Abstract
Background/Objectives
Curzerenone is a sesquiterpene primarily isolated from Curcuma zedoaria rhizomes, identified for its diverse therapeutic efficacy. Recent studies have highlighted curzerenone’s potential anti-cancer, inflammatory attenuating, and antioxidant properties. Current research was conducted to investigate the anti-cancer potency of curzerenone on hepatic carcinoma cells.
Materials and Methods
Curzerenone antioxidant potency was initially assessed in vitro using ferric reducing antioxidant power (FRAP) and 2,2-diphenyl-1-picrylhydrazyl (DPPH) assays. Human liver cancer cell line (HepG2) and Vero cell lines were utilized to assess cytotoxic effects via 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. 2′,7′-Dichlorofluorescin diacetate (DCFH-DA) staining quantified reactive oxygen species (ROS) generation. Thiobarbituric acid reactive substances (TBARS) assay and antioxidant quantification analyzed curzerenone’s impact. Cell adhesion assay evaluated anti-metastatic effects. Apoptotic signaling proteins and phosphatidylinositol 3-kinase/protein kinase B/mammalian target of rapamycin (PI3K/AKT/mTOR) proteins were quantified in curzerenone-treated HepG2 cells.
Results
Curzerenone exhibited strong antioxidant activity and tumor cell-specific cytotoxicity while remaining non-cytotoxic to normal Vero cells. Treatment increased intracellular ROS levels, decreased antioxidant levels, and induced lipid peroxidation in HepG2 cells. It elevated apoptotic signaling and attenuated PI3K/AKT/mTOR signaling, preventing cancer cell viability and division while increasing cell death.
Conclusion
Results suggest that curzerenone induces oxidative stress, thereby reducing cancer cell survival and promoting apoptosis in hepatocellular carcinoma cells.
Keywords
Introduction
Hepatic cancers tend to be the most prevalent type of cancer, ranking sixth in global incidence and third in cancer-related demises (Rumgay et al., 2022), with hepatocellular carcinoma (HCC) accounting for 80%–90% of cases (Asrani et al., 2019). It is also the second most significant cause of early cancer-related mortality (Ferlay et al., 2018). Current estimates suggest that the hepatitis B virus is responsible for 56% of liver cancer cases, hepatitis C virus for 20%, and other factors like smoking and alcohol also play significant roles (GBD 2019 Risk Factors Collaborators, 2020; Kulik & El-Serag, 2019; Maucort-Boulch et al., 2018). The global burden of liver cancer remains substantial, highlighted by over 9.6 million cancer deaths in 2020 (Sung et al., 2021). By 2025, liver cancer cases could surpass 1 million annually, with rising associations to metabolic syndrome and non-alcoholic steatohepatitis contributing to this trend (Llovet et al., 2021).
HCC management is highly dependent on disease stage and liver function, with early-stage cases treated via surgical resection, liver transplantation, or ablative therapies like radiofrequency or microwave ablation (Schlachterman et al., 2015). Advanced HCC often requires systemic therapies such as targeted therapy, immunotherapy, or chemotherapy, with drugs like pembrolizumab and lenvatinib showing improved survival rates (Liu et al., 2024). Despite advancements, treatments face challenges, including drug resistance, adverse effects, recurrence, and limited efficacy in advanced stages (Forner et al., 2019). Liver transplantation offers the potential for a cure but is hindered by high recurrence and complication rates (Zhou et al., 2009). Other options, such as trans-arterial chemoembolization (TACE) and sorafenib, provide moderate survival benefits but are limited by toxicity and resistance (Anwanwan et al., 2020; El-Serag et al., 2008).
Natural antioxidant therapies have garnered attention as potential adjunct treatments for HCC because of their capacity to counteract redox imbalance and inflammatory response, which play critical roles in cancer initiation and progression (Hassan et al., 2016; Li, Cao et al., 2023). Research has examined the efficacy of herbal extracts and phytochemicals, such as curcumin, resveratrol, and green tea polyphenols, in preclinical and clinical settings. For instance, curcumin inhibits liver cancer cell proliferation and induces apoptosis (Wang et al., 2023); likewise, resveratrol modulates inflammatory responses and enhances antioxidant defenses (Karabekir & Özgörgülü, 2020). Epigallocatechin gallate (EGCG) has demonstrated the ability to suppress tumor growth and metastasis in liver cancer models (Li, Yu et al., 2023). These natural compounds not only provide antioxidant benefits but also may enhance the efficacy of conventional therapies, suggesting a potential role for antioxidant-rich foods and natural supplements in comprehensive liver cancer management (Maiuolo et al., 2021).
Curcuma sp., belonging to Zingiberaceae family, is innate to India and extensively grown in tropical regions (Tung et al., 2019). In traditional Chinese medicine (TCM), it is prescribed for cancer, diabetes, respiratory issues, arthritis, skin conditions, and so on (El-Kenawy et al., 2018). Research indicates that turmeric and its active compounds exhibit several medicinal properties, such as free radical scavenging (Uzunhisarcikli & Aslanturk, 2019), hepatoprotective (Lee et al., 2017), attenuating inflammation (Kinney et al., 2015), anti-cancer (Jia et al., 2014), anti-diabetic (Essa et al., 2019), and neuroprotective effects (Xu et al., 2019), among others. Curzerenone is a bioactive sesquiterpene compound present in Curcuma zedoaria and Curcuma aeruginosa plants with several pharmacological properties (Poudel et al., 2022). It has been already reported that curzerenone demonstrated anti-cancer activity against lung cancer (Zheng & Chen, 2019) and cervical cancer (Sun et al., 2024) cells. Furthermore, it also showed antioxidant and anti-bacterial properties (Joshi & Mathela, 2012). However, there are not many reports on the anti-cancer properties of the curzerenone against liver cancer. Therefore, the present has explored the anti-cancer efficacy of curzerenone against HCC cell lines.
Materials and Methods
Chemicals
Curzerenone, Dulbecco’s modified Eagle medium (DMEM), and other primary chemicals were obtained from Sigma–Aldrich, USA. All the diagnostic kits for the biochemical markers were purchased from R&D Systems and Abcam, USA, respectively.
Antioxidant Evaluation
Ferric Reducing Antioxidant Power (FRAP) Assay
Curzerenone samples free radical scavenging capacity was evaluated using the FRAP test Benzie and Strain (1996). About 180 µL of the freshly prepared FRAP reagent and 24 µL of curzerenone of varied concentrations ranging from 0.5 to 25 µM, a standard solution, or distilled water as a blank, were added to 96-well plates. After allowing the mixture to incubate at 37°C for 10 min, the final OD was quantified at 593 nm. A standard curve for Trolox was established, and results were reported in mM of Trolox equivalent. The test was conducted with three replicates.
2,2-Diphenyl-1-Picrylhydrazyl (DPPH) Assay
Brand-Williams et al.’s (1995) protocol was followed for the DPPH evaluation. About 190 µL of 0.12 mM DPPH was combined with 10 µL of curzerenone of varied concentrations ranging from 0.5 to 25 µM in a 96-well plate. After allowing the mixture to incubate for 30 min, the final OD was quantified at 517 nm. The test was conducted with three replicates.
Cell Culture
Human liver cancer cell line (HepG2) and Vero cells are typically cultured in 10% fetal bovine serum supplemented DMEM and incubated at 37°C in a humidified incubator with 5% CO2. When the cells reached 70%–80% confluence, they were passaged by washing with phosphate-buffered saline (PBS), treating with trypsin to detach the cells, and then re-suspending them in fresh medium before being split at a ratio of 1:3 for subculturing into new flasks.
MTT Assay
HepG2 and Vero cells were cultured in 96-well plates at a cell density of 5 × 103 cells/well and treated with curzerenone (0.5–25 µM). Control cells were treated only with DMEM medium. The cells were incubated in a 5% CO₂ humidified chamber at 37°C for 24 h. Following treatment, the cells were treated with MTT solution (5 mg/mL dissolved in PBS) for 4 h. DMSO was added to dissolve the formazan crystals, and the final OD was quantified at 550 nm. The 50% cytotoxic concentration (CC50) was determined using GraphPad Prism software based on the dose-response curves generated from the absorbance readings (Mosmann, 1983).
Intracellular Reactive Oxygen Species Quantification
HepG2 cells (5 × 105 cells/well) were treated with 7.5 µM curzerenone or doxorubicin (standard drug) and incubated for another 24 h. Control cells were treated only with DMEM medium. ROS levels were measured using 10 µM DCFH-DA (10 µM concentration prepared in DMSO), incubated for 30 min in the dark, and fluorescence was recorded. For visualization, cells grown on coverslips were similarly treated and examined using a confocal laser scanning microscope (Eruslanov & Kusmartsev, 2010).
Thiobarbituric Acid Reactive Substances Assay
HepG2 cells were cultured in 6-well plates and treated with curzerenone and doxorubicin for 24 hours. The cells were then lysed with a protease inhibitor-containing buffer, and the lysate was centrifuged to obtain the supernatant. The supernatant was mixed with TBA solution (prepared by dissolving 0.67% (w/v) thiobarbituric acid in 0.05 M NaOH), heated at 95°C for 1 h, cooled, and the absorbance of the pink complex was measured at 532 nm using a spectrophotometer (de Godoi et al., 2017; Ohkawa et al., 1979).
Quantification of Antioxidants
Superoxide dismutase (SOD) evaluation was performed as per the protocol of Marklund and Marklund (1974). The cell lysates were centrifuged at 6,000 rpm for 15 min, and the obtained supernatant was treated with a reaction mixture containing xanthine (100 mM), xanthine oxidase (0.1 U/mL), and nitroblue tetrazolium (NBT) (100 µM) and the OD was quantified at 560 nm, results were indicated as SOD activity in units per milligram of protein. For the glutathione assay, Ellman’s (1959) protocol was followed; the supernatant was treated with DTNB, the final OD was quantified at 412 nm, and glutathione levels were expressed as µmol/mg protein.
Cell Adhesion Assay
A cell adhesion assay was performed with HepG2 cells treated with curzerenone (7.5 µM) and doxorubicin (2 µg) for 24 h. Control cells were treated only with DMEM medium. After treatment, 100 µL of cell suspension (1 × 10⁶ cells/mL) was loaded onto Matrigel-coated plates and incubated at 37°C in a humidified CO2 (5%) chamber for 2 h. Suspended cells were removed by gentle washing with PBS, and attached cells were fixed with 4% formaldehyde, stained with 0.2% crystal violet for 15 min, and photographed at 10× magnification for analysis (Wu et al., 2013).
Assessment of Apoptotic Signaling
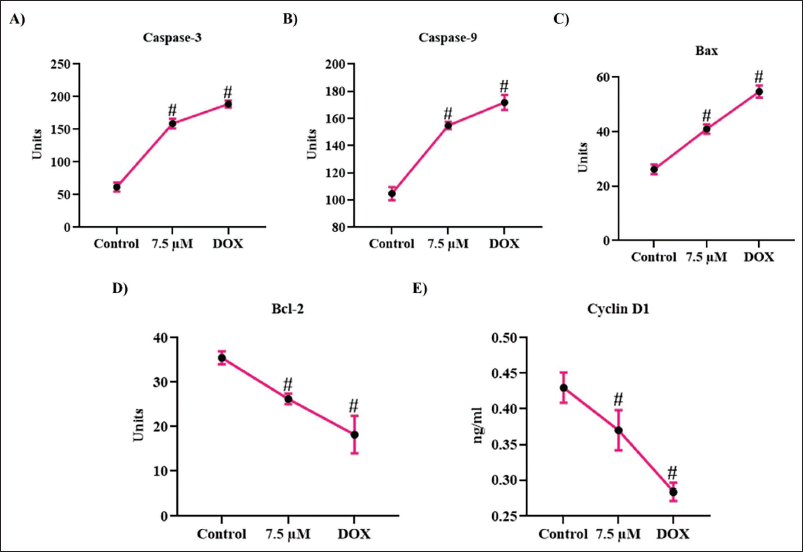
Apoptotic proteins Bax, Bcl2, caspases 3, 9, and cyclin D1 were measured in curzerenone and doxorubicin-treated HepG2 cells using an ELISA kit (R&D Systems). The assay was performed in triplicates as per the guidelines provided in the kit manual.
Assessment of Phosphatidylinositol 3-Kinase/Protein Kinase B/Mammalian Target of Rapamycin (PI3K/AKT/mTOR) Signaling
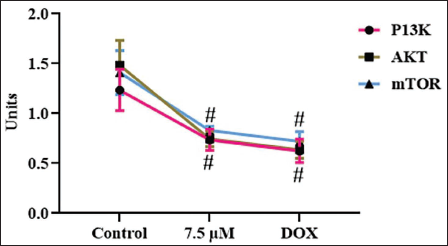
PI3K, AKT, and mTOR protein were measured in the HepG2 cells treated with curzerenone and doxorubicin using the ELISA kits obtained from Abcam. The assay was done as per the guidelines prescribed in the kit manual, and the final absorbance was estimated at 450 nm.
Statistics
GraphPad Prism 8.0 (USA) was used to analyze the results obtained. Data are expressed as Mean ± SD. Group comparisons were made using one-way analysis of variance (ANOVA), and within-group differences were evaluated using Tukey’s test.
Results
Antioxidant Efficacy of Sesquiterpene Curzerenone
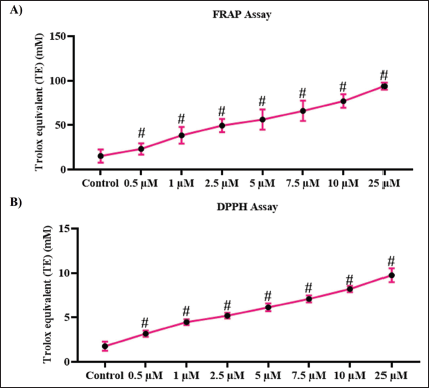
The antioxidant potency of curzerenone at different concentrations was assessed using FRAP and DPPH assays, and the data were illustrated in Figure 1. Curzerenone potency to reduce ferric ions to ferrous ions was measured and represented as Trolox equivalents in Figure 1A. Curzerenone effectively reduced ferric ions to ferrous ions, indicating its antioxidant capacity. The minimum dose of 0.5 µM curzerenone showed 22 ± 2 mM TE, while the maximum dose of 25 µM curzerenone exhibited 78 ± 1.2 mM TE. Figure 1B illustrates the free radical scavenging ability of curzerenone, quantified by DPPH assay. 3.8 ± 0.005 mM TE was observed at the 0.5 µM concentration, and 8.4 ± 0.009 mM TE was recorded at the 25 µM concentration of curzerenone treatment.
Cytotoxic Potency of Sesquiterpene Curzerenone
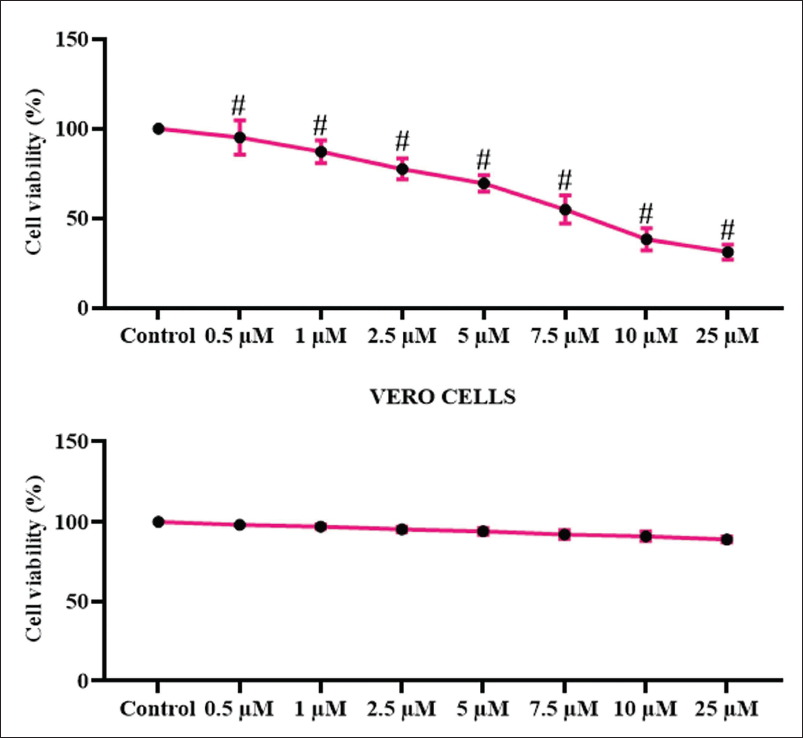
Figure 2 depicts the cytotoxic effect of curzerenone on HepG2, HCC cells, and Vero, normal kidney epithelial cells. Curzerenone treatment rendered a significant cytotoxic effect on HepG2 cells, depending on the dosage of treatment. About 3% ± 0.07% of dead cells were quantified with 0.5 µM treatment, whereas it is increased to 17% ± 0.05% and 26% ± 0.06%, with 2.5 and 5 µM treatment respectively. The IC50 dose was obtained with 7.5 µM curzerenone treatment, which exhibited 48% ± 0.09% of cell death. The cell death was further increased to 55% ± 0.04% and 63% ± 0.05% with 10 and 25 µM curzerenone treatment (Figure 2A). Curzerenone treatment exhibited a minimal cytotoxic effect on normal kidney epithelial cells even at the maximum dose of 25 µM, which rendered 63% ± 0.05% of cell death in HepG2 cells exhibited only 14.5% ± 0.05% of cell death in Vero cells (Figure 2B).
Intracellular ROS Triggering Efficacy of Sesquiterpene Curzerenone
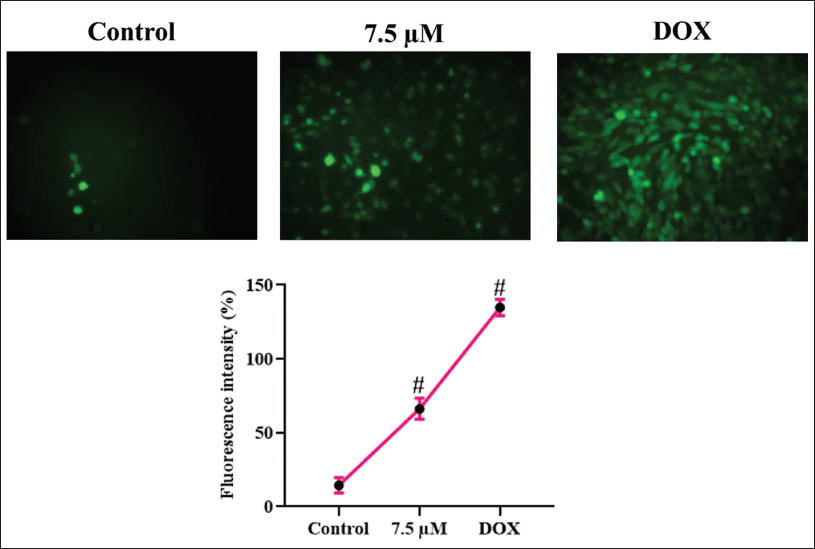
The efficacy of curzerenone to enhance intracellular ROS synthesis was quantified in the HepG2 cells (Figure 3). An increase in green fluorescence emission was witnessed in the HepG2 cells treated with curzerenone and doxorubicin than in the control cells. The standard chemotherapeutic drug doxorubicin exhibited enhanced green fluorescence, which was quantified to be 135% ± 0.06%, whereas the curzerenone-treated cells exhibited 59% ± 0.08%, and the control untreated HepG2 cells exhibited minimum fluorescence intensity of 3% ± 0.04%.
Antioxidant Attenuating Potency of Sesquiterpene Curzerenone
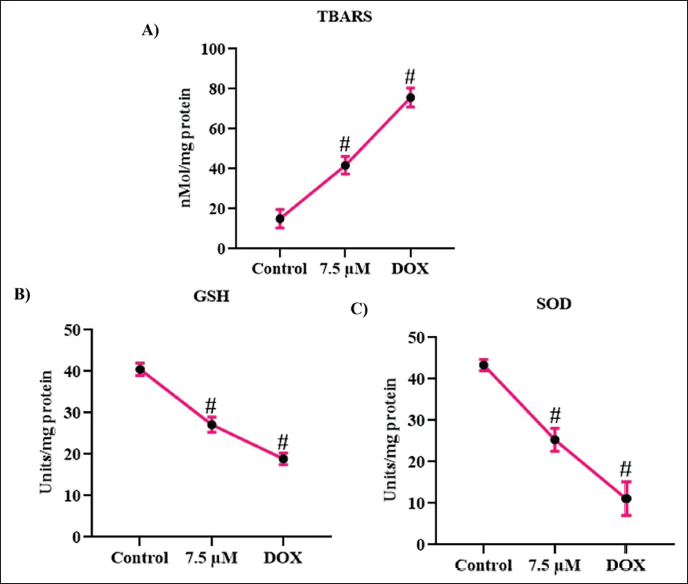
Figure 4 illustrates the impact of sesquiterpene curzerenone on the antioxidant status of HepG2 cells. Lipid peroxidation triggered by curzerenone in HepG2 cells was quantified, and the results were depicted in Figure 4A. Both curzerenone and doxorubicin treatment considerably increased the lipid peroxidation in HepG2 cells. 76.4 ± 0.04 and 39.7 ± 0.06 nMol/mg protein of MDA were quantified in the doxorubicin and curzerenone treated cells, whereas only 18.2 ± 0.05 nMol/mg protein of MDA were measured in the control untreated cells. Curzerenone treatment attenuated the antioxidant levels in the HepG2 cells. 27.6 ± 0.05 units/mg protein of glutathione and 27.3 ± 0.04 units/mg protein of SOD were quantified in the curzerenone-treated cells, whereas the control cells exhibited 42.3 ± 0.06 units/mg protein and 46.5 ± 0.05 units/mg protein of glutathione and SOD, respectively. Doxorubicin-treated cells exhibited 21.2 ± 0.04 units/mg protein of glutathione and 14.5 ± 0.05 units/mg protein of SOD.
Inhibitory Potency of Sesquiterpene Curzerenone Against HepG2 Cell Adhesion
The impact of curzerenone on HepG2 cell adhesion, which is a key phenomenon of cancer cell proliferation and metastasis, is illustrated in Figure 5. Curzerenone effectively inhibited the adhesion of HepG2 cells, which was evidenced with the microscopic analysis.

Apoptosis Stimulating Efficacy of Sesquiterpene Curzerenone on HepG2 Cells
Figure 6 depicts curzerenone impact on apoptotic proteins in HepG2 cells. Curzerenone and doxorubicin treatment significantly elevated the levels of apoptotic stimulating protein Bax, and it considerably decreased the levels of apoptotic attenuating protein Bcl2. It also significantly decreased the levels of cyclin D1 protein, which regulates the cell cycle progression. The caspase levels were also increased in the curzerenone and doxorubicin-treated cells than in control cells. Significant enhancement in both caspases 3 and 9 was noted in the curzerenone-treated cells.
Attenuating Effect of Sesquiterpene Curzerenone on PI3K/AKT/mTOR Signaling in HepG2 Cells
Figure 7 illustrates the curzerenone efficacy of attenuating PI3K/AKT/mTOR signaling in HepG2, which is often a targeted signaling pathway of anti-cancer drugs. Curzerenone and chemotherapeutic drug doxorubicin treatment attenuated the PI3K, AKT, and mTOR proteins in HepG2 cells. HepG2 cells treated with curzerenone exhibited a decrease in PI3K, AKT, and mTOR proteins than the control untreated cells.
Discussion
ROS has been widely examined in relation to several ailments, particularly cancers (Cairns et al., 2012; Chio & Tuveson, 2017). As natural byproducts of cellular processes, including oxygen metabolism, ROS levels can be modulated as a potential anti-cancer strategy (Sena & Chandel, 2012). Research indicates that ROS possesses a “double-edged sword” role in cancer (Forman & Zhang, 2021; Okon & Zou, 2015). Minimal concentrations of ROS facilitate processes such as cancer cell survival and drug resistance (Gorrini et al., 2013). Conversely, high ROS levels can be detrimental, leading to cell death. Cancer cells frequently acclimate to increased ROS levels by activating antioxidant pathways, thereby improving their capacity to neutralize and remove ROS (Schafer et al., 2009). Additionally, cancer cells exhibit heightened sensitivity to therapies that induce ROS, as many chemotherapeutic agents are known to increase ROS production within these cells (Dougan et al., 2008; Nakamura & Takada, 2021; Siomek et al., 2006). The phytochemical compound curzerenone anti-cancer effect on HCC cells via generating ROS was examined.
Multiple studies have demonstrated the significant free radical scavenging activity of extracts from the rhizomes of C. zedoaria. Previous research employed various assays, including DPPH, ABTS, and reducing power assays, to highlight the scavenging potential of essential oils derived from these Curcuma extracts and C. zedoaria (Angel et al., 2013; Sudipta et al., 2012). Additionally, Sumathi et al. (2013) utilized DPPH, ABTS, and hydrogen peroxide inhibition to further substantiate the radical scavenging activity (Sumathi et al., 2013). Our results also correlate with the above findings. FRAP and DPPH assays performed on curzerenone depicted its strong antioxidant capacity.
Even though numerous therapeutic agents have been approved to treat cancer, patients still face challenges in accessing effective treatments due to drug resistance, which significantly limits the clinical efficacy of even the most advanced therapies (Lopez & Banerji, 2017; Scott et al., 2023). This resistance may arise from the enhanced activity of detoxification mechanisms, such as glutathione-S-transferase, which facilitates the removal of reactive compounds using glutathione (Townsend & Tew, 2003). Additionally, glutathione peroxidase plays a role in converting hydrogen peroxide to water and reducing lipid peroxides to alcohol, thereby mitigating the harmful effects of ROS. Therefore, anti-cancer drugs have been designed to target glutathione for chemotherapy or to serve as detoxifying agents in chemoprevention strategies (Lubos et al., 2011). In our study, we assessed the capacity of curzerenone to generate intracellular ROS and inhibit antioxidant synthesis. Curzerenone effectively increased the levels of ROS and attenuated the antioxidants GSH and SOD. Increased thiobarbituric acid reactive substances (TBARS) levels in the curzerenone-treated HepG2 cells confirm the ROS-induced cellular damage in HCC cells.
Reactive oxygen species levels are implicated in the facilitation of tumors, but an elevation in ROS levels can also activate anti-tumor signaling pathways and induce redox imbalance, leading to apoptosis in tumor cells via proapoptotic mechanisms (Redza-Dutordoir & Averill-Bates, 2016; Zhao et al., 2023). Research has shown that free radicals produced in tumor cells can trigger cell death through apoptotic signaling pathways, contributing to tumor inhibition (Wang et al., 2018). Elevated mitochondrial ROS can trigger intrinsic apoptosis. Apoptotic protease activating factor-1 interacts with cytochrome c in the cytosol, resulting in the apoptosome that activates caspase 9 (Kumar et al., 2022). Caspase 9, an essential element of the intrinsic apoptotic pathway, activates effector caspases such as caspases 3, 6, and 7, which results in the cleavage of vital cellular proteins and ultimately results in apoptosis (Fan et al., 2005). Curzerenone treatment had significantly elevated the ROS and increased the proapoptotic protein Bax expression and the caspases 3 and 9, which eventually led to apoptosis. The MTT assay results of curzerenone-treated HepG2 cells confirm the induction of apoptosis in HCC cells. Curzerenone also inhibited the cyclin D1 expression, which is a pivotal player in cellular proliferation and progression of several cancers.
The PI3K/AKT/mTOR signaling pathway plays a pivotal role in regulating fundamental cellular processes, including cell growth, proliferation, survival, metabolism, and autophagy, thereby exerting a profound influence on the development and progression of cancer (Hoxhaj & Manning, 2020). The PI3K pathway is modulated by reactive oxygen species (ROS), as ROS-induced oxidation of protein phosphatases activates various signaling pathways. Dysregulation of this pathway, frequently observed in HCC, the most prevalent form of liver cancer, can arise from various genetic and epigenetic alterations, including mutations, amplifications, and aberrant expression of its key components (Grabinski et al., 2012; Ye et al., 2019). The PI3K/AKT pathway integrates signals from external cellular stimuli to regulate essential cellular functions and is frequently aberrantly activated in human cancers. Specifically, aberrant activation of the PI3K/AKT/mTOR pathway can promote uncontrolled cell proliferation by upregulating the expression of genes involved in cell cycle progression and inhibiting apoptosis by modulating the activity of proapoptotic proteins. The pathway’s intricate network of interacting proteins makes it a crucial player in cancer biology, and its dysregulation has far-reaching consequences for tumorigenesis and treatment resistance (Golob-Schwarzl et al., 2017). In HCC, the PI3K/AKT/mTOR pathway is commonly activated, offering a significant foundation for the development of targeted therapies. In liver cancer, the dysregulation of the PI3K/AKT/mTOR pathway can disrupt the delicate balance between cell survival and cell death, favoring the survival and proliferation of cancerous cells (Tian et al., 2023). Given the pivotal role of the PI3K/AKT/mTOR pathway in liver cancer, targeting this pathway has emerged as a promising therapeutic strategy. Inhibitors targeting different components of the PI3K/AKT/mTOR pathway have been developed and are being evaluated. Consequently, researchers have developed anti-HCC therapies aimed at this signaling pathway, with clinical trials demonstrating their effectiveness (Yan et al., 2022). Curzerenone treatment had attenuated the PI3K, AKT, and mTOR signaling proteins in HepG2 cells, which proves curzerenone had effectively suppressed cancer cell survival and progression. Our cell adhesion results further confirmed the tumor cell inhibition potency of curzerenone.
Conclusion
In conclusion, this study demonstrates that curzerenone, a sesquiterpene derived from C. zedoaria, possesses notable anti-cancer properties, particularly against HCC cells. The compound’s strong antioxidant activity was confirmed through FRAP and DPPH assays, yet it paradoxically elevated intracellular ROS levels and lipid peroxidation in HepG2 cells, leading to oxidative stress. Curzerenone exhibited selective cytotoxicity towards cancer cells, sparing normal epithelial cells, and effectively triggered apoptotic signaling and attenuated the PI3K/AKT/mTOR pathway. These findings indicate that curzerenone’s potency to trigger oxidative stress and promote apoptosis can significantly impair HCC cell proliferation and survival, emphasizing its potential as a therapeutic candidate in the treatment of liver cancer. Further in vivo studies and investigations using other cancer cell lines are needed to fully elucidate curzerenone’s anti-cancer effects.
Footnotes
Abbreviations
AKT: Protein kinase B; ANOVA: Analysis of variance; Bax: BCL2 associated X; Bcl2: B-cell lymphoma 2; DCFH-DA: 2′,7′-Dichlorofluorescin diacetate; DMEM: Dulbecco’s modified Eagle medium; DMSO: Dimethyl sulfoxide; DNA: Deoxyribonucleic acid; DOX: Doxorubicin; DPPH: 2,2-Diphenyl-1-picrylhydrazyl; ERK: Extracellular signal-regulated kinases; FRAP: Ferric reducing antioxidant power; GSH: Glutathione; HCC: Hepatocellular carcinoma; IC50: Half maximal inhibitory concentration; MAPK: Mitogen-activated protein kinase; MDA: Malondialdehyde; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; mTOR: Mammalian target of rapamycin; NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells; PBS: Phosphate-buffered saline; PI3K: Phosphatidylinositol 3-kinase; RNA: Ribonucleic acid; ROS: Reactive oxygen species; SEM: Standard error of mean; SOD: Superoxide dismutase; TACE: Trans-arterial chemoembolization; TCM: Traditional Chinese medicine; TBARS: Thiobarbituric acid reactive substances.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
