Abstract
Background
Polygonatum polysaccharide has certain limitations in drug delivery and cellular uptake. To overcome these problems, polystyrene nanoparticles serve as an effective carrier to improve its bioavailability and anti-oxidant effects.
Objectives
To this end, we explored the role of polystyrene nanoparticle-mediated polysaccharide complex in liver cancer.
Materials and Methods
Polygonatum sibiricum polysaccharide-polystyrene (PSP-PS) nanomaterials were prepared, and liver cancer mouse models were constructed. Antioxidant enzymes and proteins in the mouse spleen and thymus were detected. The levels of reactive oxygen species (ROS), superoxide dismutase (SOD), glutathione (GSH), malondialdehyde (MDA), and total antioxidant capacity (T-AOC) in the mouse spleen and thymus were detected. Western blot detects the expression levels of p38, JNK, and ERK1/2 proteins in the mitogen-activated protein kinase (MAPK) signaling pathway; it also detects the expression levels of Keap1, Nrf-2, and HO-1 proteins in the Nrf-2 and HO-1 signaling pathways.
Results
PSP-PS nanomaterials were successfully prepared, and a liver cancer mouse model was successfully constructed. Under the intervention of PSP-PS, MDA in the spleen and thymus of mice decreased, SOD in the spleen and thymus increased, T-AOC in the spleen increased, GSH in the spleen decreased, GSH in the thymus increased, Keap1 in the spleen decreased, Keap1 in the thymus increased, and Keap1 in the spleen and thymus increased. Nuclear factor erythroid 2-related factor 2 (Nrf2) and heme oxygenase 1 (HO-1) were both elevated in the thymus. p38, JNK, and ERK protein levels in the MAPK signaling pathway were significantly reduced.
Conclusion
PSP-PS upregulates SOD expression by inhibiting the MAPK/Nrf2-HO-1 signaling, thereby protecting against oxidative damage to organs in mice with liver cancer.
Keywords
Introduction
Liver cancer has attracted widespread attention due to its difficult treatment and low cure rate. The current level of treatment is still difficult to meet people’s expectations for treatment results, so it is of great significance to study better and more effective treatment methods. Research has found that Polygonatum polysaccharide may inhibit the formation of new blood vessels in tumors (angiogenesis is one of the key processes in tumor growth), thus preventing the blood supply and nutrition to tumors (Xu et al., 2023). In addition, Polygonatum polysaccharide can inhibit the inflammatory response by regulating the mitogen-activated protein kinase (MAPK) pathway, reduces the production of inflammatory factors in the tumor microenvironment, and thereby inhibits the development of liver cancer (Zhou et al., 2022). Moreover, Polygonatum polysaccharide can inhibit its kinase activity by interacting with MAPK kinase, thereby reducing the activity of the MAPK signaling pathway (Zhang et al., 2019). However, the specific interaction mechanism between Polygonatum sibiricum polysaccharides and MAPK is still unclear. Further in-depth research is needed on its potential in cancer treatment.
Studies have found that, in liver cancer, the MAPK pathway can be activated by growth, inflammatory, and other stimulating factors, thereby promoting cell proliferation, migration, invasion, and inhibiting apoptosis. Moreover, the abnormally activated MAPK pathway is related to the invasion and metastasis of tumor cells, allowing cancer cells to penetrate tissue boundaries and migrate to other sites (Wang, Feng, et al., 2022). In addition, some studies have pointed out that the activation of MAPK members, such as ERK, JNK, and p38, can promote the activity of nuclear factor erythroid 2-related factor 2 (Nrf2), thereby regulating the antioxidant and detoxification responses of cells (Ma et al., 2021). High-level Nrf2 activation may make cancer cells more able to adapt to stress conditions in the tumor microenvironment, thereby promoting the survival and proliferation of cancer cells (He et al., 2020). Additionally, Nrf2 activation may also cause tumor cells to become resistant to chemotherapy drugs and radiotherapy (Sánchez-Ortega et al., 2021). In addition, some studies have shown that activating Nrf2 can upregulate heme oxygenase 1 (HO-1), and Nrf2 and HO-1 have a synergistic effect in cells in resisting oxidative stress and inflammation (Wei et al., 2021). Therefore, it is critical to assess the regulation of Polygonatum polysaccharide in treating liver cancer by regulating the MAPK/Nrf2-HO-1 signaling pathway.
Multiple studies have revealed the close relationship between superoxide dismutase (SOD) expression and oxidative damage to organs (Ding et al., 2019; Guo et al., 2019; Huo et al., 2021). In disease states such as heart disease, liver disease, and kidney disease, the expression level of SOD can be significantly reduced, leading to the occurrence and aggravation of oxidative damage, causing harmful free radicals such as hydrogen peroxide (H2O2) to accumulate in tissues, thereby affecting the integrity of cell membranes, cell metabolism, regular progress, and cell viability (Lin et al., 2019). On the other hand, some studies have shown that by promoting the synthesis of SOD or increasing its activity, cells’ resistance to oxidative stress can be enhanced, thereby reducing the generation of oxides and the degree of tissue damage (Balendra & Singh, 2021). However, the relationship between SOD and oxidative damage to organs is not a simple cause-and-effect relationship. In addition to changes in SOD expression levels, many other factors, such as the production rate of free radicals and the activities of other antioxidant enzymes, can also affect the development of oxidative damage to organs. Although SOD is related to oxidative damage to organs, its specific mechanism is unclear, and it requires further analysis.
Polygonatum polysaccharide is easily degraded in the body and is unstable under some environmental conditions, such as light and humidity. Wrapping Polygonatum polysaccharides in polystyrene nanoparticles can protect the drug from the external environment’s influence and prolong the drug’s stability. Moreover, polysaccharide acts as a protective layer for the drug inside the polystyrene nanoparticles, preventing the drug from prematurely degrading in the body. At the same time, the structure of nanoparticles can also control the release rate of drugs and achieve sustained drug release, thereby improving drug efficacy and reducing drug side effects. This study provides a theoretical basis for the development of new antioxidant therapy strategies and opens up new paths for further exploration of efficient and low-toxicity anti-cancer therapies. In addition, the study also suggests the potential application of polystyrene nanoparticles as drug carriers in cancer treatment, especially in improving the stability and efficacy of traditional herbal ingredients, and it shows broad prospects.
Materials and Methods
Instruments and Reagents
Polygonatum polysaccharide (Chuzhou Shinuoda Biotechnology); specific pathogen-free (SPF) clean-grade mice (Shenzhen Haodi Huatuo Biotechnology); polystyrene nanoparticles (Zhongke Leiming (Beijing) Technology); fetal bovine serum (Yizefeng Biotechnology, Shanghai)); Nrf2 antibody (Yaibo (Wuhan)); β-catenin antibody (Beijing Biolab), goat anti-rabbit IgG (Beijing Borsi); dual luciferase (Beijing Xishengyuan Biotechnology); Transwell chamber (Bensen (Tianjin) Health); hematoxylin and eosin (H&E) staining kit (Shanghai Wenshi Biotechnology); polymerase chain reaction (PCR) primers and kits (Guangzhou Ruibo Biotechnology); ribonucleic acid (RNA) extraction kit (Shanghai Haifang Biotechnology).
Preparation of Polygonatum sibiricum Polysaccharide-Polystyrene (PSP-PS) Nanoparticles
Dissolve polystyrene (20 mg/mL) in xylene, add 2.312 g, 10% polyvinylpyrrolidone (PVP), and stir thoroughly to dissolve the polystyrene evenly. The polystyrene solution is added dropwise to the water phase to form an emulsion. Dissolve the polystyrene monomer in the organic solvent, add 5% cetyltrimethylammonium bromide (CTAB) for mixing, and slowly add the ethanol solution dropwise while stirring until the polystyrene forms particles and precipitates. A centrifuge is used to precipitate and separate polystyrene nanoparticles, and the nanoparticles are washed and purified with an appropriate amount of absolute ethanol to obtain polystyrene nanoparticles. Then, slowly add the Polygonatum polysaccharide extract to the polystyrene precursor solution and stir thoroughly. Put the mixed solution into an ultrasonic cleaner for 24 h to stabilize the combination of the two. Finally, the mixed solution is appropriately concentrated and purified to remove the solvent. Moreover, impurities to obtain polystyrene nanoparticles carrying Polygonatum polysaccharide.
Nano Characterization
Dip a small amount of the nanoparticle dispersion liquid and evenly drop it on the conductive adhesive. After natural drying, spray gold for 1 min, and then observe the morphology of the nanoparticles with a transmission electron microscope (TEM).
Liver Cancer Mouse Modeling and Liver Cancer Mouse Organ Oxidative Damage Modeling
Fifteen healthy male mice were randomly assigned to five groups (n = 3 in each group). The control group did not receive any treatment. The rest used diethylnitrosamine (DEN) to induce mice and successfully constructed a liver cancer mouse model. By controlling the diet of mice, they suffer from malnutrition, vitamin deficiency, or protein deficiency, which leads to oxidative damage to the liver. The mouse organs were tested, and it was found that all liver cancer mice had oxidative damage, indicating that the organ oxidative damage modeling of liver cancer mice was successfully constructed (Xue et al., 2022).
Group Intervention
Mice that were successfully modeled were assigned to the negative control group (NC group), PSP group, PS group, and PSP-PS group, with three mice in each group. The PSP group was treated on the 1st, 3rd, 6th, 9th, and 13th days after vaccination. PSP was injected intraperitoneally at 3 mg/kg. The PS and PSP-PS groups were given 3 mg/kg PS and 3 mg/kg PSP-PS, respectively, at the same time. The NC group was given 1 mL/100 g body weight of sterile normal saline. The other three healthy mice were healthy controls (control group) and fed normally.
Biochemical Marker Measurement
SOD activity was detected. Malondialdehyde (MDA), formed by the decomposition of polyunsaturated fatty acids, can be used as the peroxide-absorbance value to calculate the concentration.
Analysis of Reactive Oxygen Species (ROS), SOD2, and Glutathione Peroxidase 1 (GPX1)
Optimal cutting temperature (OCT) embedded tissues were fixed with 4% paraformaldehyde for 30 min. Acquire images by fluorescence microscopy (200× magnification). Use a detection kit to measure tissue/cell H2O2. Analyze glutathione (GSH) using the GSH-Glo™ GSH assay kit.
Organ Coefficient Detection
After the mice were sacrificed, the blood stasis, fat, and connective tissue on the surface of each organ were removed. Cut the tissue along the midline, wash the contents with physiological saline, absorb the blood or liquid on the surface of each organ with paper, and then weigh it. Organ coefficient = organ weight/body weight ×100%.
Quantitative Reverse Transcription Polymerase Chain Reaction (qRT-PCR) Detection
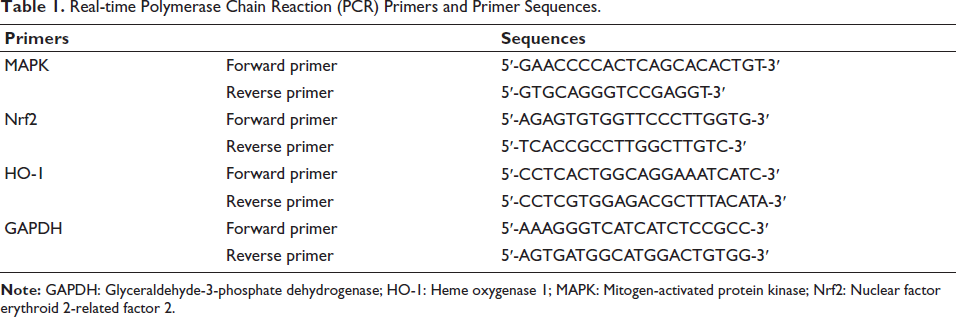
Total RNA from mouse tissue cells in each group was collected using the TRIzol method and then reverse-transcribed into complementary DNA (cDNA). Reverse transcription quantitative polymerase chain reaction (RT-qPCR) was performed using SYBR fluorescent PCR technology. Using glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as an internal control, the qRT-PCR results were analyzed. Relative levels were estimated using the 2−∇∇Ct method. Table 1 lists the primers and their sequences.
Real-time Polymerase Chain Reaction (PCR) Primers and Primer Sequences.
Western Blot Detection
Bicinchoninic acid (BCA) method determination, gel loading, electrophoresis, and membrane transfer. Blocked with skim milk powder and incubated with corresponding primary antibodies (MAPK, Nrf2, GAPDH, 1:1,000) overnight, and with secondary antibodies (goat anti-rabbit 1:2,000) for 2 h. GAPDH served as an internal reference. After interaction with enhanced chemiluminescence reagents, the blots are analyzed by software.
H&E Pathology of Thymus Tissue
Tissues were removed from the lesions. They were prepared by paraffin sectioning, H&E staining, and eosin acid staining. The pathological changes of the tissues were observed under a microscope through the staining of cell nuclei, cytoplasm, and cell matrix.
Sirius Red Staining
Take the liver tissue of the model mouse. In short, the cell samples are stained and fixed, and the fixed cells are put into Sirius red staining solution. After staining for a certain period of time, the cells are washed with a wash buffer and observed under a microscope.
Statistical Analysis
The data obtained in each of the above experiments were analyzed using Statistical Package for the Social Sciences (SPSS) 21.0 and GraphPad Prism software. If there are no special requirements, p < .05 is used as the test standard.
Results
Successful Preparation of PSP-PS Nanoparticles
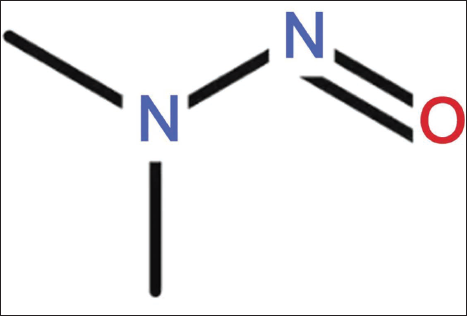
The molecular structure formula of DEN is shown in Figure 1. We successfully prepared PSP-PPS, which has a regular circular shape with a specific number distribution size (611.72 ± 11.27) d.nm, which indicates the stability of the nanoparticles (Figure 2).
Diethylnitrosamine (DEN) Molecular Structure Formula.
Polygonatum sibiricum Polysaccharide-Polystyrene (PSP-PS) Nano.

Successful Modeling of Liver Cancer in Mice
The hepatocellular carcinoma (HCC) mouse model was induced by intraperitoneal injection of DEN three times/week for a total of 8 weeks. The NC group was given the same dose of 0.9% NaCl.
It was shown that the liver in the model group showed atypical lesions at 4 weeks, with more tumor lesions and bulges visible on the surface (Figure 3A). The cancer tissue was a typical cord-like invasion of the normal tissue of the liver (Figure 3B), and the cancer cells were numerous. After PSP-PS intervention, a decrease in tumor cell density was observed (Figure 3C). As shown by Tianliangxing staining (200×), the liver showed obvious fibrosis (Figure 3D), which progressed to portal vein fibrosis at 8 weeks (Figure 3E).
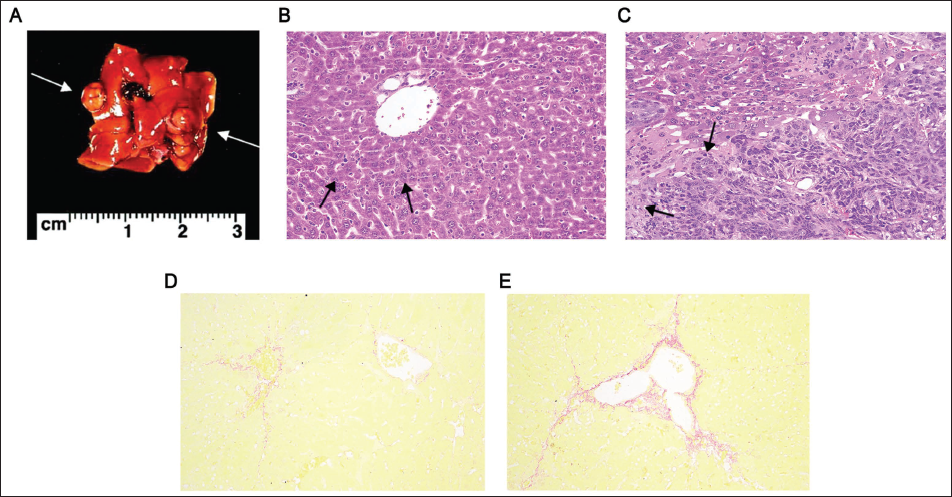
PSP-PS can Improve Liver Cancer Mouse Modeling, and MAPK/Nrf2-HO-1 is Involved in the Process
We injected PSP-PS into the resulting liver cancer mouse model and used drugs to intervene and group them. Levels of serum IgG and IgM were increased under PSP intervention (Figure 4A–4B), along with elevated spleen SOD levels and thymus ROS (Figure 4C–4D). Further Western blot testing found that the expression of P-p38 and p38 was significantly increased (Figure 4E); the expression of P-ERK and ERK proteins in the spleen was reduced, and P-JNK/JNK was decreased (Figure 4F–4G). Under PSP-PS intervention, this effect was more significant. It shows that PSP-PS can improve the oxidative damage of organs in mice with liver cancer, and the MAPK/Nrf2-HO-1 is involved in this process.
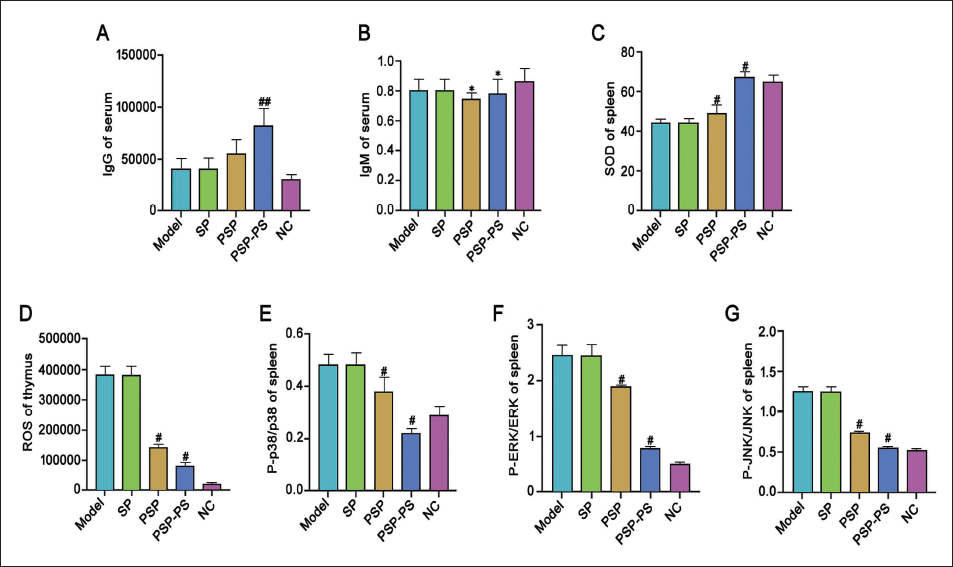
PSP-PS Upregulates SOD Expression and Protects Organs from Oxidative Damage in Liver Cancer Mice
In terms of organ coefficients, compared with other model groups, the coefficients of the liver, kidney, and spleen of the PSP-PS group were the lowest (p < .05), which were still higher than the NC group, suggesting that PSP-PS is beneficial in alleviating organ damage in mice. Still, it also shows that PSP-PS has positive biological effects (Table 2).
Mouse Organ Coefficients.
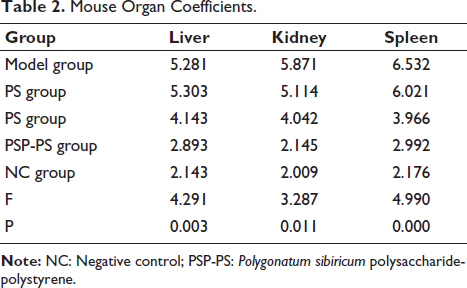
PSP-PS Upregulates SOD Expression and Protects Organs from Oxidative Damage in Liver Cancer Mice
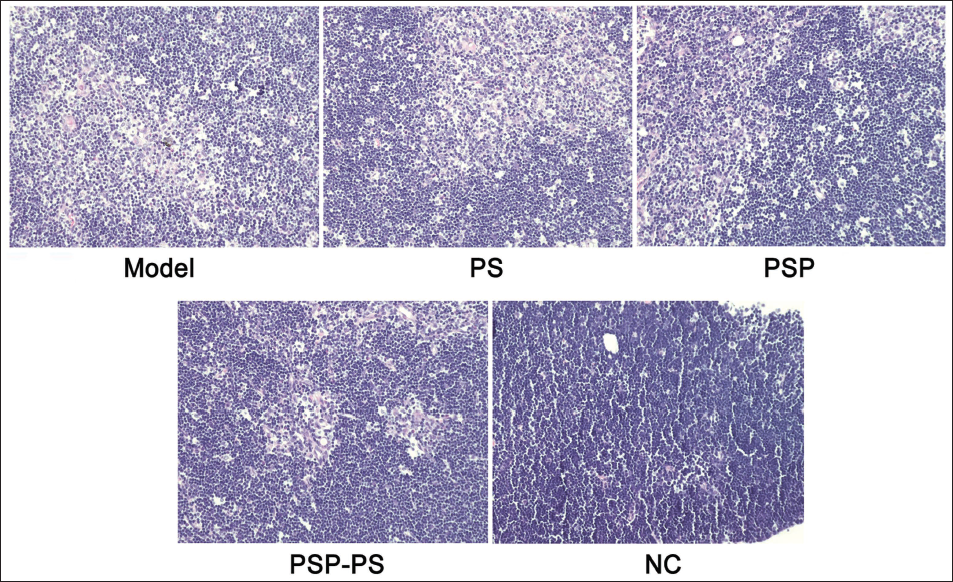
We tested the spleens of mice and found that under the intervention of SP, PSP, and PSP-PS, compared with the model group, MDA and GSH levels in the PSP group were significantly reduced, and SOD and total antioxidant capacity (T-AOC) levels increased, indicating that PSP has good biological effects; while no change was found in the SP group; the PSP-PS group had the most significant change, which shows that the nanocomposite actively protects organ immunity. It points out further research directions for our in-depth study of the mechanism of PSP-PS. The PSP-PS group showed a significant protective effect against thymus damage. Compared with the model group, its thymocytes and thymocytes increased, the phagocytosis of macrophages was weakened, and the thymus damage was alleviated (p < .05). The PSP group: Second, no macrophages were seen under the microscope in the NC group, and scattered thymic corpuscles were visible. Thymic epithelial cells were densely distributed, and the local cortex and medulla in the tissue were visible. It shows that PSP-PS can protect the organs from oxidative damage in liver cancer mice by upregulating SOD expression (Figures 5 and 6).
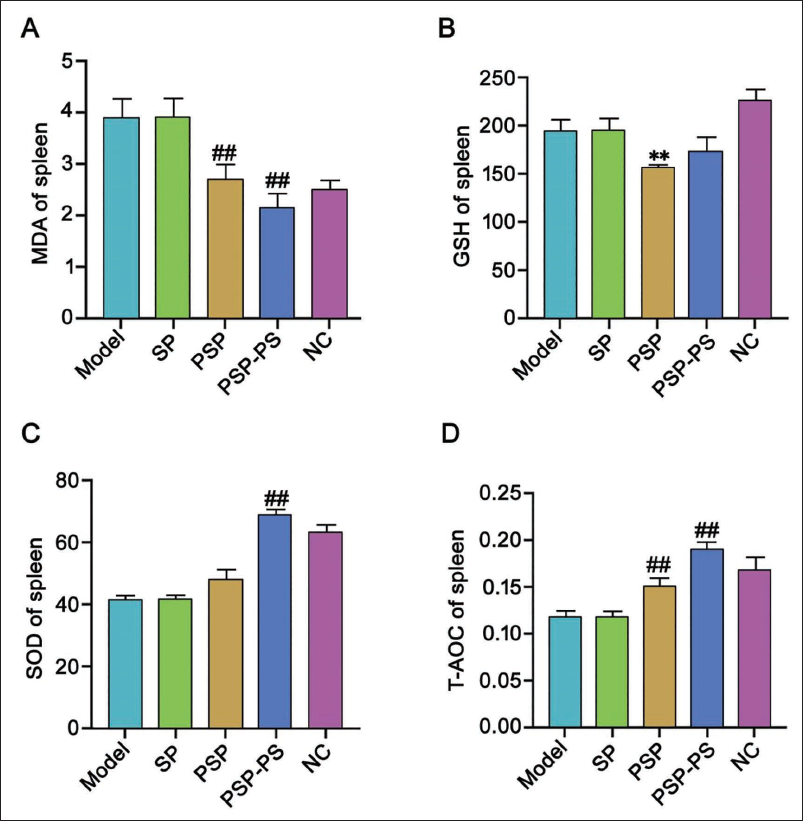
Polygonatum sibiricum Polysaccharide-Polystyrene (PSP-PS) Improves Thymus Tissue in Mice with Liver Cancer [Hematoxylin and Eosin (H&E), ×200].
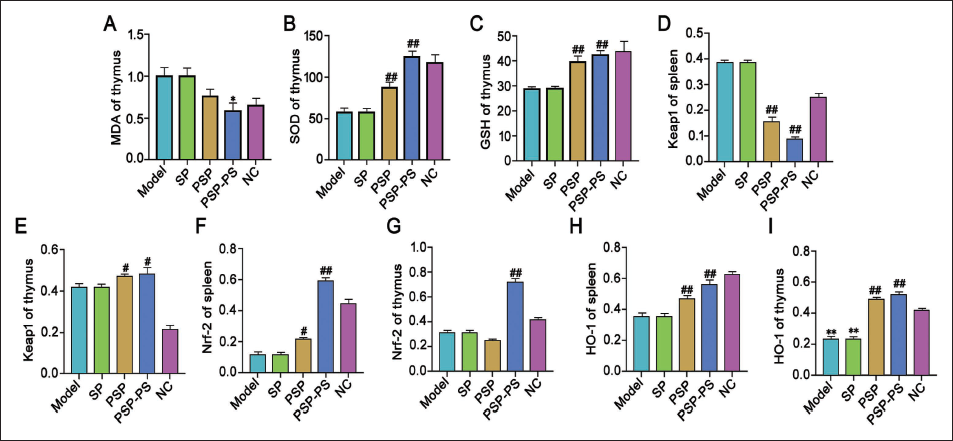
MAPK/Nrf2-HO-1 Signaling Pathway is Involved in the Process of PSP-PS Protecting Organs from Oxidative Damage in Mice with Liver Cancer
MAPK/Nrf2-HO-1 signaling may be involved in PSP-PS. We observed the protein change trends related to oxidative damage in the mouse thymus and found that under PSP intervention, MDA decreased, GSH increased, and SOD increased, and the effect of PSP-PS was the most significant. This result is encouraging. As the above results indicate, our further protein detection results also showed a similar trend, and Keap1 in the spleen was reduced, Keap1 in the thymus was increased, and Nrf2 and HO-1 were both increased in the spleen and thymus. Interestingly, this was still the case in the PSP-PS group. The changes are the most obvious. It illustrates that the MAPK/Nrf2-HO-1 signaling is involved in PSP-PS’s role in protecting organs from oxidative damage in mice with liver cancer. Specifically, it inhibits phosphorylation of p38, JNK, and ERK in the MAPK signaling, while activating the Nrf-2/HO-1 oxidative stress defense signaling pathway, thereby enhancing the body’s antioxidant capacity (Figure 7).
Discussion
Polygonatum polysaccharide has a positive effect in improving oxidative damage, but it has high biochemical activity and biodegradability, and low solubility in water, resulting in limited bioavailability (Wang, Fu, et al., 2021). In addition, based on the above advantages, we successfully prepared PSP-PS nanocomposites. In order to clarify the effect of PSP-PS on liver cancer, through the detection and change results of various index levels in the thymus and spleen of mice, it was found that PSP can improve the oxidative damage of organs in mice with liver cancer, especially under the intervention of PSP-PS. The protective effect is more significant, mainly because polystyrene nanoparticles serve as carrier materials, which can protect Polygonatum polysaccharide from environmental conditions and improve its stability, thus enhancing the storage and delivery capabilities of the drugs (Baird & Yamamoto, 2020). It also increases the surface area of Polygonatum polysaccharide in contact with water, improves its solubility in vitro and in vivo, and enhances the dissolution kinetics of the drug. In addition, during this process, p38, JNK, and ERK levels in the MAPK signaling pathway were significantly reduced, suggesting that the MAPK/Nrf2-HO-1 is involved in the process of PSP-PS improving oxidative damage in mice with liver cancer. Analysis suggests that AP-1 can interact with transcription factors such as Nrf2 and FoxO to form a transcription activation complex, thereby changing the chromatin structure and making the promoter region of the SOD gene more accessible to transcription machinery such as RNA polymerase, thereby increasing the transcriptional activity of SOD genes to combat intracellular oxidative stress (Wang, Botchway, et al., 2022). In addition, astragalus can affect the gene expression level of host cells, thereby regulating the activity of Nrf2 and AP-1, increasing the expression level of SOD messenger ribonucleic acid (mRNA), increasing the synthesis of SOD, increasing the activity of SOD, and enhancing the cells’ response to oxidative stress (Yang et al., 2023).
To explore the specific mechanism of PSP-PS in protecting organs from oxidative damage in mice with liver cancer, we found that under the action of PSP, MDA and GSH decreased, and SOD and T-AOC increased. Under the intervention of PSP-PS, this change showed a more consistent trend. It shows that PSP-PS can upregulate SOD expression and protect the organs from oxidative damage in liver cancer mice. This is because Polygonatum polysaccharide can directly interact with Kea1 or Nrf2 (Xu et al., 2022), changing the interaction between them, causing Keap1 to be unable to effectively promote the ubiquitination and degradation of Nrf2, thereby causing Nrf2 to accumulate (Li et al., 2019), gradually and increasing its binding with antioxidant response element (ARE) to upregulate antioxidant enzymes and detoxification enzymes such as SOD and catalase (CAT) to resist oxidative stress (Lai et al., 2022). In addition, Polygonatum polysaccharide can directly bind to IKK and prevent its phosphorylation of the IκB protein, thereby preventing NF-κB activation, reducing the release of inflammatory mediators such as TNF-α, IL-1β, and thus protecting cells from oxidative damage (Ray et al., 2024). The targeted delivery of polystyrene nanoparticles can increase the enrichment of Polygonatum polysaccharide in diseased areas, reduce the adverse effects on normal tissues, and enhance the therapeutic effect (Wang, Hu, et al., 2021). In addition, another study found that under the stimulation of oxidative stress, MAPK is activated, and Nrf2 enters the nucleus to initiate the HO-1 antioxidant response and promote the cell’s resistance to oxidative stress (Deng et al., 2020). In addition, when Nrf2 binds to ARE, it interacts with co-activator proteins such as CBP and p300 and initiates the transcription of antioxidant response genes such as SOD and GST by recruiting transcription factors and co-activator proteins, thereby enhancing intracellular antioxidant activity (Jahan et al., 2021). We tested the thymus and spleen of mice and found that under PSP intervention, MDA in the thymus decreased, and GSH and SOD increased. Keap1 was increased in the thymus and decreased in the spleen, and Nrf2 and HO-1 were both increased in the spleen and thymus. And the intervention effect of PSP-PS is more obvious than that of PSP. This indicates that the MAPK/Nrf2-HO-1 signaling is involved in PSP-PS’s role in protecting organs from oxidative damage in liver cancer mice. However, more detailed experiments are currently needed to study the chemical properties, pharmacokinetics, and interactions with tumor cells of Polygonatum polysaccharide, as well as to conduct more detailed research on the biocompatibility, biodistribution, metabolism, and long-term safety of nanoparticles. We have explored the mechanism in vivo, but whether we can show the same results in vitro needs more verification. Next, we will elaborate on the cell line.
Conclusion
In summary, PSP-PS upregulates SOD expression by inhibiting the MAPK/Nrf2-HO-1 signaling, thereby protecting against oxidative damage to organs in mice with liver cancer. This study will explore new strategies for the antioxidant treatment of liver cancer and provide a theoretical basis for further developing effective antioxidant treatments.
Footnotes
Abbreviations
GSH: Glutathione; HO-1: Heme oxygenase 1; MAPK: Mitogen-activated protein kinase; MDA: Malondialdehyde; Nrf2: Nuclear factor erythroid 2-related factor 2; PSP: Polygonatum sibiricum polysaccharide; ROS: Reactive oxygen species; SOD: Superoxide dismutase; T-AOC: Total antioxidant capacity.
Acknowledgments
The authors gratefully acknowledge the Pingyang Hospital Affiliated to Wenzhou Medical University for providing the necessary equipment for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethics Committee of Pingyang Hospital Affiliated to Wenzhou Medical University.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
