Abstract
Background
Bupleurum is an active ingredient extracted from the Chinese medicine bupleurum, which has been proven to have various biological effects, such as anti-tumor, anti-inflammatory, and immune regulation. However, its application is limited by factors such as low bioavailability and poor stability. In addition, extracellular vesicle secretion by liver cancer cells is involved in the formation and progression of liver cancer. The p38 mitogen-activated protein kinase (MAPK) and interleukin (IL)-6 pathways are two signaling pathways related to the development of liver cancer.
Objectives
By modifying SiNPs and bupleurum, we tried to inhibit these two signaling pathways to achieve a therapeutic effect on liver cancer.
Materials and Methods
HepG2 cells were cultured and used in group experiments to explore how bupleurum regulates the p38 MAPK/IL-6 pathway and exerts inhibition by using SB202190, p38 MAPK agonists, and inhibitors and agonists of IL-6 in liver cancer.
Results
SiNPs-SS has a stronger inhibitory effect on liver cancer than SS and inhibits the secretion of extracellular vesicles and progression of liver cancer by inhibiting the p38 MAPK/IL-6 pathway.
Conclusion
SiNPs-SS inhibits the secretion of extracellular vesicles and the progression of liver cancer by inhibiting the p38 MAPK/IL-6 pathway.
Introduction
China currently relies mainly on surgical resection combined with drug-assisted therapy to deal with cancer. However, these methods have high recurrence rates, significant drug side effects, and low cure rates (Man et al., 2021), and there is an urgent need to explore new treatment methods and drugs. Extracellular vesicles (EVs) released by cancer cells are rich in bioactive molecules. They play an important role in promoting the development of cancer by interacting with target cells to transmit signals. For example, according to the study by Wu et al. (2022), EVs regulate the tumor microenvironment in liver immunity and therapy; Wang et al. (2023) pointed out that EVs in fatty liver can promote the development of a metastatic tumor microenvironment, specifically manifested as an increasing trend of 1209.e1213–1226.e1213. In addition, Tan et al. (2023) emphasized the application of EVs in tumor diagnosis and treatment, demonstrating their potential as therapeutic targets. At the same time, some EVs can inhibit the killing effect of immune cells and help liver cancer cells escape from immune surveillance. As Liu, Chen, et al. (2022) and Liu, He, et al. (2022) stated, in pancreatic cancer, EVs promote immune escape through various mechanisms. Therefore, studying how to effectively inhibit the secretion of EVs by cancer cells is of great significance for cancer treatment.
Chaihu Su, as an active ingredient extracted from the traditional Chinese medicine Chaihu, has been proven to have various biological effects in anti-tumor, anti-inflammatory, and immune regulation. However, its application effect is limited due to its low solubility and easy degradation
p38 MAPK is closely related to various cancers. It exerts different effects on different types of tumors by regulating key signaling pathways such as cell proliferation, apoptosis, invasion, and angiogenesis (Nie et al., 2019, 2022; Xin et al., 2022). In addition, p38 MAPK can regulate the DNA repair ability, drug efflux pump expression, and apoptosis pathway of tumor cells, leading to resistance to anti-cancer drugs (Colomer et al., 2019; Sanz-Ezquerro & Cuenda, 2021). Moreover, p38 MAPK inhibits the function of immune cells by regulating the tumor microenvironment, affecting the ability of tumor cells to evade immune recognition and attack (Han et al., 2019). Given that p38 MAPK signaling can activate the production of interleukin (IL)-6 (Natani et al., 2022), which not only promotes inflammatory responses, stimulates immune cells, and releases inflammatory cytokines, but also affects the cell cycle and cell survival by activating signaling pathways such as JAK/STAT and PI3K/AKT, promoting tumor growth and spread (Hirano, 2021), p38 MAPK and IL-6 are considered potential markers in tumors. It is particularly important to investigate the relationship between Chaihu and p38 MAPK and their potential in cancer treatment as therapeutic targets.
To improve the water solubility and bioavailability of Chaihu Su, silica nanoparticles were used for modification, which not only increased the solubility of the drug but also made the nanoparticles more easily absorbed by specific cells or tissues, increased the drug concentration in the target area, and reduced the impact on normal tissues (Jiang et al., 2022; Liu et al., 2019; Zhu et al., 2022). At the same time, by modifying the nanoparticles with bupivacaine, the behavior of the drug molecules was changed, reducing the impact on normal cells and reducing toxic side effects. Therefore, using silica nanoparticles to modify Chaihu Su provides a new idea and technical means for cancer treatment.
Materials and Methods
Instruments and Reagents
The following instruments and reagents were used: human liver cancer HepG2 cells (Shanghai Hanbo Biotech); bupleurum (Shanghai Jizhi Biochemistry); silica nanoparticles (Shanghai Xikerui Biotech); vascular endothelial growth factor (VEGF) immunohistochemistry kit (Shanghai Yaji Biotech); epidermal growth factor receptor (EGFR) immunohistochemistry reagent kit (Shanghai Caiyou Industrial); Cell Counting Kit (CCK)-8 kit (Yisheng Biotechnology (Shanghai)); Transwell chamber (Beijing Xinshengyuan Biotechnology); flow cytometer (Shanghai Ranzhe Instrument Equipment); microscope (Shanghai Fu Lai Optical Technology); transmission electron microscope (Wuxi Bohe Biotechnology); polymerase chain reaction (PCR) primers and kits (Guangzhou Ruibo Biotechnology) and ribonucleic acid (RNA) extraction kit (Shanghai Haifang Biotechnology).
Preparation of SiNPs-SS
Add an appropriate amount of SS to distilled water, stir to fully dissolve, and obtain an SS solution with a concentration of 20 mg/mL. Then, add the silica precursor ethyl orthosilicate to 30 mL of ethanol, stir, and perform ultrasonic treatment to form a uniform silica precursor solution. Add an appropriate amount of catalyst (such as ammonia or ammonium nitrate) dropwise to the silica precursor solution while continuing to stir and keeping the reaction temperature within a suitable range. Slowly add the SS solution dropwise to the silica precursor solution, stir thoroughly while adding, and mix evenly. During this process, the silica precursors will gradually aggregate to form nanoparticles, and the solution is allowed to stand for a while until the SS is successfully wrapped inside the SiNPs. Unreacted substances were removed by ultracentrifugation to obtain SiNPs-SS.
Culture and Transfection of Human Liver Cancer HepG2 Cells
HepG2 cells were routinely cultured and set as the blank group; SS was added to the HepG2 cell culture medium and incubated to make it in full contact with the cells, which was set as the SS group; SiNPs-SS was added to the HepG2 cell culture medium and incubated, and was set as the SiNPs-SS group.
Cell Transfection
HepG2 was cultured routinely in Dulbecco’s modified Eagle medium (DMEM) at 37°C, 5% CO2, and 10% bovine serum RPM11640 as the medium. Transfection was performed according to the steps of the Lipofectamine 3000 kit. After the transfection, the culture medium was replaced, the culture was continued, and the pc-p38 MAPK plasmid and pc-IL-6 plasmid were transfected into the cells.
Proliferation Ability Detection
Inoculate “Cancer HepG2 cells” into a 96-well plate at 1 × 103 (200 µL). After 4 h, aspirate the cell culture medium, add 10% CCK-8, and incubate in the dark for 1 h to measure the growth of cells at 24, 48, and 72 h, and the growth curve was measured.
Migration and invasion ability detection. Prepare “Cancer HepG2 cells” to 1 × 105 cells/mL, inoculate them into the upper chamber of Transwell, put serum-free culture medium containing SDF-1α into the lower chamber, incubate for 12 h, take out and wash, fix with paraformaldehyde, and crystallize. After staining with purple, absorb the floating color, observe and detect the absorbance at 565 nm. Cell migration and invasion after culture were observed using conventional microscopy.
Detection of Apoptotic Changes by Flow Cytometry
The liver cancer cells of each group were made into a cell suspension, centrifuged, washed with PBS, resuspended, and then added to the buffer. An appropriate amount of Annexin V-FITC and PI (2:1) suspension solution was added, cultured for 15 min, and flow cytometry was used. Apoptotic changes in each group were detected.
Quantitative Reverse Transcription Polymerase Chain Reaction (qRT-PCR) Detection
Total RNA was extracted for complementary deoxyribonucleic acid (cDNA) synthesis. RT-qPCR was performed using SYBR fluorescent PCR technology. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) is an intrinsic control between ROCK/MLCK and miRNA, and the qRT-PCR results were analyzed. Relative levels were estimated using the 2−∇∇Ct method (Livak & Schmittgen, 2001). Table 1 lists the primers and primer sequences.
Real-time Polymerase Chain Reaction (PCR) Primers and Primer Sequences.

Statistical Analysis
The data obtained in the above experiments were analyzed using SPSS 21.0 and GraphPad Prism software. If there are no special requirements,
Results
Successfully Constructed SiNPs-SS
When observed under a transmission electron microscope, the nanoparticles appear spherical, and the particle distribution is relatively uniform (Figure 1).
Nanoscale Characterization (Magnification ×50,000).

SiNPs-SS Affects EVs Secretion by Liver Cancer Cells and Liver Cancer Progression
We found that VEGF was reduced through cell experimental culture (Figure 2A), and the expression level of EGFR also showed a decreasing trend (Figure 2B). Further comparison results showed that SiNPs-SS intervention. The expression of related factors secreted by EVs of liver cancer cells under the conditions was lower; with the extension of intervention time, SiNPs-SS intervention showed a phenomenon of liver cancer proliferation inhibition (Figure 2C), and at the same time, weakened HepG2 cells were observed. The migration and invasion ability (Figure 2D and E), and the apoptosis rate of the SiNPs-SS group also confirmed this phenomenon (VS SS group,
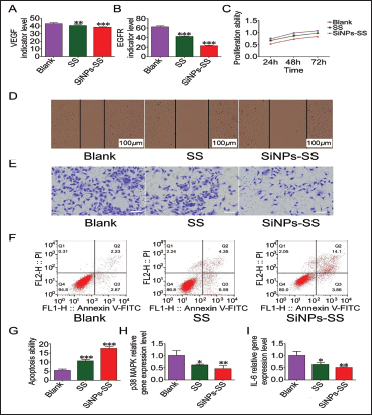
p38 MAPK/IL-6 Pathway is Involved in the Process of SiNPs-SS Inhibiting the Secretion of EVs and the Progression of Liver Cancer
Based on the above research, this topic will further explore the mechanism of SiNPs-SS in EVs secretion by liver cancer cells and liver cancer progression. After intervention with p38 MAPK agonist, we found that both VEGF and EGFR were increased (VS blank group,
To further explore the role of IL-6 in this process, we used IL-6 agonists and IL-6 inhibitors to intervene in HepG2 cells under SiNPs-SS intervention conditions and IL-6 inhibitor intervention conditions. The relevant results and their changing trends are consistent with those under SB 202190 intervention conditions (Figure 3A–3G). The above experimental results reflect that downregulating the expression of the p38 MAPK/IL-6 pathway can promote SiN to a certain extent. The inhibitory effect of Ps-SS on the secretion of EVs by liver cancer cells is beneficial in inhibiting liver cancer progression.
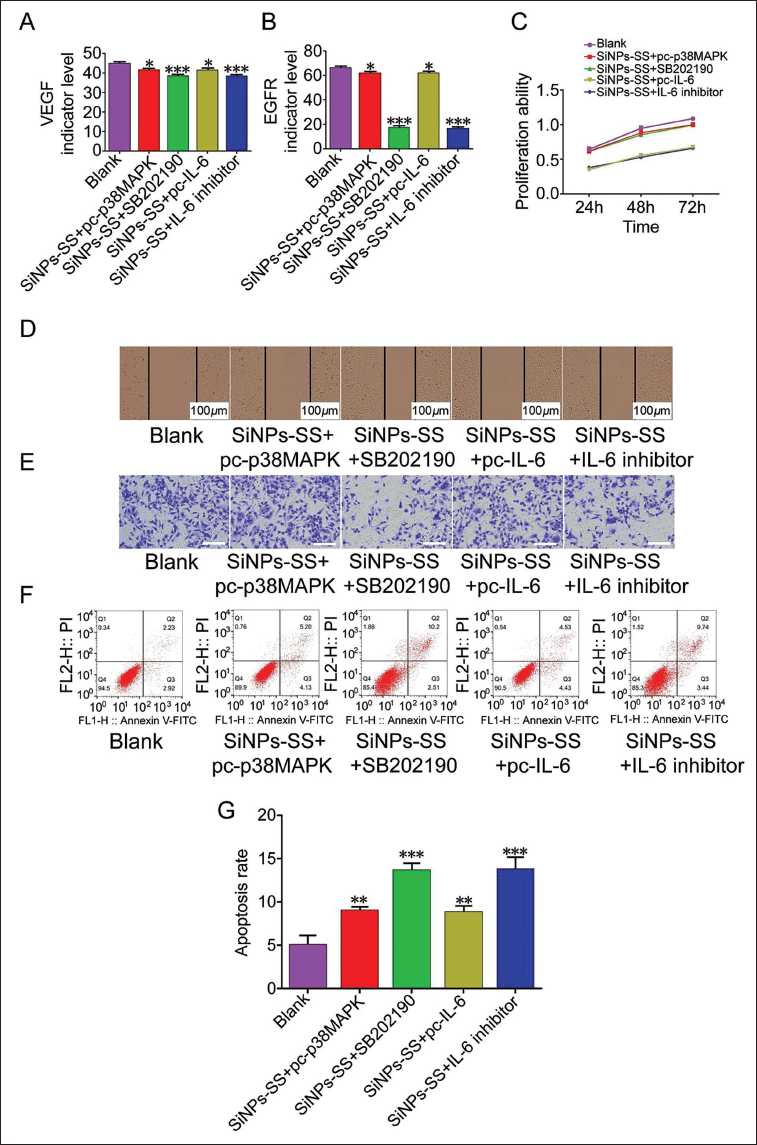
Role of SiNPs-SS in Inhibiting the Secretion of EVs and the Progression of Liver Cancer by Regulating p38 MAPK/IL-6 Pathway
We added IL-6 agonist to the intervention of SB 202190 + IL-6 inhibitor and found that the index levels of VEGF and EGFR showed an upward trend (Figure 4A and B). We further investigated the biological behavior of liver cancer HepG2 cells. It was observed that the SB 202190 group and the SB 202190 + IL-6 inhibitor + IL-6 agonist group had the strongest abilities in terms of proliferation, migration, and invasion (Figure 4C–4E). At the same time, the apoptosis ability was also significantly reduced. The apoptosis rate decreased significantly (VS SB 202190,
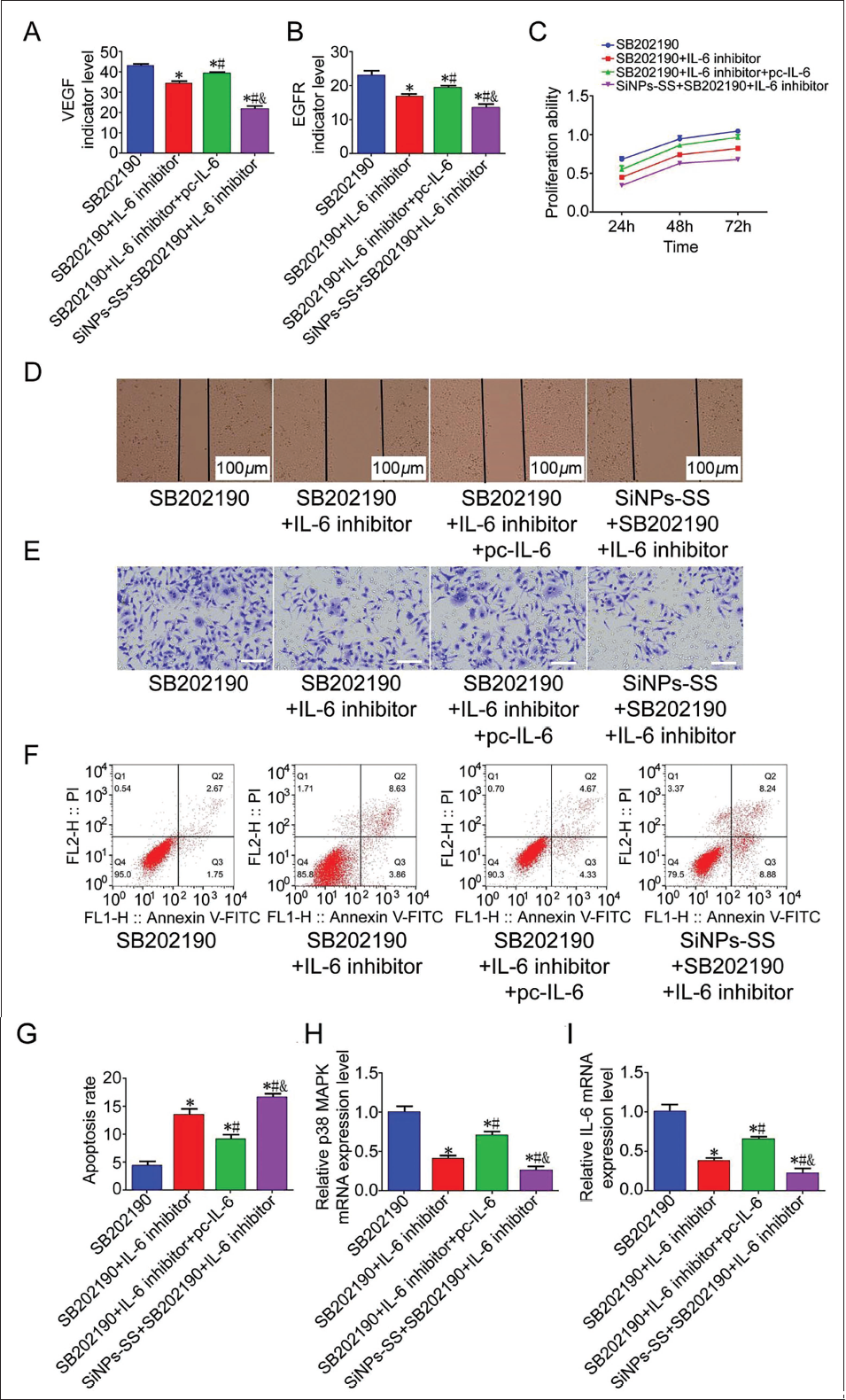
Discussion
Bupleurum has low solubility in the body and a low oral absorption rate, so its efficacy is limited. To improve the solubility, bioavailability, and efficacy of bupleurum, we use silica nanoparticles to modify and transport it, which can effectively improve the solubility of bupleurum in water or biological media and help enhance the efficacy of bupleurum. The solubility of the hormone makes it easier for the body to absorb and utilize. In addition, a previous study (Livak & Schmittgen, 2001) pointed out that silica nanoparticles have significant biodegradability and can be effectively absorbed and metabolized
Some studies have pointed out that VEGF can activate the receptor tyrosine kinase on the cell membrane through its binding to the VEGF receptor, thereby initiating the p38 MAPK signaling pathway, causing it to be phosphorylated, to enter the cell nucleus, to promote gene transcription, and to participate in angiogenesis, cell proliferation, cell migration, and inflammatory response and other processes (Wang et al., 2022). In addition, p38 MAPK also regulates HSF1 activity by phosphorylating CREB (Liu, Chen, et al., 2022). The phosphorylated HSF1 forms a complex with AP-1 (Ishikawa & Mori, 2023), which jointly regulates cell survival, proliferation, and apoptosis, further affecting the heat shock response and abnormal-stress-cell physiological processes. In our experiments, after using SB 202190 based on the intervention of SiNPs-SS, we found that the biological behavior of HepG2 cells was inhibited and the apoptosis rate increased. However, p38 MAPK agonist intervention showed the opposite results. Further use of IL-6 inhibitors found that the results were consistent with the intervention conditions of SB 202190. Therefore, it shows that downregulating the expression of the p38 MAPK/IL-6 pathway can, to a certain extent, promote the inhibitory effect of SiNPs-SS on the secretion of EVs of liver cancer cells. Analysis suggests that p38 MAPK can phosphorylate and activate Mnk1, thereby promoting Mnk1 to phosphorylate eIF4E, enhancing eIF4E-mediated mRNA translation, and thereby increasing the synthesis of AP-1 protein, thereby inhibiting the development of cancer. MSK1 can be phosphorylated and activated by p38 MAPK, thereby phosphorylating and activating multiple transcription factors such as CREB and histone H3 in the nucleus to modulate gene expression and thereby inhibit cancer. p38 MAPK can downregulate cyclin D by activating the transcription factors p53 and E2F, while IL-6 can reduce cyclin D1 and CDK4 by inhibiting the STAT3 transcription factor, thereby preventing cells from entering S phase from the G1 phase. SiNPs-SS further strengthens this effect and further reflects its good biological efficacy.
We added an IL-6 agonist to the intervention of SB 202190 + IL-6 inhibitor and found that the inhibitory effect of SB 202190 + IL-6 inhibitor on liver cancer was significantly reversed. To further study the effect of SiNPs-SS on p38 MAPK/IL-6, we combined SiNPs-SS with SiNPs-SS + SB 202190 + IL-6 inhibitor and found that it has the most substantial inhibitory effect on liver cancer. Therefore, it is proved that SiNPs-SS can downregulate the expression of p38 MAPK/IL-6, inhibit the secretion of EVs, and slow down liver cancer progression. However, this study requires analysis of more animal models and clinical samples. It is helpful to evaluate the efficacy and safety of silica nanoparticle-modified bupleurum in the overall biological system. In addition, the effects of modified bupleurum on normal cells and tissues need to be evaluated to better understand its toxicity and side effects.
Conclusion
In summary, SiNPs-SS can inhibit the p38 MAPK/IL-6 pathway, inhibiting the secretion of EVs and liver cancer progression. This provides an important theoretical basis for further research and development of liver cancer treatment strategies.
Footnotes
Abbreviations
IL-6: Interleukin-6; MAPK: Mitogen-activated protein kinase.
Acknowledgments
The authors gratefully acknowledge the Chongqing Red Cross Hospital Laboratory for providing the necessary equipment for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Informed Consent
This study was approved by the Chongqing Red Cross Hospital.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
