Abstract
Background
Pneumonia is a serious respiratory illness that impacts the lungs, leading to inflammation and fluid accumulation in the alveoli, the tiny air sacs essential for gas exchange. Mycoplasma pneumonia is a severe respiratory infection caused by the atypical bacterium Mycoplasma pneumoniae (MP).
Objective
The primary objective of this work is to explore the therapeutic effects of ferulic acid against MP-infected pneumonia in an experimental mice model.
Materials and Methods
In this work, BALB/c mice were subjected to 100 µL of MP challenge via nasal drips to trigger pneumonia. The mice were then administered 50 mg/kg of ferulic acid for 3 days. After experimentation, the lung weight, myeloperoxidase (MPO), and nitric oxide (NO) concentrations in the experimental mice were assessed. The inflammatory cytokines and oxidative stress biomarkers were analyzed using appropriate diagnostic kits. The total cell count in the bronchoalveolar lavage (BAL) fluid and deoxyribonucleic acid (DNA) concentration in the lungs were assessed in the experimental mice. The histopathology examination of the lungs was conducted to evaluate the histological abnormalities.
Results
The ferulic acid administration significantly decreased lung weight, MPO, and NO concentrations in MP-infected mice. Ferulic acid treatment resulted in a diminution of malondialdehyde (MDA) and an enhancement of glutathione (GSH) and superoxide dismutase (SOD) concentrations. The ferulic acid treatment significantly diminished the inflammatory marker concentrations in the MP-challenged mice. Furthermore, ferulic acid reduced the DNA content and total cell counts in the MP-infected mice. In addition, the outcomes of the histological examination indicated a significant reduction in alveolar damage in the mice with pneumonia.
Conclusion
The findings of this work confirm the salutary properties of ferulic acid against MP-infected pneumonia in mice. Consequently, ferulic acid may serve as an effective therapeutic drug candidate for pneumonia treatment.
Keywords
Introduction
Pneumonia remains a major public health issue, specifically in low- and middle-income nations, where the burden of the disease is disproportionately high. Inadequate access to healthcare, limited availability of diagnostic tools, and the high prevalence of risk factors such as malnutrition, indoor air pollution, and comorbidities contribute to the high mortality rates associated with pneumonia in these regions (Bolluyt et al., 2024). Strategies to address the global burden of pneumonia must involve a multifaceted approach, including the development and distribution of effective vaccines, improving access to healthcare and diagnostic services, and addressing the underlying social and environmental determinants of health (Wang et al., 2023). Mycoplasma pneumonia is a severe respiratory infection caused by a group of bacteria known as Mycoplasma. Mycoplasma pneumoniae (MP) is a pivotal cause of community-acquired pneumonia (CAP), responsible for up to 33% of hospitalized cases. Unlike typical bacterial pneumonia, Mycoplasma pneumonia often presents with milder symptoms, making it challenging to diagnose and manage (Georgakopoulou et al., 2024). M. pneumoniae is a unique microorganism that lacks a cell wall, which distinguishes it from other bacteria. This poses a challenge in the diagnosing, treating, and monitoring of individuals with Mycoplasma pneumonia. MP is one of the primary causes of CAP, along with other potential pathogens (Hu et al., 2022). Mycoplasma pneumonia is generally a self-limiting respiratory condition, while it has infrequently been linked to extrapulmonary consequences, such as neurological, cardiac, and immunological symptoms. Patients with Mycoplasma pneumonia often present with symptoms such as fever, cough, and difficulty breathing. The diagnosis and therapy of pneumonia, if diagnosed, are determined by the patient’s risk factors, physical examination results, and laboratory test outcomes (Song et al., 2023). Pneumonia is categorized into three subtypes based on the characteristics in which the patient shows signs of infection: CAP, hospital-acquired pneumonia, and ventilator-associated pneumonia. Mycoplasma pneumonia is typically considered a CAP, as it is acquired by a patient in a non-hospitalized condition (Zhan et al., 2022).
The pathogenesis of Mycoplasma pneumonia encompasses a multifaceted interplay between the host’s immunity and the evasive techniques employed by the Mycoplasma pathogen. The pathogenesis of pneumonia begins with the colonization of the mucosal surfaces of the respiratory tract. M. pneumoniae can persist at the site of infection by employing various evasive techniques, like changes in surface antigens, inappropriate activation of alveolar macrophages, and neutrophil infiltrations at the inflammatory sites (Li et al., 2023). The discharge of inflammatory markers, like cytokines and chemokines, by the host’s immune cells in response to the Mycoplasma infection leads to the characteristic symptoms of Mycoplasma pneumonia, including cough, fever, and respiratory distress. The challenge in the treatment of pneumonia is not in making the diagnosis of pneumonia but rather in identifying the causative agent so that the appropriate anti-biotic treatment can be employed in a timely manner (Zhang et al., 2023). Current treatment options often involve the use of anti-biotics, but the occurrence of anti-biotic-resistant pathogens has resulted in the exploration of alternative methods, including plant-based approaches.
One of the key challenges in the treatment of Mycoplasma pneumonia is the unique characteristics of the Mycoplasma species, which lack a cell wall and have a hydrophobic cell structure, contributing to their innate anti-biotic resistance (Lee et al., 2024). In addition, Mycoplasma species have been reported to employ various strategies to escape the host’s immunity like varying surface antigens, inhibiting the respiratory burst of neutrophils, and enhancing the Th2-biased immune response (Lorentzen et al., 2023). The current therapies for pneumonia are challenging due to multidrug-resistant pathogens, which highlight the need for innovative approaches. Plant-based therapies may offer a promising alternative or adjuvant to traditional anti-microbial treatments (Açar et al., 2023; Alcorta et al., 2024; Cerqua et al., 2022; Ferrarini et al., 2022; Sardar et al., 2024). There are several plant-derived compounds of different classes that are specifically investigated for their effects on respiratory conditions and inflammation (Goni et al., 2021; Khan et al., 2020; Küpeli Akkol et al., 2021; Vieira et al., 2020). Ferulic acid is a 4-hydroxy-3-methoxycinnamic acid that extensively occurs in the cell walls of monocotyledonous plants, barley, rice, oranges, apples, wheat, peanuts, and coffee (Klepacka & Fornal, 2006). Several previous reports have already highlighted the pharmacological properties of ferulic acid, including antioxidant (Zduńska et al., 2018), anti-aging (Neopane et al., 2023), cardioprotective (Neto-Neves et al., 2021), nephroprotective (Nouri et al., 2022), anti-diabetic (Li et al., 2022), and anti-cancer (Fahrioğlu et al., 2016) properties. In addition to these biological activities, the salutary properties of ferulic acid against Mycoplasma pneumonia have not been assessed yet. Therefore, this study was focused on exploring the therapeutic activities of ferulic acid against MP-infected pneumonia in the experimental mice model.
Materials and Methods
Reagents
The azithromycin, ferulic acid, and others were procured from Sigma–Aldrich, USA. The diagnostic kits for estimating the biochemical markers were acquired commercially from Abcam, USA; Elabscience, USA; and Thermo Fisher Scientific, USA.
Anti-bacterial Assay
The anti-bacterial properties of ferulic were assessed using the well-diffusion technique. The test pathogens, including Streptococcus pneumoniae, Staphylococcus aureus, Escherichia coli, and Klebsiella pneumoniae, were spread over the agar medium surface. Following the inoculation of each strain into the culture medium, 6 mm wells were created on the surface, and these wells were subsequently filled with varying dosages (40, 50, and 60 µM/mL) of ferulic acid. Amoxicillin (30 µg/well), a widely used anti-biotic, was employed as standard anti-biotic. Following the incubation period, the results were evaluated, and the inhibition zones were measured.
Experimental Animals
The current study employed BALB/c mice with a body weight of 23 ± 5 g. The mice were maintained in sterile polypropylene enclosures under controlled conditions at 23°C ± 2°C temperature, with humidity levels between 50% and 60%, and a 12-h light/dark series. The animals were granted free access to food and water throughout the experimental periods. Before initiating the experiments, a 7-day interval was designated for the mice to acclimate to the laboratory environment.
Treatment Groups
Acclimated mice were allocated into four experimental groups, each consisting of six mice. Group I consisted of controlled animals. Group II consisted of mice that were administered 100 µL of MP via nasal drips for 2 days to trigger pneumonia. Group III mice received an MP challenge and were then administered 50 mg/kg of ferulic acid for 3 days. Group IV consisted of MP that received azithromycin (100 mg/kg) for 3 days. Following treatments, the experimental mice were euthanized, and subsequently, samples were collected for further experimentation. The excised lungs were meticulously measured for their wet and dry weights.
Analysis of Inflammatory Markers
The excised lung tissues of the experimental mice were homogenized with an iced saline solution and subsequently centrifuged at 10,000 rpm for 10 min. The supernatant was employed to evaluate nitric oxide (NO) and myeloperoxidase (MPO) activities utilizing the respective kits, adhering meticulously to the instructions provided by the manufacturer (Abcam, USA). All tests were conducted with three replicates.
Analysis of Oxidative Stress Markers
The concentrations of malondialdehyde (MDA), glutathione (GSH), and superoxide dismutase (SOD) in the lung tissues of both control and treated animals were evaluated using appropriate kits. All tests were performed in triplicate as per the guidelines provided by the manufacturer’s kit (Elabscience, USA).
Preparation of Bronchoalveolar Lavage (BAL) Fluid and Cell Count
The saline (30 mL) was injected into the right middle lobe of the experimental mice to collect the BAL fluid samples. The sample was subsequently centrifuged at 6,000 rpm for 10 min. The obtained BAL fluid was subsequently changed to other tubes for additional tests. A hemocytometer was utilized along with a microscope to count the total cells in the acquired BAL fluid sample.
Analysis of Deoxyribonucleic Acid (DNA) Amount of MP
The excised lungs from the mice were homogenized and treated with DNA-extracting reagents for 10 min. The fluid was subsequently centrifuged for 5 min at 13,000 rpm to isolate the DNA. The total amount of DNA in the sample was evaluated using the polymerase chain reaction (PCR) technique (Yamashita et al., 1995).
Analysis of Inflammatory Cytokine Levels
The acquired BAL fluid samples from the mice were employed to evaluate inflammatory cytokines, including transforming growth factor (TGF), tumor necrosis factor-alpha (TNF-α), interleukin (IL)-1, IL-6, and IL-8, utilizing the kits as per the manufacturer’s recommended guidelines (Thermo Fisher, USA).
Analysis of Extracellular Signal-regulated Kinase (ERK)1/2, c-Jun N-terminal Kinase (JNK)1/2, and Nuclear Factor Kappa B (NF-κB) Level
The diagnostic kits acquired from Abcam, USA, were employed to evaluate the ERK1/2, JNK1/2, and NF-κB concentrations in the lungs of experimental mice, following the manufacturer’s recommended protocols.
Histopathological Analysis
A histopathological study was performed on lung tissue samples to identify histological alterations. The lungs were treated with formalin (10%) and subsequently preserved in paraffin. The paraffin-embedded tissue was subsequently sectioned into 5 µm slices. The tissues were stained with eosin and hematoxylin and thereafter examined microscopically to assess histopathological abnormalities.
Statistical Analysis
The results are statistically scrutinized using GraphPad Prism, and the values are presented as Mean ± Standard Deviation (SD) of three measurements (n = 6). The data are examined with a one-way analysis of variance (ANOVA) and Tukey’s post hoc assay to study the significance level. A p < .05 is employed to ascertain statistical significance.
Results
Anti-bacterial Activity of Ferulic Acid
The anti-bacterial efficacy of ferulic acid against S. pneumoniae, S. aureus, E. coli, and K. pneumoniae strains was assessed using the good diffusion method, with results presented in Table 1. The results indicated that ferulic acid possesses significant anti-bacterial capabilities, as it likely suppressed the growth of the examined pathogens. The elevated inhibitory zones surrounding the increased dosages of ferulic acid (40, 50, and 60 µM/mL)-loaded wells are evident on the growth plates. The augmented zones of inhibition were noted against K. pneumoniae and E. coli when treated with ferulic acid.
Anti-bacterial Activity of Ferulic Acid.
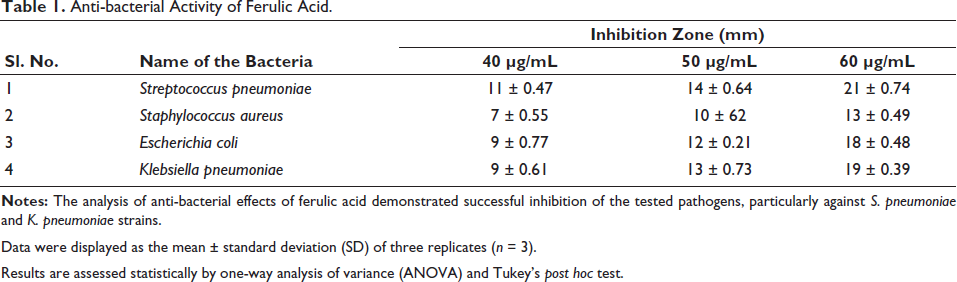
Data were displayed as the mean ± standard deviation (SD) of three replicates (n = 3).
Results are assessed statistically by one-way analysis of variance (ANOVA) and Tukey’s post hoc test.
Effect of Ferulic Acid on the Lung Weight and Inflammatory Markers in Experimental Mice
Figure 1 summarizes the findings of ferulic acid treatment on the lung weight, NO, and MPO concentrations of experimental animals. The MP-challenged mice exhibited a substantial elevation in lung weight, NO, and MPO concentrations in comparison to the control group. Nonetheless, ferulic acid (50 mg/kg) treatment remarkably diminished NO, MPO, and lung weight in the MP-infected mice.
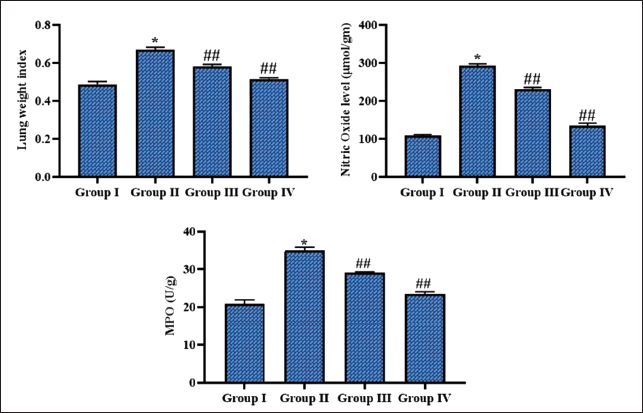
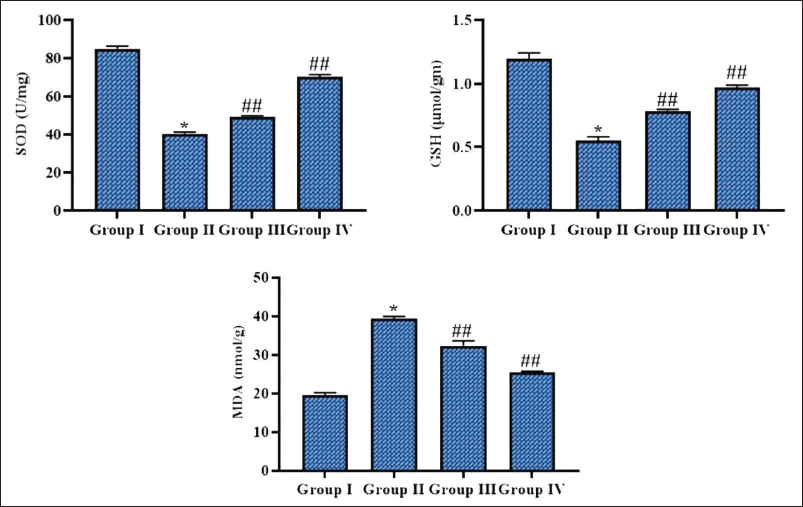
Effect of Ferulic Acid on the Oxidative Stress Marker Levels in the Experimental Mice
Figure 2 illustrates a noteworthy elevation in MDA levels and a subsequent decrease in GSH and SOD concentrations in the MP-infected mice relative to the control group. Fascinatingly, ferulic acid (50 mg/kg) treatment substantially decreased MDA levels and notably increased GSH and SOD concentrations in MP-induced mice. Likewise, a diminution in MDA levels and an elevation of antioxidant levels were seen in the azithromycin-treated mice, corroborating the antioxidant capabilities of ferulic acid.
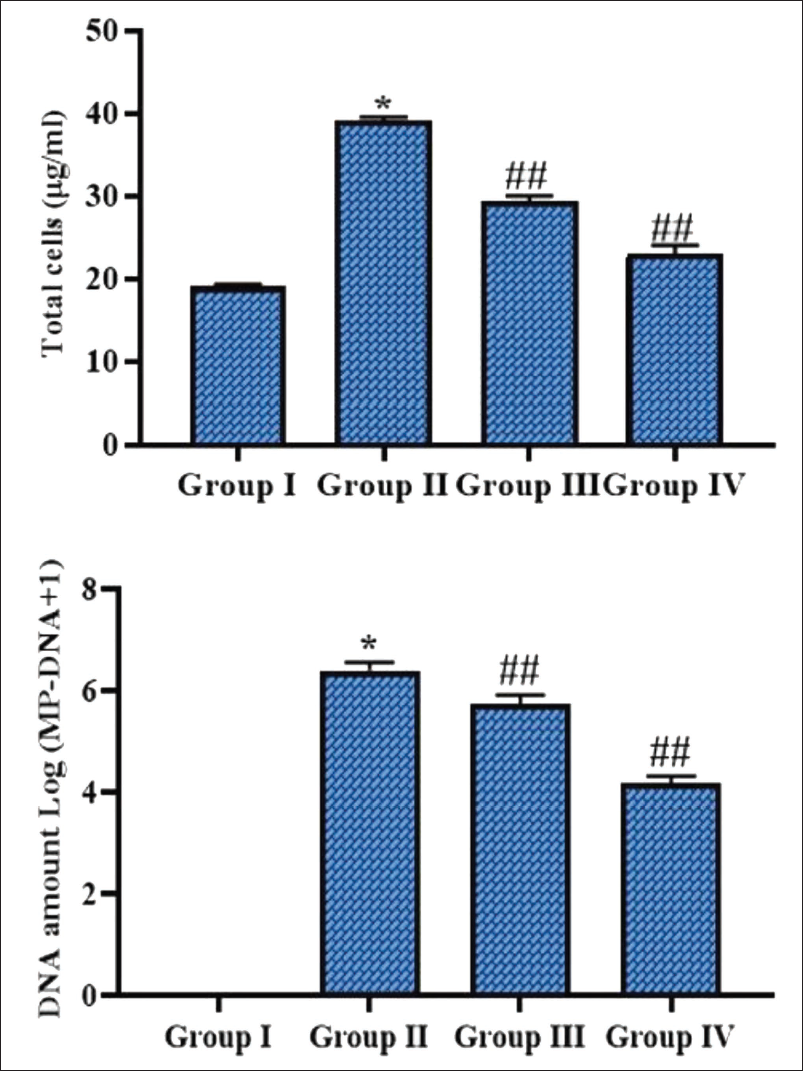
Effect of Ferulic Acid on the Cell Counts and DNA Level in Experimental Mice
The total cell count level and DNA quantities were assessed in the experimental mice, with the results depicted in Figure 3. An elevation in cell counts and DNA quantities was noted in MP-infected mice relative to the control group. Intriguingly, ferulic acid (50 mg/kg) treatment exhibited a substantial diminution in DNA quantity and cell counts. The efficacy of ferulic acid was further corroborated by the results of azithromycin, which similarly reduced these markers in MP-induced mice.
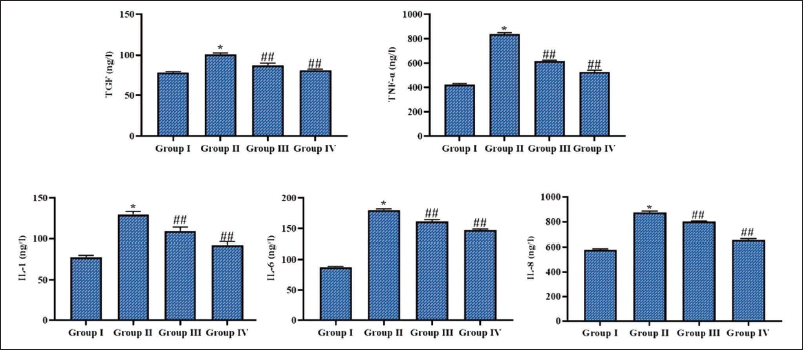
Effect of Ferulic Acid on the Inflammatory Cytokines in Experimental Mice
The concentrations of inflammatory cytokines in the BAL fluid samples of experimental mice were assessed, and the findings are revealed in Figure 4. A significant increase in TGF, TNF-α, IL-1, IL-6, and IL-8 was noted in the BAL fluid sample of pneumonia-induced mice relative to the control. In contrast, the 50 mg/kg ferulic acid treatment significantly reduced all those cytokine concentrations in the MP mice. Similar findings were also seen in the azithromycin treatment, thereby confirming the anti-inflammatory activities of ferulic acid.
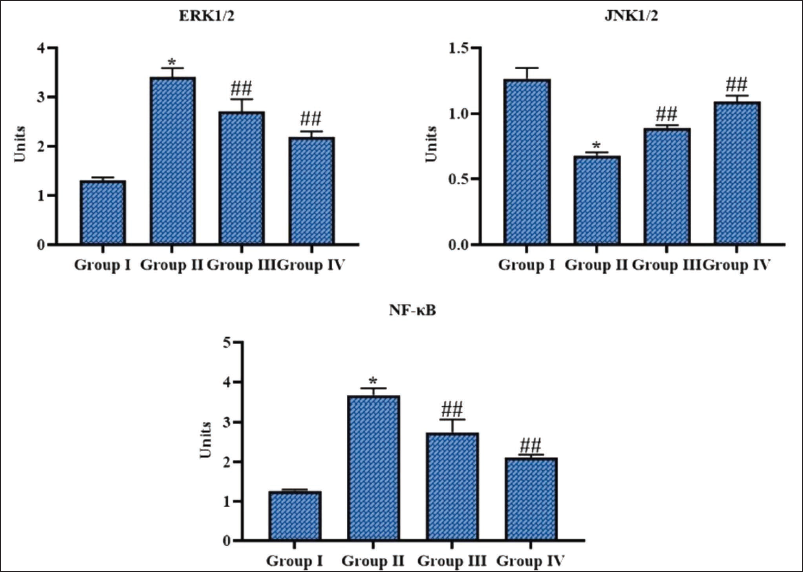
Effect of Ferulic Acid on ERK1/2/JNK1/2/NF-κB in the Lung Tissues
The concentrations of ERK1/2/JNK1/2/NF-κB in the lungs of experimental mice were analyzed, with the findings depicted in Figure 5. A substantial elevation in ERK1/2/JNK1/2/NF-κB concentrations was seen in the lungs of the MP-infected mice relative to the control group. The treatment of 50 mg/kg ferulic acid substantially lessened the ERK1/2/JNK1/2/NF-κB levels in the MP-induced mice, which is also corroborated by the results of azithromycin.
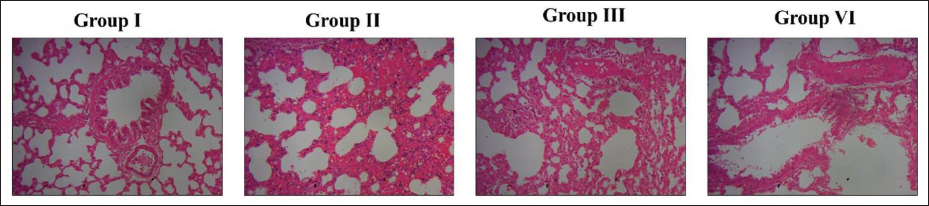
Effect of Ferulic Acid on the Lung Histopathology of Experimental Mice
The results of lung histopathology in experimental mice are presented in Figure 6. The control mice exhibited normal configurations of alveolar cells, devoid of inflammation or lung tissue injury. Contrastingly, pneumonia-induced mice exhibited significant histological alterations, including thickening of alveolar walls, constriction of bronchial tubes, and increased inflammatory cell infiltrations in their lungs. Notably, ferulic acid (50 mg/kg) significantly reduced lung histological alterations. The findings of azithromycin showed a significant reduction in lung histological alterations, hence corroborating the therapeutic efficacy of ferulic acid.
Discussion
Pneumonia is a serious respiratory illness that impacts the lungs, resulting in inflammation and fluid accumulation in the alveoli, the tiny air sacs essential for gas exchange in the lungs. This condition may be induced by several causes and pathogens and can vary in severity from moderate to life-threatening (Candel et al., 2023). Over the centuries, pneumonia has been a pivotal cause of hospitalization, morbidity, and mortality. The pathogenesis of pneumonia is a complex process that involves the interplay between the host’s immune system and the invading pathogen. The specific pathogen responsible for an individual case of pneumonia can have a significant impact on the disease severity and the course of therapy (Anderson & Feldman, 2023). Mycoplasma pneumonia is a major respiratory infection caused by the atypical bacterium M. pneumoniae. While advancements in anti-biotic therapies and intensive care have improved outcomes, the mortality rate remains high, emphasizing the need for a deeper understanding of the underlying pathogenic mechanisms (Carugati et al., 2020). One key factor in the pathophysiology of pneumonia is the participation of MPO and NO, which have been shown to play a critical part in the pathology of numerous types of pneumonia. NO, a potent vasodilator and signaling molecule, has been implicated in the regulation of inflammatory processes during respiratory infections (Pellegrini et al., 2024). MPO, an enzyme found in neutrophils, has been shown to catalyze the production of hypochlorous acid, a potent oxidant that can contribute to tissue damage and pathogen clearance. The interplay between these various factors, including NO and MPO, creates a complex and dynamic landscape that ultimately determines the progression and severity of pneumonia (Bezerra et al., 2023). The current findings demonstrated a significant increase in both MPO and NO contents in the MP-infected mice. Surprisingly, ferulic acid significantly reduced the NO and MPO amounts in the MP-infected mice.
Oxidative stress and an imbalance of antioxidant systems play a crucial role in the onset of pneumonia. MDA is a reliable marker of oxidative stress and lipid peroxidation. During pneumonia, the host’s immune response generates an abundance of ROS, leading to increased lipid peroxidation and the consequent production of MDA. The increased MDA accumulation can further exacerbate oxidative damage and contribute to the pathogenesis of the disease (Muscolo et al., 2024). SOD is a critical antioxidant enzyme that neutralizes the superoxide radical, a highly reactive oxygen species. In pneumonia, the activity of SOD may be upregulated as a compensatory mechanism to combat the increased oxidative stress. However, the effectiveness of this enzyme may be compromised if the overall antioxidant capacity of the host is overwhelmed by the magnitude of oxidative stress (Kozlov et al., 2024). GSH is another crucial antioxidant that plays a central role in maintaining cellular redox balance. During pneumonia, the depletion of GSH levels can lead to an imbalance between oxidant and antioxidant mechanisms, further exacerbating oxidative stress and implicating the onset of the disease (Hernandez-Morfa et al., 2023). It has been already demonstrated the role of oxidative stress and antioxidant imbalance in the onset of various diseases, including pneumonia (Xu et al., 2020). Similarly, increased oxidative stress and the subsequent alterations in antioxidants may contribute to the pathogenesis of pneumonia (Albano et al., 2022). In this work, significantly increased MDA levels and a subsequent lessening in GSH and SOD were found in the MP-infected mice. Captivatingly, ferulic acid remarkably diminished the MDA level and notably elevated both SOD and GSH concentrations in pneumonia-induced mice, which corroborates its antioxidant capabilities.
A key aspect in the progression of pneumonia is the role of inflammation and inflammatory cytokines. Mycoplasma species are known to evade the host’s immune defenses and persist within the respiratory tract. During infection, the host’s immune cells, like macrophages and T cells, are activated and secrete various pro-inflammatory cytokines (Long et al., 2022). TNF-α and IL-1 are two primary cytokines implicated in the initial inflammatory response to M. pneumoniae infection. These cytokines stimulate the recruitment of additional immune cells, like neutrophils, to the site of infection, leading to further inflammation and tissue damage (Mizgerd et al., 2018). IL-6 is another important cytokine that plays a critical role in the onset of pneumonia. Elevated IL-6 concentrations have been observed in pneumonia patients, and it is thought to contribute to the onset of systemic inflammation and complications, such as acute respiratory distress syndrome (Pandey et al., 2023). IL-8, a chemokine, is also upregulated in response to M. pneumoniae infection. IL-8 is responsible for the recruitment of neutrophils, further worsening inflammation. TGF-β, on the other hand, has been shown to have a more complex role in pneumonia, with both pro- and anti-inflammatory properties, depending on the stage of the infection (Keskinidou et al., 2022). The overproduction of these inflammatory cytokines, coupled with the persistent nature of M. pneumoniae infection, can lead to a sustained and exaggerated inflammatory response, resulting in tissue damage and the development of complications associated with pneumonia (Korkmaz & Traber, 2023). Understanding the role of these cytokines in the pathogenesis of pneumonia is critical to developing effective interventions and improved management strategies for this respiratory infection (Rendon et al., 2016). The current study has evidenced a significant increase in TGF, TNF-α, IL-1, IL-6, and IL-8 concentrations in pneumonia-induced mice. Captivatingly, ferulic acid substantially lessened all of these inflammatory cytokines in MP-induced mice, subsequently confirming the anti-inflammatory activities of ferulic acid.
The pathophysiology of MP infection involves complex signaling cascades that regulate the host’s inflammatory response. Understanding the complex signaling pathways involved in the pathogenesis of this disease is crucial for developing effective treatment strategies. JNK signaling, part of the MAPK family, has been extensively studied for its role in stress responses. Upon activation by various stimuli, such as TNF and growth factors, the JNK cascade culminates in the phosphorylation and activation of the transcription factor c-Jun, which controls the expression of genes implicated in inflammation and cell survival (Yan et al., 2024). Similarly, the ERK1/2 pathway is another MAPK signaling cascade that is responsive to extracellular signals and plays a critical role in modulating inflammation (Li et al., 2024). A wide range of stimuli, including bacterial products, are known to stimulate the NF-κB signaling, leading to the transcription of genes implicated in the immune response. NF-κB signaling is a critical mediator of the inflammatory response in pneumonia, driving the generation of numerous pro-inflammatory cytokines, as well as the generation of ROS and NO, all of which contribute to the exacerbation of inflammation (Roberti et al., 2022). It has been shown that the MAPK and NF-κB pathways are pivotal in the development of acute lung injury (Millar et al., 2022). In the context of pneumonia, it is likely that these signaling cascades are also implicated in orchestrating the host’s inflammatory reaction to the infection. The modulation of these signaling pathways and their downstream effectors may demonstrate a potential therapeutic method to manage Mycoplasma pneumonia (Guo et al., 2024). The present findings demonstrated that ERK/JNK/NF-κB concentrations were drastically elevated in the lungs of MP-infected mice. Fascinatingly, ferulic acid treatment significantly reduced the ERK/JNK/NF-κB concentrations in the pneumonia-induced mice.
Conclusion
The findings of this work confirm the salutary activities of ferulic acid against MP-infected pneumonia in mice. Treatment with ferulic acid led to a reduction in lung weight, diminished inflammatory cytokines and oxidative marker levels, enhanced antioxidant concentrations, and lowered ERK/JNK/NF-κB concentrations in mice with pneumonia. These data demonstrate the therapeutic efficacy of ferulic acid against pneumonia. Consequently, ferulic acid may serve as an effective therapeutic drug candidate for pneumonia treatment. Furthermore, additional studies are highly advised to be conducted in the future to thoroughly understand the therapeutic qualities of ferulic acid against pneumonia.
Footnotes
Abbreviations
AMPK: AMP-activated protein kinase; ANOVA: Analysis of variance; BAL: Bronchoalveolar lavage; BALB/c: Bagg albino/c (mouse strain); CAP: Community-acquired pneumonia; DNA: Deoxyribonucleic acid; ERK: Extracellular signal-regulated kinase; GSH: Glutathione; HO-1: Heme oxygenase-1; IL-1: Interleukin-1; IL-6: Interleukin-6; IL-8: Interleukin-8; JNK: c-Jun N-terminal kinase; MAPK: Mitogen-activated protein kinase; MDA: Malondialdehyde; MEK: Mitogen-activated protein kinase; MP: Mycoplasma pneumoniae; MPO: Myeloperoxidase; NF-κB: Nuclear factor kappa B; NO: Nitric oxide; Nrf2: Nuclear factor erythroid 2-related factor 2; PCR: Polymerase chain reaction; ROS: Reactive oxygen species; SD: Standard deviation; SOD: Superoxide dismutase; TGF: Transforming growth factor; Th2: T helper type 2; TNF-α: Tumor necrosis factor-alpha.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This work has been approved by the Institutional Animal Ethical Committee at Xi’an International Medical Center Hospital, Xi’an, Shaanxi 710100, China.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
