Abstract
Background
Lung cancer is a serious disease affecting millions of individuals globally, defined by the unregulated proliferation of abnormal cells in the lungs. It is the primary cause of cancer-associated fatalities worldwide, representing a significant portion of cancer incidence and mortality rates.
Objectives
This work was dedicated to assessing the anti-cancer activity of α-mangostin against benzo[a]pyrene (B[a]P)-induced lung cancer in mice model.
Materials and Methods
Lung tumorigenesis was initiated in Swiss mice with oral treatment of B[a]P for 4 consecutive weeks. Mice were treated with α-mangostin following B[a]P induction in mice for 16 weeks. The body and lung weights and tumor incidences were assessed in the experimental mice after the conclusion of the treatments. The concentrations of oxidant and antioxidant markers, tumor biomarkers, mitochondrial enzymes, and apoptotic protein markers in the experimental mice were assessed using the commercial diagnostic kits. The immunohistochemical analysis was performed on the lung tissues to assess the expression of the proliferating cell nuclear antigen (PCNA) protein.
Results
Both pre- and post-treatments of α-mangostin (25 mg/kg) significantly enhanced body weight while reducing lung weight and tumor incidences in the B[a]P-induced mice. The α-mangostin treatment effectively increased the antioxidants and vitamins and diminished the tumor biomarker concentrations in the mice with lung cancer. Furthermore, the α-mangostin treatment considerably increased the mitochondrial enzyme concentrations and regulated the apoptotic proteins in the lung tissues of B[a]P-induced mice. In addition, the PCNA protein expression was reduced by the α-mangostin treatment in the lung tissues.
Conclusion
In conclusion, our findings demonstrate that α-mangostin exhibits chemopreventive properties in lung cancer initiated by B[a]P in mice, suggesting its potential to enhance chemotherapy approaches for lung cancer.
Introduction
Lung cancer is a serious condition that remains a major public health issue globally. It is one of the most widely diagnosed cancers, with approximately 2.20 million new incidences reported each year and a 5-year survival rate of just 15%–20% globally. Recently, the prevalence of lung cancer has continued to rise, particularly in Asia, where it accounts for a staggering 60% of new cases and deaths globally. This alarming trend underscores the urgent need to understand the causes, recent trends, and the immense burdens associated with this disease (Siegel et al., 2022). The causes of lung cancer are multifaceted, with both environmental and genetic factors playing a crucial role. Tobacco smoking is broadly known as the major cause, attributing to most lung cancer incidences. Beyond smoking, exposure to various chemicals and environmental pollutants, such as air pollution and radon, has also been associated with lung cancer progression (Kludt et al., 2024). The link between tobacco smoking and lung cancer has been well-known for decades, with tobacco smoke containing a cocktail of carcinogens that can directly damage deoxyribonucleic acid (DNA) and promote the development of malignant cells (Romaszko-Wojtowicz et al., 2022).
The intricate interaction of environmental factors, genetic predisposition, and molecular mechanisms related to the onset and advancement of lung cancer has been well documented. Environmental factors, such as occupational exposure, air pollution, and dietary factors, have also been identified as contributors to lung cancer risk (Liu et al., 2023). Despite advances in the medical field, the burden of lung cancer remains substantial. The economic impact of lung cancer is also staggering, with the disease responsible for significant direct medical costs, indirect costs related to low productivity, and the emotional toll on patients and their families. The complexity of lung cancer development, driven by a multitude of environmental, genetic, and molecular factors, has made it a challenging disease to understand and effectively treat (Barr et al., 2024). It has been suggested that tobacco smoke components, like benzo[a]pyrene (B[a]P), may contribute to the pathogenesis of lung cancer (Petit et al., 2019). Mouse models have become increasingly crucial in cancer research, offering useful insights into pathophysiology and potential treatments of several cancers. One such model that has garnered significant attention is the B[a]P-induced lung cancer in mice (Alzohairy et al., 2021).
The heterogeneity of lung cancer, combined with the complexity of cancer cells and their propensity for drug resistance, has made effective treatment a persistent issue. Current therapies, including chemotherapy, radiotherapy, and targeted therapies, have demonstrated limited success, often accompanied by significant side effects (Pons-Tostivint & Bennouna, 2022). Recently, there has been more attention on the potential of phytochemicals as a complementary approach to lung cancer treatment. These naturally occurring compounds have demonstrated considerable effects in inhibiting tumor cell growth, inducing apoptosis, and overcoming drug resistance (Dehelean et al., 2021). α-Mangostin, a xanthone derivative compound, is predominantly present in the mangosteen fruit’s pericarp. It has been already reported that α-mangostin has extensive pharmacological properties, including anti-microbial (Narasimhan et al., 2017), immunomodulatory (Kasemwattanaroj et al., 2019), anti-tumor (Li et al., 2014), antioxidant (Suhandi et al., 2023), anti-inflammatory (Dang et al., 2023), anti-aging (Li et al., 2019), and anti-arthritis (Herrera-Aco et al., 2019) activities. Furthermore, there are not many reports about the anti-cancer properties of α-mangostin against lung cancer. Thus, this work was dedicated to assessing the anti-cancer properties of α-mangostin against B[a]P-induced lung cancer in mice.
Materials and Methods
Chemicals
The primary chemicals used in the present work, including α-mangostin and B[a]P, were obtained from Sigma–Aldrich, USA. All the biochemical markers were determined using commercial diagnostic kits obtained from MyBioSource and Abcam, USA, respectively.
Animals
Healthy BALB/c mice, weighing around 20–30 g, were acquired and maintained in clean polypropylene enclosures under conventional laboratory settings of 25°C ± 2°C temperature, a 12-h dark/light cycle, 55% ± 10% humidity, and a regular feed consisting of 18% protein, 7% fat, and 53% carbohydrates (AIN-93G, Harlan Laboratories, USA), with ad libitum access to water.
Experimental Design and Treatment Groups
The acclimated mice were allocated into four groups, namely, Group I: Control mice administered corn oil; Group II: Mice received an oral administration of 50 mg/kg of B[a]P twice daily for 4 consecutive weeks to initiate lung tumorigenesis; Group III: Mice received a pre-treatment of 25 mg/kg of α-mangostin 1 day prior to the B[a]P exposure for 16 weeks; Group IV: Mice received post-treatment with 25 mg/kg of α-mangostin starting from the 8th week of B[a]P treatment for 16 weeks. Subsequent to the cessation of therapy, the mice from all groups fasted overnight and were sacrificed under anesthesia via cervical dislocation, and blood and tissue specimens were collected for further experimentation.
Analysis of Lung and Body Weights, Tumor Incidence, and Total Number
During the study, body weight was meticulously measured, and the data were compiled. After animal sacrifice, the lung tissues were extracted, rinsed with saline, and weighed to assess alterations in lung weights. The occurrences and total tumor numbers were assessed and documented.
Analysis of Oxidative Stress Markers
The lung tissues were excised and homogenized in 10% buffered saline and subsequently centrifuged at 5,000 rpm for 15 min. The resulting supernatant was employed to assess oxidative stress markers, including catalase (CAT), superoxide dismutase (SOD), glutathione (GSH), glutathione peroxidase (GPx), glutathione reductase (GR), glutathione-S-transferase (GST), malondialdehyde (MDA), vitamin E, and vitamin C, utilizing assay kits as per the manufacturer’s guidelines (Abcam, USA).
Analysis of Cancer Marker Enzymes
The concentrations of gamma-glutamyl transferase (γ-GT), 5′-nucleotidase (5′-NT), aryl hydrocarbon hydroxylase (AHH), and lactate dehydrogenase (LDH) enzymes in the serum of mice were assessed using the commercial diagnostic kits. The assays were done in triplicates using the manufacturer’s guidelines (MyBioSource, USA). The concentrations of Carcinoembryonic antigen (CEA) and neuron-specific enolase (NSE) in the serum of experimental mice were assessed using commercial kits. The tests were performed in triplicates using the manufacturer’s protocols (Elabscience, USA).
Analysis of Mitochondrial Enzymes
The concentrations of isocitrate dehydrogenase (ICDH), alpha-ketoglutarate dehydrogenase (α-KDH), succinate dehydrogenase (SDH), and malate dehydrogenase (MDH) enzymes in the lung tissue homogenates were assessed using the commercially available test kits. The experiments were conducted in triplicates using the manufacturer’s specifications (Elabscience, USA).
Analysis of Apoptotic Proteins
The concentrations of apoptotic proteins Bcl-2, Bax, cytochrome c, caspase-3, and caspase-9 in lung tissue homogenates were evaluated utilizing commercially available assay kits. The studies were performed in triplicate as per the manufacturer’s protocols (Abcam, USA).
Immunohistochemical Analysis
The collected lung tissue was preserved in 4% paraformaldehyde and subsequently paraffinized. The paraffinized tissue specimens were sectioned into 5-µm thickness and exposed to thermal pre-treatment at 60°C for 1 h. After quenching 3% H2O2, slices were incubated with 10% goat serum (Merck Millipore) for 1 h at 37°C. The tissue slides were then incubated with a primary antibody for the proliferating cell nuclear antigen (PCNA; Abcam, USA) at 4°C overnight and subsequently probed with an HRP-conjugated secondary antibody (Abcam, USA) at 37°C for 30 min. The slides were then exposed to 3,3-diaminobenzidine and stained using hematoxylin at 37°C for 5 min. The slides were then imaged using an optical microscope.
Statistical Analysis
Statistical tests were conducted utilizing GraphPad software (version 10.3.0). Data are revealed as the Mean ± Standard Deviation (SD) from three separate assays. The disparities between the two groups were examined using unpaired analysis. The differences across more than two groups were assessed using one-way analysis of variance (ANOVA) with Tukey’s correction. A p < .05 was deemed to signify significance. Data normality was tested using the Shapiro–Wilk test, and the results confirmed that our data were normally distributed, justifying the use of ANOVA for statistical analysis.
Results
Effect of α-Mangostin on Body Weight, Lung Weight, and Tumor Incidence in the Experimental Mice
The present work observed reduced body weight, elevated lung weight, and tumor incidence in the B[a]P-induced mice relative to the other groups. In contrast, the treatment of α-mangostin at 25 mg/kg concentrations significantly elevated the body weight and diminished both lung weight and tumor incidences, as depicted in Table 1. The results suggest that treating α-mangostin prior to B[a]P exposure may be advantageous for mitigating lung tumorigenesis, as seen in the mouse model.
Effect of α-Mangostin on the Body Weight, Lung Weight, and Tumor Incidence in the Experimental Mice.
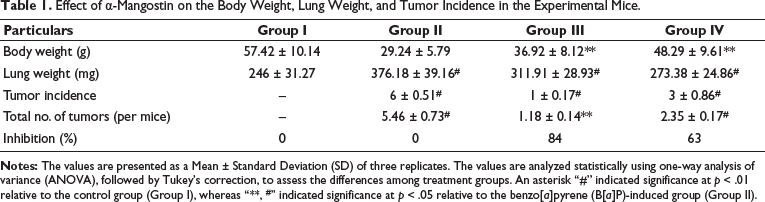
Effect of α-Mangostin on Oxidative Stress Markers in Experimental Mice
The existence of an oxidative stress condition in the experimental mice was validated by assessing the levels of oxidative stress biomarkers. Figure 1 demonstrates that B[a]P-treated mice with lung cancer exhibited a significant increase in MDA and a subsequent reduction in primary antioxidants, like SOD, CAT, GSH, GPx, GR, GST, vitamin E, and vitamin C concentrations. Interestingly, the mice with α-mangostin (25 mg/kg) treatment effectively reduced MDA levels and subsequently enhanced the antioxidant concentrations in the B[a]P-induced mice, as depicted in Figure 1.
Effect of α-Mangostin on Oxidative Stress Markers in Experimental Mice. The Values are Presented as a Mean ± Standard Deviation (SD) of Three Replicates. The Values are Analyzed Statistically Using One-way Analysis of Variance (ANOVA), Followed by Tukey’s Correction, to Assess the Differences Among Treatment Groups. An Asterisk “#” Indicated Significance at p < .01 Relative to the Control Group (Group I), Whereas “**” Indicated Significance at p < .05 Relative to the Benzo[a]pyrene (B[a]P)-induced Group (Group II).
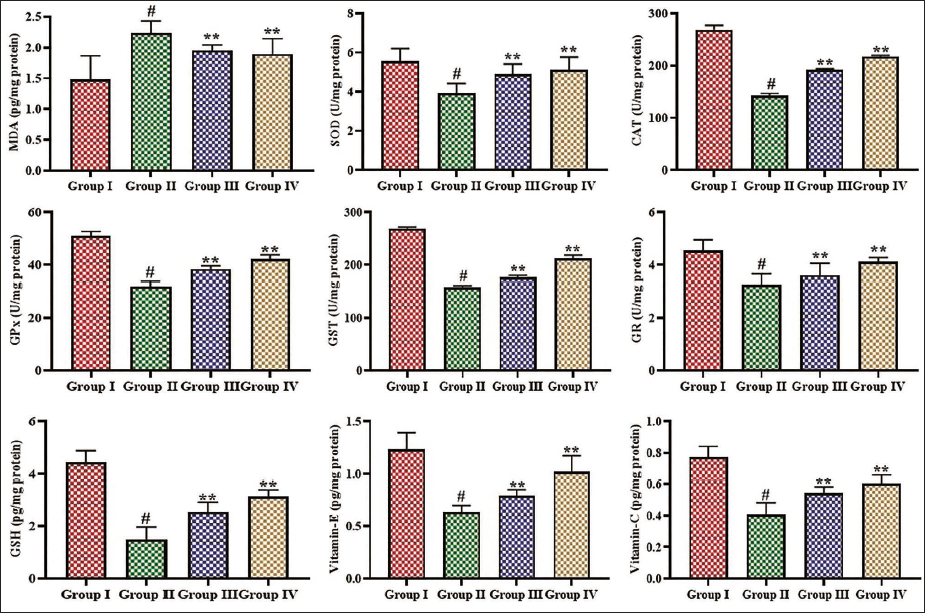
Effect of α-Mangostin on the Tumor Biomarkers in the Experimental Mice
Figure 2 depicts the results of α-mangostin on the concentrations of tumor biomarkers in the experimental mice. The B[a]P-treated mice with lung cancer revealed increased serum concentrations of GGT, 5′-NT, AHH, LDH, CEA, and NSE in comparison to the control group. Remarkably, these alterations were successfully controlled by the α-mangostin treatment (Figure 2). The pre- and post-treatments of 25 mg/kg of α-mangostin significantly decreased these tumor biomarker concentrations in the B[a]P-induced mice.
Effect of α-Mangostin on the Tumor Biomarkers in the Experimental Mice. The Values are Presented as a Mean ± Standard Deviation (SD) of Three Replicates. The Values are Analyzed Statistically Using One-way Analysis of Variance (ANOVA), Followed by Tukey’s Correction, to Assess the Differences Among Treatment Groups. An Asterisk “#” Indicated Significance at p < .01 Relative to the Control Group (Group I), Whereas “**” Indicated Significance at p < .05 Relative to the Benzo[a]pyrene (B[a]P)-induced Group (Group II).
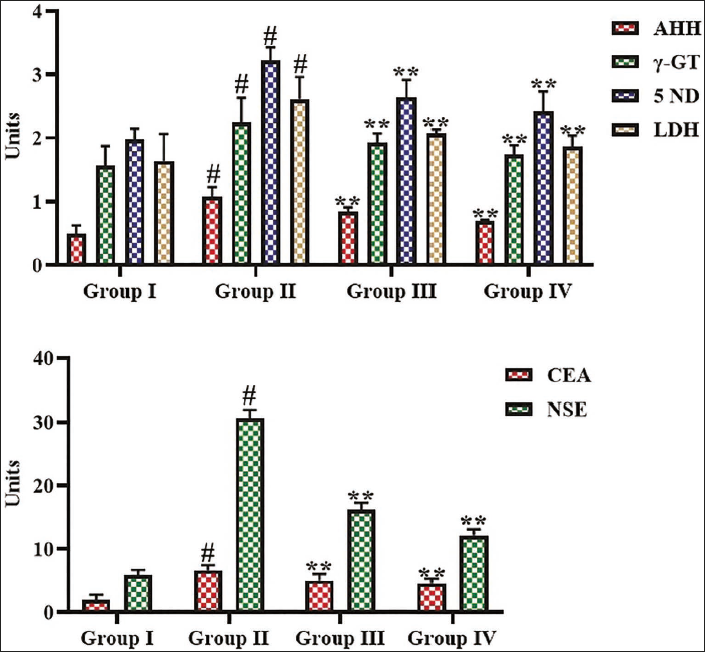
Effect of α-Mangostin on Mitochondrial Enzymes in Lung Tissues of Experimental Mice
The concentrations of mitochondrial marker enzymes, namely, α-KDH, SDH, ICDH, and MDH, in the lung tissues were assessed, and the data are depicted in Figure 3. The decreased concentrations of α-KDH, SDH, ICDH, and MDH were seen in the lung tissues of B[a]P-induced mice. However, the treatment with 25 mg/kg of α-mangostin significantly enhanced the concentrations of α-KDH, SDH, ICDH, and MDH in lung tissues of B[a]P-induced lung cancer (Figure 3).
Effect of α-Mangostin on the Mitochondrial Enzymes in Lung Tissues of Experimental Mice. The Values are Presented as a Mean ± Standard Deviation (SD) of Three Replicates. The Values are Analyzed Statistically Using One-way Analysis of Variance (ANOVA), Followed by Tukey’s Correction, to Assess the Differences Among Treatment Groups. An Asterisk “#” Indicated Significance at p < .01 Relative to the Control Group (Group I), Whereas “**” Indicated Significance at p < .05 Relative to the Benzo[a]pyrene (B[a]P)-induced Group (Group II).
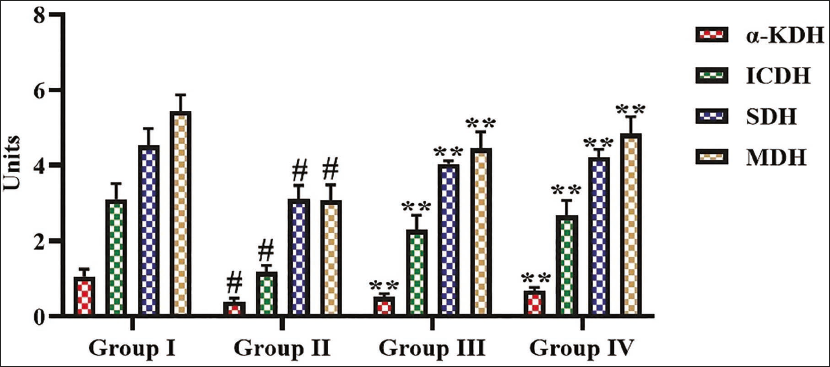
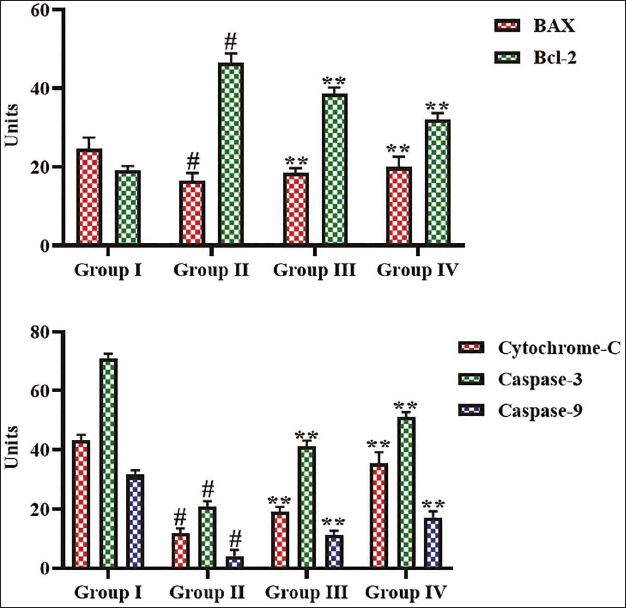
Effect of α-Mangostin on Apoptotic Proteins in Experimental Mice
Figure 4 demonstrates a substantial reduction in the concentrations of Bax, cyt-c, caspase-3, and caspase-9, accompanied by an increase in Bcl-2 levels in the lung tissues of B[a]P-induced mice, contrasting with the values recorded in the control. Notably, the 25 mg/kg concentration of α-mangostin effectively increased the Bax, cyt-c, caspase-3, and caspase-9 concentrations and reduced the anti-apoptotic Bcl-2 level in the lung tissues of B[a]P-induced mice.
Effect of α-Mangostin on the Apoptotic Protein Levels in Lung Tissues of Experimental Mice. The Values are Presented as a Mean ± Standard Deviation (SD) of Three Replicates. The Values are Analyzed Statistically Using One-way Analysis of Variance (ANOVA), Followed by Tukey’s Correction, to Assess the Differences Among Treatment Groups. An Asterisk “#” Indicated Significance at p < .01 Relative to the Control Group (Group I), Whereas “**” Indicated Significance at p < .05 Relative to the Benzo[a]pyrene (B[a]P)-induced Group (Group II).
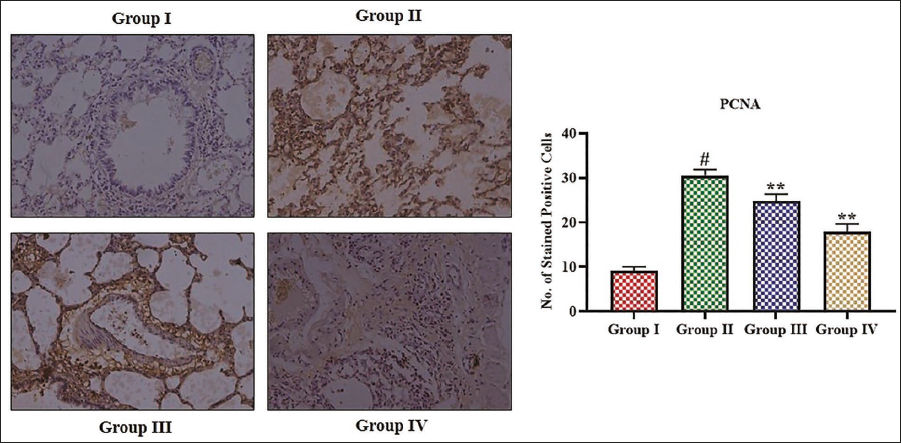
Effect of α-Mangostin on PCNA Expression in Lung Tissues of Experimental Mice
The findings of the immunohistochemical analysis revealed that the B[a]P-induced mice elevated the PCNA protein expression in their lung tissue relative to the control, whereas the treatment of α-mangostin at a 25 mg/kg concentration led to a substantial diminution in PCNA protein expression in the lung tissues of B[a]P-induced mice (Figure 5).
Effect of α-Mangostin on the Proliferating Cell Nuclear Antigen (PCNA) Protein Expression in Lung Tissues of Experimental Mice. The Values are Presented as a Mean ± Standard Deviation (SD) of Three Replicates. The Values are Analyzed Statistically Using One-way Analysis of Variance (ANOVA), Followed by Tukey’s Correction, to Assess the Differences Among Treatment Groups. An Asterisk “#” Indicated Significance at p < .01 Relative to the Control Group (Group I), Whereas “**” Indicated Significance at p < .05 Relative to the Benzo[a]pyrene (B[a]P)-induced Group (Group II).
Discussion
Lung cancer is a major public health issue, and understanding the mechanisms underlying its development is essential for improving prevention and treatment strategies. One of the broadly utilized approaches to investigating lung cancer is the animal models, such as the B[a]P-induced lung cancer in mice (Hassanin et al., 2019). The evaluation of body and lung weights and tumor incidences in these mouse models can provide useful knowledge into the progression and impact of lung cancer. These parameters are important in understanding the disease pathogenesis (Kasala et al., 2016). One key aspect of the B[a]P-initiated lung cancer is the effect on body weight. Lung cancer, like many other cancers, can lead to significant weight loss and cachexia, which can further exacerbate the disease. In the B[a]P-initiated model, researchers have observed a decrease in the body weight of the affected mice, which may be linked to the metabolic changes and energy demands associated with tumor growth (Goal et al., 2024). The lung weight of the affected mice is another imperative factor. As the tumor develops, the weight of the lungs can increase due to the presence of the growing tumor. Furthermore, lung inflammation and edema associated with the cancer can also contribute to an increase in lung weight (Wang et al., 2021). The tumor incidence in the B[a]P-induced model is a crucial measure of the severity and progression of the disease. The frequency and size of the tumors can provide insights into the carcinogenic potential of B[a]P and the efficacy of potential interventions or treatments. These factors, when analyzed in the context of the lung cancer model, can help to understand the onset of the disease (Salem et al., 2021). In a similar manner, the present findings revealed the reduced body weight and increased lung weight and tumor incidence in the mice with B[a]P-induced lung cancer. Whereas, the α-mangostin treatment significantly elevated the body weight and diminished the lung weight and tumor incidences in the B[a]P-induced mice, which evidences that α-mangostin shows potential in mitigating lung tumorigenesis and associated complications.
Oxidative stress is a significant contributor to the onset and advancement of numerous diseases, including cancer. In the realm of lung cancer, comprehending the significance of oxidative stress and its related indicators is essential for developing effective treatment methods. Oxidative stress is defined by a disproportion between the generation of ROS and the body’s capacity to neutralize them through antioxidant defense systems (Stefanou et al., 2022). In the context of cancer, oxidative stress can participate in the initiation, promotion, and advancement of the disease by inducing DNA damage, altering cellular signaling pathways, and promoting angiogenesis and metastasis. Lipid peroxidation can also result in the development of reactive aldehydes, such as MDA, which can further exacerbate cellular damage (Li et al., 2024). The primary antioxidants, like SOD, CAT, and the GSH-related enzymes, play an essential role in neutralizing the harmful outcomes of oxidative stress. SOD promotes the conversion of superoxide radicals into H2O2, which is then broken down by CAT or GPx. GSH, a ubiquitous antioxidant, is involved in the detoxification of various compounds, including ROS, and its depletion is often associated with increased oxidative stress (Gu et al., 2024). Vitamin E and vitamin C are also important antioxidants that can help to mitigate the damaging effects of oxidative stress. Vitamin E is a lipid-soluble antioxidant that safeguards cells from lipid peroxidation, whereas vitamin C is a water-soluble antioxidant that neutralizes various ROS and regenerates other antioxidants (Fagbohun et al., 2023). The importance of analyzing oxidative stress markers in the lung tissues are crucial for understanding the pathogenesis of various lung diseases (Bodduluru et al., 2015). The present results also evidenced the increased MDA level and decreased antioxidant concentrations in the lung tissues of the mice with B[a]P-exposed lung cancer. However, the treatment with α-mangostin substantially reduced the MDA and elevated the antioxidant concentrations in the lung tissues of B[a]P-initiated mice.
The analysis of specific serum biomarkers can provide useful insights into the mechanisms and early detection of lung cancer development, particularly in the context of B[a]P exposure. γ-GT is an enzyme involved in GSH metabolism and has been related to several cancers, including lung cancer. Its activity in the serum can serve as an indicator of oxidative stress and cellular damage, which are hallmarks of cancer development (Wang et al., 2024). 5′-NT plays a major role in nucleotide metabolism and has been observed to be altered during lung cancer development (Toffalorio et al., 2018). AHH is an enzyme responsible for the metabolic activation of benzopyrene into reactive intermediates that can lead to DNA damage and carcinogenesis (Shivanna et al., 2022). LDH, a glycolytic enzyme, has been broadly utilized as a non-specific indicator for several cancers, including lung cancer, due to its involvement in energy metabolism and cellular proliferation (Wang, Li, et al., 2022; Wang, Lv, et al., 2022). CEA and NSE are two well-established tumor biomarkers that have been extensively analyzed in lung cancer. CEA is a glycoprotein that participates in cell-cell adhesion and has been found to be elevated during lung cancer progression. NSE, a glycolytic enzyme, has been associated with lung cancer and can offer prognostic information for this subtype of lung cancer (Yang et al., 2023). The analysis of these serum biomarkers can offer useful information into the underlying mechanisms of disease development and progression. Early detection and monitoring of these biomarkers may help in the advancement of more effective strategies for lung cancer, ultimately improving patient outcomes. In the present study, the findings proved that the B[a]P-exposed mice demonstrated elevated serum tumor biomarker concentrations, including GGT, 5′-NT, AHH, LDH, CEA, and NSE. Considerably, the α-mangostin treatment remarkably reduced these tumor biomarker concentrations in the B[a]P-induced mice.
Mitochondrial dysfunction and respiratory impairment are hallmarks of cancer cells, and the activity and regulation of the key mitochondrial enzymes can provide useful insights into the metabolic alterations that participate in cancer development and progression (Selvendiran et al., 2005). ICDH is pivotal in the tricarboxylic acid cycle, facilitating the oxidative decarboxylation of isocitrate to α-ketoglutarate. Reduced activity of this enzyme has been linked to various neurodegenerative diseases, as well as cancer, suggesting its importance in maintaining cellular energy metabolism and redox balance (Carosi et al., 2024). Similarly, α-KDH, SDH, and MDH are all key enzymes in the mitochondrial respiratory chain and the tricarboxylic acid cycle, and their dysregulation can contribute to the metabolic reprogramming observed in cancer cells (Chang et al., 2022). It has already been shown that many cancer cells, including those in lung cancer, maintain the ability to respire through mitochondrial oxidative phosphorylation, despite the Warburg effect, which is characterized by a high rate of glycolysis and lactate production (Ashton et al., 2018). This suggests that mitochondrial function is not necessarily impaired in cancer cells, but rather that the metabolic shift toward aerobic glycolysis is an active process driven by signaling pathways that control glucose uptake and utilization (Li et al., 2021). By analyzing the activity and regulation of these mitochondrial enzymes, researchers can gain a better understanding of the metabolic rewiring that occurs in cancer cells and the potential therapeutic targets that may be exploited to disrupt these processes. Supportively, the present work evidenced the decreased concentrations of α-KDH, SDH, ICDH, and MDH in the lung tissues of mice with B[a]P-treated lung cancer. Whereas the α-mangostin elevated the α-KDH, SDH, ICDH, and MDH concentrations in mice with B[a]P-treated lung cancer.
Bcl-2 and Bax are the pivotal proteins of the Bcl-2 family that play an essential role in regulating the intrinsic apoptotic pathway, with Bcl-2 acting as an anti-apoptotic and Bax as a pro-apoptotic protein. The balance between these two proteins is a critical determinant of cellular fate, as an imbalance can lead to either cell survival or apoptosis (Alam et al., 2022; Dakkak et al., 2024). Cytochrome c, on the other hand, is a key player in the mitochondrial apoptotic pathway, as its release from the mitochondria into the cytosol can trigger caspase-9, which eventually activates the caspase-3, leading to the execution of apoptosis. Caspase-3 and caspase-9 are essential components of the apoptotic machinery, and their activation is a hallmark of apoptosis (Pessoa, 2022). The analysis of these apoptotic proteins in lung cancer can offer valuable insights into the mechanisms by which carcinogen induces cell death or promotes cell survival in the lung (Wang, Li, et al., 2022; Wang, Lv, et al., 2022). The interplay between Bcl-2 and Bax can determine the susceptibility of lung cancer cells to apoptosis, with an increased ratio of Bax to Bcl-2 being associated with a greater propensity for cell death. Additionally, the analysis of caspase-9 activation can help to understand the involvement of the intrinsic apoptotic pathway in lung cancer (Xu et al., 2023). The findings of this work demonstrated the substantial reduction in the Bax, cytochrome c, caspase-3, and caspase-9 concentrations and subsequently increased the Bcl-2 concentration in the lungs of B[a]P-exposed mice. Interestingly, the α-mangostin treatment effectively increased the pro-apoptotic protein concentrations and reduced the Bcl-2 level in the lungs of B[a]P-treated mice.
Apart from these findings, our study has some limitations that need to be acknowledged. The present study was conducted in a mouse model, and the results may not be directly translatable to humans. The study focused on the chemopreventive effects of α-mangostin on lung cancer but did not explore its potential therapeutic effects on established tumors. The study relied on a single dose of α-mangostin, and the optimal dosing regimen remains to be determined. The study did not examine the potential interactions between α-mangostin and other chemotherapeutic agents. Therefore, further in-depth molecular studies are needed to fully elucidate the mechanisms by which α-mangostin exerts its chemopreventive effects. These studies should investigate the effects of α-mangostin on key signaling pathways and molecular targets involved in lung cancer development.
Conclusion
In conclusion, our findings demonstrate that α-mangostin exhibits chemopreventive properties in lung cancer induced by B[a]P in mice. The chemopreventive potential of α-mangostin in B[a]P-exposed cancer growth is linked to its anti-tumorigenic and antioxidant activities. Moreover, α-mangostin reduced tumor incidence, tumor markers, and PCNA protein expression in the B[a]P-exposed mice. The α-mangostin treatment also elevated the concentrations of mitochondrial enzymes in the lung tissues. Consequently, it was evident that α-mangostin possesses chemopreventive properties and may enhance chemotherapy approaches for lung cancer.
Footnotes
Abbreviations
5′-NT: 5′-Nucleotidase; AHH: Aryl hydrocarbon hydroxylase; AKT: Protein kinase B; ANOVA: Analysis of variance; B[a]P: Benzo[a]pyrene; Bax: Bcl-2-associated X protein; Bcl-2: B-cell lymphoma 2; CAT: Catalase; ccRCC: Clear cell renal cell carcinoma; CEA: Carcinoembryonic antigen; cyt-c: Cytochrome c; DCFH-DA: 2′,7′-Dichlorofluorescin diacetate; DMEM: Dulbecco’s modified Eagle medium; DMRT: Duncan’s multiple range test; DMSO: Dimethyl sulfoxide; DOX: Doxorubicin; FBS: Fetal bovine serum; FTIR: Fourier-transform infrared spectroscopy; GPx: Glutathione peroxidase; GR: Glutathione reductase; GSH: Glutathione; GST: Glutathione-S-transferase; HIF: Hypoxia-inducible factor; ICDH: Isocitrate dehydrogenase; LDH: Lactate dehydrogenase; MDA: Malondialdehyde; MDH: Malate dehydrogenase; mTOR: Mammalian target of rapamycin; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; NSE: Neuron-specific enolase; PCNA: Proliferating cell nuclear antigen; PI: Propidium iodide; PI3K: Phosphatidylinositol 3-kinase; RCC: Renal cell carcinoma; ROS: Reactive oxygen species; SDH: Succinate dehydrogenase; SOD: Superoxide dismutase; TBARS: Thiobarbituric acid reactive substances; TNF-α: Tumor necrosis factor-alpha; VHL: Von Hippel-Lindau; α-KDH: Alpha-ketoglutarate dehydrogenase; γ-GT: Gamma-glutamyl transferase.
Authors’ Note
Yuan Feng and Jinxiu Guo have contributed equally to this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This work has been approved by the Institutional Animal Ethical Committee by the Department of Medical Oncology, Ankang Hospital of Traditional Chinese Medicine, Ankang, Shaanxi 725000, China.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
