Abstract
Background
As diabetes continues to be a global epidemic, the prevalence of retinopathy, a frequent microvascular complication, is anticipated to increase significantly. Effective management of blood glucose, blood pressure, and lipid levels may reduce the risk of diabetic retinopathy (DR); however, maintaining optimal control remains challenging in clinical practice. In advanced stages, therapeutic interventions such as anti-vascular endothelial growth factor (VEGF) medication, laser photocoagulation, and vitrectomy are necessary to preserve vision; however, they render side effects, and vision recovery is not 100%.
Objectives
We aimed to investigate the efficacy of bioactive alkaloid noscapine to mitigate DR.
Materials and Methods
Hyperglycemia was triggered in male Wistar rats with streptozotocin (STZ) and treated with noscapine. The impact of noscapine on regulating STZ-induced hyperglycemia was analyzed by quantifying the glycated hemoglobin and blood glucose levels. The body weight, feed intake, and cholesterol levels of the experimental animals were monitored to assess the lipid-lowering efficacy of noscapine. To assess the inhibitory potency of noscapine against hyperglycemia-triggered oxidative stress, the malondialdehyde (MDA) levels and the antioxidant levels were quantified. Inflammatory proteins were measured to evaluate the efficacy of noscapine in hyperglycemic rats. VEGF, MCF-1, and matrix metallopeptidase 9 (MMP-9) protein levels were quantified in the experimental rats to analyze the mitigating potency of noscapine against hyperglycemia-triggered DR. The retinal histopathological analysis and apoptotic protein levels in the experimental animals were performed to confirm the ameliorative efficacy of noscapine against hyperglycemia-induced retinopathy.
Results
Results from various analyses proved noscapine treatment effectively attenuated hyperglycemia and hypercholesterolemia in the STZ-administered rats. It also scavenged free radicals, enhanced antioxidant status, and thereby inhibited ROS-induced inflammation and apoptosis in hyperglycemia-triggered rats. The levels of VEGF, MCF-1, and MMP-9 were significantly decreased with noscapine treatment, which signifies its anti-angiogenic effect in treating DR, and it is further confirmed with our retinal histopathological analysis.
Conclusion
Overall, our study demonstrates that noscapine is a potent anti-hyperglycemic and antioxidant agent, showing significant ameliorative effects against retinopathy in a diabetic rat model.
Keywords
Introduction
The global diabetes epidemic is rapidly escalating, with 529 million individuals affected in 2021, translating to a prevalence rate of 6.1% (GBD 2021 Diabetes Collaborators, 2023). Recent estimates from the International Diabetes Federation (IDF) indicate that approximately 10.5% of the adult population, or over half a billion people, currently live with diabetes (Sun et al., 2022). Projections suggest that this number could rise to nearly 800 million by 2045 (Unnikrishnan et al., 2017). The prevalence of diabetes among adults was 8.5% in 2014, and it has been steadily increasing over the past 30 years, especially in low and middle-income countries (Chisha et al., 2017). Type 2 diabetes is particularly concerning among youth, as it tends to manifest more aggressively and is associated with early onset of complications such as hypertension, nephropathy, polycystic ovary syndrome, and dyslipidemia (Dabelea et al., 2017; Ke et al., 2022).
Diabetic retinopathy (DR) is the most prevalent microvascular complication of diabetes mellitus and represents a major cause of vision impairment and blindness globally (Stitt et al., 2016). The rising incidence of type 2 diabetes has significantly contributed to the increasing burden of DR, establishing it as a leading cause of preventable blindness (Gupta & Chhikara, 2018). Currently, DR affects an estimated 30%–40% of individuals with diabetes, with a global prevalence of approximately 103 million cases, a figure projected to rise to 161 million by 2045 (Teo et al., 2021). Although the overall prevalence of visual impairment has declined, the number of individuals experiencing blindness due to DR has doubled, increasing from 0.2 million to 0.4 million. Notably, approximately 80% of individuals with type 2 diabetes are expected to develop some degree of retinopathy (Abdulhussein & Abdul Hussein, 2023; Zegeye et al., 2023).
DR is primarily caused by hyperglycemia, which triggers a range of metabolic pathways leading to retinal microvascular damage (Morya et al., 2024). Key mechanisms include the polyol pathway, advanced glycation end products (AGEs) accumulation, activation of the protein kinase C (PKC) pathway, and the hexosamine pathway (Haydinger et al., 2023). Hyperglycemia initially causes retinal blood vessel dilation and altered blood flow; a response aimed at enhancing retinal metabolism (Bek, 2017). Additionally, diabetes leads to increased mitochondrial superoxide production in vascular endothelial cells, resulting in oxidative stress and enhanced flux through various damaging pathways (Wang & Lo, 2018). This situation promotes inflammation, elevated levels of reactive oxygen species (ROS), and harmful epigenetic modifications (Reddy et al., 2015). Furthermore, growth factors like VEGF and insulin-like growth factor-1 (IGF-1), changes in hemodynamics, activation of the renin–angiotensin–aldosterone system (RAAS), and leukostasis intertwine with these pathways, contributing to the disease’s development and progression (Tarr et al., 2013).
DR treatments encompass various modalities aimed at preventing vision loss and managing disease progression. Laser therapy, anti-VEGF injections, and corticosteroid injections reduce the risk of severe vision loss but may lead to transient visual disturbances, elevated intraocular pressure, and pain (Gonzalez et al., 2016; Wang & Lo, 2018; Wroblewski et al., 2016). Phytochemicals such as alkaloids, polyphenols, terpenoids, and flavonoids exert various pharmacological effects by targeting multiple metabolic pathways simultaneously. Additionally, these bioactive compounds enhance intercellular drug delivery, a crucial factor in achieving optimal therapeutic outcomes (Dal & Sigrist, 2016; Efferth & Koch, 2011; Parveen et al., 2018). Noscapine is an isoquinoline alkaloid compound extracted from opium sourced from the Papaver somniferum plant. Noscapine already showed various biological activities, including anti-tumor (Chougule et al., 2011), neuroprotective (Jayaraj et al., 2021), and anti-fibrotic (Kach et al., 2014) properties. In this study, we analyzed noscapine’s mitigating efficacy against DR in rats.
Materials and Methods
Experimental protocols performed on the rats were presented to the ethical committee and were approved. Adult Wistar rats of the male species weighing about 230 ± 10 g were utilized for the study. The animals were acclimatized for a week in an ambient environment with a temperature of about 23°C ± 2°C, 55% ± 5% relative humidity, and 12 h/12 h artificial circadian lighting was provided. The rats were provided 24 h of free access to food and water.
DR Induction and Grouping
DR was induced in the animals via intravenous injection of streptozotocin (STZ) (60 mg/kg bwt). Blood glucose levels were quantified 48 h after the STZ induction. Animals exhibiting glucose levels of 250 mg/dL or higher were categorized as diabetic. Acclimatized animals were allocated into four clusters, each containing six animals. Group I served as the control group was given a 0.1 M saline solution without STZ or noscapine. In Group II, rats were administered STZ at a dosage of 60 mg/kg to induce diabetes-related retinopathy (DR). Group III included diabetic rats that received an oral treatment of 50 mg/kg noscapine for 9 weeks. In Group IV, diabetic rats were treated with metformin at a dosage of 350 mg/kg, which served as the standard treatment for a duration of 60 days. The body weight of the experimental animals was monitored regularly, and the weights were recorded. The food intake of the animals was measured and recorded daily. At the end of the treatment period, the animals were anesthetized, and the blood sample and eyeballs were collected.
Diabetic Profile Analysis
Diabetic induction in experimental animals was quantified with blood glucose and glycated hemoglobin (HbA1c) levels. Blood glucose levels were quantified with the blood samples collected from the tail vein of the experimental animals. The glucose diagnostic kit AccuCheck was used to quantify the glucose levels in the blood samples of experimental animals. HbA1c was measured using the HbA1c assay kit procured from Crystal Chem. The assay was performed as per the manufacturer’s protocol.
Lipid Profile Analysis
Total cholesterol, high-density lipoprotein (HDL)-cholesterol, and triglycerides were measured enzymatically in the serum samples using the respective diagnostic kits procured from Randox test kits. Low-density lipoprotein (LDL)-cholesterol was estimated using the method outlined by Friedewald et al. (1972), and the atherogenic index was calculated by the Muruganandan et al. (2005) method.
Oxidative Stress Analysis
Malondialdehyde (MDA) concentration as an indicator of lipid peroxidation was quantified through its reaction with thiobarbituric acid (TBA) in a spectrophotometric assay using the protocol of Ohkawa et al. (1979). The final absorbance of the reaction mixture was recorded at 535 nm. Reduced glutathione (GSH) and oxidized glutathione (GSSG) were measured using the protocol described by Linillos-Pradillo et al. (2023). The fluorescence emitted by the sample upon final reaction was measured at an excitation wavelength of 350 nm and an emission wavelength of 420 nm.
The activities of glutathione peroxidase (GPx), glutathione reductase (GR), catalase (CAT), and superoxide dismutase (SOD) were evaluated in the experimental animals using the respective diagnostic kits. The enzymatic activities were analyzed spectrophotometrically following the protocols provided by the manufacturer (Cayman Chemical, Ann Arbor, MI, USA).
GPx activity was evaluated by coupling it with the oxidation of nicotinamide adenine dinucleotide phosphate (NADPH) via GR. The reduction of organic hydroperoxides by GPx produces GSSG, which is subsequently recycled to GSH by GR in the presence of NADPH. This process results in a decrease in absorbance at 340 nm, which corresponds to NADPH oxidation and is directly proportional to GPx activity. GR activity was determined using an assay that measures the rate of NADPH oxidation. The reduction of NADPH to NADP⁺ was monitored by the corresponding decline in absorbance at 340 nm, which reflects GR activity. CAT activity was measured based on its reaction with methanol in the presence of an optimal concentration of hydrogen peroxide (H2O2). The resulting formaldehyde was quantified spectrophotometrically using 4-amino-3-hydrazino-5-mercapto-1,2,4-triazole as a chromogenic reagent, with absorbance recorded at 540 nm. SOD activity was determined by assessing the dismutation of superoxide radicals generated by the xanthine oxidase and hypoxanthine reaction system.
Inflammatory Signaling Analysis
Enzyme-linked immunosorbent assay (ELISA) kits were used to quantify the inflammatory-stimulating proteins nuclear factor kappa B (NF-κB), tumor necrosis factor-alpha (TNF-α), interleukin (IL)-6, and IL-1β in the experimental animals. ELISA kits were procured from Thermo Fisher Scientific, USA. The assay was performed as per the protocol prescribed by the manufacturer. A standard curve plot was drawn with OD of known concentration to quantify the levels of inflammatory proteins.
Assessment of DR Induction
The induction of DR in the experimental animals was assessed by quantifying the proteins vascular endothelial growth factor (VEGF), matrix metallopeptidase 9 (MMP-9), and monocyte chemoattractant protein-1 (MCP-1). Abcam (USA) ELISA kits were utilized for the quantification of the proteins, and the assay was performed following the instructions of the manufacturer. Upon termination of the reaction with the stop solution, the final absorbance of the samples and standard was quantified at 450 nm.
Examination of Retinal Histology
Eyeballs extracted were subjected to paraformaldehyde fixation for 48 h at 4°C, followed by embedding in paraffin. Paraffin sections were deparaffinized and rehydrated through graded alcohols. The sections, cut to a thickness of 5 µm, were stained using hematoxylin and eosin (H&E). Digital images of the retina were captured at 400× magnification using a light microscope. For each eye, four sections were analyzed, and the results were averaged. Total retinal thickness was assessed at 400× magnification by taking two measurements per section at reference points located 1 mm away from the optic nerve on both the superior and inferior sides. The number of retinal ganglion cells (RGCs) was also determined at 400× magnification, following the method described by Zhang et al. (2018).
Apoptotic Protein Quantification
Proapoptotic proteins BCL-2 associated X protein (Bax) and B-cell lymphoma 2 (Bcl2) were quantified in the experimental animals to measure the retinal cell apoptosis induced by diabetic conditions. Thermo Fisher Scientific (USA) ELISA kits were utilized to quantify the levels of apoptotic proteins. To assess the severity of apoptosis Bax/Bcl2 ratio was calculated with the obtained level of Bax and Bcl2 proteins in the experimental animals.
Statistics
Data obtained from at least three independent experiments were analyzed using Statistical Package for the Social Sciences (SPSS) software (version 16.0). Results were presented as the mean ± standard deviation. Comparisons among multiple groups were performed using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test. A p value of <.05 was considered statistically significant.
Results
Noscapine Prevented Polyphagia in DR-triggered Rats
Diabetic-triggered untreated rats’ feed intake was significantly increased to 46 ± 0.08 g/day, whereas the control rats’ feed intake was exhibited to be 25 ± 0.06 g/day. Treatment with noscapine in diabetic-triggered rats reduced polyphagia, and the feed intake was reduced to 34 ± 0.05 g/day. Metformin treatment also reduced the feed intake to 28 ± 0.05 g/day in diabetic-triggered rats (Figure 1B). The body weight was reduced with diabetic induction to 157 ± 0.9 g compared to control rats, which exhibited a body weight of 235.24 ± 12 g whereas treatment with noscapine and metformin increased the body weight to 171 ± 1.2 g and 183 ± 1.9 g, respectively (Figure 1A).
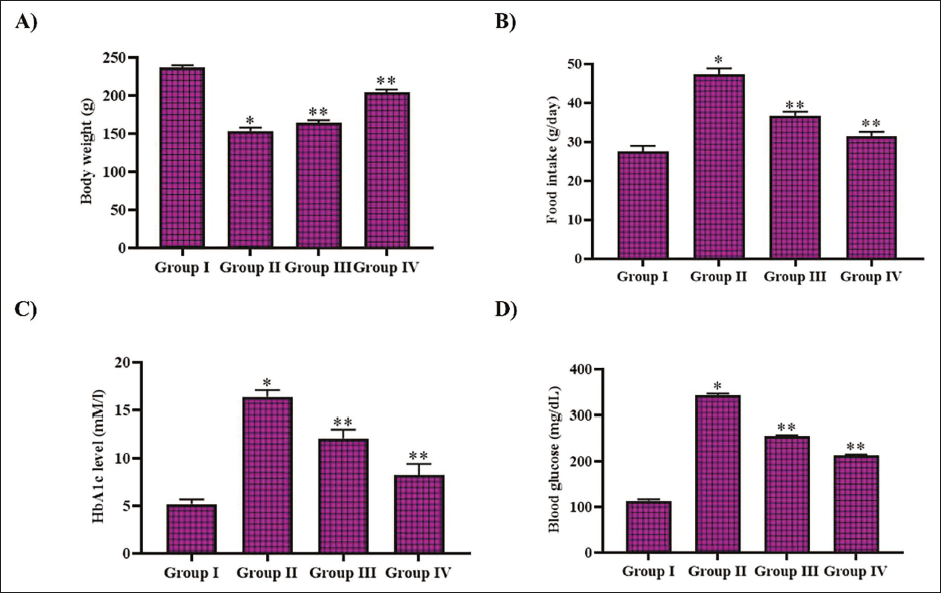
Noscapine Attenuated Hyperglycemia in DR-triggered Rats
Figure 1B depicts the effect of noscapine against hyperglycemia in diabetic-triggered rats. Increased HbA1c and blood glucose levels were increased to 14.9 ± 0.0004 mM/L and 320 ± 0.4 mg/dL in the diabetic-triggered untreated rats, whereas it exhibited to be 4.9 ± 0.0003 mM/L and 110 ± 0.2 mg/dL, respectively, in control animals. Furthermore, our results showed a significant reduction in the levels of HbA1c and blood glucose following treatment with noscapine and metformin. HbA1c was decreased to 9.8 ± 0.0007 mM/L and 5.4 ± 0.0002 mM/L with noscapine and metformin treatment. The blood glucose also decreased to 240 ± 0.4 and 185 ± 0.4 mg/dL with noscapine and metformin treatment.
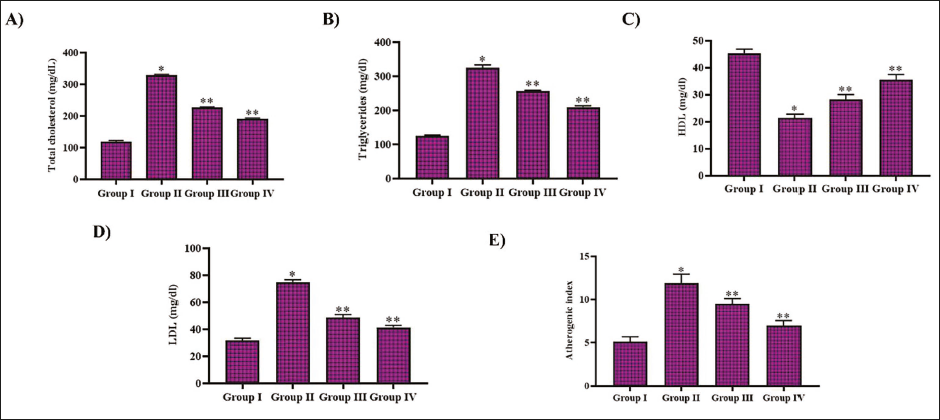
Noscapine Inhibited Hypercholesterolemia in DR-triggered Rats
Figure 2 illustrates the impact of noscapine against STZ treatment-triggered hypercholesterolemia in experimental animals. STZ treatment increased the levels of total cholesterol and the triglycerides in the untreated diabetic-triggered and noscapine-treated rats compared to the control rats. Treatment with both noscapine and metformin decreased the levels of total cholesterol and triglycerides. The LDL cholesterol levels also increased with STZ treatment in experimental animals. Noscapine treatment decreased the LDL cholesterol and increased the HDL cholesterol levels in the STZ-treated rats. Control rats exhibited an atherogenic index of 4.7 ± 0.0003, whereas it increased to 10.2 ± 0.0003. The noscapine-treated rats showed a decreased atherogenic index of 7.3 ± 0.0005 and 5.2 ± 0.003 with metformin treatment.
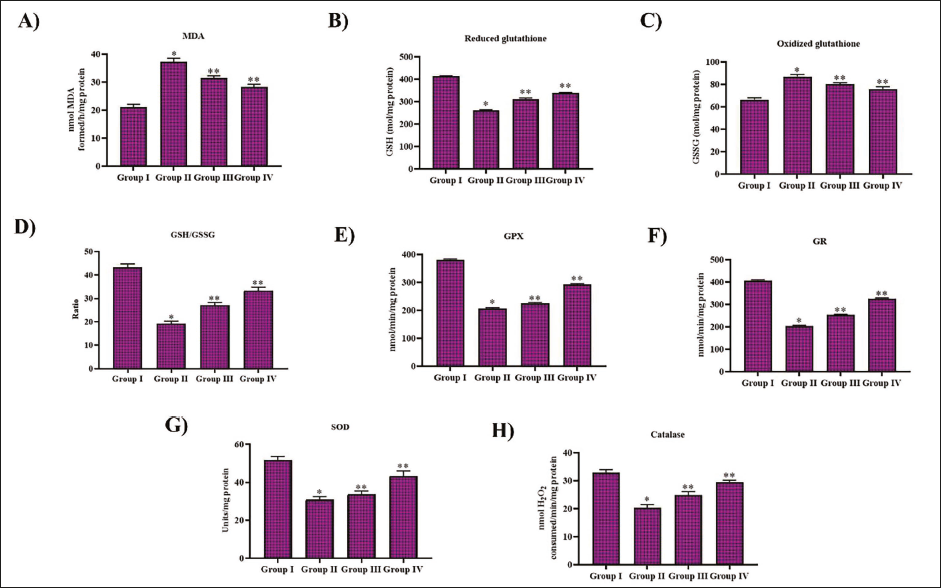
Noscapine Scavenged Free Radicals in DR-triggered Rats
STZ administration in the experimental animals increased the free radicals and inhibited the antioxidant defense system. MDA levels and GSSG levels significantly increased with the STZ treatment, whereas treatment with noscapine reduced the glutathione oxidation and lipid peroxidation in the STZ-treated rats. Noscapine-treated rats exhibited enhanced antioxidant status compared to the untreated diabetic-triggered rats. Reduction of glutathione and GSH/GSSG levels was significantly enhanced with noscapine and metformin treatment. It also increased the enzymes GPx and GR levels in the diabetic-triggered rats. The enzymatic antioxidants SOD and CAT levels were also considerably enhanced with noscapine and metformin treatment (Figure 3).
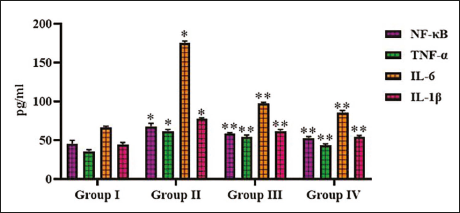
Noscapine Prevented the Inflammatory Signals in DR-triggered Rats
The impact of noscapine against STZ-triggered inflammatory proteins was quantified, and the levels were illustrated in Figure 4. STZ administration enhanced the production of inflammatory protein NF-κB and TNF-α, which was attenuated with noscapine and metformin treatment. Compared to NF-κB, TNF-α, the levels of IL were significantly elevated with STZ administration. Treatment with noscapine inhibited the synthesis of both IL-1β and IL-6 in STZ-administered rats (Figure 4).
Noscapine Treatment Inhibited DR Induction in STZ-treated Rats
STZ administration in the experimental animals elevated the levels of VEGF, MCF-1, and MMP-9 to 151 ± 0.003, 11.6 ± 0.0007, and 78 ± 0.0002 ng/mL, whereas the control rats exhibited 52 ± 0.002 ng/mL of VEGF, 4.7 ± 0.0003 ng/mL of MCF-1, and 30 ± 0.0002 ng/mL of MMP-9. VEGF, MCF-1, and MMP-9 levels were decreased to 126 ± 0.05, 7.7 ± 0.0004, and 57 ± 0.003 ng/mL, respectively, with noscapine treatment. Metformin-treated rats exhibited 78 ± 0.04 ng/mL of VEGF, 5.2 ± 0.0002 ng/mL of MCF-1, and 36 ± 0.0003 ng/mL of MMP-9 in diabetic-triggered rats (Figure 5).
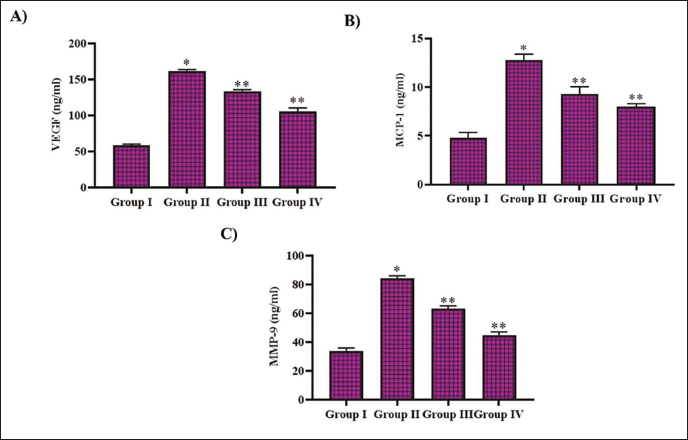
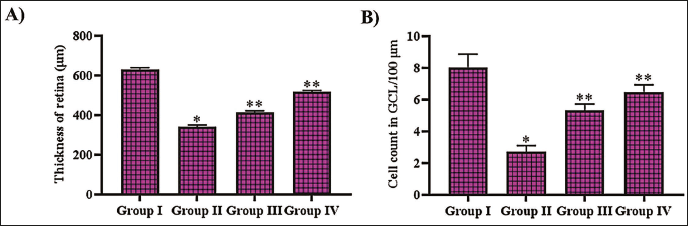
Noscapine Mitigated Retinopathy in Diabetic-triggered Rats
Figure 6 depicts the impact of noscapine against STZ-triggered retinopathy in experimental rats. The retinal thickness was significantly reduced to 310 ± 1.7 µm in the diabetic triggered, whereas the thickness of the control rat’s retina was 595 ± 2.5 µm. Noscapine and metformin treatment attenuated STZ-induced retinopathy, and the retinal thickness was exhibited to be 395 ± 2.4 and 480 ± 3.1 µm, respectively. The ganglion cell count was severely decreased in the STZ-administered untreated rats (2.7 ± 0.0006 GCL/100 µm) compared to the control rats (6.9 ± 0.0004 GCL/100 µm). 4.9 ± 0.0004 GCL/100 µm and 5.7 ± 0.0006 GCL/100 µm were counted in the STZ-administered rats treated with noscapine and metformin, respectively.
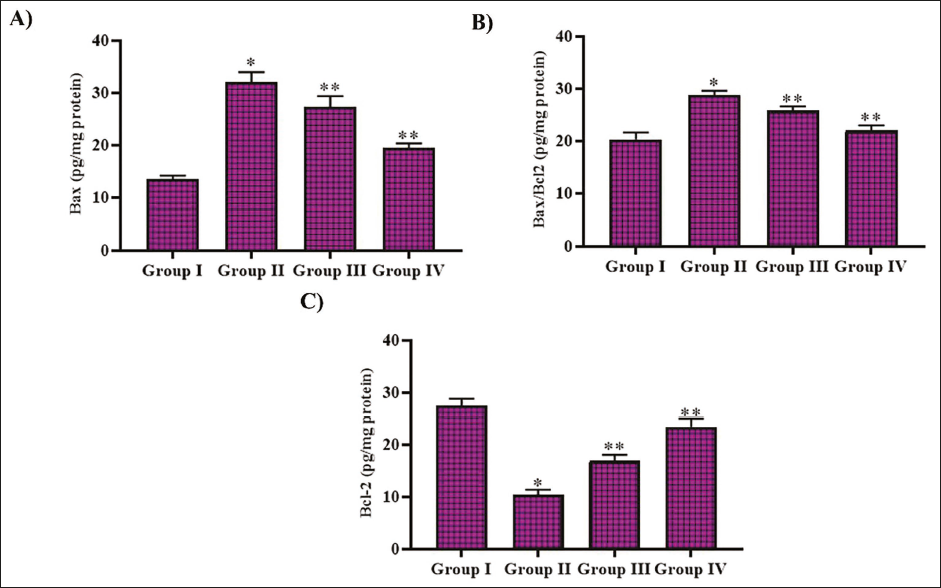
Noscapine Treatment Attenuated Apoptosis in DR-triggered Rats
The levels of pro-apoptotic protein Bax and anti-apoptotic protein Bcl2 were quantified in experimental animals, and the levels are illustrated in Figure 7. The levels of pro-apoptotic protein Bax and Bax/Bcl2 ratio were significantly enhanced to 27 ± 0.0005 protein and 28 ± 0.0007 pg/mg protein, respectively, with STZ administration, whereas treatment with metformin and noscapine decreased the Bax levels to 18 ± 0.0003 and 15 ± 0.0002 pg/mg protein, respectively, in the experimental rats. The apoptotic inhibitor protein Bcl2 levels were increased to 14 ± 0.0004 and 18 ± 0.0003 pg/mg protein with noscapine and metformin treatment, respectively, whereas untreated STZ-administered rats exhibited 12 ± 0.0007 pg/mg protein. Control rats exhibited 13 ± 0.0002 pg/mg protein of Bax, 26 ± 0.0004 pg/mg protein of BCl2, and 17 ± 0.0003 pg/mg protein of Bax/Bcl2.
Discussion
Noscapine is a benzylisoquinoline alkaloid naturally present in plants belonging to the Papaveraceae, Ranunculaceae, and Berberidaceae families (Karlsson et al., 1990; Rida et al., 2015). Despite being derived from alkaloid-rich sources like papaverine, noscapine exhibits minimal to no effects on the central nervous system and does not induce respiratory depression, euphoria, or addiction (Chen et al., 2015; Martindale, 1977). Traditionally used as a cough suppressant (Kumar et al, 2022; Segal et al., 1957), recent research has highlighted its potential as an anti-cancer agent due to its ability to inhibit tumor growth (Tomar et al., 2017). Noscapine has been shown to induce apoptosis and exert anti-tumor effects in both murine and human solid tumors (Altinoz et al., 2019; Zhou et al., 2002). Additionally, it has demonstrated neuroprotective properties, effectively reducing cerebral injury in neonatal rats subjected to hypoxic-ischemic conditions (Altinoz et al., 2022). Clinical findings also suggest that oral administration of noscapine significantly decreases mortality in ischemic stroke patients when administered within 12 h of symptom onset (Mahmoudian et al., 2003). Since noscapine possesses varied pharmacological properties and exerts nil or minimal toxicity, we investigated its efficacy against hyperglycemia-induced retinopathy.
Elevated glucose levels enhance glycolytic activity, resulting in increased production of pyruvate, nicotinamide adenine dinucleotide (NADH), and flavin adenine dinucleotide (FADH2), which contribute to oxidative stress in retinal neurons (Metea & Newman, 2006). These neurons exhibit heightened vulnerability due to their metabolic demands for glucose and oxygen, coupled with weak antioxidant defenses and limited regenerative capacity (Ola et al., 2012). Insulin resistance and hyperlipidemia further compromise cellular organelles, such as mitochondria and lysosomes, leading to the release of excessive ROS and pro-inflammatory enzymes (Ahmed et al., 2021; Stinson et al., 2024). This cascade results in neurovascular dysfunction, inflammation, and neuronal degeneration in the retinal cells (Tecce et al., 2024). To assess the mitigating potency of noscapine against DR, we created a rat model with STZ administration. STZ administration had successfully induced hyperglycemia and hypercholesterolemia in experimental animals. Noscapine treatment eventually prevented the induction of STZ-induced hyperglycemia and hypercholesterolemia, thereby attenuating free radical generation in the retinal cells. Hyperglycemia-induced oxidative stress plays a crucial role in enhancing lipid peroxidation in DR (Robles-Rivera et al., 2020). Furthermore, lipid peroxidation is positively correlated with both the severity and duration of diabetes (Gupta & Chari, 2005). Noscapine treatment enhanced the antioxidant system, which eventually protected the cells from hyperglycemia-induced lipid peroxidation in the hyperglycemia-triggered rats.
Chronic hyperglycemia induces oxidative stress, activates various signaling pathways, and promotes epigenetic modifications, leading to inflammation in DR. Elevated levels of pro-inflammatory cytokines, including TNF-α, IL-1β, IL-6, and MCP-1, have been detected in the vitreous, serum, and retinal tissues of DR patients (Rubsam et al., 2018). TNF-α, a key mediator of inflammatory responses, is associated with increased disease susceptibility due to genetic polymorphisms (Demircan et al., 2006). Additionally, IL-1β and its downstream effector, caspase 1, are upregulated in diabetic retinas, while IL-6 levels correlate with DR progression (Eguchi & Nagai, 2017; Kowluru & Odenbach, 2004; Vincent & Mohr, 2007). TNF-α contributes to retinal vascular permeability; however, pharmacological interventions, such as PKCζ inhibition and TNF-α blockade with Etanercept, effectively prevent blood-retinal barrier disruption and mitigate inflammation in experimental models of diabetes (Aveleira et al., 2010; Joussen et al., 2002). In our analysis, noscapine treatment in hyperglycemia-triggered rats effectively attenuated the levels of TNF-α, IL-1β, and IL-6 and thereby prevented the rats from diabetic-induced retinal damage.
NF-κB activation is a key pro-inflammatory and proapoptotic signaling pathway that mediates hyperglycemia-induced inflammation and cellular apoptosis (Moscat & Diaz-Meco, 2009; Oeckinghaus et al., 2011). As a redox-sensitive nuclear transcription factor, NF-κB critically regulates inflammatory responses while also repressing antioxidant enzymes (Nakajima & Kitamura, 2013). It has been shown that NF-κB triggers proapoptotic processes in retinal pericytes under high glucose conditions (Romeo et al., 2002). Moreover, NF-κB can activate MMP-9, which eventually damages the mitochondrial gap junction protein, enhancing pore permeability and facilitating the leakage of cytochrome c into the cytosol (Chen et al., 2019; Kowluru & Mishra, 2015). STZ administration in the experimental rats activated NF-κB and triggered apoptotic signaling, whereas treatment with noscapine attenuated NF-κB and pro-apoptotic protein Bax thereby preventing retinal cells from hyperglycemia-induced cellular death. Noscapine treatment also inhibited MCP-1 and MMP-9 proteins and mitigated hyperglycemia in rats from blood–retinal–brain breakdown and vascular cell degeneration.
Clinical trials have established the effectiveness of intravitreal anti-VEGF agents in managing DR. While evidence supports the use of repeated intravitreal anti-VEGF injections to control the progression of proliferative DR, the associated follow-up requirements and costs remain considerable (Bahr et al., 2023). Additionally, systemic administration of anti-VEGF drugs in cancer therapy has been linked to an increased risk of hypertension (An et al., 2010), thromboembolic events (Scappaticci et al., 2007), myocardial ischemia (Ranpura et al., 2010), and impaired wound healing (Scappaticci et al., 2005). Noscapine, the bioactive compound, may be a potent alternative for currently available anti-VEGF drugs in treating DR. Noscapine treatment had effectively inhibited the VEGF synthesis, thereby preventing retinal cell degeneration in hyperglycemic-triggered rats. The ameliorative effect of noscapine was confirmed with our retinal histopathological analysis, which exhibited increased retinal thickness and RGC count.
Conclusion
This study demonstrates noscapine’s therapeutic potential in DR. In a STZ-induced diabetic rat model, noscapine effectively reduces hyperglycemia and hypercholesterolemia while enhancing antioxidant defenses and minimizing oxidative stress. It also suppresses inflammation by lowering VEGF, MCP-1, and MMP-9 levels. Histopathological analysis confirms its protective role against retinal damage. These findings suggest that noscapine could be a promising treatment for DR, complementing existing therapies. Further research is needed to explore its clinical applications and underlying mechanisms.
Footnotes
Abbreviations
AGEs: Advanced glycation end products; ANOVA: Analysis of variance; Bax: BCL-2 associated X protein; Bcl2: B-cell lymphoma 2; CAT: Catalase; DR: Diabetic retinopathy; ELISA: Enzyme-linked immunosorbent assay; FADH2: Flavin adenine dinucleotide; GBD: Global Burden of Disease; GCL: Ganglion cell layer; GPx: Glutathione peroxidase; GR: Glutathione reductase; GSH: Reduced glutathione; GSSG: Oxidized glutathione; HDL: High-density lipoprotein; H&E: Hematoxylin and eosin; H2O2: Hydrogen peroxide; IDF: International Diabetes Federation; IGF-1: Insulin-like growth factor-1; IL-1β: Interleukin-1 beta; IL-6: Interleukin-6; LDL: Low-density lipoprotein; MCP-1: Monocyte chemoattractant protein-1; MDA: Malondialdehyde; MMP-9: Matrix metallopeptidase 9; NADH: Nicotinamide adenine dinucleotide; NADPH: Nicotinamide adenine dinucleotide phosphate; NF-κB: Nuclear factor kappa B; PKC: Protein kinase C; RAAS: Renin–angiotensin–aldosterone system; RGCs: Retinal ganglion cells; ROS: Reactive oxygen species; SOD: Superoxide dismutase; SPSS: Statistical Package for the Social Sciences; STZ: Streptozotocin; TBA: Thiobarbituric acid; TNF-α: Tumor necrosis factor-alpha; VEGF: Vascular endothelial growth factor.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This work was approved by the Institutional Ethical Committee Xi’an Aier Eye Hospital, Xi’an, Shaanxi 710000, China.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
