Abstract
Background
Polydopamine (PDA) shows promising potential in disease diagnosis and targeted drug delivery.
Objectives
This study aimed to investigate the role of Paeonol (PAE)-modified PDA nanoparticles (NPs) in the treatment of colitis-associated cancer (CAC).
Materials and Methods
First, PAE-modified PDA NPs were prepared, and then the mouse model of the CAC model was set up. The mice were divided into a blank control group, a CAC model group, a PAE group, and PAE + nano group (
Results
PAE alleviated the pathological injury of colonic tissue in CAC mice and promoted the apoptosis of CAC cells, and a PDA NPs carrying PAE exhibited a greater curative effect when dramatically decreasing IL-6 and STAT-3 expression. The presence of STAT-3 inhibitors decreased the CAC cell proliferation and induced apoptosis. A combination of PAE + nano and WP1066 resulted in the most significant difference.
Conclusion
PAE-modified PDA NPs can attenuate injuries of the colon tissue in CAC mice and promote the apoptosis of CAC cells by inhibiting the IL-6/STAT-3 pathway and increasing the expression level of Bcl-2, thereby improving the condition of CAC.
Keywords
Introduction
Morbidity and mortality of colorectal cancer (CRC) keep increasing, and CRC has become one of the most common cancers worldwide, as well as an important cause of death (Zygulska & Pierzchalski, 2022). Long-term inflammation is one of the characteristics of tumors, and many cancers originate from chronic inflammation or present long-term inflammation throughout the development process (Bai et al., 2021). Inflammatory bowel disease, namely, Crohn’s disease and ulcerative colitis, is closely related to the development of CRC. Inflammatory bowel disease with inflammation can stimulate the colonic mucosa for a long time, thereby elevating the risk of CRC. Despite significant progress in the treatment and technologies, advanced colitis-associated cancer (CAC) is hardly cured, and those patients have a low survival rate (Zhao & Jiang, 2021). And there are still great challenges in curing advanced CRC. Therefore, it is urgent to develop new therapeutic methods or drugs with better efficacy and safety to control the progression of CAC.
Tissue engineering (TE) aims at developing biological substitutes for replacing, repairing, or enhancing tissue functions. As one of the most important components in TE, biomaterials (or scaffolds) stimulate and regulate target cells to regenerate new tissues (Venkatesan et al., 2022). In biomaterials, nanoparticles (NPs) have become a powerful tool for improving scaffolds’ mechanical and biological properties. Recent decades have witnessed the rapid development of nanotechnology, which has provided new opportunities for tumor therapy (Mitchell et al., 2021). As an ideal drug carrier, NPs have small particle size, high surface area, and adjustable release, which can strengthen the solubility, stability and targeting of drugs, thereby improving therapeutic efficacy and reducing side effects (Peng et al., 2021). The advent of polydopamine (PDA), a multifunctional biomaterial, improves biomedical applications of NPs (Jin et al., 2020). PDA is derived from sea mussels. With good biocompatibility, biodegradability and photothermal conversion, PDA might be used for disease diagnosis and drug delivery (Zhu et al., 2021). PDA-loaded NPs are characterized by strong stability, high drug-loading capacity, good biocompatibility, convenient surface functionalization, and anti-tumor effects, so they might be a potential drug-delivery platform (Zhang et al., 2021).
Increasingly, researchers are focusing on natural products as potential alternatives for cancer treatment. Paeonol (PAE) is a phenolic compound derived from Paeoniae Alba with anti-cancer, anti-inflammatory, anti-oxidation, and other biological activities (Liu et al., 2021). PAE is indicated as an effective anti-neoplastic agent and might serve as an alternative to conventional treatment (Lv et al., 2022). Therefore, this study aims to explore the mechanism whereby polydopamine nanoparticles (PDA NPs) carrying PAE mediate apoptosis of CAC cells. This evidence might provide evidence for the potential application value of PAE-modified PDA NPs in the treatment of CAC and provide new novel insight into CAC treatment.
Materials and Methods
Experimental Animals, Materials and Instruments
Animals
The C57Bl/6J male mice (6–8 weeks, weight: 15–20 g) were purchased from Beijing Huafukang Biological Company and raised in an SPF laboratory with free access to standard food and water.
Materials and Instruments
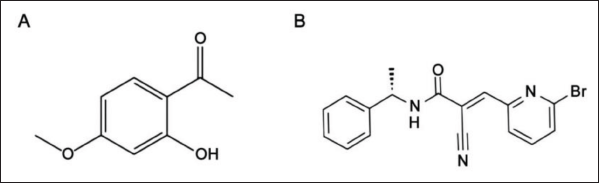
Azomethane (AOM, batch number A5486) and Dextran Sodium Sulfate (DSS, batch number 42867) were purchased from Sigma; PAE (batch number S28004, 2.5/10 g) from Shanghai Yuanye Biological Co. Ltd., with molecular structure diagram of PAE shown in Figure 1A. Other materials included signal transducer and activator of transcription 3 (STAT-3) inhibitor WP1066 (Beijing, Biolabs, batch number M00377-DJG, purity 99.67%, Figure 1B), Cell Counting Kit (CCK)-8 (Shanghai Yihui Trade Co. Ltd.); hematoxylin and eosin (HE) staining kit (Yeasen Biotechnology (Shanghai) Co. Ltd.); polymerase chain reaction (PCR) primers (iCell Bioscience Inc, Shanghai); Trizol Reagents (Invitrogen, USA); primary and secondary antibodies (Abcam, USA) and an inverted microscope (Nikon, Japan).
Molecular Structure Diagram. (A) Molecular Structure Diagram of Paeonol (PAE); (B) Molecular Structure Diagram of WP1066.
Methods
Preparation of PAE-loaded Polydopamine Nanoparticles
Dopamine powder was put into the aqueous solution and stirred at high speed with a magnetic stirrer (50°C) to fully dissolve it in water to 50 mL (pH = 9) to obtain a dopamine solution (concentration: 0.2 mg/mL). The solution gradually turned dark brown. After centrifugation, the precipitate was collected, washed with ion water, and then centrifuged. The bottom precipitate was dispersed, and the supernatant was used to obtain PDA NPs. PAE powder was added to the solution of PDA NPs and stirred using a magnetic stirrer. The modified NPs were separated from the solution using a centrifuge and washed with ionized water to obtain PAE-modified PDA NPs (Chen et al., 2021).
Observation of Nanoparticle Morphology
The NP dispersion was dropped evenly on the conductive adhesive and sprayed for 1 min after natural drying. Then, the morphology of PAE-loaded PDA NPs was observed with a scanning electron microscope (SEM) and a transmission electron microscope (TEM).
Particle Size Observation
The particle size was detected using dynamic light scattering. The freeze–dried blank and PAE-loaded NPs were weighed, ultrasonically dispersed in distilled water, and placed in a quartz cuvette to measure the particle size distribution of NPs.
Loading Capacity
PDA NPs solution (0.5 mg/mL) was prepared, PAE (0.1–2 µg) was stirred in double steaming water, and the PAE released in PDA NTP-PAE at different time points was collected in the water bath solution.
Construction and Grouping of the CAC Mouse Model
According to previous studies (Arnesen et al., 2021; Wang et al., 2021), AOM/DSS induction was used to construct a mouse model of CAC by stimulating sustained damage of colonic mucosal epithelium and inducing heteroplastic proliferation and mutation of colonic epithelial tissue. For the establishment of the CAC model, mice were intraperitoneally injected with 10% AOM solution (10 mg/kg), and 7 days later, they drank 2.5% DSS solution for five consecutive days and then sterile water for 2 weeks. There are three cycles of intervention (Ishikawa & Herschman, 2010). After the modeling, the general condition and changes in the colonic histopathological structure of mice were observed to determine whether the CAC mouse model was successfully constructed.
The mice were divided into blank control group (mice without any intervention), CAC model group, PAE group [CAC model mice were subcutaneously injected with PAE (20 mg kg−1)], PAE + nano group (CAC model mice were subcutaneously injected with PAE-modified PDA NPs (
Observation of Mouse
The Disease Activity Index (DAI) of the mice was detected at the same time every day, including stool properties, mental activity, bloody feces prolapse, fur gloss, and so on. The DAI was calculated with the formula body mass index + stool properties and hematochezia index/3 (Kohno et al., 2005).
Specimen Collection
After administration of the last dosage of the drug, collection of specimens began. The night before sample collection, the mice were fasted but allowed to drink water for about 12 h. On the second day, the mice were weighed and sacrificed under anesthesia with an intraperitoneal injection of amobarbital sodium (0.1 mL/10 g). The abdomen of the mice was sterilized with alcohol and cut longitudinally. The colon tissue at 6 cm–8 cm above the anus was taken and placed in 10% neutral formalin for later use.
Histopathological Examination
The colon tissues of the mouse were immersed in ethanol (70%) for 30 min and then washed repeatedly three times, each time for 5 min. After dehydration, the colon tissues of each group were marked and placed vertically on the bottom of paraffin, embedded in paraffin, cut into slices (5 µm), spread flat at 43°C, and baked at 65°C for half an hour. The sections were then immersed in hematoxylin and stained with eosin for 1–2 min. Afterwards, the sections were dehydrated in 70%, 80%, and 95% ethanol for 3–5 min, and then they were sealed with neutral gum and observed.
CCK-8 Assay
The primary colorectal cells of the P4 generation with a growth density of 80% were inoculated with 1 × 105 cells/mL on a 96-well plate and cultured until adherent to the wall. PDA NFS-PAE NPs were added to the well plate, and a blank control hole was set up without any substance being added. The cells were cultured for 12, 24, and 48 h, and the medium was discarded after culture. CCK-8 solution was incubated for 1 h; the OD value was measured at 450 nm, and cell proliferation activity was calculated.
Transwell Assay
Transwell cells were inoculated with serum-free medium, supplemented with Matrigel matrix glue, then inoculated with fetal bovine serum medium, set up multiple pores and cultured at 37°C and 5% of CO2 for 24 h. Transwell cells were taken, the culture medium was removed, PBS was used for cleaning, crystal purple was given again, non-migrating cells were removed with cotton swabs, and PBS was used for cleaning.
Flow Cytometry
Cells treated with PBS were cultured in a CO2 incubator. After culturing, the cells (1 × 106) were collected and fixed with 500 µL of 70% ethanol for 12 h. The fixed cells were washed with PBS solution and centrifuged to remove the liquid. Following the addition of 0.5 mL of 10 mg/L PI, the cells were placed for 60 min, and the apoptosis rate was detected by flow cytometry after staining.
Reverse Transcription Quantitative Polymerase Chain Reaction (RT-qPCR)
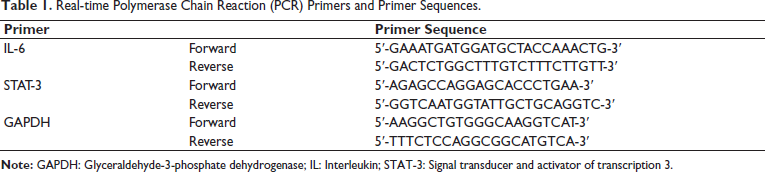
The total RNA in the cells was extracted by TRIzol, and RT-PCR prepared complementary deoxyribonucleic acid (cDNA) double strands according to Roche Company’s RT-PCR method. With cDNA as a template and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as an internal reference, RT-qPCR was conducted to determine gene expression using the 2−∇∇CT method and primers (Table 1). Table 1 lists the primers and their sequences.
Real-time Polymerase Chain Reaction (PCR) Primers and Primer Sequences.
Western Blot
The total protein was extracted, and its content was determined using the BCA method. The proteins were separated by SDS-PAGE (80 V, time 30 min; 120 V, time 60 min), transferred to PVDF film (100 min) by wet transfer method, and sealed with 5% skimmed milk for 1 h. The membrane was then incubated overnight with 1:400 monoclonal antibodies and 1:5,000 secondary antibodies. Afterward, the membrane was exposed to a luminescent agent, and Grayscale processing was performed using ImageJ software (internal reference GAPDH).
Statistical Analysis
Data were processed by SPSS 26.0, and GraphPad Prism and the calculated data were expressed as [mean ± standard deviation (SD)], following normal distribution and homogeneity of variance. All data calculations were performed using the
Results
Successful Establishment of a CAC Mouse Model and PAE-modified Polydopamine Nanoparticles
Compared with the mice in the blank control group, the CAC mice appeared to have lost stools and rectal prolapse, and they moved slowly with dark fur color. These systems indicate that the mice were successfully modeled. After the intervention with PAE and PAE NPs, the above symptoms were all relieved. In this experiment, a total of two mice died, all of which were mice in the CAC model group during DSS feeding. After dissection, it was found that the mice died due to ulcer adhesion and severe intestinal mucosal edema. The modeling success rate is 90%.
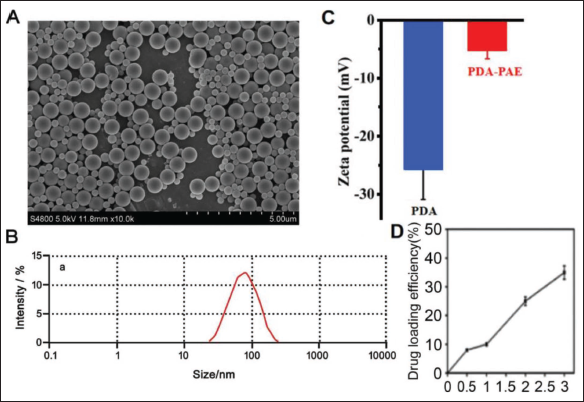
It can be seen by SEM that the surface of PDA NPs is smooth and spherical, the shell is light gray or white, the arrangement is tight, and there is no adhesion between particles (Figure 2A). The particle size distributions of these two NPs are very concentrated, indicating that they have a narrow particle size distribution range. In addition, the PDI of about 0.2 indicates that the NPs are small in size and concentrated in distribution and can avoid being phagocytized by Kupffer cells (Figure 2B). The zeta potential values for PDA and PDA NTP-PAE were −(25.7 ± 5.2) and −(5.1 ± 1.53) mV, respectively (Figure 2C). The PAE drug load ratio (PAE:PDA) increases with the amount of PAE added (Figure 2D). Meanwhile, the encapsulation rate of PDA NPs-PAE is (43.06 ± 5.29)%. The results show that PDA NPs-PAE is successfully constructed.
Detection Results of Paeonol (PAE)-modified Polydopamine Nanoparticles (n = 10). (A) Scanning Electron Microscope (SEM) Images of Blank Polydopamine Nanoparticles and PAE-modified Polydopamine Nanoparticles (NPs); (B) Particle Size Diagram of PAE-modified Polydopamine NPs; (C) Polydopamine (PDA) NPs-PAE Zeta Potentiogram; (D) PDA NPs-PAE Drug Load Rate.
PAE-loaded PDA NPs Alleviate Colon Tissue Injuries in CAC Mice and Promote the Apoptosis of CAC Cells Through Downregulation of Interleukin (IL)-6 and STAT-3 expression
In terms of the activity status of the mice in the four groups, it was found that the CAC model group had a higher DAI (compared with the blank control group,
Effect of Paeonol (PAE)-modified Polydopamine Nanoparticles on Apoptosis of Colitis-associated Cancer (CAC) Cells (n = 10). (A) Mouse Disease Activity Index Score; (B) Representative Hematoxylin and Eosin (HE) Staining Images of Pathological Changes in Mouse Colon Tissue; (C) Cell Counting Kit (CCK)-8 Assay of Relative Cell Viability; (D) Transwell Assay of Migrated Cells; (E) Flow Cytometry of Cell Apoptosis Activity; (F) Reverse Transcription Quantitative Polymerase Chain Reaction (RT-qPCR) Analysis of Interleukin (IL)-6 Gene Expression in Cells; (G) RT-qPCR Analysis of Signal Transducer and Activator of Transcription 3 (STAT-3) in Cells; (H) Western Blot Detection of IL-6, STAT-3 and Bcl-2 Protein Expression in Cells; Compared with Model Group, #p < .05.
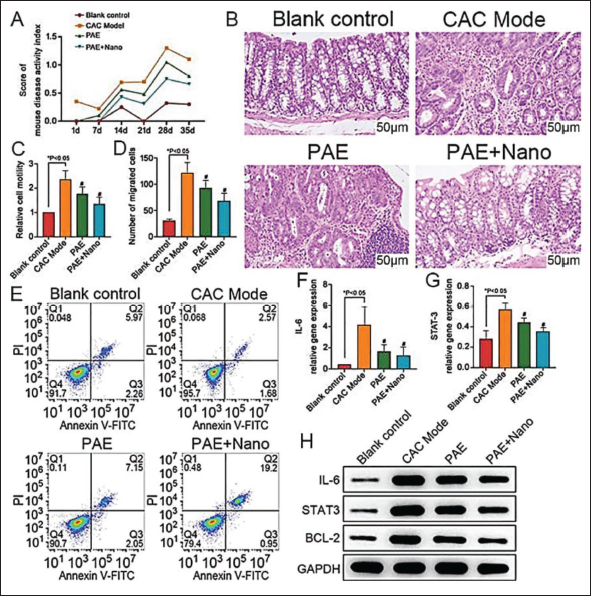
Furthermore, we evaluated the biological behavior of mice in the four groups. We found that cell proliferation and migration in the CAC model group were increased, while cell apoptosis was decreased (
Further PCR detection of IL-6 and STAT-3 messenger ribonucleic acid (mRNA) expression of mice in each group showed that the expression levels of IL-6 and STAT-3 mRNA in CAC mice were significantly higher than those in the blank control group (
STAT-3 Inhibitors Decrease the Proliferation Ability and Apoptosis of CAC
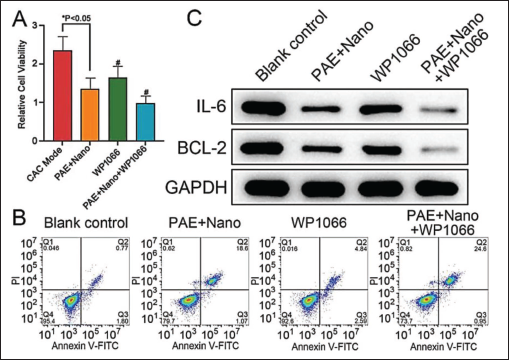
In order to further observe the effect of the IL-6/STAT-3 signaling pathway on CAC cell viability, this study used a STAT-3 inhibitor (WP1066) to intervene. It is shown that PAE-modified PDA NPs can play a synergistic role with STAT-3 inhibitors to reduce the activity of the IL-6/STAT-3 signaling pathway. Meanwhile, the cell viability of the WP1066 group was lower than that of the CAC model group, and the cell apoptosis was increased; moreover, the apoptosis was further accelerated in the PAE + nano + WP1066 group (Figure 4A and 4B). These indicate that STAT-3 is involved in the process of CAC cell apoptosis. Western blot results show that the protein expression of Bcl-2 in the WP1066 group was significantly higher than that of the CAC model group. The combination of NP-carrying PAE and WP1066 achieved the most significant effect on Bcl-2 protein expression (see Figure 4C). In addition, STAT-3 protein in the WP1066 group was significantly inhibited. Moreover, IL-6 protein expression was significantly lower than that in the CAC model group and was lower in the PAE + nano + WP1066 group (see Figure 4C). It shows that PAE-modified PDA NPs promote the apoptosis process of CAC cells, probably through the inhibition of STAT-3.
Effect of Paeonol (PAE)-modified Polydopamine Nanoparticles on Apoptosis of Colitis-associated Cancer (CAC) Cells Via Interleukin (IL)-6/Signal Transducer and Activator of Transcription 3 (STAT-3) (n = 10). (A) Cell Counting Kit (CCK)-8 Assay of Relative Cell Viability Upon Treatments; (B) Apoptotic Activity in Groups; (C) Western Blot Detection of Bcl-2 Protein Expression in Cells; Compared with Model Group, #p < .05.
Discussion
As new and promising technology, nanomedicine and nano delivery systems are rapidly developing, among which materials in the nanoscale range serve as means for diagnostic tools or to deliver therapeutic agents to specific target sites (Bhatia et al., 2022; Mishra, Mishra, Nand Rai, Singh, et al., 2023; Mishra, Mishra, Nand Rai, Vamanu, et al., 2023a, 2023b). NPs with specific functions can be prepared by introducing functional groups on PDA molecules or modifying them with other substances (Fu et al., 2021). At the same time, previous studies have found that as a multifunctional nanomaterial, the surface of PDA is rich in phenolic hydroxyl and amino groups, which can be covalently bonded with a variety of drugs to achieve high-efficiency loading of drugs (Yang et al., 2023). Moreover, it has good biocompatibility, and its stability and targeting
The results show that PAE alleviated the colon tissue injuries in CAC mice and promoted the apoptosis of CAC cells, and the PAE + nano group had a better effect on enhancing injury healing and apoptosis, which is in line with the research results of Xiao et al. (2021). At the same time, it can be found in the results of this study that the expression of Bcl-2 and IL-6 in the PAE + nano group is significantly reduced. This may be because PAE can block tumor cells in the S phase, thus reducing cell mitosis and inhibiting cell proliferation, while Bcl-2, as a marker protein of cell apoptosis, is significantly downregulated during the process of cell apoptosis. IL-6 can drive the proliferation, invasion, and metastasis of tumor cells through signaling pathways such as AK/STAT-3 and hinder the anti-tumor immune response. When its expression is reduced, it can effectively inhibit the growth and metastasis of CAC tumors, thereby enhancing the anti-tumor immune response and improving the overall anti-tumor efficacy of treatment. Ultraviolet light is one of the important factors that lead to drug degradation. PDA has good light absorption characteristics, which protect PAE from direct ultraviolet radiation by absorbing ultraviolet light. At the same time, phenolic hydroxyl groups in PDA can interact with PAE to form hydrogen bonds and other non-covalent bonds. This interaction helps to stabilize the chemical structure of PAE, maintain its molecular integrity under high-temperature conditions and prevent chemical bond breakage or rearrangement due to thermal energy. Moreover, it was found (Wu, Wang, Dong, et al., 2023) that PDA can form a hydrophobic layer under certain conditions to further reduce the direct contact of water to PAE. This hydrophobic layer can effectively prevent water from penetrating the interior of drug molecules, avoid drug failure caused by hydrolysis reactions and improve the stability of PAE in different humidity environments. Through the above-mentioned protective effects, drug-loaded NPs can effectively protect PAE from external factors such as light, temperature, humidity, and so on, thereby extending the storage life of drugs and enhancing their bioavailability and safety (Szewczyk et al., 2023). However, the effect of different environmental factors on NPs has not been observed in this study, which is also a shortcoming of this study. The performance of PAE-PDA NPs should be further observed in subsequent experiments to improve the bioavailability of nanomaterials. At the same time, as a hydrophobic drug, PAE has the characteristics of low melting point, poor water solubility, and so on. By interacting with the hydrophobicity of PDA, PAE can be stably wrapped in the PDA-based nanocoliths and reduce the early release of drugs in the blood circulation process so as to improve the stability of drugs effectively. Moreover, PDA contains a large number of phenol hydroxyl groups; these phenol hydroxyl groups can form hydrogen bonds with the hydroxyl group in PAE, which can enhance the binding force between PAE and PDA so as to improve the stability of the drug on the PDA carrier while preventing the drug from falling off and losing during delivery. In addition, the benzene ring in PAE molecules can be combined with the ύ-electron system of PDA through ύ–ύ stacking, which can stably adsorb drugs in a neutral environment. When reaching acidic environments such as tumors, the ύ–ύ stacking may be weakened, thus promoting drug release and improving drug delivery efficiency (Wang et al., 2017). Also, the presence of PDA reduces the leakage and early release of drugs, thereby improving the delivery efficiency and anti-inflammatory and anti-tumor effects of PAE. The surface of dopamine is rich in functional groups, chemically modified or functionalized, which allows drug-loaded NPs to pass through the surface to achieve targeted delivery and controlled release, thereby improving the local efficacy and reducing systemic side effects (Wu et al., 2022). In addition, PDA itself has anti-tumor activity, and it can elevate the concentration of copper ions in cells and activate autophagy pathways. It may trigger oxidative stress, damage the DNA of cancer cells, and induce apoptosis and autophagy, as PDA might reduce the viability of cancer cells and inhibit tumor growth (Han et al., 2021). Therefore, the drug-loading mechanism of PDA can improve the therapeutic effect of PAE and alleviate the pathological damage of colonic tissue in CAC mice.
Previous studies have found that excessive activation of the IL-6/STAT-3 signaling pathway can promote the epithelial-mesenchymal transformation of tumor cells, thereby reducing the adhesion between cells, resulting in the release of a large number of tumor cells and promoting the invasion and migration of tumor cells. Moreover, inhibition of IL-6/STAT-3 pathway activity can effectively inhibit the epithelial-mesenchymal conversion of colon cancer stem cells, thus providing a new target for the treatment of colon cancer (Noguchi & Shimonishi, 2025). At the same time, inhibition of IL-6/STAT-3 could play a protective role in the occurrence of Escherichia coli-associated tumors in mice (Grivennikov et al., 2009). This also suggests that IL-6/STAT-3 may play an important regulatory role in the treatment of CAC (Zhang et al., 2024; Zheng et al., 2018). WP1066, as a novel STAT-3 inhibitor with strong cell permeability, can significantly inhibit tumor survival
Conclusion
In conclusion, this study successfully prepared PAE-modified PDA NPs and conducted follow-up experiments to draw the following conclusions: PAE-loaded PDA NPs promote the apoptosis of CAC cells and relieve the colon tissue injuries through inhibiting IL-6/STAT-3 pathway and increasing the expression level of Bcl-2. On the basis of previous studies, this study further constructed new nano-targeted drugs, thus providing more possibilities for the treatment of CAC patients and having certain clinical application values.
However, this study also has some limitations: (a) The biological characteristics of PDA–PAE NPs were not observed in detail in this study, and its effects on cells have not been clarified, and whether the NPs are toxic to normal cells needs further investigation; (b) this study was only analyzed at the animal level and the specific mechanism of action of NPs could not be clarified; (c) the conventional treatment of CAC has not been compared and observed in this study, which cannot reflect the advantages of clinical application of this NP; (d) this study did not observe the long-term application effect of PDA–PAE NPs in CAC and could not determine the recurrence of disease after intervention. Based on the above shortcomings, it is necessary to expand the research program further, deeply understand the physical properties of PDA–PAE NPs, and observe their biological toxicity and long-term effects on CAC. At the same time, further cell experiments and clinical observation are needed to improve the scientific results of the research.
Footnotes
Abbreviations
CAC: Colitis-associated cancer; NPs: Nanoparticles; PAE: Paeonol; PDA: Polydopamine; STAT-3: Signal transducer and activator of transcription 3.
Acknowledgments
The authors gratefully acknowledge Jiayuguan Traditional Chinese Medicine for providing the necessary equipment for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the ethics committee of Jiayuguan Traditional Chinese Medicine.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
