Abstract
Background
Sika deer antler is a traditional Chinese medicine, and its mechanism of action on immune regulation is still unclear. Therefore, this study aims to explore its related mechanism.
Objectives
To investigate the mechanism by which velvet antler peptides (VAPs) from Sika deer regulate immune function in mice through the toll-like receptors 2 (TLR2)/toll-like receptors 4 (TLR4) and tumor necrosis factor-alpha (TNF-α)/interleukin-3 (IL-3)/recombinant cytotoxic T-lymphocyte-associated-antigen-4 (CTLA-4) signaling pathways.
Materials and Methods
Male ICR mice were randomly divided into five groups: blank control (BC), positive control (PC), low dose (LD, 60 mg/kg), medium dose (MD, 120 mg/kg), and high dose (HD, 180 mg/kg). The BC group was administered saline for 1 week, while the other groups received dexamethasone (25 mg/kg) for 1 week. The BC and PC groups were gavaged with distilled water, while the experimental groups were gavaged with different doses of VAPs. After 21 days, serum levels of immunoglobulin M (IgM), immunoglobulin G (IgG), T lymphocyte CD8+ (CD8+), TNF-α, and spleen gene expression levels of TLR2, TLR4, TNF-α, IL-3, and CTLA-4 were measured.
Results
Compared to the PC group, mice in the LD, MD, and HD groups exhibited a significant increase in body weight. Enzyme-linked immunosorbent assay (ELISA) results showed that TNF-α levels in the serum were significantly decreased, while IgM, IgG, and CD8+ levels were significantly increased in the LD, MD, and HD groups. Gene expression analysis revealed that TNF-α levels were significantly decreased, and IL-3 and CTLA-4 levels were significantly increased in the spleen of the LD, MD, and HD groups compared to the PC group. Additionally, the expression levels of TLR2 and TLR4 were significantly elevated.
Conclusion
VAPs improve immune status in mice by modulating the expression of TLR2/TLR4 and TNF-α/IL-3/CTLA-4 signaling pathways, revealing the underlying mechanism of their immunoregulatory effects.
Introduction
Cervid velvet antler, which refers to the unossified, growing juvenile antlers of male deer such as the Sika deer or wapiti, undergoes a cyclical process each year, including stages of antler cap formation, velvet growth, ossification, shedding, and regeneration (Price & Allen, 2004; Wu et al., 2013). As a traditional medicinal material, velvet antler possesses various benefits, such as enhancing tendons, strengthening bones, and promoting renal function (Sun et al., 2024). Velvet antler peptides (VAPs) constitute 50%–60% of the wet weight of velvet antlers and exhibit persistent expression and accumulation during the regeneration process, containing abundant substances such as glycine, leucine, lysine, alanine, and glutamic acid (Xue et al., 2022; Zhou et al., 2001). Molecular weight determination and amino acid sequence analysis of VAPs indicate that peptides with a confidence level greater than 90% have molecular weights ranging between 420.46 and 749.4 Da (Zhao et al., 2011). Immunomodulation is critical to maintaining immune system balance and function, which is crucial for health and disease management. The search for natural compounds that can modulate immune responses has garnered significant attention due to their potential therapeutic applications (Ali et al., 2021). One such compound of interest is velvet antler, which has been used in traditional medicine across various cultures, particularly in East Asia, for its purported health benefits (Li et al., 2024). Velvet antler, harvested from the growing antlers of cervid species such as the Sika deer, contains a complex mixture of bioactive peptides that are believed to influence various physiological processes, including immune regulation (Sui et al., 2014).
Recent studies have highlighted the potential of VAPs in modulating immune responses. Velvet antlers can activate immune functions by enhancing both cellular and humoral immunity. Supplementation with VAPs significantly increases the serum immunoglobulin G (IgG) levels in mice, effectively promotes the proliferation of T and B lymphocytes, and activates macrophages to secrete interleukin (IL)-12, thereby enhancing immune function (Dai et al., 2011; Liu et al., 2024). Significantly promote the proliferation of splenocytes and regulate the mouse immune system by increasing T lymphocyte CD4+ (CD4+)/T lymphocyte CD8+ (CD8+) lymphocyte subpopulations (Lincoln et al., 1984; Zha et al., 2016). Toll-like receptors (TLRs), especially TLR2 and TLR4, play a key role in recognizing pathogen-associated molecular patterns and initiating inflammatory responses. These receptors are essential for maintaining the balance between immune activation and tolerance (Sameer & Nissar, 2021). In addition, red deer antler has certain efficacy in regulating immunity, cytokines tumor necrosis factor-alpha (TNF-α) and IL-3, and immune checkpoint molecules such as recombinant cytotoxic T-lymphocyte-associated-antigen-4 (CTLA-4) are essential for regulating immune cell function and preventing excessive inflammation or autoimmunity.
Although more and more evidence supports the immunomodulatory effect of VAPs, the research is still focused on the immune regulation of red deer antler peptides. The number of research reports on the mechanism is limited, especially on the composition and efficacy mechanism of the Sika deer antler. Understanding the mechanism of action of Sika deer antlers as a natural product will help develop new therapies for immune-related diseases. This study used sika deer antler as raw material, used liquid chromatography–mass spectrometry/medium dose (LC–MS/MS) to identify peptide sequences, and used enzyme-linked immunosorbent assay (ELISA) and polymerase chain reaction (PCR) techniques to reveal the immune regulation mechanism of Sika deer antler peptides, providing theoretical support for the comprehensive utilization of Sika deer antler.
Materials and Methods
Experimental Materials
Cervid Velvet Antler: Sika deer velvet antler from Changchun Shuangyang. Trypsin (1:250): Beijing BioTeke Corporation. Acetonitrile, formic acid, ammonium bicarbonate: All were of mass spectrometry grade. Hydrochloric acid, sodium hydroxide, dithiothreitol, and iodoacetamide: All were of analytical grade. ICR mice, bedding, basic feed: Changchun Yisi Experimental Animal Technology Co., Ltd., ELISA kits for immunoglobulin M (IgM), IgG, CD8+, TNF-α: Nanjing Jiancheng Bioengineering Institute. KR116-cDNA First Strand Synthesis Kit, FP209-Talent Fluorescent Quantitative Detection Kit (SYBR Green): TianGen Biochemical Technology (Beijing) Co., Ltd., Primer Synthesis: Jilin Kumei Biotechnology Co., Ltd.
Experimental Instruments
JJ124BF Electronic Balance: Changshu Shuangjie Testing Instrument Factory. HH-6 Digital Constant Temperature Water Bath: Shanghai Lichenbangxi Instrument Technology Co., Ltd., CJ-040ST Ultrasonic Cleaner: Shenzhen Chaojie Technology Industry Co., Ltd., H3-16KR Desktop High-Speed Refrigerated Centrifuge: Hunan Keceng Instrument Equipment Co., Ltd., NP-30S Vortex Mixer: Changzhou Enpei Instrument Manufacturing Co., Ltd., DH3600A Electric Constant Temperature Incubator: Tianjin Test Instrument Co., Ltd., LC–MS System: Agilent Technologies Inc. Tecan Spark Microplate Reader: Shanghai Laboratory Equipment Co., Ltd., LightCycler®96 Roche Fluorescent Quantitative PCR System: Hoffmann-La Roche Ltd., Thermo Nucleic Acid Concentration Analyzer: Thermo Fisher Scientific.
Experimental Methods
Preparation and Determination of Velvet Antler Peptides
Accurately weigh 10 g of Sika deer velvet antler powder and mix it with 100 mL of ultrapure water. Adjust the pH to 7 using 1.0 mol/L NaOH, then add 6,000 U/g trypsin and incubate at 50°C for 4 h to terminate the reaction. Inactivate the enzyme by heating it in a 95°C water bath for 15 min, then cool rapidly. After cooling, ultrasonicate at 250 W and 10°C for 10 min. Centrifuge the extract at 10,000 rpm for 10 min, collect the supernatant, and desalinate using a self-filled desalting column. Concentrate the solvent using a vacuum concentrator at 45°C. Analyze peptide distribution using the following conditions: mobile phase A: 0.1% formic acid aqueous solution; mobile phase B: 80% acetonitrile/0.1% formic acid aqueous solution; flow rate: 600 µL/min.
Animal Experiment
Thirty male ICR mice were acclimated under conditions of temperature (24 ± 1)°C, relative humidity (60 ± 5)%, and a 12-h light/dark cycle for 1 week. They were randomly divided into five groups (n = 6): blank control (BC) group, positive control (PC) group, low dose (LD) group, middle dose (MD) group, and high dose (HD) group. The blank group was injected with physiological saline for 1 week, while the other groups received dexamethasone (25 mg/kg) for 1 week. Afterward, the blank and PC groups were gavaged with distilled water, and the experimental groups were gavaged with different doses of VAPs (60, 120, and 180 mg/kg). Mice weights were measured every 3 days. After 21 days, mice were fasted for 12 h, and blood was collected for serum separation by centrifugation at 3,000 rpm, 4°C for 15 min. Spleens were excised, cleaned of connective tissues, and stored at –80°C until analysis.
Serum Biomarker Measurement
According to the protocols of the ELISA kits, the levels of IgM, IgG, CD8+, and TNF-α were measured.
Real-time PCR Measurement of Gene Expression
Total ribonucleic acid (RNA) was extracted from spleen tissue using the RNA iso Plus reagent and reverse transcribed to complementary deoxyribonucleic acid (cDNA). Quantitative PCR was performed using SYBR Green PCR reagent in the LightCycler®96 system, with β-actin as the internal reference. Relative gene expression was calculated, and primer sequences are listed in Table 1.
Primers for Polymerase Chain Reaction (PCR) Amplification.

Data Analysis
Data are presented as mean ± standard deviation. Differences between groups were analyzed using one-way analysis of variance (ANOVA) with SPSS 23 (IBM, USA). Duncan’s multiple extreme difference test was employed, and differences between the superscript letters (a, b, c, and d), where a > b > c > d in the graphs were statistically significant (p < .05).
Results
Peptide Sequence Identification
The peptide components of velvet antler were analyzed for amino acid sequences using LC–MS/MS, and the data were analyzed with the Byonic software database. The total ion chromatogram is shown in Figure 1. Fifty-four peptide sequences were obtained from the velvet antler powder, with molecular weights ranging from 200 to 2,000 Da. These peptides are high molecular weight biological substances composed of 8–20 amino acid residues, which benefit human absorption. The primary amino acid components identified include glycine, leucine, aspartic acid, phenylalanine, and glutamic acid, with glycine being the most frequently occurring.
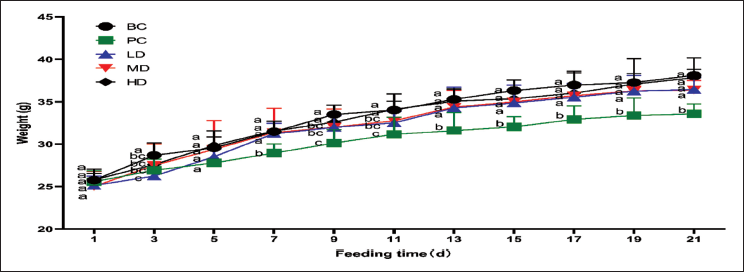
Changes in Mouse Body Weight
As shown in Figure 2, compared to the PC group, body weight significantly increased in the LD, MD, and HD groups after supplementation with VAPs (p < .05). Among these, the HD group exhibited a higher body weight than the LD and MD groups.
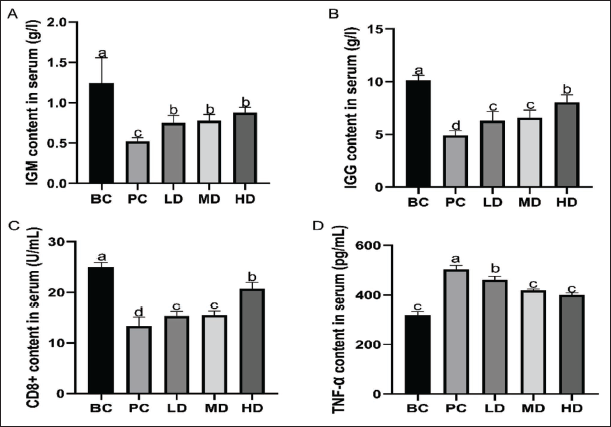
Serum Levels of IgM, IgG, CD8+, and TNF-α
The results are shown in Figure 3. Compared to the PC group, the levels of IgM, IgG, and CD8+ in the serum were significantly increased in the LD, MD, and HD groups (p < .05). Among these, the HD group demonstrated a more pronounced improvement than the LD and MD groups. In contrast, supplementation with VAPs significantly reduced the serum levels of TNF-α compared to the PC group (p < .05).
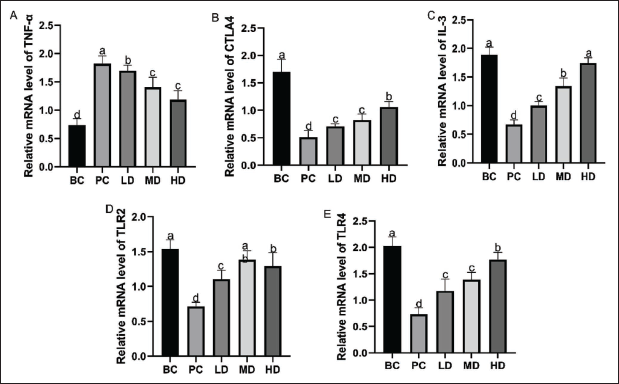
Expression of Immune Regulation-related Messenger Ribonucleic Acid (mRNA) in Mouse Spleen
The gene expression results are shown in Figure 4. Compared to the PC group, the expression levels of immune regulation-related genes CTLA-4, IL-3, TLR2, and TLR4 were significantly increased in the spleens of the LD, MD, and HD groups (p < .05). Notably, the expression of IL-3 did not differ significantly from that in the BC group. Compared to the PC group, supplementation with VAPs significantly reduced the TNF-α levels in the spleens of the LD, MD, and HD groups (p < .05).
Discussion
In this study, we investigated the immunomodulatory effects of VAPs from Sika deer and elucidated their mechanisms of action. The results indicate that VAPs substantially impact immune system modulation, as evidenced by changes in body weight, serum cytokine levels, and the expression of key immune-related genes. Our findings show that mice administered VAPs significantly increased body weight compared to the control group. This increase in body weight may reflect improved overall health and enhanced physiological function, which can be attributed to the immunomodulatory properties of VAPs. Similar observations have been reported in other studies, where natural compounds improved general health parameters through immune modulation (Radek & Gallo, 2007).
The study demonstrates that Sika deer velvet antler has immunomodulatory effects, with VAPs being the primary active components (Zang et al., 2016; Zha et al., 2013). ELISA results revealed a significant decrease in serum TNF-α levels in mice treated with VAPs, while IgM, IgG, and CD8+ T cell levels were significantly increased. TNF-α is a proinflammatory cytokine often associated with systemic inflammation and immune dysregulation (Travis & Sheppard, 2014). The reduction in TNF-α levels suggests that VAPs may help alleviate inflammation, potentially contributing to improved immune balance. The increase in IgM, IgG, and CD8+ T cells further supports the notion that VAPs enhance immune responses by boosting humoral and cellular immunity (Napodano et al., 2021). CD8+ and TNF-α play broad roles in immune regulation (Borriello et al., 2016; Kawai & Akira, 2010).
TLRs are pattern recognition receptors widely present in the innate immune system. TLR2 and TLR4 are crucial for mediating macrophage activation and are closely related to immune regulation (Brightbill & Modlin, 2000; Van Coillie et al., 2020). Our study shows that after supplementation with VAPs, the expression levels of TLR2 and TLR4 in the spleen were significantly increased, which plays a key role in macrophage regulation. TLR2 and TLR4 are essential for recognizing pathogen-associated molecular patterns and initiating appropriate immune responses (Wicherska-Pawlowska et al., 2021). The upregulation of these receptors indicates that VAPs might enhance the capacity of the immune system to detect and respond to potential threats, thus contributing to improved immune surveillance and response.
Monocytes or macrophages primarily secrete TNF and can induce the production of itself and other cytokines such as IL-3 and IL-6, promoting the proliferation of T and B lymphocytes and the synthesis of immunoglobulins, ultimately playing a vital role in immune regulation (Aggarwal, 2003; Van Der Meer et al., 1998). IL-3 is crucial for the growth and differentiation of immune cells, while CTLA-4 plays a role in immune checkpoint regulation, preventing excessive activation of T cells (Zaidi et al., 2019). The observed increase in IL-3 and CTLA-4 suggests that VAPs may promote immune system balance and tolerance, reducing the risk of excessive inflammatory responses or autoimmunity. Our research shows that after supplementation with VAPs, the expression level of TNF-α was significantly downregulated, while the expression levels of IL-3 and CTLA-4 were increased considerably. The improvement in the TLR2/TLR4 and TNF-α/IL-3/CTLA-4 signaling pathways may represent the potential mechanisms by which Sika deer VAPs regulate immunity.
Conclusion
In summary, VAPs significantly improved serum IgM, IgG, CD8+, and TNF-α levels. They enhanced the immune status of immunocompromised mice by modulating the TLR2/TLR4 and TNF-α/IL-3/CTLA-4 signaling pathways. These findings suggest that VAPs have promising potential as a traditional Chinese medicine for immune regulation.
Footnotes
Abbreviations
BC: Blank control; CD8+: T lymphocyte CD8+; CTLA-4: Recombinant cytotoxic T-lymphocyte-associated-antigen-4; HD: High dose; IgG: Immunoglobulin G; IgM: Immunoglobulin M; IL-3: Interleukin-3; LC–MS: Liquid chromatography–mass spectrometry; LD: Low dose; MD: Medium dose; PC: Positive control; TNF-α: Tumor necrosis factor-alpha; TLR2: Toll-like receptors 2; TLR4: Toll-like receptors 4; VAPs: Velvet antler peptides.
Author Contributions
Methodology resources, formal analysis, investigation, and writing original draft: Lan-Yue Yin. Writing review and editing and methodology resources: Jia-Ying Li. Formal analysis and conceptualization: Xue-Yue Tai and Ming-Ran Luan. Conceptualization and investigation: Guo-Qi Zhang and Wu-Yang Hua. Resources and data curation resources: Chao Liu. Supervision and funding acquisition: Bao Zhong. Project administration, writing review and editing, supervision, and funding acquisition: Feng-Lin Li.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the Animal Ethics Committee of Jilin Agriculture Science and Technology University (Approval Number: LLSC202305025).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National College Student Science and Technology Innovation and Entrepreneurship Project: Research on the Preparation of Velvet Antler Peptides and their Immunomodulatory Mechanism (Project No: 202311439007); Key R&D Project of Jilin Provincial Science and Technology Department: Research on the Deep Processing Technology of Sika Deer and Development of Series Health Products (Project No: 20220304002YY); Jilin Agricultural Science and Technology College, Key Discipline in Food Science and Engineering (2023).
