Abstract
Background
Hepatocellular carcinoma (HCC) is a major global health threat with a high mortality rate. Sorafenib, the standard treatment for advanced HCC, is often rendered ineffective by tumor drug resistance, particularly under hypoxic conditions that promote resistance and epithelial–mesenchymal transition (EMT) in cancer cells. Traditional Chinese medicine (TCM), with its anti-tumor components, offers a potential avenue for overcoming resistance and inhibiting EMT.
Objectives
Rhaponticin (RA), an active compound from TCM, is investigated in this study for its ability to counter hypoxia-induced sorafenib resistance and EMT in HCC cells.
Materials and Methods
HepG2 and Huh7 cells were cultivated under hypoxic conditions to evaluate sorafenib resistance, with cell proliferation assessed using 5-ethynyl-2′-deoxyuridine (EdU) staining and EMT markers analyzed post-treatment. Then, RA’s capacity to diminish cell viability, enhance sorafenib sensitivity, and reverse EMT was examined. The EMT inhibitor C19 was employed to verify the EMT-dependent nature of RA’s effects.
Results
Hypoxia increased sorafenib half maximal inhibitory concentration (IC50) values in HCC cells, indicating sorafenib resistance. Its inhibitory effect on EMT was reduced under hypoxia, as shown by altered EMT marker expression. RA treatment decreased HCC cell viability without toxicity to normal liver cells, enhanced sorafenib’s sensitivity, and reversed EMT under hypoxia, evidenced by increased E-cadherin and decreased Vimentin and N-cadherin. The EMT inhibitor negated the nullified RA effect on sorafenib sensitivity, confirming RA EMT-dependent action.
Conclusion
RA can re-sensitize HCC cells to sorafenib by inhibiting hypoxia-mediated EMT, presenting a promising therapeutic approach for HCC.
Keywords
Introduction
Hepatocellular carcinoma (HCC) ranks as the fifth most prevalent malignant tumor globally and is the third leading cause of cancer-related mortality worldwide, with primary HCC constituting over 90% of all HCC cases (Vogel et al., 2022). Despite advancements in diagnostic and therapeutic approaches for liver cancer, the incidence of primary liver cancer continues to rise, and the overall prognosis remains grim, with an estimated 5-year survival rate of merely 15% (Zhou & Song, 2021). Sorafenib, a multikinase inhibitor with multitarget capabilities, exerts dual effects by inhibiting both angiogenesis and tumor cell proliferation, and it has become the standard first-line treatment for advanced HCC (Tang et al., 2020). However, the development of sorafenib resistance significantly impacts the prognosis of patients with primary liver cancer. Hypoxia, a shared characteristic of many solid tumors, induces a series of changes in tumor cells that can lead to tumor proliferation, invasion, distant metastasis, and resistance to radiochemotherapy (Bao & Wong, 2021). Prior research suggests that tumor resistance may be associated with various factors, including the tumor microenvironment, hypoxia, cancer stem cells, epithelial–mesenchymal transition (EMT), and multiple cellular signaling pathways (Bukowski et al., 2020; Hashemi et al., 2022; Tao et al., 2021). Literature has documented that hypoxic conditions can induce chemoresistance in various cancers, including those of the breast, ovary, esophagus, and lung (Luan et al., 2021; Lyu et al., 2023; McAleese et al., 2021; Wang et al., 2022). Murakami et al. (2014) have shown through experiments that downregulating hypoxia-inducible factor-1α (HIF-1α) can reverse hypoxia-induced resistance to gefitinib in lung cancer cells. The hypoxic microenvironment is a critical factor contributing to sorafenib resistance in HCC cells, with HIF-1α enhancing drug resistance by upregulating the expression of multidrug resistance-associated genes (Wang et al., 2021). Additionally, hypoxia can activate the Yes-associated transcriptional regulator (YAP), further promoting cell survival and evading apoptosis, which contributes to sorafenib resistance (Zhou et al., 2016). Therefore, targeting sorafenib resistance induced by the hypoxic environment may represent a viable strategy for treating HCC.
Previous studies have indicated that hypoxia directly stimulates EMT in liver cancer cells and promotes EMT through diverse signaling pathways and molecular mechanisms (Aventaggiato et al., 2022). In HCC, HIF-1α activates various transcription factors, including Snail and Twist1, to facilitate EMT (Børretzen et al., 2021; Yang et al., 2024). Moreover, the Wnt/β-catenin pathway, under hypoxic conditions, upregulates β-catenin through a glycogen synthase kinase 3 beta (GSK-3β)-mediated mechanism, further advancing EMT (Xue et al., 2024). Activating these signaling pathways enhances the invasive and metastatic potential of tumor cells and plays a significant role in the development of sorafenib resistance. Thus, we hypothesize that targeted therapeutic strategies against EMT and its associated signaling pathways under hypoxic conditions may help overcome sorafenib resistance and improve the prognosis of patients with HCC. Consequently, exploring new drugs that can target EMT and restore the sensitivity of HCC cells to sorafenib under hypoxic conditions is essential.
Traditional Chinese medicine (TCM) has achieved significant advancements in anti-tumor activity and mechanism research (Xiang et al., 2019). In the context of HCC treatment, TCM not only directly impacts tumor cells to prolong patients’ progression-free and overall survival but also mitigates toxicity and enhances therapeutic outcomes, proving safe and effective (Xu et al., 2024). For example, TCM components such as polyphenols, quercetin, and ligustrazine have been identified to effectively reduce the expression of multidrug resistance-associated proteins (like P-glycoprotein) in resistant cells, thereby reversing resistance (Li et al., 2023; Liu et al., 2020; Qian et al., 2021). In terms of EMT, TCM has shown promising effects. Certain TCM components have been shown to inhibit EMT, potentially serving as targets to overcome tumor drug resistance. For instance, the combination of regorafenib with cisplatin can downregulate the expression levels of N-cadherin and matrix metalloproteinase-9 (MMP-9) in HepG2 liver cancer cells while upregulating E-cadherin, thus inhibiting EMT and suppressing cell migration and invasion (Chen et al., 2019). This suggests that TCM components can suppress EMT and enhance chemosensitivity, offering novel strategies for clinical HCC treatment.
Rhaponticin (RA), a diphenylethylene monomer compound isolated from Rheum palmatum native to northern China, exhibits significant anti-tumor bioactivity (Tang et al., 2019). Studies have indicated that RA shows potential therapeutic effects across various cancer types, particularly in inhibiting tumor growth and inducing apoptosis. Research by Kim and Ma (2018) has shown that RA significantly suppresses the HIF-1α signaling pathway, thereby reducing the proliferation and metastasis of breast cancer cells under hypoxic conditions. This finding highlights RA’s potent anti-metastatic and anti-angiogenic properties and its non-cytotoxic nature. However, specific research outcomes regarding RA’s role in hypoxia-mediated drug resistance and EMT in cancer are yet to be fully elucidated.
In this study, we initially assessed whether the sensitivity of HCC cells to sorafenib is diminished under hypoxic conditions and its correlation with EMT expression. Following this, we investigated the impact of sorafenib sensitivity in HCC cells after treatment with RA in combination.
Materials and Methods
Cell Culture and Treatment
The human HCC cell lines HepG2 and Huh7 and the human normal liver cell lines QSG-7701 and LO-2 are preserved in the General Surgery Department, Hui Ya Hospital of the First Affiliated Hospital. Cells were grown in Dulbecco’s modified Eagle medium (DMEM)/F12 (Sigma–Aldrich, St. Louis, MO, USA), with the addition of 10% fetal bovine serum (v/v) (FBS, Invitrogen, Waltham, MA, USA) and 1% penicillin–streptomycin solution (v/v) (Gibco, USA). These cultures are maintained in a humidified chamber at 37°C with a 5% carbon dioxide (CO2) environment. For hypoxic cultivation, HCC cells were grown in a complete medium within a tri-gas incubator, which was set to maintain an O2 concentration of 1%, a CO2 concentration of 5%, and N2 at 94%. The medium is changed every 24 h, and for experiments involving hypoxic treatment, the cells are subjected to cultivation at hypoxic conditions for 36 h.
To elucidate whether the mechanism underlying RA’s restoration of sorafenib sensitivity in hypoxia-induced resistance is associated with EMT, an EMT inhibitor (C19, at a concentration of 60 µM) was introduced. In brief, HepG2 and Huh7 cells were maintained in C19-containing cultured media under hypoxic conditions, followed by treatment with sorafenib alone or combined with half the half maximal inhibitory concentration (IC50) concentration of RA for HepG2 or Huh7 cells. RA (purity ≥ 98%) was purchased from ChemFaces (Wuhan, China). Both RA and sorafenib at various concentrations were dissolved in DMSO with a final concentration of 0.1% (Sigma–Aldrich).
Cell Viability Assay
As previously described (Zhang et al., 2018), the Cell Counting Kit-8 (CCK-8, Thermo Fisher Scientific, Carlsbad, CA, USA) was utilized to analyze cell viability. HepG2, Huh7, QSG-7701, and LO-2 cells were respectively seeded in a 96-well plate at a density of 5 × 103 cells per well, each well receiving 100 µL of the growth medium, and the cells were given 24 h to adhere. Subsequently, the medium was changed to a 10% FBS-enriched medium with varying concentrations of the experimental drugs (sorafenib or RA, ranging from 0 to 96 µM). After 48 h of incubation, 10 µL of CCK-8 reagent was added to each well, and the plates were incubated for an additional 3 h. The optical density was recorded at 450 nm using an MRX II plate reader (Dynex Technologies, USA). Cell viability was calculated as a percentage compared to the control group without treatment. The IC50 concentrations of sorafenib or RA were calculated based on the cell viability of cells with varying concentrations of the corresponding drug.
5-Ethynyl-2′-deoxyuridine (EdU) Analysis
To evaluate the cell proliferation ability, the EdU analysis was performed as previously described (Zhang et al., 2021). In brief, cells were seeded in 96-well plates at a density of 5 × 103 cells per well to ensure a healthy and adherent monolayer. Once the cells reached the desired confluence, they were treated with various concentrations of the test compounds, including sorafenib or RA, to assess their impact on cell proliferation. Following treatment, the Click-iT EdU Imaging Kit (Thermo Fisher Scientific) was employed to label and detect cell deoxyribonucleic acid (DNA) replication. EdU-positive cells were subsequently investigated under a fluorescence microscope. The number of EdU-positive cells was quantified and used to calculate the proliferation rate, which was then compared to that of the control group to determine the inhibitory effect of the test compounds on cell proliferation.
Western Blot Analysis
A western blot is an approach for investigating the protein levels in different types of samples using a specific antibody (Schmittgen & Livak, 2008). Total proteins were extracted with RIPA buffer (Invitrogen) containing 25 mM Tris–HCl (pH 7.6), 150 mM NaCl, 1% NP-40, 1% sodium deoxycholate, and 0.1% sodium dodecyl sulfate (SDS). Lysates were separated by SDS-PAGE (10%) and transferred to polyvinylidene difluoride (PVDF) membranes (Sigma–Aldrich). After blocking with 8% milk in tris-buffered saline with Tween (TBS-T) for 1 h at 37°C, to investigate the expression levels of EMT-related markers, membranes were incubated with primary antibodies against E-cadherin (1:1,000; #AF0138; Beyotime, Shanghai, China), Vimentin (1:1,000; #AF0318; Beyotime), and N-cadherin (1:1,000; #ab280375; Abcam, Waltham, MA, USA), with β-actin as a loading control. Horseradish peroxidase (HRP)-conjugated goat anti-mouse immunoglobulin G [IgG(H+L)] secondary antibodies (1:10,000; #AF0216; Beyotime) were used, and bands were detected using an enhanced chemiluminescence (ECL) kit (Pierce, Waltham, MA). Band quantification was performed using ImageJ software after film scanning.
Ribonucleic Acid (RNA) Isolation and Quantitative Reverse Transcription Polymerase Chain Reaction (qRT-PCR)
RNA was isolated using TRIzol reagent (Invitrogen) following the protocol provided. Complementary DNA was generated from 2 µg of RNA via reverse transcription with oligo(dT) primers and SuperScript II reverse transcriptase (Invitrogen). qRT-PCR targeted E-cadherin, Vimentin, N-cadherin, and glyceraldehyde 3-phosphate dehydrogenase (GAPDH), with GAPDH as the reference gene. Amplifications were performed in a SYBR Green mix (Qiagen, Valencia, CA, USA) in 20 µL volumes, with triplicate measurements for each sample. Gene expression levels were calculated using the 2−∆∆Ct method to find relative quantities, and the standard computed tomography (CT) approach determined the fold changes (Schmittgen & Livak, 2008). Primer sequences are listed in Supplementary Table S1.
Statistical Analysis
The experimental data were subjected to analysis using GraphPad Prism version 8.0 (GraphPad, USA), with results presented as the mean ± standard error of the mean (SEM). Statistical evaluations were performed utilizing Student’s t-test or one-way and two-way analysis of variance (ANOVA), followed by post hoc comparisons with Dunnett’s or Bonferroni’s tests, respectively. A p value of less than .05 was established as the threshold for statistical significance.
Results
Hypoxia Confers Sorafenib Resistance on HCC Cells
To determine whether hypoxia affects the hypoxia-inducible factor (HIF) in HCC cells against sorafenib, HepG2 and Huh7 cells were individually cultured under normoxic and hypoxic conditions, followed by treatment with varying concentrations of sorafenib. Then, cell viability was evaluated using the CCK-8 assay after a 24-h incubation. The results in Figure 1A and 1B indicate that the IC50 values for sorafenib in HepG2 and Huh7 cells were approximately fivefold higher under hypoxic conditions than normoxic ones.
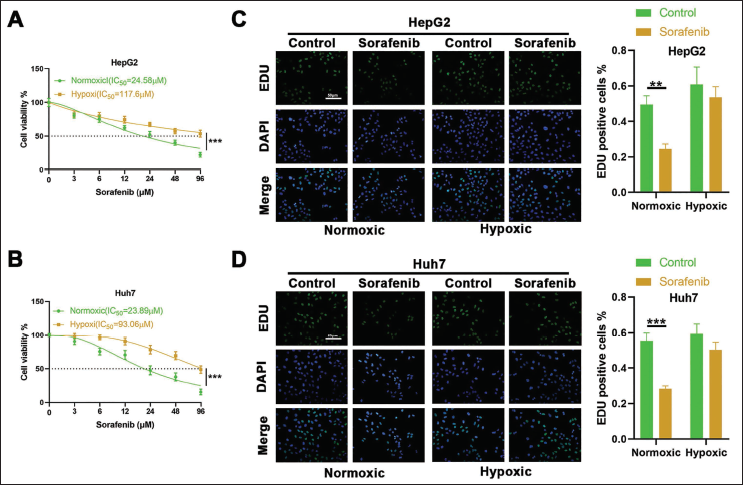
Next, HepG2 and Huh7 cells were again cultured under both normoxic and hypoxic environments and treated with a fixed concentration of 30 µM sorafenib. After 24 h, cell proliferation was assessed using EdU staining. Following sorafenib treatment, the findings revealed a significant decrease in EdU fluorescence intensity in both HepG2 and Huh7 cells under normoxic conditions. In stark contrast, the cytotoxic effect of sorafenib was markedly reduced under hypoxic conditions, as indicated by only a marginal decrease in EdU fluorescence levels in cells cultured under hypoxia after sorafenib treatment (Figure 1C and 1D). The above indications suggest that hypoxic conditions can confer sorafenib resistance to HCC cells.
Hypoxia Leads to Impaired Inhibition of EMT in HCC Cells by Sorafenib
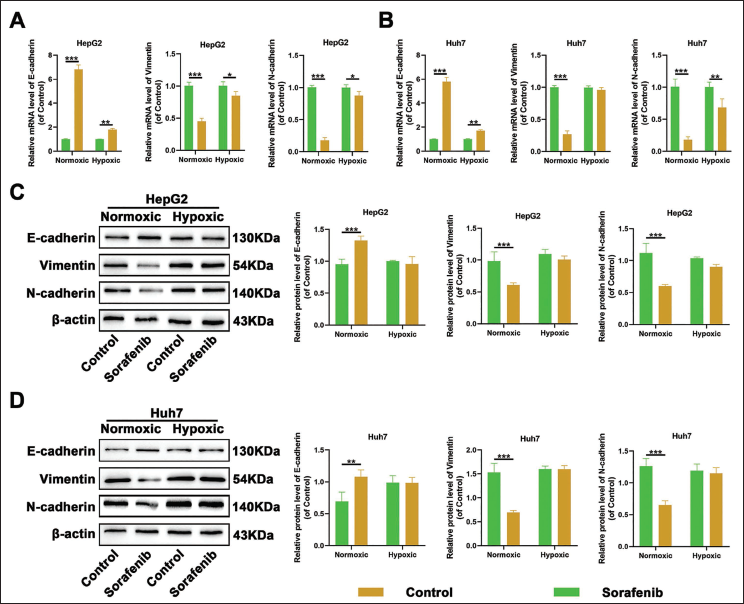
Subsequently, HepG2 and Huh7 cells were individually cultivated under normoxic and hypoxic environments and then exposed to 30 µM sorafenib. Following a 24-h incubation, the messenger ribonucleic acid (mRNA) and protein levels of EMT-associated proteins-E-cadherin, Vimentin, and N-cadherin were quantified using qRT-PCR and Western blotting. The findings indicated that in normoxic conditions, sorafenib treatment significantly enhanced the mRNA and protein levels of E-cadherin and concurrently diminished the mRNA and protein levels of Vimentin and N-cadherin, suggesting that sorafenib effectively suppresses the EMT process in HCC cells. In contrast, under hypoxic conditions, sorafenib treatment led to only a modest increase, or even no significant change, in the mRNA and protein expression of E-cadherin. Likewise, the mRNA and protein expression of Vimentin and N-cadherin showed a modest decrease or remained unchanged (Figure 2A–2D). Collectively, these observations imply that hypoxia attenuates the inhibitory effects of sorafenib on the EMT process in HCC cells.
RA Reversed Hypoxia-induced Sorafenib Resistance and EMT in HCC Cancer Cells
To assess whether RA could reverse hypoxia-induced sorafenib resistance, this study initially treated HCC cells with a range of concentrations of RA alone. The findings revealed that the IC50 of RA for HepG2 cells was 48.87 µM, and for Huh7 cells was 35.46 µM (Figure 3A). This suggested that a specific concentration of RA alone can significantly reduce the viability of HCC cells, thereby inhibiting their growth.
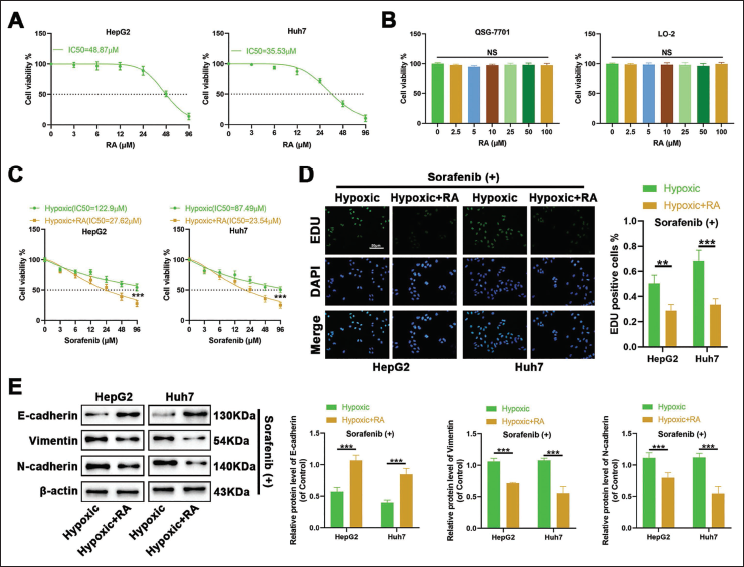
Subsequently, a range of RA concentrations was applied to normal human liver cells QSG-7701 and LO-2 to establish the safety profile. The results indicated that RA concentrations from 0 to 100 µM did not markedly decrease cell viability, thus confirming its biosafety (Figure 3B).
Thereafter, under hypoxic conditions, cells were treated with a series of sorafenib concentrations, followed by intervention with half the IC50 concentration of RA for both HepG2 and Huh7 cells. Cell viability was then assessed using the CCK-8 assay. Figure 3C demonstrates that the sensitivity to sorafenib was enhanced when combined with RA.
Moreover, under hypoxic conditions, cells were treated with 30 µM sorafenib and then intervened to half the IC50 concentration of RA for HepG2 and Huh7 cells, respectively. EdU assay results indicated that the inhibitory effect of sorafenib on the proliferation of HCC cells under hypoxia was markedly potentiated in the presence of RA (Figure 3D).
Finally, we further examined the expression of EMT-related proteins under the same treatment conditions. Figure 3E illustrates that sorafenib significantly increased the protein expression of E-cadherin and markedly decreased the protein expression of Vimentin and N-cadherin when combined with RA.
These research findings suggest that treatment with RA can restore the sensitivity of HCC cells to sorafenib under hypoxic conditions.
EMT Inhibitors Render RA’s Restoration of Sensitivity to Sorafenib in HCC Cells Ineffective
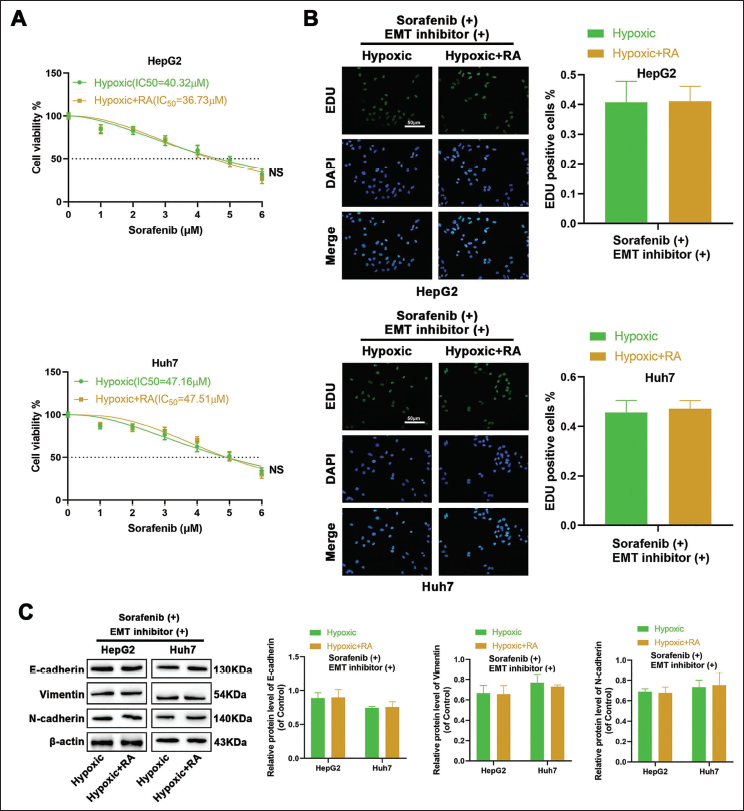
Then, whether EMT is involved in the reversive role of RA in hypoxia-induced sorafenib resistance was further explored. As depicted in Figure 4, our data demonstrated that introducing the EMT inhibitor (C19) negated the significant impact of RA treatment on the sensitivity of HCC cells to sorafenib under hypoxic conditions. There were no notable alterations in cell viability (Figure 4A), cell proliferation capacity (Figure 4B), or the expression of EMT-related proteins (Figure 4C) when RA was combined with sorafenib. These observations suggest that RA can re-sensitize liver cancer cells to sorafenib by inhibiting hypoxia-mediated EMT. Once EMT is suppressed, RA loses its efficacy, indicating that the reversal of sorafenib resistance by RA is indeed EMT-dependent.
Discussion
The current study provides a comprehensive analysis of the impact of hypoxia on sorafenib resistance and EMT in HCC cells, as well as the potential of RA to reverse these effects. Our findings contribute to the understanding of the complex interplay between hypoxia, drug resistance, EMT, and the therapeutic potential of RA in HCC treatment.
Hypoxia is a pivotal factor contributing to the development of sorafenib resistance in HCC cells. A growing amount of evidence suggests that the hypoxic microenvironment in HCC fosters sorafenib resistance through many mechanisms (Méndez-Blanco et al., 2018). HIFs, particularly HIF-1α and HIF-2α, play a significant role in HCC progression, enhancing tumor cell survival under hypoxic conditions and correlating with resistance to various anti-cancer drugs (Xiong et al., 2017). It is noteworthy that studies have confirmed that hypoxic environments can lead to sorafenib resistance in HCC cells through various mechanisms. Studies have identified hypoxia-induced YAP activation as a critical factor in sorafenib resistance among HCC cells. Under hypoxic conditions, YAP is predominantly nuclear, promoting cell survival and evading apoptosis by transactivating target genes like CTGF and CRY61, which leads to sorafenib resistance (Zhou et al., 2016). Statins have been shown to inhibit YAP’s nuclear accumulation and transcriptional activity, overcoming hypoxia-mediated sorafenib resistance (Yan et al., 2019). Furthermore, prolonged sorafenib treatment can intensify intratumoral hypoxia, accelerate tumor growth, and exacerbate the development of resistance. Targeting the hypoxic microenvironment with therapeutic strategies may help overcome sorafenib resistance and enhance treatment efficacy. Consistent with these previous studies (Yan et al., 2019; Zhou et al., 2016), our study confirmed that hypoxia is a significant factor in developing sorafenib resistance in HCC cells. The increased IC50 values for sorafenib under hypoxic conditions indicate a reduced sensitivity to this drug. EdU staining results further substantiate this resistance, demonstrating that the cytotoxic effects of sorafenib are attenuated under hypoxia. These insights underscore the clinical challenges in treating HCC within hypoxic tumor microenvironments, where the efficacy of sorafenib is significantly impaired.
Advanced research has uncovered that hypoxia not only induces sorafenib resistance in HCC cells but also weakens the ability of the drug to inhibit EMT, a pivotal process in cancer progression that bestows upon tumor cells the traits of invasion and metastasis (Saxena et al., 2020). In this research, sorafenib potently inhibits the EMT process in HCC cells under normoxic conditions, as indicated by the enhanced expression of E-cadherin and the reduced levels of Vimentin and N-cadherin. However, the impact of sorafenib on EMT is considerably diminished under hypoxic conditions, suggesting that hypoxia mitigates the inhibitory effects of sorafenib on EMT. Herein, E-cadherin expression is progressively silenced, while the expression of N-cadherin and Vimentin is upregulated by hypoxia. Our data was supported by a previous study, which demonstrated that hypoxia not only fosters sorafenib resistance but also diminishes sorafenib’s capacity to curb EMT in HCC cells, resulting in alterations of EMT markers (Xia et al., 2020). These insights underscore the significance of targeting EMT and its associated signaling pathways in the treatment of HCC, particularly under hypoxic conditions. Ultimately, hypoxia emerges as a critical factor in the development of sorafenib resistance in HCC cells, augmenting tumor cell survival and drug resistance through the activation of EMT.
In our study, the incorporation of RA has emerged as a promising strategy to counteract hypoxia-induced sorafenib resistance and EMT in HCC cells. RA, a diphenylethylene glycoside derived from various medicinal plants, has demonstrated notable anti-cancer properties (Chen et al., 2020). It has been shown to suppress tumor growth and trigger apoptosis in cancer cells effectively. Specifically, RA has exhibited proliferation-inhibiting effects in osteosarcoma cells and the ability to induce apoptosis (Mickymaray et al., 2021). Furthermore, RA has been identified to inhibit the proliferation of lung cancer cells with an IC50 value of 25 µM (Wang et al., 2021). Our research has revealed that RA effectively reverses hypoxia-induced sorafenib resistance and EMT in HCC cells. When administered alone, RA significantly reduced the viability of HCC cells, with an IC50 value of 48.87 µM for HepG2 cells and 35.46 µM for Huh7 cells. Crucially, RA demonstrated biosafety when applied to normal human liver cells QSG-7701 and LO-2 at concentrations ranging from 0 to 100 µM. Under hypoxic conditions, treatment with RA enhanced the sensitivity of HCC cells to sorafenib, as evidenced by increased cytotoxic effects and increased inhibition of cell proliferation when sorafenib was combined with RA. Moreover, following combined treatment with RA and sorafenib, there was a marked increase in E-cadherin protein expression and a decrease in Vimentin and N-cadherin protein expression, indicating a reversal of EMT. These findings suggest that RA may overcome sorafenib resistance by inhibiting EMT in HCC cells under hypoxic conditions. Given the widespread resistance to sorafenib in HCC treatment, which may be linked to EMT (van Malenstein et al., 2013), the ability of RA to effectively inhibit EMT could be instrumental in overcoming this resistance. Similar findings have been shown in several previous studies. For example, regorafenib has been reported to reverse sorafenib resistance by inhibiting EMT in HCC (Chen et al., 2019). A more recent study demonstrated that emodin, a widely studied TCM compound, could suppress EMT to overcome sorafenib resistance in HCC (Wang & Zhang, 2025), which supports our hypothesis. To confirm EMT’s role in RA’s sorafenib sensitivity restoration, this study introduced an EMT inhibitor, C19, under hypoxic conditions. The results indicated that when EMT was suppressed, the efficacy of RA in enhancing sorafenib sensitivity was negated, as there were no significant changes in cell viability, proliferation, or expression of EMT-related proteins. This suggests that the capacity of RA to reverse sorafenib resistance is contingent upon its impact on EMT, underscoring the significance of targeting EMT in overcoming drug resistance in HCC.
In summary, this study delves into the effects of hypoxia on sorafenib resistance and EMT in HCC cells, as well as the potential of RA to counteract these influences. However, certain limitations warrant acknowledgment. First, the in vitro nature of our experiments, conducted with cell lines, may not accurately replicate the complexities of the in vivo environment. Subsequent studies should incorporate in vivo models to corroborate our findings and provide a more comprehensive understanding. Second, although we have explored the role of RA in reversing sorafenib resistance and EMT, precise molecular mechanisms are required for further exploration. Future research should aim to pinpoint the specific molecular targets of RA and elucidate its interactions with hypoxia-induced pathways and EMT regulators. Additionally, assessing the safety and efficacy of RA in clinical settings is crucial. Clinical trials should be considered to evaluate RA’s potential as a therapeutic agent for HCC, either as a standalone treatment or in conjunction with sorafenib. The potential for RA to synergize with other therapeutics, such as radiotherapy or immunotherapy, should also be investigated to enhance treatment outcomes.
Conclusion
In conclusion, our research delves into the effects of hypoxia on sorafenib resistance and EMT in HCC cells, as well as the potential of RA to counteract these influences. Hypoxia enhances resistance to sorafenib and weakens its capacity to inhibit EMT. RA has been shown to reverse these effects by suppressing hypoxia-induced EMT, exhibiting both biosafety and efficacy. These discoveries underscore the significance of novel therapeutic targets and the promise of RA. However, further investigation into the underlying mechanisms, additional clinical assessments, and the development of more effective HCC treatment strategies are warranted.
Footnotes
Abbreviations
ANOVA: Analysis of variance; CCK-8: Cell Counting Kit-8; CO2: Carbon dioxide; CRY61: Cysteine rich angiogenic inducer 61; CTGF: Connective tissue growth factor; DMEM: Dulbecco’s modified Eagle medium; DMSO: Dimethyl sulfoxide; DNA: Deoxyribonucleic acid; ECL: Enhanced chemiluminescence; EdU: 5-Ethynyl-2′-deoxyuridine; EMT: Epithelial–mesenchymal transition; FBS: Fetal bovine serum; GAPDH: Glyceraldehyde 3-phosphate dehydrogenase; GSK-3β: Glycogen synthase kinase 3 beta; HCC: Hepatocellular carcinoma; HIF-1α: Hypoxia-inducible factor-1α; HRP: Horseradish peroxidase; IC50: Half maximal inhibitory concentration; IgG: Immunoglobulin G; MMP-9: Matrix metalloproteinase-9; N2: Nitrogen; O2: Oxygen; PVDF: Polyvinylidene difluoride; qRT-PCR: Quantitative reverse transcription polymerase chain reaction; RA: Rhaponticin; RIPA: Radioimmunoprecipitation assay; RNA: Ribonucleic acid; SDS-PAGE: Sodium dodecyl sulfate-polyacrylamide gel electrophoresis; SEM: Standard error of the mean; TBS-T: Tris-buffered saline with Tween; TCM: Traditional Chinese medicine; YAP: Yes-associated transcriptional regulator.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
