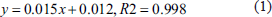Abstract
Background
Cough variant asthma (CVA) is a distinct type of asthma manifested as dysregulation of fluid metabolism and modulation of ferroptosis.
Objectives
The aim of this study was to demonstrate the regulatory effects of Mulberry Apricot Coughing Granules on fluid metabolism dysregulation in CVA patients and elucidate its mechanism in inhibiting ferroptosis.
Materials and Methods
In this study, Mulberry Apricot Coughing Granules were prepared and analyzed for their major active components. Subsequently, in vitro experiments were conducted using human bronchial epithelial cells (BEAS-2B), which were divided into the following groups: control group (CG, basal medium), RSL3 group (treated with Ras-selective lethal substance 3), 0.2 mg/mL group, 0.1 mg/mL group, and 0.05 mg/mL group (treated with Mulberry Apricot Coughing Granules extract at concentrations of 0.2, 0.1, and 0.05 mg/mL, respectively), as well as the RSL3 + 0.2 mg/mL group (treated with RSL3 + 0.2 mg/mL Mulberry Apricot Coughing Granules extract). The levels of inflammatory factors, including interleukin (IL)-4, IL-6, and tumor necrosis factor (TNF)-α, were measured in each group, and reactive oxygen species (ROS) and malondialdehyde (MDA) levels were determined using assay kits. In addition, the expression levels of glutathione peroxidase 4 (GPX4) and acyl-CoA synthetase long-chain family member 4 (ACSL4) were assessed. Subsequently, a CVA mouse model was established using a sensitizing solution, with groups including the normal group (NG, physiological saline), CVA group (sensitizing solution), and treatment group (TG, sensitizing solution + Mulberry Apricot Coughing Granules treatment). Urine output and water intake were observed, and plasma anti-diuretic hormone (ADH) levels and urinary aquaporin-2 (AQP-2) content were measured. The expression levels of aquaporin-5 (AQP5), mucin 5AC (MUC5AC), and cystic fibrosis transmembrane conductance regulator (CFTR) were evaluated using quantitative real time polymerase chain reaction (qRT-PCR) and Western blotting techniques.
Results
The chromatographic peaks of the water extract from Mulberry Apricot Coughing Granules and the amygdalin standard exhibited identical positions with clear separation. The distribution of active ingredients in Mulberry Apricot Coughing Granules was uniform. In the 0.2, 0.1, and 0.05 mg/mL groups, concentrations of IL-4, IL-6, and TNF-α were markedly inferior to CG (p < .05), with the 0.2 mg/mL group showing the lowest levels (p < .05). The levels of ROS and MDA, as well as the mRNA and protein expression of ACSL4, in the RSL3 group were significantly higher than those in the CG (p < .05), while the mRNA and protein expression of GPX4 were decreased (p < .05). In contrast, the levels of ROS and MDA and the mRNA and protein expression of ACSL4 were reduced in the 0.2 mg/mL group and the RSL3 + 0.2 mg/mL group, while the mRNA and protein expression of GPX4 were increased compared to the RSL3 group, with the 0.2 mg/mL group showing a better effect (p < .05). In the CVA model, relative to NG, urine volume, water intake, AQP5, MUC5AC mRNA, and protein expression level were decreased, while plasma ADH concentration, urine AQP-2 content, CFTR mRNA, and protein expression level were increased in the CVA group (p < .05). Mice in TG showed increased urine volume, water intake, AQP5, MUC5AC mRNA, and protein expression level, as well as decreased plasma ADH concentration, urine AQP-2 content, CFTR mRNA, and protein expression level versus CVA group (p < .05).
Conclusion
Mulberry Apricot Coughing Granules regulate oxidative stress and lipid peroxidation by upregulating the antioxidant protein GPX4 and reducing ACSL4 expression level, thereby inhibiting ferroptosis and improving inflammatory responses. Moreover, Mulberry Apricot Coughing Granules may improve fluid metabolism abnormalities in CVA by enhancing the expression levels of AQP5 and MUC5AC while reducing CFTR expression levels. These findings suggest that Mulberry Apricot Coughing Granules may exert therapeutic effects on CVA through multiple mechanisms.
Introduction
Cough Variant Asthma (CVA) is a distinct type of asthma manifested as cough without prominent wheezing or dyspnea (Abi-Ayad et al., 2023; Enseki et al., 2019). Traditional Chinese medicine (TCM) attributes dysregulation of fluid metabolism as a key pathological mechanism leading to cough and asthma (Hussain-Alkhateeb et al., 2021; Watchorn et al., 2021). Pulmonary dysfunction can induce fluid metabolism disorders, potentially resulting in phlegm-damp syndrome (Liu et al., 2018). Modern research has also linked fluid metabolism disturbances to respiratory diseases such as airway hyperresponsiveness and inflammation (Frippiat et al., 2023). Ferroptosis, an iron-dependent form of cell death, has garnered attention in various diseases in recent years (Amaral & Namasivayam, 2021; Feng et al., 2022; Yu et al., 2021). Studies suggested that ferroptosis may participate in airway injury and remodeling, and modulating ferroptosis could be a strategy for asthma, particularly in managing oxidative stress and airway inflammation (Liu et al., 2023).
Mulberry Apricot Coughing Granules, a Traditional Chinese herbal compound formulation, originates from the TCM system and is applied for respiratory issues such as CVA (Li et al., 2021; Zhang et al., 2023). This formulation primarily includes medicinal materials such as mulberry leaf (Moraceae family, Morus genus), apricot kernel (Rosaceae family, Prunus genus), Fritillaria (Liliaceae family, Fritillaria genus), white root (Campanulaceae family, Platycodon genus), Platycodon (Campanulaceae family, Platycodon genus), and licorice (Fabaceae family, Glycyrrhiza genus), each of which possesses unique pharmacological properties. Together, they synergistically regulate the body’s internal environment, effectively alleviating symptoms of CVA. Mulberry leaf is renowned for its ability to dispel wind-heat, clear lung heat, and relieve cough by moistening the lungs, which is particularly beneficial for asthma patients with associated cough. The apricot kernel has anti-tussive properties that help alleviate respiratory spasms and coughing attacks in asthma patients. Fritillaria thunbergia and Fritillaria thunbergii are used to clear heat, transform phlegm, moisten the lungs, and stop coughing, effectively alleviating symptoms of asthma characterized by excessive phlegm and frequent coughing. Platycodon grandiflorus, a crucial component of the formulation, acts to ventilate the lungs, benefit the throat, and promote pus drainage, significantly aiding in improving airway patency and reducing inflammation in asthma patients. Licorice is commonly used in formulations to harmonize ingredients and enhance the efficacy of medications while reducing their side effects. Modern pharmacological research has shown that Mulberry Apricot Coughing Granules exert therapeutic effects against CVA through multiple mechanisms (Sun et al., 2022). First, various medicinal herbs in their composition exert anti-inflammatory effects to alleviate airway inflammation and reduce mucus secretion, thereby improving respiratory difficulties and frequency of coughing in asthma patients (Chang et al., 2021; Deng et al., 2020). Second, active ingredients in Mulberry Apricot Coughing Granules possess significant antioxidant effects, which can mitigate oxidative stress-induced damage to lung tissues and protect the function of respiratory epithelial cells (Abbas et al., 2024). Additionally, Mulberry Apricot Coughing Granules modulate immune function by balancing Th1/Th2 cell immune responses, thereby reducing airway hyperresponsiveness and allergic reactions, leading to improved long-term control of asthma.
However, despite demonstrating certain therapeutic efficacy in clinical practice, the underlying mechanisms of Mulberry Apricot Coughing Granules remain incompletely elucidated. Particularly in the regulation of fluid metabolism disorders and inhibition of ferroptosis, relevant studies are scarce. Therefore, this study aimed to explore the mechanisms by which Mulberry Apricot Coughing Granules treat CVA, specifically its effects on fluid metabolism and ferroptosis. The findings aim to provide scientific rationale and theoretical support for further clinical adoptions.
Materials and Methods
Fabrication of Mulberry Apricot Coughing Granules
The preparation method for Mulberry Apricot Coughing Granules is as follows: 13 traditional Chinese medicinal herbs were used for extraction in the laboratory. These herbs include: 15 g mulberry leaf (Morus alba L., Moraceae), 9 g Platycodon root (P. grandiflorus (Jacq.) A. DC., Campanulaceae), 9 g apricot kernel (Prunus armeniaca L., Rosaceae), 10 g processed Fritillaria (Pinellia ternata (Thunb.) Breit., Araceae), 9 g Lophatherum gracile (Adenophora triphylla (Thunb.) Schult., Campanulaceae), 10 g processed Perilla seeds (Perilla frutescens (L.) Britton, Lamiaceae), 10 g loquat leaf (Eriobotrya japonica (Thunb.) Lindl., Rosaceae), 10 g Salvia root (Salvia miltiorrhiza Bunge, Lamiaceae), 6 g cicada slough (Cryptotympana facialis (Fabricius), Cicadidae), 12 g broom leaf (Kochia scoparia (L.) Schrad., Amaranthaceae), 15 g elderberry (Sambucus chinensis Lindl., Caprifoliaceae), 15 g purple root (Folium asiaticum), and 3 g raw licorice (Glycyrrhiza uralensis Fisch., Fabaceae) (Beijing Tongrentang, China). These herbs were extracted utilizing a traditional decoction method. Specifically, 200 g of the herbal formula was soaked in water (12 times the mass of herbs) in a 5,000 mL round-bottom flask (Pyrex, Corning, USA) for 30 min. The extraction was then performed by reflux heating for approximately 2 h in a single extraction cycle. The filtrate was obtained through cooling, replenishing, and filtering processes. After combining and filtering the filtrates, the supernatant was generated by centrifugation at 3,000 rpm for 40 min at 10°C. The obtained Mulberry Apricot Coughing Granules extract was transferred utilizing a Pipetman P200 pipette (Gilson, France) into a dried evaporating dish (100 mL Porcelain Evaporating Dish, Corning, USA). The pipette containing the aqueous extract was then placed in a water bath (WB-22, Memmert, Germany) for evaporation. After evaporation, the dish was placed in a drying oven (UN55, Memmert, Germany) at 105°C for approximately 3 h to obtain a dry paste. The paste was then finely ground employing an analytical mill (IKA A11 Basic Analytical Mill, IKA Works, Germany). In addition, 9 g of white-flowered snake tongue grass (F. thunbergii Siebold & Zucc., Liliaceae, sourced from Beijing Tongrentang) was finely ground into a powder and uniformly mixed with the dry extract powder at a 1:4 ratio. 5‰ of steviol glycosides (PureCircle, Malaysia) were applied as a sweetener. Subsequently, 93% ethanol (Sigma-Aldrich, USA) was applied as a binder to form Mulberry Apricot Coughing Granules, with a drug particle concentration of 20 mg/100 g.
Determination of Amygdalin Content in Bitter Almonds
The study assessed the content of amygdalin in the aqueous extract of Mulberry Apricot Coughing Granules during preparation, comparing it with amygdalin standard and negative control.
Preparation of amygdalin standard: 5 mg of amygdalin standard (Sigma-Aldrich, USA) was weighed and placed in a volumetric flask of 10 mL. The amygdalin standard was dissolved in chromatographic methanol to obtain a stock solution with a concentration of 0.5 mg/mL. Subsequently, aliquots of the 0.5 mg/mL amygdalin standard were further diluted with chromatographic methanol to form various concentrations of amygdalin standard (0.5, 0.25, 0.125, 0.0625, and 0.03125 mg/mL).
Negative control: Aqueous extract without amygdalin was prepared according to the method used for Mulberry Apricot Coughing Granules.
A high-performance liquid chromatography (HPLC) system (Waters Alliance HPLC, Waters Corporation, USA) equipped with a C18 reverse-phase column (250 × 4.6 mm, 5 µm) was employed. The mobile phase consisted of acetonitrile and 0.1% phosphoric acid (2:98, v/v), with a flow rate of 1.0 mL/min. The column temperature was 29°C. A sample volume of 10 µL was used, and detection was conducted at a wavelength of 208 nm. Standard curves for amygdalin were constructed based on peak areas obtained from different concentrations of standard solutions. Subsequently, amygdalin concentration was calculated utilizing the standard curve method, analyzing 20 Mulberry Apricot Coughing Granules samples for concentration determination.
Determination of Total Flavonoid Content
The study employed UV detection to quantify the total flavonoid content in the aqueous extract, comparing it with a rutin standard.
Preparation of rutin standard: 10 mg of rutin standard (Sigma-Aldrich, USA) was weighed and placed in a 25 mL beaker. The rutin standard was dissolved in water and then diluted. Subsequently, 5 mL of the diluted solution was transferred into a volumetric flask of 10 mL, where it was further diluted with water to achieve a concentration of 0.2 mg/mL of rutin standard solution. It was then diluted again to generate different concentrations of rutin standard solutions (0.05, 0.04, 0.03, 0.02, and 0.01 mg/mL).
The absorbance of each solution was measured using a UV-visible spectrophotometer (Cary 60, Agilent Technologies, USA) with the wavelength set at 510 nm. A rutin standard curve was constructed based on the absorbance values of rutin standard solutions at different concentrations. The concentration range of the rutin standard solutions was 0–100 µg/mL. The equation for the rutin standard curve is:
Here, y represents the absorbance, and x represents the concentration of rutin (µg/mL). Subsequently, the total flavonoid content in the sample solutions was calculated using the absorbance values and the standard curve method. This analysis involved testing the concentration of 20 samples of Mulberry Apricot Coughing Granules, with the flavonoid content expressed as rutin equivalents.
In Vitro Experiments
Cell Culture
BEAS-2B cells (ATCC, USA) were selected for experimental research. The cells were cultured in RPMI-1640 medium with 10% fetal bovine serum (Thermo Fisher Scientific, USA). The conditions were at 37°C, 95% humidity, and a 5% CO2 atmosphere. Subculturing was performed when the cells reached 80–90% confluence to ensure the logarithmic growth phase of cells.
Preparation of Mulberry Apricot Coughing Granule Extract
Preparation of Mulberry Apricot Coughing Granules extract: An appropriate amount of Mulberry Apricot Coughing Granules was taken and mixed with methanol. The mixture was subjected to ultrasound extraction for 30 min utilizing an ultrasonic cleaner. After extraction, a solution was centrifuged at 4,000 rpm for 10 min to collect supernatant. The clarified Mulberry Apricot Coughing Granules extract was filtered through a sterile membrane filter (Millex-GP, Millipore (Merck), USA) to obtain the original extract. The original extract’s concentration was determined utilizing UV–vis spectrophotometry, quantified as 0.2 mg/mL of flavonoid compounds. Portions of the original extract were diluted with methanol to prepare different concentrations (0.2, 0.1, and 0.05 mg/mL). Each concentration of Mulberry Apricot Coughing Granules extract was aliquoted into labeled sterile tubes (Corning, USA) and stored in a light-protected refrigerator at 4°C (Thermo Fisher Scientific, USA) for subsequent research use.
Cell Grouping
Control group (CG): RPMI-1640 medium supplemented with an equal volume of RPMI-1640 medium without Mulberry Apricot Coughing Granules extract.
RSL3 group: RPMI-1640 medium supplemented with an equal volume of Ras-selective lethal 3 (RSL3) (Sigma-Aldrich, USA) without Mulberry Apricot Coughing Granules extract.
Mulberry Apricot Coughing Granules group: RPMI-1640 medium supplemented with different concentrations (0.2, 0.1, and 0.05 mg/mL) of Mulberry Apricot Coughing Granules extract, designated as the 0.2, 0.1, and 0.05 mg/mL groups.
RSL3 + 0.2 mg/mL group: RPMI-1640 medium supplemented with an equal volume of RSL3 and 0.2 mg/mL concentration of Mulberry Apricot Coughing Granules extract.
All cells in the RPMI-1640 medium grew under these conditions for 24 h to visualize the short-term effects of various treatments.
In this study, the RSL3 group was used as a positive control to investigate the effects of Mulberry Apricot Coughing Granules on cellular oxidative stress and ferroptosis. RSL3 is a well-known ferroptosis inducer, widely used to study the mechanisms of ferroptosis. It induces ferroptosis by inhibiting the activity of glutathione peroxidase 4 (GPX4), a process closely associated with oxidative stress. Therefore, the RSL3 group, as a positive control, can simulate the occurrence of ferroptosis and provide a comparative basis for evaluating the potential protective effects of Mulberry Apricot Coughing Granules. By comparing with the RSL3 group, this study can assess whether the granules play a role in inhibiting oxidative stress and ferroptosis, thereby validating their therapeutic effects.
Inflammatory Factors Detection
After treatment, supernatants from CG and Mulberry Apricot Coughing Granules groups (0.2, 0.1, and 0.05 mg/mL) were collected and clarified by centrifugation at 2,000 rpm for 10 min (Allegra X-15R, Beckman Coulter, USA) to remove cellular debris. The cleared supernatants were then subjected to enzyme-linked immunosorbent assay (ELISA) analysis (Abcam, UK). A 96-well ELISA plate was prepared with standards and samples were loaded accordingly. Levels of interleukin (IL)-4, IL-6, and tumor necrosis factor (TNF)-α were quantified by measuring absorbance at 450 nm via a microplate reader (SpectraMax M5, Molecular Devices, USA). The concentrations of IL-4, IL-6, and TNF-α in the samples were calculated based on the standard curve. The differences between the groups were compared to determine the optimal concentration of Mulberry Apricot Coughing Granules for subsequent studies.
Detection of Oxidative Stress and Iron Death Markers
Oxidative Stress Detection
Utilizing reactive oxygen species (ROS) assay kits (Cell Signaling Technology, USA), intracellular ROS levels were measured in each experimental group. Fluorescence intensity indicating ROS production was observed and recorded utilizing a fluorescence microscope (Eclipse Ti2, Nikon, Japan), followed by quantitative analysis to assess differences between groups. Additionally, malondialdehyde (MDA) levels within the cells were measured utilizing MDA assay kits (Cayman Chemical, USA) to evaluate the extent of lipid peroxidation across different treatment conditions.
Detection of Substances Related to Ferroptosis
After 24 h of cell culture, total RNA was extracted from CG, RSL3 group, and RSL3 + 0.2 mg/mL group. RNA concentration and quality were assessed utilizing NanoDrop 2000 (Thermo Fisher Scientific, USA). Subsequently, 1–2 µg of RNA was reverse transcribed into cDNA utilizing a reverse transcription kit (Invitrogen, USA). GAPDH was used as an internal reference gene. Employing a qRT-PCR kit (Applied Biosystems, USA), reaction conditions were as follows: initial denaturation (95°C, 30 s), denaturation (95°C, 5 s), and annealing/extension (60°C, 30 s) for 40 cycles. Melting curve analysis lasted from 65°C to 95°C with a 1°C increment to verify PCR product specificity. Ct values were analyzed utilizing qRT-PCR software to calculate relative mRNA expression levels of GPX4 and acyl-CoA synthetase long-chain family member 4 (ACSL4), normalized to the internal reference. Primer design information for target genes and the reference gene is shown in Table 1.
Primer Sequence Information.

Western Blotting
After 24 h of cell culture, cells were lysed using radioimmunoprecipitation assay (RIPA) buffer containing 1% phenylmethanesulfonyl fluoride (PMSF, Sigma-Aldrich, USA) as a protease inhibitor. The lysate was mixed with 5× sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) loading buffer (Bio-Rad Laboratories, USA) and heated at 100°C for 10 min to ensure complete protein denaturation. The lysate was centrifuged to remove cell debris, and the supernatant was collected for protein quantification. Protein samples were loaded onto a 10% SDS-PAGE gel and separated by electrophoresis. After electrophoresis, proteins were transferred onto a polyvinylidene fluoride (PVDF) membrane (0.45 µm pore size, Bio-Rad Laboratories, USA) under conditions of 100 V for 1 h. After transfer, the membrane was washed three times with Tris-buffered saline with Tween (TBST) for 5 min each to remove non-specific bindings. Following protein transfer, the membrane was blocked with 5% non-fat milk in TBST buffer and incubated at room temperature for 1 h to prevent non-specific antibody binding. Subsequently, the membrane was incubated with primary antibodies against GPX4 and ACSL4. GPX4 antibody (1:1,000 dilution, DuPont, USA) and ACSL4 antibody (1:1,000 dilution, DuPont, USA) were applied to the membrane and incubated overnight at 4°C. The following day, the membrane was washed three times with TBST buffer for 5 min each to remove unbound antibodies. Next, an appropriate secondary antibody (1:5,000 dilution, anti-rabbit immunoglobulin G (IgG) secondary antibody, horseradish peroxidase (HRP)-conjugated, Santa Cruz Biotechnology, USA) was added, and the membrane was incubated at room temperature for 1 h. After incubation, the membrane was washed three times with TBST buffer for 5 min each. Finally, the membrane was developed using an enhanced chemiluminescence (ECL) reagent (Thermo Fisher Scientific, USA) according to the manufacturer’s instructions, and images were captured using a chemiluminescence imaging system (Bio-Rad Laboratories, USA). The band intensity was quantified by calculating the grayscale values, and β-actin was used as the internal reference protein for normalization.
In Vivo Experiments
Animals
Thirty clean-grade healthy adult male BALB/c mice, provided by the Shanghai Experimental Animal Center, were selected for this study. The mice were all 8 weeks old (20 ± 2 g), kept at a temperature of 20 ± 1°C, relative humidity 50–60%, with a 12-h light–dark cycle, and a ventilation rate of 8–15 air changes per hour.
Model Construction and Grouping
The study used 100 µg of ovalbumin and 20 µg of aluminum hydroxide (Sigma-Aldrich, USA) dissolved in saline to prepare the sensitizing solution. The ovalbumin was aerosolized to induce the chronic CVA model. Thirty mice were rolled into a normal group (NG), CVA group, and treatment group (TG), with 10 mice in each group. After acclimatization under stable environmental conditions for 1 week, the mice were treated as follows:
NG: Each mouse received 200 µL of saline via intraperitoneal injection on days 1, 7, and 14.
CVA group and TG: Each mouse received 200 µL of sensitizing solution via intraperitoneal injection on days 1, 7, and 14. One week after the final injection, mice were exposed to 1.5% OVA solution via ultrasonic nebulization for 30 min per day continuously for 2 weeks.
One hour prior to each nebulization session, mice in the NG and CVA groups received intragastric gavage of 2 mg/kg of saline. Mice in TG received an equivalent dose of Mulberry Apricot Coughing Granules dissolved and administered via gavage.
Following sensitization, airway reactivity, inflammatory cell infiltration, and cytokine levels were assessed to evaluate the progression of asthma pathology.
Observation of Urine Output and Water Intake
The urine volume and water intake of mice in NG, CVA group, and TG were observed before model induction and after treatment. Before model induction, daily water intake and urine volume were measured based on the total collected over 24–48 h within the first 7 days of acclimatization feeding. After model induction, measurements were based on the total collected over 24–48 h following successful model induction.
Detection of Plasma Anti-diuretic Hormone (ADH) Concentration and Urine Aquaporin 2 (AQP-2) Content
Collecting the total urine output of each group of mice within 24 h after successful model induction was performed as the detection specimen. During this period, mice in each group had free access to water. Subsequently, the specimens were processed utilizing the methods of mouse femoral artery bleeding (Makley et al., 2010). The concentration of AQP-2 in mouse urine was measured using the Bradford assay (Takeda-Uchimura et al., 2022), while the concentration of ADH in mouse plasma was determined by radioimmunoassay (RIA) (Hou et al., 2023).
qRT-PCR Detection
After 2 weeks of treatment, mice were euthanized utilizing cervical dislocation, and lung tissue samples were collected. Each lung tissue sample (100 mg) was homogenized in 1 mL TRIzol reagent (Invitrogen, USA) in a precooled Eppendorf tube. The homogenate was then processed utilizing a homogenizer to extract RNA. Following RNA extraction, cDNA synthesis was performed. The relative expression levels of aquaporin-5 (AQP5), mucin 5AC (MUC5AC), and cystic fibrosis transmembrane conductance regulator (CFTR) were quantified utilizing qRT-PCR, followed by statistical analysis. Primer design details are provided in Table 2.
Primer Sequence Information.

Western Blotting
The lung tissue homogenate was subjected to cell lysis, protein denaturation, and electrophoresis separation. The samples were incubated with primary antibodies against AQP5, MUC5AC, and CFTR, sourced from PTFE (DuPont, USA). The membrane was rinsed and then incubated with goat anti-rabbit IgG H&L (HRP) secondary antibody. Finally, the ECL reagent was used for visualization to detect changes in the protein expression level of AQP5, MUC5AC, and CFTR, normalized to β-actin as the internal reference protein.
Statistical Analysis
Employing SPSS 20.0, quantitative data were presented as Mean ± Standard Deviation. One-way analysis of variance (ANOVA) was applied to datasets that met the assumptions of normality and homogeneity of variances among groups. Marked differences identified by ANOVA were further analyzed utilizing Bonferroni post hoc tests to determine specific group variations. Statistical significance was α = .05, with p < .05 indicating significant differences.
Results
Main Active Ingredient Detection Results
In this study, HPLC and UV spectroscopy were employed to analyze amygdalin and total flavonoid contents in Mulberry Apricot Coughing Granules. HPLC analysis revealed that the chromatographic peak of amygdalin in the aqueous extract of Mulberry Apricot Coughing Granules matched that of the standard, showing distinct separation from other peaks (Figure 1A–C). Further analysis involved preparing and analyzing different concentrations of amygdalin standards, yielding a regression equation between concentration and peak area of y = 1 × 10–7X + 0.0082 (r = .999) (Figure 1D). According to the regression analysis, the concentration of amygdalin in Mulberry Apricot Coughing Granules ranged smoothly between 0.52 and 0.50 mg/mL (Figure 1E).
Detection of Amygdalin and Total Flavonoid Concentrations. (A) Chromatogram of Amygdalin Standard Solution; (B) Chromatogram of Water Extract from Mulberry Apricot Coughing Granules; (C) Chromatogram of Negative Control; (D) Amygdalin Standard Solution; (E) Mulberry Apricot Coughing Granules; (F) Rutin Standard Solution; (G) Mulberry Apricot Coughing Granules.
On another aspect, the total flavonoid content in Mulberry Apricot Coughing Granules was determined utilizing UV spectroscopy. Standard solutions of rutin at various concentrations were prepared and their absorbance was measured, resulting in a regression equation of y = 9.65X + 0.1075 (r = 1.000), as depicted in Figure 1F. According to this regression equation, the concentration of total flavonoids in Mulberry Apricot Coughing Granules remained stable between 0.20 and 0.23 mg/mL (Figure 1G).
In Vitro Results
Immunofluorescence (IF) Detection Results
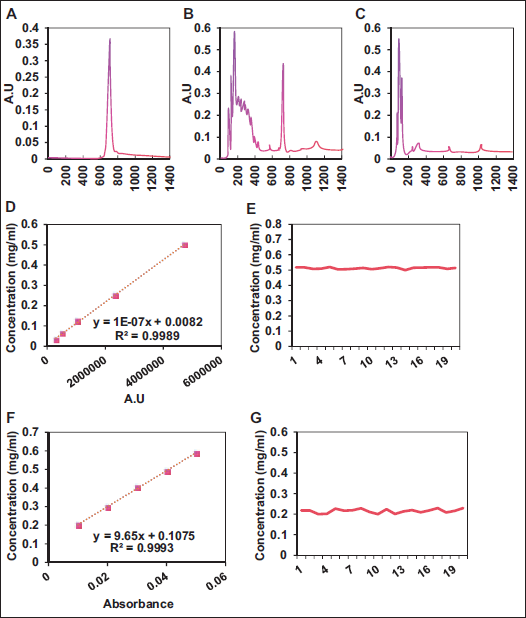
The study compared the concentrations of IL-4, IL-6, and TNF-α in BEAS-2B cells among CG, 0.2, 0.1, and 0.05 mg/mL groups (Figure 2). Results indicated that the concentrations of IL-4, IL-6, and TNF-α in 0.2, 0.1, and 0.05 mg/mL groups were considerably inferior to CG (p < .05). Further comparisons revealed that the concentrations of IL-4, IL-6, and TNF-α in 0.2 mg/mL group were markedly inferior to those in the 0.1 and 0.05 mg/mL groups (p < .05). These findings suggest that Mulberry Apricot Coughing Granules at a concentration of 0.2 mg/mL exhibit the most optimal effects in vitro experiments.
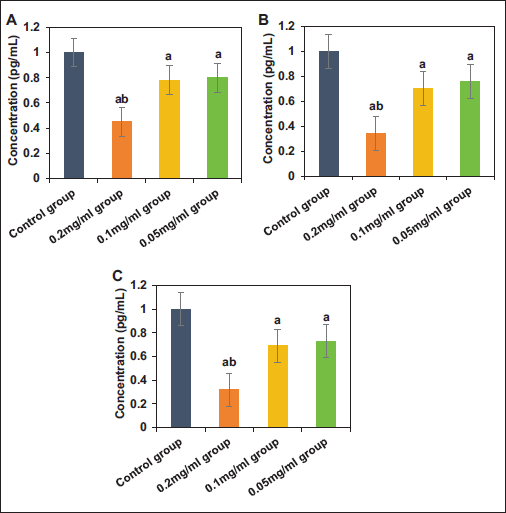
Changes in Oxidative Stress and Iron Death Indicators
The ROS and MDA levels in BEAS-2B cells from the CG, 0.2 mg/mL group, RSL3 group, and RSL3 + 0.2 mg/mL group are shown in Figure 3A–B. Compared to the CG, the ROS and MDA levels in BEAS-2B cells of the RSL3 group were significantly increased (p < .05). Compared to the RSL3 group, the ROS and MDA levels in the 0.2 mg/mL group and RSL3 + 0.2 mg/mL group were significantly decreased, with the 0.2 mg/mL group showing lower levels than the RSL3 + 0.2 mg/mL group (p < .05). The expression levels of GPX4 and ACSL4 mRNA and protein in BEAS-2B cells from the CG, 0.2 mg/mL group, RSL3 group, and RSL3 + 0.2 mg/mL group are shown in Figure 3C–G. Compared to the CG, the GPX4 mRNA and protein levels in BEAS-2B cells of the RSL3 group were significantly decreased, while the ACSL4 mRNA and protein expression levels were significantly increased (p < .05). Compared to the RSL3 group, the GPX4 mRNA and protein levels in the 0.2 mg/mL group and RSL3 + 0.2 mg/mL group were significantly elevated, while the ACSL4 mRNA and protein levels were significantly reduced, with the 0.2 mg/mL group showing higher GPX4 mRNA and protein expression and lower ACSL4 mRNA and protein expression than the RSL3 + 0.2 mg/mL group (p < .05).
In Vivo Results
Comparison of Urine and Water Intake Among Different Groups of Mice
The study observed changes in urine volume and water intake in mice before and after modeling in NG, CVA group, and TG. Before modeling, urine volume and water intake differed slightly among the groups (p > .05). After modeling and treatment, mice in the CVA group showed drastic decreases in urine volume and water intake relative to NG (p < .05), while mice in TG exhibited great increases in these versus the CVA group (p < .05) (Figure 4).
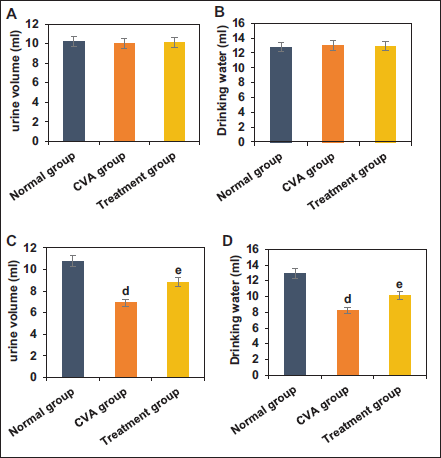
Comparison of Blood ADH and Urine AQP-2 Detection Results Among Different Groups
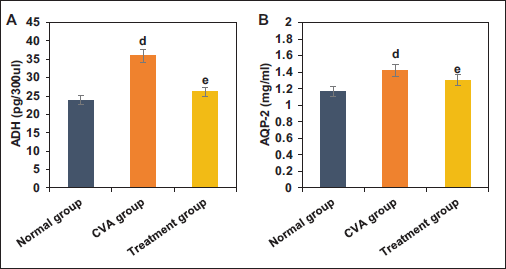
The study collected and analyzed the blood ADH concentration and urine AQP-2 levels in mice from NG, CVA group, and TG. Relative to NG, mice in the CVA group showed substantially increased blood ADH concentration and urine AQP-2 levels (p < .05). In contrast, mice in TG exhibited drastically decreased blood ADH concentration and urine AQP-2 levels versus the CVA group (p < .05) (Figure 5).
AQP5, MUC5AC, and CFTR Test Results
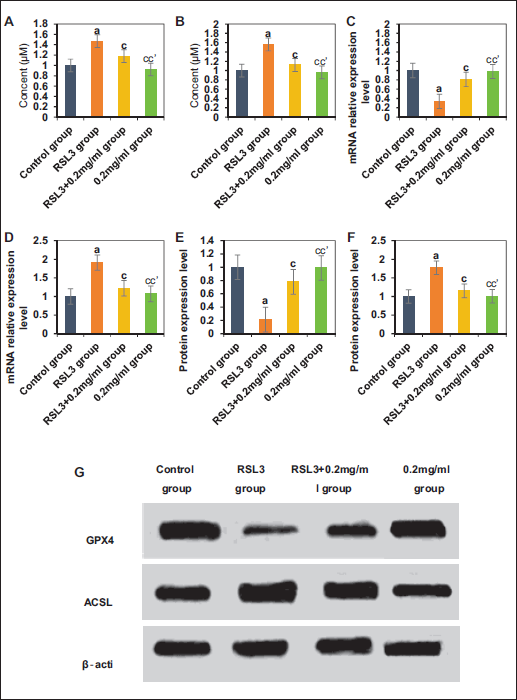
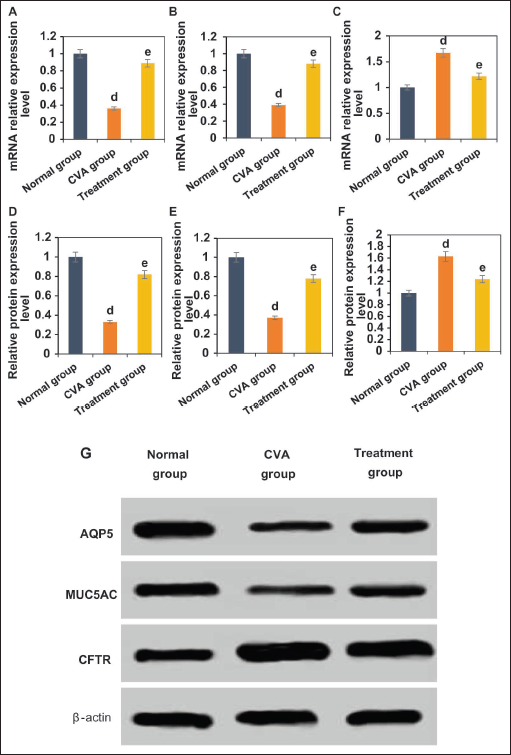
The study collected mRNA and protein expression level levels of AQP5, MUC5AC, and CFTR in mice from the NG, CVA group, and TG (Figure 6). Mice in the CVA group exhibited notably decreased levels of AQP5 and MUC5AC mRNA and protein expression level versus NG, while CFTR mRNA and protein expression level levels were greatly increased (p < .05). Mice in TG showed increased levels of AQP5 and MUC5AC mRNA and protein expression level, and decreased levels of CFTR mRNA and protein expression level versus CVA group (p < .05).
Discussion
This study systematically investigated the therapeutic effects of Mulberry Apricot Coughing Granules on CVA and its underlying mechanisms, focusing on drug component analysis, in vitro inflammation and oxidative stress, and regulation of fluid metabolism in vivo. First, utilizing HPLC and UV spectrophotometry, stable levels of amygdalin and total flavonoids in Mulberry Apricot Coughing Granules were confirmed, providing a chemical basis for its pharmacological activity. Second, in vitro studies demonstrated marked anti-inflammatory effects of Mulberry Apricot Coughing Granules, effectively suppressing IL-4, IL-6, and TNF-α secretion in BEAS-2B cells, with optimal efficacy observed at a concentration of 0.2 mg/mL. Additionally, it alleviated oxidative stress and ferroptosis by reducing ROS and MDA levels, upregulating GPX4 expression level, and downregulating ACSL4 expression level. Lastly, in vivo experiments showed that Mulberry Apricot Coughing Granules improved fluid metabolism in CVA mice, evidenced by increased urine output and water intake, and decreased blood ADH concentration and urine AQP-2 content. These effects may be associated with its regulation of AQP5, MUC5AC, and CFTR expression level, thereby alleviating fluid metabolism disorders induced by CVA. In summary, Mulberry Apricot Coughing Granules significantly reduce levels of inflammatory factors, regulate fluid metabolism, and influence ferroptosis processes.
The study found that in the 0.2, 0.1, and 0.05 mg/mL groups, the concentrations of IL-4, IL-6, and TNF-α were inferior to CG, indicating that Mulberry Apricot Coughing Granules effectively reduce levels of these inflammatory factors (p < .05). Mulberry Apricot Coughing Granules contain herbal ingredients such as mulberry leaf and apricot kernel, which previous research has shown possess anti-inflammatory properties capable of significantly reducing the release of IL-4, IL-6, TNF-α, and other inflammatory factors, thereby aiding in the improvement of respiratory inflammatory responses (Tang et al., 2023). Furthermore, the study found that a concentration of 0.2 mg/mL of Mulberry Apricot Coughing Granules exhibited optimal anti-inflammatory effects, effectively reducing the release of inflammatory factors.
This study innovatively explored the impact of Mulberry Apricot Coughing Granules on fluid metabolism. TG of mice exhibited increased urine and water intake, as well as elevated expression levels of AQP5 and MUC5AC, and decreased levels of blood ADH concentration, urine AQP-2 content, and CFTR expression level relative to the CVA group. TCM attributes properties to mulberry leaves for clearing heat, detoxifying, and diuresis, while apricot kernels are believed to moisten the lungs, resolve phlegm, and alleviate coughing and wheezing. Studies by Zhang et al. (2015) indicated that Mulberry Apricot Coughing Granules can improve phlegm-dampness and heat evils in the body through heat-clearing and phlegm-dispelling actions, thus regulating fluid metabolism and promoting urine production, which aligns with the findings of this study. Modern pharmacology has confirmed that components such as mulberry leaves and apricot kernels have diuretic effects, increasing renal urine excretion (Bolda Mariano et al., 2023; Liu et al., 2021). ADH promotes water reabsorption in renal tubules, reducing urine volume and concentrating urine (Holst & Nejsum, 2019). AQP-2 is an important water channel protein in renal collecting duct epithelial cells, responsible for rapid water transport, thereby influencing urine concentration and dilution (Abdelhamid et al., 2023). Research indicated that asthma patients experience water loss due to respiratory difficulties and airway inflammation, leading the body to compensate by increasing secretion of ADH and AQP-2 (Wang et al., 2021; Yadav et al., 2020). Additionally, increased expression level of AQP5 and MUC5AC was observed in TG of mice. Previous studies confirmed the critical roles of AQP5 and MUC5AC proteins in water regulation and mucus secretion (Zhang et al., 2024). AQP5 functions as a water channel protein facilitating water entry and exit from cells, while MUC5AC is primarily expressed in respiratory epithelial cells and enhances mucus secretion, thereby protecting and lubricating epithelial surfaces. Mutations and dysfunction of CFTR are closely associated with fluid imbalance in various diseases; reduced CFTR expression level helps decrease mucus secretion and viscosity. Studies by Shi et al. (2019) demonstrated that Mulberry Apricot Coughing Granules promote the expression level of these key proteins to enhance fluid secretion and regulate fluid metabolism. However, this research was conducted utilizing animal models, necessitating further studies to confirm its applicability and safety in humans.
The mechanism by which Mulberry Apricot Coughing Granules inhibited ferroptosis revealed that relative to CG, the ROS and MDA levels in RSL3-treated BEAS-2B cells were elevated, while GPX4 mRNA and protein expression level levels were reduced. RSL3 is a compound known to induce ferroptosis by inhibiting GPX4 activity, thereby promoting cell death (Taufani et al., 2023). Studies by Wang et al. (2024) proposed that ferroptosis leads to an increase in lipid peroxidation products such as MDA and 4-hydroxynonenal, thereby increasing oxidative stress levels in the airways, consistent with the findings of this study. Further investigation into the mechanism of Mulberry Apricot Coughing Granules in treating CVA revealed that the RSL3 + 0.2 mg/mL group exhibited notably lower ROS and MDA levels versus the RSL3 group. Moreover, treatment with 0.2 mg/mL Mulberry Apricot Coughing Granules extract markedly increased GPX4 mRNA and protein expression level levels while significantly decreasing ACSL4 expression level. Studies confirmed that ACSL4 is a crucial enzyme that promotes lipid peroxidation and ferroptosis, whereby its increased expression level exacerbates membrane lipid peroxidation and facilitates the onset of ferroptosis (Li et al., 2023; Wu et al., 2024). Thus, Mulberry Apricot Coughing Granules may alleviate RSL3-induced ferroptosis by upregulating GPX4 and reducing ACSL4 expression level, suggesting a potential therapeutic effect on respiratory diseases such as CVA. However, the specific molecular mechanisms of action of Mulberry Apricot Coughing Granules and their effects in complex in vivo environments still require further investigation.
In summary, Mulberry Apricot Coughing Granules, as a TCM formulation, holds promise as a potential therapeutic agent for treating respiratory diseases such as chronic bronchitis, owing to its multifaceted pharmacological effects. Particularly, its roles in modulating inflammation, improving fluid metabolism, and alleviating oxidative stress are important. Future research should further explore the therapeutic mechanisms of Mulberry Apricot Coughing Granules in various respiratory disease models, especially its modulation of ferroptosis and oxidative stress. Additionally, clinical trials will be crucial to validate its efficacy and safety in humans, providing robust scientific evidence for its clinical application.
Conclusion
In conclusion, Mulberry Apricot Coughing Granules improved oxidative stress and lipid peroxidation by upregulating the antioxidant protein GPX4 and downregulating ACSL4 expression level, thereby inhibiting ferroptosis and ameliorating inflammatory responses. Moreover, Mulberry Apricot Coughing Granules enhanced the expression levels of AQP5 and MUC5AC while decreasing CFTR expression level, thereby improving abnormal fluid metabolism in CVA. These findings suggest that Mulberry Apricot Coughing Granules may exert therapeutic effects on CVA through multiple mechanisms. This study provides a theoretical basis for further exploration of Mulberry Apricot Coughing Granules in the treatment of CVA. Although the animal experiment results suggest the potential efficacy of Mulberry Apricot Coughing Granules in alleviating inflammation, oxidative stress, and ferroptosis, the current study still lacks supporting clinical trial data. Animal models provide valuable preliminary evidence; however, due to the complexity of human physiological and pathological conditions, the results from animal studies may not be directly translatable to clinical outcomes. Therefore, translating these experimental findings into clinical applications requires further clinical research and validation. Future studies should design larger-scale clinical trials to assess the safety, efficacy, and long-term effects of Mulberry Apricot Coughing Granules, as well as explore its indications in different patient populations. Additionally, further research is needed to investigate its pharmacological mechanisms, dose–response relationships, and potential side effects, thereby providing more robust evidence for its clinical use.
Footnotes
Abbreviations
ADH: Anti-diuretic hormone; AQP-2: Aquaporin-2; AQP5: Aquaporin-5; CFTR: Cystic fibrosis transmembrane conductance regulator; CG: Control group; CVA: Cough variant asthma; ECL: Enhanced chemiluminescence; ELISA: Enzyme-linked immunosorbent assay; GPX4: Glutathione peroxidase 4; HPLC: High-performance liquid chromatography; IL: Interleukin; MDA: Malondialdehyde; MUC5AC: Mucin 5AC; NG: Normal group; ROS: Reactive oxygen species; TCM: Traditional Chinese medicine; TG: Treatment group; TNF: Tumor necrosis factor.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All animal experiments were approved by the Ethics Committee of the Fourth Clinical Medical College of Xinjiang Medical University (Xinjiang, China). All animal experiments were carried out in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Project Approval Number: 82060794).
Informed Consent
Not Applicable.