoclonal antibody to IFN-1 receptor subunit 1 that inhibits signalling by all type I interferons. 28 was used in a phase-II trial. 29 That trial showed efficacy across several endpoints. Encouraging results in phase-II trial led to its phase-III trial called ‘TULIP-1 (Treatment of Uncontrolled Lupus via the Interferon Pathway [TULIP])’. Most disappointingly, the trial did not show a significant effect on the primary endpoint. 30 However, the trialists noted that some prespecified secondary endpoints in that trial, showed significant response. That led to the use of a modified primary endpoint called ‘BICLA’ (The British Isles Lupus Assessment Group (BILAG)–based Composite Lupus Assessment)
(BICLA) in the subsequent trial called ‘TULIP-2’, a second phase-III trial of anifrolumab in active SLE. 31 The results showed a significantly higher percentage of patients with a clinically meaningful response at week 52 as compared to placebo. This trial led to the FDA approval for the use of anifrolumab in active SLE patients. 17
Nature’s Experiment – Mutated Toll-like Receptor 7 (TLR-7) Gain-in-function – B Cells and IFN-1 Pathway Activation
In the context of the involvement of IFN-1 in the pathogenesis of SLE, a ‘Nature’s experiment’ is worth recalling. 32 In this article, Brown and colleagues describe a seven-year-old girl of Spanish ancestry with SLE, whose genome sequencing revealed a change in a single-base in the gene that codes for toll-like receptor-7 (TLR-7). This receptor plays a crucial role in protecting against viral infections by acting as a sensor of viral RNA that binds to guanosine. The researchers used advanced laboratory techniques, including a mouse model, to establish the mechanism of causation of SLE by a TLR7 gain-of-function variant. They found that the variant led to enhanced TLR7 signalling, which drove abnormal survival of B cell receptor accumulation of CD11c+ age-associated B cells and germinal centre B cells. The study identified a previously unreported missense TLR7Y264H in the child with severe SLE, which was confirmed in additional patients with SLE. The study showed the importance of TLR7 and guanosine-containing self-ligands in human SLE pathogenesis, paving the way for therapeutic targeting of TLR7 or MyD88. TLR7 and MyD88 are upstream from IFN-1. Blocking these upstream molecules may effectively block additional inflammatory molecules, making them effective in a broader spectrum of SLE patients. An oral small molecule compound (M5049) that targets TLR7 and TLR8 is currently undergoing early stages of drug trial in SLE. 17
Discovery of Drugs for Complex Disease with Heterogeneous Clinical Phenotypes – Recent Advances
Discovery of effective drugs for clinically well-defined and easily diagnosable diseases that are inherently heterogeneous with different clinical phenotypes, is an arduous task. As discussed, vide supra, SLE is a prime example of such a disease. It shows considerable patchiness across subjects. Such heterogeneity presents a major barrier both to deciphering pathogenesis and translating the same for designing, conducting, and interpreting clinical trials of disease-modifying agents. Candidate drugs with well-defined mechanisms of action that would theoretically control the SLE disease activity are repeatedly failing in phase-III trials after showing promising results in phase-II trials (vide supra). Therefore, ‘a homogeneous single disease’ (phenotype, i.e., observable characteristics) approach for drug discovery for treating SLE is untenable. Sub-classifying a mix of SLE patients based upon underlying biological mechanisms (i.e., endotyping, vide infra) is likely to identify a homogeneous subgroup of patients. Thus, for an increased success rate of new drug trials in SLE, patients stratified with clearly defined endotypes would be able to generate compelling outcome data for a faster transition to market for explicitly identified disease subsets. Incorporating the endotype concept into routine clinical and laboratory practice may be the right way forward. This approach is likely to fast-track the implementation of precision medicine by impacting the approach to translational research, trial design, and clinical management.
Understanding Phenotype, Endotype and Theratype
Phenotype
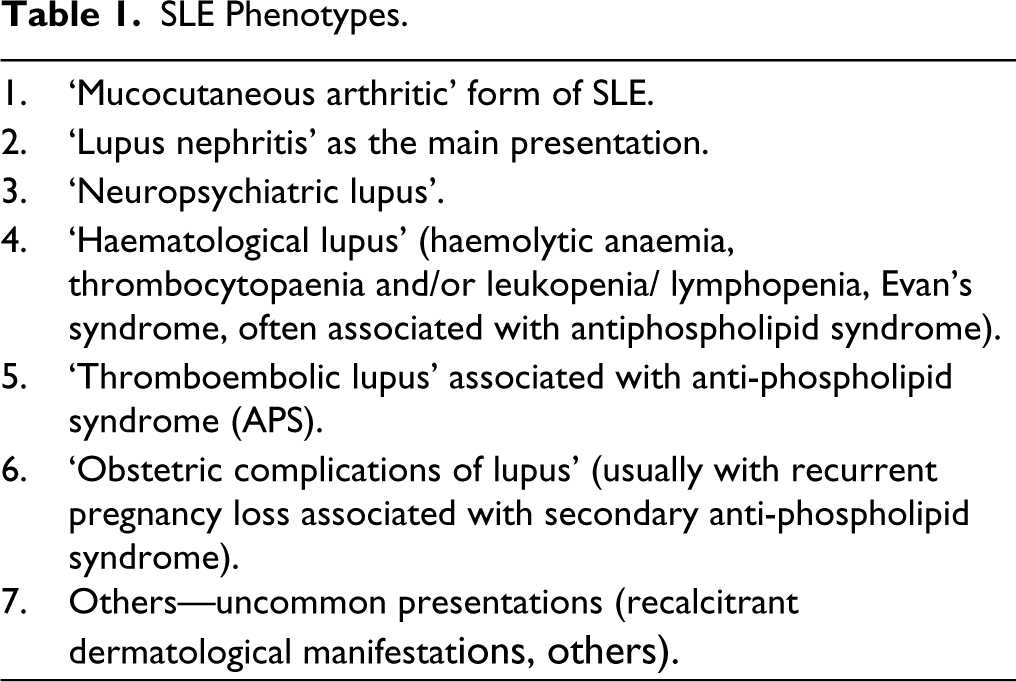
In biology, the term ‘phenotype’ is used to define the observable traits or characteristics of a living being, which specify the species of that organism. These traits include physical appearances as well as other traits that are easily recognisable. Specific phenotypes are the result of the interaction of genes with environmental factors. In the context of SLE, the clinically recognisable phenotypes are given in Table 1.
SLE Phenotypes.
Endotype
Over the years, it has been observed that the different phenotypes do not offer insight into the underlying pathobiological mechanism; these may or may not have the same underlying abnormalities in disease-causing biological pathways. For an appropriate understanding of that, a much deeper analysis of the biological mechanisms involved in causing the pathobiology, is required. The evolving understanding of the physiology of the biological systems has discovered a variety of biochemical, metabolic, inflammatory, immunological cells and subcellular milieu that can be exploited to understand their role in health and disease. Studying the heterogeneity in type-1 diabetes, it was demonstrated that traits are often linked (e.g., age and HLA-specific autoimmunity) in such a way that suggests associations, which could be built into distinct pathobiological entities. 33 Using molecular and omics techniques, these abnormalities can be characterised with high precision. Using these techniques, a particular clinically diagnosable disease, especially those with protean manifestations, can be further categorised in clearly defined subsets. This approach of precisely subtyping such diseases is called ‘Endotyping’33,34 that further stratifies any clinical disease into different ‘endotypes’. As endotypes characterise the disease at subcellular/biochemical/molecular level, it becomes possible to correct that abnormality with targeted treatment. The current popular term for such a treatment approach is called ‘precision medicine’.
Theratype
In practice, whether a trial succeeds or fails in meeting its primary objective(s), there are often small subgroups of subjects who appear to benefit from the therapy. Such patients within a large group, who uniformly respond to a certain drug, are called ‘theratypes’.35,36 ‘Theratyping’ the trial subjects could be exploited to circumvent disease heterogeneity that negatively influences drug trials. Using endotyping method, ‘thertyped’ patients could be well characterised. Trying the same drug on patients stratified accordingly would fast-track its approval in such a subset of patients. Despite the heterogeneity of SLE, this approach would be the way forward for the rapid development of drug discovery in this disease.
In recent times researchers have been intensely investigating the cellular/molecular pathways that may be involved in the pathobiology of different subsets of SLE patients to accurately stratify them into distinct endotypes. Such stratification of patients may help to identify more specific and tailored treatment approaches.
Methods used for Endotyping Patients with SLE—An Update
Till date, six different approaches have been reported in the literature for endotyping patients with SLE in different subgroups. Table 2 provides a list of methods that have been reported for the endotyping of SLE in recent years.
Different Methods Used for Endotyping Patients with SLE.

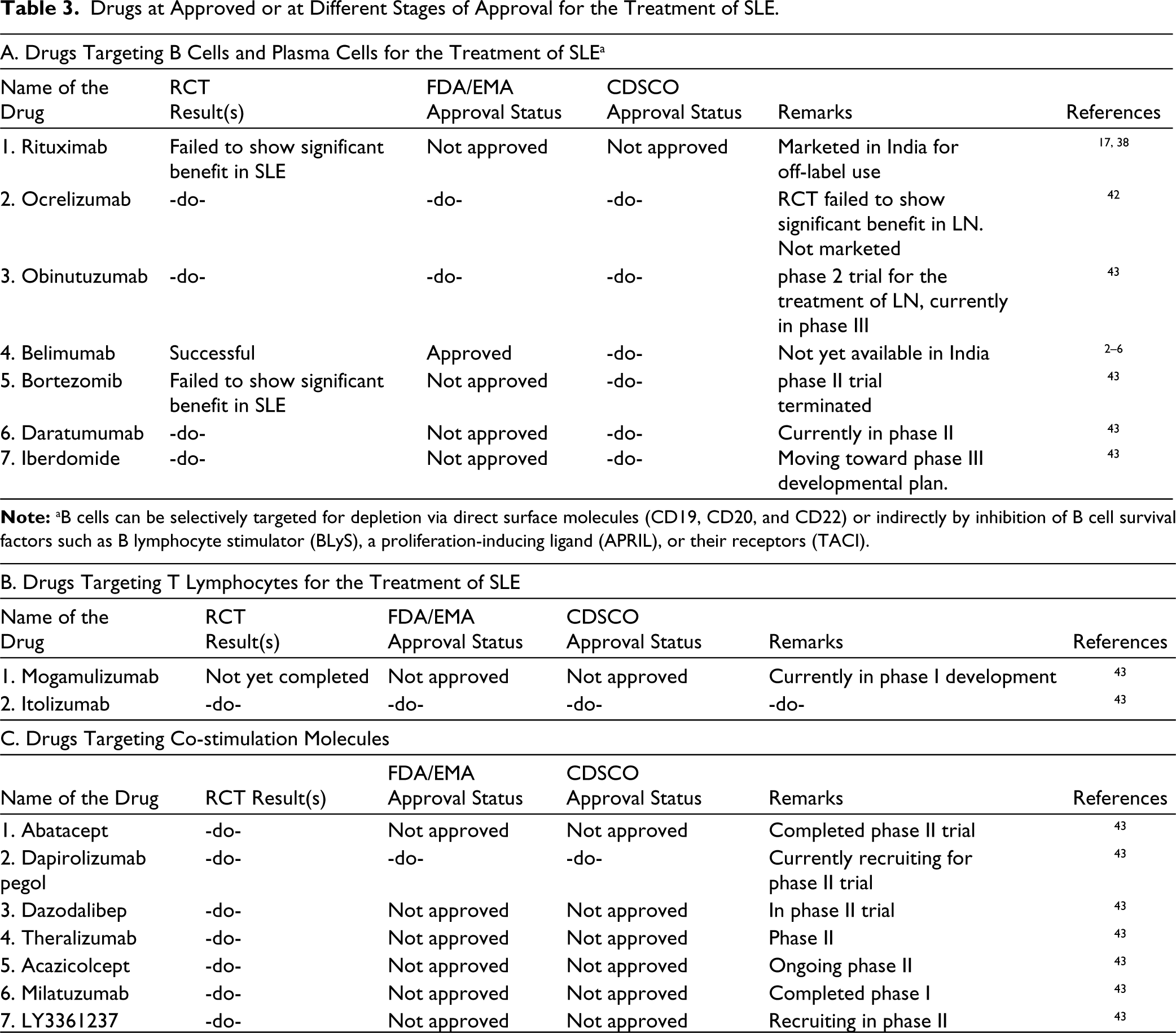
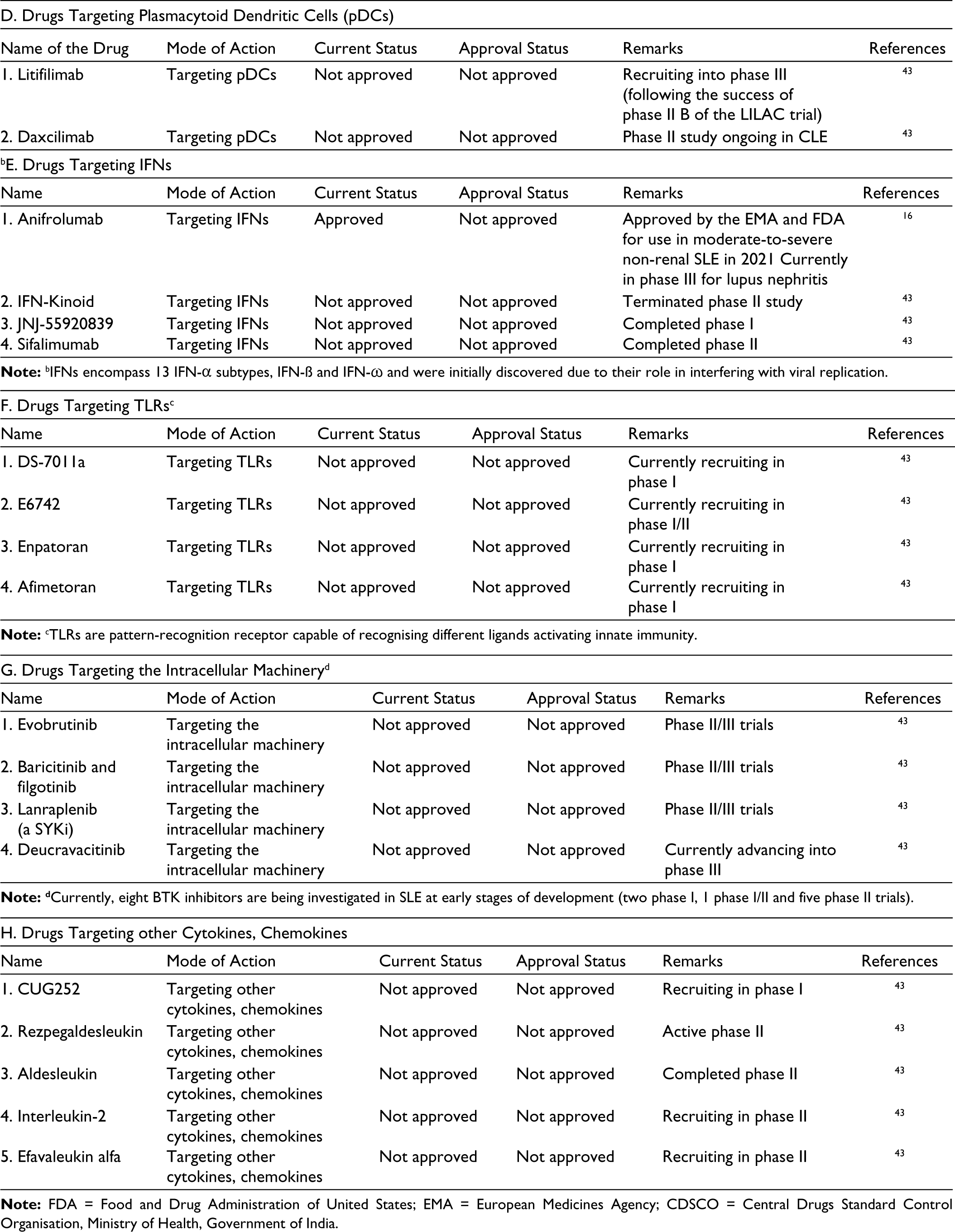
Table 3 gives an extended list of drugs for the treatment of SLE categorised as ‘approved’, ‘under trial/stage of trial’ and purported ‘mechanism of action’.
Drugs at Approved or at Different Stages of Approval for the Treatment of SLE.
Conclusion
In conclusion, the heterogeneity of patients with SLE has been a major challenge in developing effective targeted therapies. However, recent research has focused on stratifying SLE patients with different phenotypes into endotypes based on different biological parameters such as BAFF and IFN-I bioactivity, immunological biomarker profile, gene signature fingerprinting, transcriptome molecular characters, molecular endotypes, and molecular patterns of mouse models. Accurate endotyping on appropriately theratyped patients would be able to stratify them into homogenous groups with similar underlying pathobiology. Such an approach will facilitate drug trials in SLE with a high chance of success and help fast-track its approval. 44 It is exciting to see progress being made in this area of research. With this background, if any future trial shows promise in an early phase but fails in phase-III trials, the workers may not have to ask: ‘What did not work: the drug or the trial?’ 45
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Not applicable
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
