Abstract
Background
The turmeric rhizome yields a medicinal compound called curcumin. Curcumin’s anticancer methods mostly involve blocking cell invasion and proliferation, controlling gene expression, preventing angiogenesis, triggering apoptosis, and inducing ferroptosis. A brand-new nonapoptotic kind of planned cell death is called ferroptosis. Ferroptosis can be initiated through two major pathways: the extrinsic or transporter-dependent pathway, and the intrinsic or enzyme-regulated pathway. It is unknown, therefore, how curcumin and its derivatives prevent cancer by controlling ferroptosis.
Purpose
Through this review, we concentrate on the possible anticancer mechanisms of curcumin by means of its effect on ferroptosis; the regulation of iron metabolism, the synthesis of reactive oxygen species, and the targeting of antioxidant pathways represent a few of these processes.
Methodology
Retrieve English literature of curcumin and its derivatives in the treatment of tumor by ferroptosis from databases such as Web of Science, etc.
Conclusion
This review provides a rationale for the clinical use of curcumin and its derivatives as anticancer agents.
Introduction
Curcumin
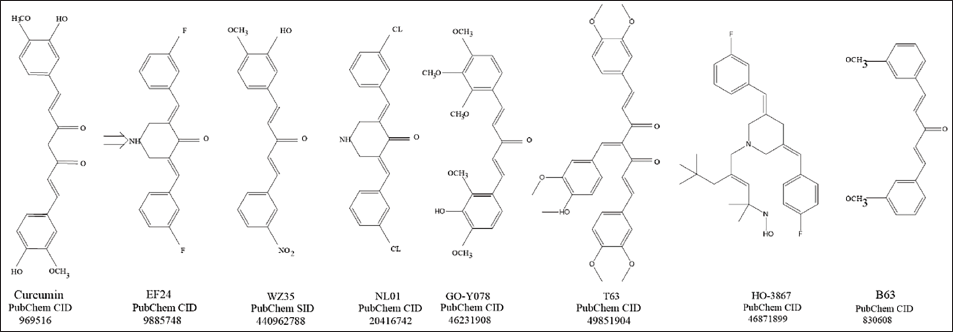
Compared to chemical drugs, plant species are cheap and easily available, safe and effective, and have few adverse effects, which makes them potentially valuable in the development of new antitumor drugs (Imran et al., 2018). Because curcumin is a lipophilic molecule, it may permeate the cell membrane and function in the nucleus, mitochondria, and endoplasmic reticulum (Jaruga et al., 1998; Sharma et al., 2022). Remarkably, curcumin inhibits the synthesis of reactive oxygen species (ROS) at low concentrations while stimulating the formation of ROS at high ones (Chen et al., 2005). Curcumin has been proven in multiple investigations to inhibit the spread, invasion, and migration of cancerous cells, inhibit angiogenesis, regulate gene expression, induce apoptosis of tumor cells through multiple mechanisms (Kunnumakkara et al., 2017), and sensitize and reverse drug resistance to chemical drugs (Abadi et al., 2022). According to recent research, curcumin can inhibit colorectal and breast cancers by modulating ferroptosis (Cao et al., 2022; Chen, Tan et al., 2023). Despite all of these benefits, curcumin’s restricted water solubility also contributes to its low chemical stability (Nagahama et al., 2016). Since curcumin is hydrophobic, it tends to pass through cell membranes, thereby decreasing its availability in the cytoplasm (Barry et al., 2009). To increase curcumin’s overall anticancer activity, researchers are working to enhance its bioavailability and pharmacological properties through various approaches, including the development of novel curcumin derivatives (Kanai et al., 2012; Mahal et al., 2017) (Figure 1).
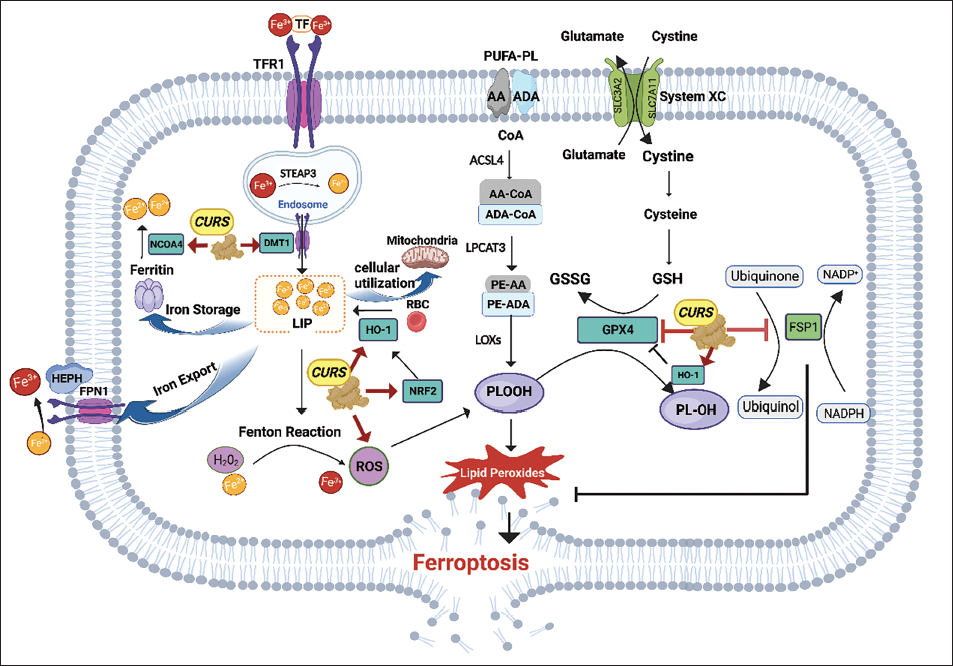
Ferroptosis and Its Mechanism of Occurrence
In 2022, Hanahan (2022) proposed that malignant tumor features include persistent proliferative signals, genomic instability, immune escape, and programmed cell death (PCD) resistance. PCD is characterized as cell death resulting from the activation of one or more signaling modules. Both pathological and physiological conditions depend on the maintenance of organismal homeostasis through these signaling modules. Disruption of PCD regulatory mechanisms will cause tumor cell survival and unlimited proliferation (Galluzzi et al., 2018; Green, 2017). This iron-dependent, peroxide-driven kind of PCD was named ferroptosis by Dixon et al. in 2012; it is associated with several in vivo metabolic disorders. The morphology of ferroptosis cells showed: cell membrane disruption; formation of double-membrane vesicles; mitochondria crumpling; volume reduction; increased density of bilayer membranes; inner cristae were blurred, reduced, or disappeared; complete nuclear membrane; no DNA fragmentation (Friedmann Angeli et al., 2014; Li, Cao et al., 2020); and numerous divalent iron ions were seen in the endoplasmic reticulum and mitochondria (Dixon et al., 2012, 2014). Biochemically, there was a rise in the concentration of divalent iron ions, a substantial buildup of lipid peroxides and ROS, and a reduction in the activity of glutathione peroxidase 4 (GPX4) among other things (Li, Cao et al., 2020; Wang, Liu et al., 2020). The executing molecule for the onset of ferroptosis is lipid-based ROS/phospholipid hydroperoxide (PLOOH). Surrounding the synthesis and metabolism of PLOOH, oxidative damage and antioxidant protection are at the core of ferroptosis regulatory mechanisms. Under normal conditions, the two mechanisms maintain a dynamic balance to ensure cell metabolism. Ferroptosis can be induced when antioxidant protection is reduced and oxidative damage is enhanced. Lipid peroxidation and an accumulation of iron are two crucial mechanisms in the occurrence of ferroptosis, which combined cause oxidative degradation of the cell membranes. Cellular antioxidant mechanisms can reduce PLOOH and inhibit oxidative damage, thus preventing the occurrence of ferroptosis. The antioxidant mechanism of ferroptosis mainly includes the classical GPX4-dependent pathway and the non-GPX4-dependent pathway. Thus, lipid peroxidation, antioxidant defense, and intracellular iron metabolism are associated with the incidence of ferroptosis (Jiang et al., 2021). Because tumor cells have larger concentrations of iron ions and engage in strong metabolic activity that raises ROS levels, malignant cells are more prone to ferroptosis than ordinary cells (Li & Li, 2020).
The Extrinsic Pathway of Curcumin-induced Ferroptosis for Cancer Treatment
Induction of Ferroptosis through Lipid Peroxidation Reactions
Via the activation of many cell death pathways, including necrosis, apoptosis, autophagy, and ferroptosis, high ROS levels can offer a route for cancer treatment. Consequently, one of the main anticancer methods is to increase ROS (Gorrini et al., 2013). Elevated levels of ROS are fundamental for the occurrence of ferroptosis. Curcumin induces ROS production in cervical, colorectal, breast, and prostate cancer fibroblasts ultimately leading to cell death (Li et al., 2021; Tong et al., 2020; Wang et al., 2022; Wang, Wu et al., 2020; Zeng et al., 2020). Curcumin causes ROS generation, which inhibits glioblastoma growth, sphere-forming capacity, and colony-forming capability of glioblastoma stem cells (Gersey et al., 2017). A new 4-arylidene curcumin analogueT63 activates ROS and thus inhibits the proliferation of lung cancer cells (Liu et al., 2012). Curcumin is shown to decrease human gastric cancer cell (hGCC) proliferation, colony formation, and migration in a dose- and time-dependent mode in research on gastric cancer cells. Curcumin at high concentrations (≥20 µmol/L) elevated ROS levels, caused damage to mitochondria, DNA damage, and destroyed hGCC (Tong et al., 2020). Liang et al. (2014) also found that 20 µmol/L curcumin induced ROS production in human gastric cancer BGC-823 cells. Through a ROS-ER stress-mediated pathway, the curcumin derivative WZ35 demonstrated beneficial effects on colon cancer cells both in vitro and in vivo (Zhang et al., 2017). Another curcumin derivative HO-3867 induced ROS accumulation in P53 wild-type non-small-cell lung cancer (NSCLC) cells. Wu et al. (2023) assessed the involvement of divalent metal transporter 1 (DMT1) in the HO-3867-triggered increase in ROS production in NSCLC cells. The regulation of ferroptosis in hepatocellular carcinoma (HCC) cells by lactate is confirmed by both in vitro and in vivo studies. Zhao et al. (2020) found that liver cancer cells rich in lactate are more resistant to ferroptotic damage and that the uptake of lactate by the monocarboxylate transporter 1 (MCT1) can increase the production of adenosine triphosphate (ATP) in HCC cells and turn off AMP-activated protein kinase (AMPK), an energy sensor. This, in turn, can lead to the upregulation of sterol regulatory element-binding protein 1 (SREBP1) and the downstream of stearoyl-coenzyme A (CoA) desaturase 1 (SCD1), which, in turn, increases the synthesis of monounsaturated fatty acids that can fend against ferroptotic damage. Additionally, via activating AMPK to decrease SCD1, blocking the hydroxycarboxylic acid receptor 1 (HCAR1)/MCT1 promotes ferroptosis. This may contribute to the vulnerability to ferroptosis together with its effect on acyl-coenzyme A synthetase 4 (ACSL4). Shi et al. (2023) demonstrated that the curcumin derivative NL01 caused ovarian cancer cells to undergo ferroptosis by inhibiting lactate absorption by activating the AMPK system and reducing the expression of HCAR1/MCT1. The transporter protein solute carrier family 1 member 5 (SLC1A5) is necessary for glutamate absorption, and glutamine metabolism increases the accumulation of oxidizable cholesterol in cells, which leads to cellular ferroptosis (Xu et al., 2019). Cao et al.’s (2022) research indicates that curcumin has anticancer properties against breast carcinoma via raising intracellular Fe2+ levels, ROS levels, and the buildup of malondialdehyde (MDA), the result of lipid peroxidation. It might trigger ferroptosis in vitro and in vivo by upregulating SLC1A5 expression.
Curcumin Induces Ferroptosis by Regulating Iron Metabolism
Tumor cells require iron more than normal cells during growth (Manz et al., 2016). Numerous investigations have shown that TFR expression in malignancies is quite high (Aulbert et al., 1980; Shterman et al., 1991; Sutherland et al., 1981), indicating that cancer cells are more prone to undergo ferroptosis. The rate-limiting enzyme heme oxygenase 1 (HO-1) breaks down heme into ferrous iron, carbon monoxide, and biliverdin/bilirubin (Chiang et al., 2018). The high expression of HO-1 promotes many types of cellular cancer death, including ferroptosis (Kwon et al., 2015; Loboda et al., 2016, 2015; Nitti et al., 2017). Normal physiological activation of HO-1 results in cytoprotective actions and ROS scavenging; overactivation of HO-1, on the other hand, leads to ferroptosis by raising ROS concentrations and iron levels (Chiang et al., 2018; Jomova & Valko, 2011). One efficient promoter of HO-1 is curcumin. In addition to directly activating HO-1, curcumin also activates Nrf2, which increases the expression of HO-1 (Balogun et al., 2003; Scapagnini et al., 2002). Curcumin upregulates HO-1 expression through the generation of ROS, p38 activation, and phosphatase inhibition in various HCC cells (McNally et al., 2007). In another study, HO-1 gene expression levels were significantly elevated in all three curcumin-treated HCC cell lines (PLC, KMCH, and Huh7) (Liu et al., 2023). Curcumin has been shown by Li, Zhang et al. (2020) to induce ferroptosis in breast cancer cells by upregulating the expression of HO-1. In their tests, they discovered that curcumin drastically reduced glutathione levels while causing a buildup of intracellular iron, ROS, lipid peroxides, and MDA. Consoli et al. found that curcumin (5–50 µM) dose-dependently reduced cell viability in triple-negative breast cancers and that the cells exhibited ferroptosis. Curcumin-treated breast cancer cells demonstrated enhanced HO-1 protein expression levels, according to the enzyme-linked immunoassay (ELISA) test (Consoli et al., 2022). Chen, Li et al. (2023) discovered that curcumin upregulated HO-1 expression, which, in turn, caused ferroptosis in follicular thyroid carcinoma (FTC). They discovered that curcumin raised iron and MDA levels, lowered glutathione (GSH) levels, and enhanced HO-1 protein expression in FTC-133 and FTC-238 cells in a dose-dependent manner after treating FTC cells with 0, 10, and 20 µm curcumin for 24 hours. In the Co-IP experiment in FTC cells, there was a negative regulatory interaction between HO-1 and GPX4, and siGPX4 transfection reversed the inhibitory effect of siHO-1 on curcumin-induced ferroptosis. The same manifestation occurred in the action of EF24, a synthetic analog of curcumin, on osteosarcoma cells. In osteosarcoma cells, EF24 dose-dependently increased the expression of HO-1, which led to ferroptosis. EF24 significantly increased MDA level, ROS level, and intracellular ferri ion level; these effects were significantly attenuated by ferrostatin-1. HO-1 knockdown attenuated EF24-induced cytotoxicity (Lin et al., 2021). GO-Y078 is a newly developed synthetic derivative that exhibits greater oral absorption and anticancer efficacy in comparison to curcumin. GO-Y078 induced HO-1 gene upregulation, thereby exerting an inhibitory effect on oral squamous cell carcinoma (Chien et al., 2022). GO-Y078 transcriptionally induced upregulation of the HO-1 gene by increasing the AP-1 DNA-binding activity, which was initiated by activation of the p38/JNK1/2 pathways. Another curcumin derivative, HO-3867, can increase iron accumulation in NSCLC cells, thereby inducing ferroptosis. In NSCLC cells, treatment with HO-3867 led to an increase in DMT1 mRNA levels, and protein blotting further validated HO-3867’s dose-dependent upregulation of DMT1 (Wu et al., 2023).
The Intrinsic Pathway of Curcumin-induced Ferroptosis for Cancer Treatment
Curcumin Induces Ferroptosis through the Targeted Antioxidant Pathway
Under normal conditions, the two mechanisms of cellular oxidation and antioxidation maintain a dynamic balance to ensure cellular metabolism. When the antioxidant protection function is reduced and oxidative damage is enhanced, ferroptosis can be induced in cells. Chen, Tan et al. (2023) discovered that curcumin-treated HCT-8 cells had considerably lower levels of GSH, SLC7A11, and GPX4, which caused ferroptosis and prevented colon cancer cells from proliferating. FSP-1 is a GPX-4-independent ferroptosis inhibitor. By collaboratively suppressing GPX-4 and FSP-1, two important negative regulators of the ferroptosis defense system, curcumin and Andrographis, activated ferroptosis. When administered together, the antitumor growth effects were much greater than when administered separately. This opens new avenues for overcoming chemoresistance in colon cancer (Miyazaki et al., 2023). The curcumin analog EF24-induced ferroptosis in osteosarcoma cells inhibited GPX4 levels through upregulation of HO-1 expression, thereby making an entrance as a possible medication for osteosarcoma sufferers (Lin et al., 2021). The ovarian cancer cells, Anglne and HO8910PM, were shown to be 13-fold more susceptible to growth inhibition by the curcumin derivative NL01. NL01 promoted the downregulation of GPX4 expression and inhibited the defense pathway of ferroptosis. The ovarian cancer cell lines’ growth suppression induced by NL01 was reversed by the addition of ferrostatin-1, an inhibitor of ferroptosis. Shi et al. (2023) found that NL01 was able to reduce the expression of HCAR1/MCT1 and activate the AMPK signaling pathway, which, in turn, induced cellular ferroptosis via the SREBP1 pathway. It has been demonstrated that ALZ003, another curcumin analog, disrupts GPX4-mediated redox equilibrium to promote androgen receptor ubiquitination and inhibit the development of temozolomide-resistant glioblastoma in both in vitro and in vivo studies (Chen et al., 2020). In a study on lung cancer stem cells, curcumin caused ferroptosis in A549 CD133 cells by interfering with the GSH-GPX4 and FSP1-CoQ10-NADH procedures, which inhibited self-renewal (Zhou et al., 2023). The curcumin derivative HO-3867 induced a dose-dependent decrease in GPX4 mRNA levels in NSCLC cells thereby inducing ferroptosis (Wu et al., 2023). The thioredoxin/thioredoxin reductase (TrxR) process enables tumor cells to withstand cell death caused by oxidative stress (Kim et al., 2005). A novel analog of curcumin, B63, can increase cellular ROS levels by targeting TrxR1 protein leading to cell death in 5-fluorouracil-resistant gastric cancer cells and has been validated in in vivo experiments, thus providing a new approach to reverse 5-fluorouracil resistance (Chen et al., 2019). Under situations of cancer stress, the antioxidant protein lipocalin 2 (LCN2) can be elevated. Curcumin has been demonstrated to dramatically reduce LCN2 expression at the protein and mRNA levels (He et al., 2021; Wan et al., 2016). Liu et al. (2023) found that curcumin inhibited LCN2 levels in HCC cell lines. This implies that curcumin may control ferroptosis with LCN2.
Induction of Ferroptosis by Autophagy and lncRNA
Prior research has demonstrated that autophagy causes transferrin receptor 1 (TFR1) expression and ferritin degradation, which results in iron-dependent ferroptosis (Park & Chung, 2019). A growing body of research indicates that ferroptosis is significantly influenced by the autophagy process (Kang & Tang, 2017). Tang et al. (2021) demonstrated that curcumin-activated and ferroptosis in A549 and H1299 cells could be inhibited by pretreatment with an autophagosome lysosomal fusion inhibitor or by knockdown of the autophagy-related gene Beclin1, indicating that autophagy pathway activation can cause tumor ferroptosis in tumor cells. Through ferritin phagocytosis mediated by nuclear receptor coactivator, autophagy can provide free iron for ferroptosis, and it can also regulate intracellular iron homeostasis by degrading ferritin and promoting ROS production, leading to ferroptosis (Zhou et al., 2020). Zhang et al. (2022) found that the long non-coding RNA H19 (lncRNA H19) was significantly downregulated in lung cancer cells treated with curcumenol. Mechanistically, they showed that lncRNA H19 functioned as a competing endogenous RNA to bind to miR-19b-3p, thereby increasing the expression of ferroptosis negative regulatory components (SLC7A11, FTH1, NRF2, and GPX4).
Clinical Studies of Curcumin in Cancer Patients
Researchers have carried out several clinical research on the therapeutic benefits of curcumin in cancer patients, according to the findings of various preclinical investigations. A randomized clinical trial evaluating curcumin to improve the treatment outcome of multiple myeloma patients who were not suitable for transplantation showed that curcumin (8 g/d × 28 d) in combination with an melphalan and prednisone (MP) regimen was effective in improving the overall remission rate of patients with first-treatment multiple myeloma (75% vs. 33.3%, p = 0.009) (Santosa et al., 2022). A randomly assigned, double-blinded parallel-controlled clinical trial evaluated the safety and efficiency of the intravenous infusion of curcumin (300 mg once weekly) in addition to paclitaxel for the treatment of patients with metastatic and advanced breast cancer. After 12 weeks of therapy, in terms of overall response rate (ORR) and physical function, the combined use of curcumin and paclitaxel performed superior than the combination of paclitaxel and placebo (Saghatelyan et al., 2020).
Conclusion
Ferroptosis is a novel style of mortality. On the other hand, tumor cells that are constantly under oxidative stress have an array of defense mechanisms to withstand this stress, which contributes to tumor cell treatment resistance. Viswanathan et al. (2017) suggested that ferroptosis is expected to surpass the traditional pathway based on drug-resistant mutations as a new approach to reverse drug resistance in tumor cells.
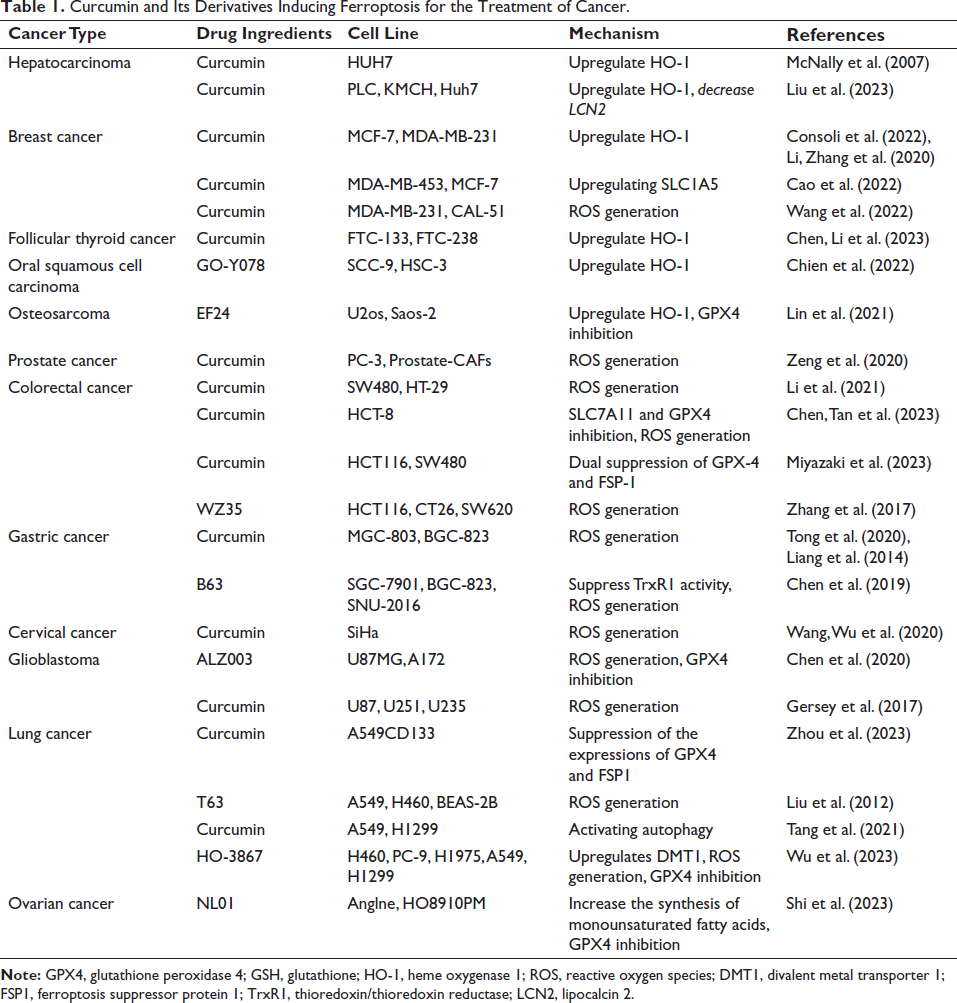
As a ferroptosis inducer, researchers have demonstrated that curcumin inhibits the development of tumors via changing ferroptosis in a variety of malignancies (Table 1). We compiled the study data and identified the main mechanisms by which curcumin suppresses tumors through ferroptosis (Figure 2). However, because of its hydrophobicity, rapid metabolism and rapid clearance by the liver limit the bioavailability of curcumin. More curcumin derivatives have been created by researchers to get around this restriction and boost curcumin’s anticancer effectiveness and bioavailability. Curcumin has also been delivered using a variety of methods, including liposomal curcumin, curcumin nanoparticles, and curcumin phospholipid compounds. These approaches may help curcumin overcome its drawbacks and increase its efficiency in cancer treatment; nevertheless, to improve these derivatives as well as assess their therapeutic effectiveness, more study is necessary.
Curcumin and Its Derivatives Inducing Ferroptosis for the Treatment of Cancer.
In addition to being extensively supported by preclinical research, curcumin has also been shown to have anticancer properties in several clinical trials including tumor patients. Still, some clinical studies have shown contradictory results (Choi et al., 2019; Gunther et al., 2022; Passildas-Jahanmohan et al., 2021), which were considered to be connected to curcumin’s low bioavailability, the small sample sizes, and the short follow-up time. More clinical trials should be conducted to provide more convincing evidence for curcumin and its derivatives to be effective oncology clinical agents.
However, the mechanisms of ferroptosis in malignant tumors still leave many questions to be explored, for example, what precise mechanism causes cell death when polyunsaturated fatty acid peroxidation occurs? and what is the correlation with the efficacy of immunotherapy in tumors? Future studies on ferroptosis in malignant tumors need to identify as many mechanisms and molecules that regulate ferroptosis as possible to provide effective targets for tumor therapy or reversal of drug resistance.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
This work was supported by the National Natural Science Foundation of China (No. 82173056) and the Beijing Natural Science Foundation Program (No. 7222032).
