Abstract
Background
Cerebral palsy (CP) is a serious childhood disease characterized by changes in inflammatory responses and synaptic plasticity. Recent evidence highlighted the anti-inflammatory properties of β-sitosterol and that the nerve growth factor (NGF) regulates neuronal and synaptic plasticity.
Objectives
This study aimed to explore the mechanism by which β-sitosterol regulates inflammatory homeostasis through NGF, thereby improving synaptic plasticity in high-risk children with CP.
Materials and Methods
A rat model of CP was constructed, and the animals were divided into a model group, β-sitosterol group, β-sitosterol group + NGF group, and β-sitosterol + K252α group. Following treatment, the Morris water maze and Bederson score were used to evaluate the behavioral performance of rats, Western blot analysis was used to detect the expression of NGF and hippocampal protein, and enzyme-linked immunosorbent assay (ELISA) was used to measure inflammatory factor levels. Additionally, immunofluorescence examined the expression of synaptophysin in rat brains, and the brain tissue was observed by hematoxylin and eosin (HE) staining.
Results
The CP rat model was successfully constructed. Of note, β-sitosterol treatment improved the synaptic plasticity of the CP rats with decreased Abnormal Involuntary Movement Scale (AIMS) and Bederson scores and a shorter latent period. Moreover, β-sitosterol inhibited the production of pro-inflammatory factors and increased the number of synapses in the hippocampus of rats while increasing the expression of brain-derived neurotrophic factor (BDNF), SYN, N-methyl
Conclusion
β-Sitosterol improves synaptic plasticity in high-risk children with CP and alleviates inflammation and nerve cell apoptosis through up-regulation of NGF expression.
Introduction
Nervous system diseases, exemplified by cerebral palsy (CP), not only impact the motor and cognitive functions of children but also significantly constrain their language abilities (Gaebler-Spira & Green, 2020). Synaptic plasticity, facilitating information transmission between neurons, is crucial to children’s neurological development (Goto, 2022). Despite recent progress in medical research, the pathogenesis of CP remains incompletely understood, and effective treatments are still lacking. Consequently, exploring novel treatments has become the focal point of current research.
β-Sitosterol, derived from soybean, corn, and other vegetable oils, exhibits anti-inflammatory, anti-oxidant, and anti-hyperlipidemia effects (Bao et al., 2022). Concurrently, it regulates immunity and inhibits tumor development with various pharmacological effects (Wang, Wang, et al., 2023). Studies indicate that β-sitosterol contributes to the improvement in the meridian as well as various brain and neurological diseases (Khan et al., 2022). Its cholesterol-lowering, anti-inflammatory, and tissue-repairing properties make it valuable not only for preventing cancer and neurological conditions such as Parkinson’s disease (Prabahar et al., 2023; Schaefer et al., 2021) but also for treating cardiovascular diseases and skin disorders like ulcers, eczema, and psoriasis (Huang et al., 2023). The promising pharmacological effects of β-sitosterol on neurological function and inflammation in degenerative diseases have garnered significant attention. β-Sitosterol can diminish the inflammatory response and enhance synaptic plasticity by inhibiting the production of inflammatory mediators such as prostaglandins (PGs), leukotrienes (LTs), and matrix proteins (MPs), or by promoting the secretion of nerve growth factor (NGF) (Ribeiro et al., 2019). Additionally, β-sitosterol improves synaptic plasticity by scavenging numerous free radicals and mitigating oxidative stress (Adebiyi et al., 2019). Therefore, through the synergy of multiple mechanisms of action, β-sitosterol holds promise in improving synaptic plasticity in children with CP. However, further research is required to validate the specific mechanism.
In brain diseases, the expression of NGF often exhibits abnormal increases (Otani et al., 2019). Studies reveal that by upregulating pathways such as mitogen-activated protein kinase (MAPK)/extracellular signal-regulated kinase (ERK), phosphoinositide 3-kinase (PI3K)/protein kinase B (Akt), phospholipase C gamma (PLCγ), and NGF can not only promote cell anti-apoptosis, safeguard neurons from stress-induced damage, but also facilitate neuronal growth and synaptic connections, thereby influencing cognitive functions such as memory (Wang, Fan, et al., 2023). NGF is also known to modulate cell movement and neuronal growth through the upregulation of signaling pathways like Rho GTPases, calcium/calmodulin-dependent protein kinase (CAMK), and nuclear factor kappa B (NF-κB) (Dai et al., 2020). Furthermore, activating calcium ion channels like voltage-dependent calcium channels (VDCC), receptor-operated channels (ROC), and N-methyl-D-aspartate (NMDA) receptors is implicated in synaptic plasticity, regulating the learning and memory process (Gulyaeva et al., 2023). It was reported that NGF inhibits the expression of inflammatory mediators, including tumor necrosis factor alpha (TNF-α), COX-2, and IL-1β, thereby reducing the production of inflammatory substances like prostaglandin E2 (PGE2), prostaglandin D2 (PGD2), and thromboxane A2 (TXA2), and mitigating the inflammatory response (Mozafarihashjin et al., 2022). NGF influences chemokines and cell adhesion molecules such as CXCL12, ICAM, and VCAM, participating in the migration of leukocytes and the inflammatory response (Minnone et al., 2017). Although several studies have explored the use of β-sitosterol in other neurological diseases, little is known about the interaction between NGF and β-sitosterol in CP and its impact on synaptic plasticity.
This study aims to delve into the mechanism by which β-sitosterol enhances synaptic plasticity in high-risk children with CP through the regulation of inflammatory homeostasis via NGF, with particular attention to the hypothesis that β-sitosterol can improve neurodevelopment and synaptic plasticity in children by regulating the NGF signaling pathway on inflammatory homeostasis. The objective is to provide a novel therapeutic strategy for enhancing synaptic plasticity in clinical medicine in high-risk children with CP.
Materials and Methods
Reagents and Instruments
β-Sitosterol was provided by Chengdu Zhichun Herbal Biotechnology Co., Ltd. (CAS: 83-46-5), NGF by Wuhan Maxi Biotechnology Co., Ltd. (CAS: GS12987), and K252α by Yisheng Biotechnology Co., Ltd., Shanghai (CAS: 99533-80-9). Twenty-four healthy male Sprague–Dawley (SD) rats, which weighed 350 ± 12.5 g, were obtained from Tianqin Biotechnology Co., Ltd., BDNF antibody was obtained from Wuhan Boute Biotechnology Co., Ltd. (CAS: orb318730), and SYN antibody was obtained from Shenzhen Xinbosheng Biotechnology Co., Ltd. (CAS: GTX82594). Other materials included N-methyl
Construction of CP Rat Model
Twenty-four healthy male SD rats were raised at a constant temperature of 18°C–26°C, avoiding light and keeping sufficient water and feed, six were taken as the blank group, and the others were subjected to operation under anesthesia by inhalation with 3.0% isoflurane. The 7-day-old SD rats were fixed post-anesthesia, while the isoflurane concentration was adjusted to a 1.5% volume. Following complete disinfection of the neck skin, a mid-neck incision exposed the common artery in the left neck, which was ligated twice at different locations and then cut, with subsequent skin suturing. One hour post-surgery, rats were placed in a closed hypoxia box (92% nitrogen, 8% oxygen) for 2.5 h to simulate hypoxic conditions. Successful modeling was assessed 24 h post-surgery based on abnormal behavioral suspension test results, abnormal involuntary movements, and a Bederson score of ≥1 point, confirming CP modeling. All rats were successfully modeled. Successfully modeled rats were randomly divided into four groups (n = 6 each): model group, β-sitosterol group, β-sitosterol + NGF group, and β-sitosterol + K252α group. The β-sitosterol group received intraperitoneal injections of 50 mg/kg, while the β-sitosterol + NGF group received 5 mg/kg NGF intraperitoneally, based on the β-sitosterol intervention. The β-sitosterol + K252α group was intraperitoneally injected with 3 mg/kg NGF inhibitor K252α, based on β-sitosterol, once a day for 14 days.
Morris Water Maze Test
Four equidistant points were marked in a pool (150 cm diameter × 60 cm height × 40 cm water depth). A rectangular platform (length × width: 12 × 30 cm) was placed under the pool. Rats were placed from the marked points facing the pool wall, and their escape latency was recorded when they found the submerged platform. Tests were conducted five times daily for five consecutive days. On the 6th day, the hidden platform was removed, and the number of times the rat crossed it was recorded after removing the platform quadrant. The platform position changed daily to evaluate memory 3 days post-platform. After each change, the above steps were repeated, and the rats were observed again. When rats have severe pathological conditions (inability to eat, difficulty breathing, and so on), excessive pain or disease symptoms, abnormal behavior or severe fatigue, or irreversible damage, they should be physically tested or euthanized.
Animal Behavior Evaluation Methods
Suspension Experiment
The rat’s falling time and score were recorded with both rat forelimbs at a height of 45 cm from the ground and a horizontally placed wooden stick (diameter of 0.1 cm). For falling time ≤10 s, 10–30 s, 30 s–2 m, and 2–5 min, scores are 1, 2, 3, and 4, respectively.
Involuntary Movements
Involuntary movements include tremors, athetosis, twitching, spasmodic twisting, and dancing. With normal conditions, 0 points, the presence of any of the above symptoms gives 1 point, and the condition is defined as abnormal.
Bederson Rating
The neurological function score of rats was classified into 4 levels using the Bederson score. Level 0 (0 points): no neurological deficit; Level I (1 point): reduced rat activity and insensitivity to external stimulation; Level II (2 points): one forelimb or side of the rat’s hind limbs is paralyzed; Level III (3 points): the rat rotates to the paralyzed side.
Postural Abnormality Scoring
Abnormal postures include (a) the body weight is mainly supported by three limbs (2 points); (b) the body weight is mainly supported by two limbs (3 points); (c) the rat is unable to stand on its limbs (5 points).
Muscle Tone Measurement
Scoring criteria: 4 points, increased muscle hardness of the limbs, limited passive joint movement; 4 points, decreased muscle hardness of the limbs, increased passive joint movement angle, muscle weakness, excessive joint extension; 2 points, increased muscle tension changes.
Walking Activities
The rats were placed in a closed, clean, and transparent container, and the rat’s tail was lightly touched, when we observed the rat’s activity status within 3 min. Scoring criteria: 0 points for motionless, 1 point for asymmetric limb movement and lameness, 2 points for lameness but coordinated movements, and 3 points for crawling quickly.
Hind Paw Angle
The rat’s hind paws were dipped in black ink and allowed to walk in a 30 × 6 × 10 cm cylindrical glass channel with a long strip of white paper at the bottom. Each rat was tested three times (the white paper was replaced in each test), and the angle between the line connecting the middle toe and the center of the heel was tested. The scoring criteria were 0 points for an angle ≤60° and 1 point for >60°.
Western Blot Analysis
In order to reduce the pain of rats, the rats were executed by decapitation. Then, the right hippocampus samples of rats were obtained and subjected to protein extraction, gel preparation, loading, electrophoresis, transfer, and blocking. The blots were then incubated with antibodies (1:200, BDNF, SYN, NR2B, Caspase-3, NGF, β-actin, GAPDH) at 4°C overnight and anti-rabbit secondary antibodies (1:10,000) for 2 h, and results were analyzed using enhanced chemiluminescence and analyzed by software.
Transmission Electron Microscope
The left hippocampus tissue was fixed with 1% osmium tetroxide for 1 h, dehydrated with concentrations of ethanol, embedded, sectioned, and comprehensively stained with uranyl acetate before observation with a transmission electron microscope.
Enzyme-linked Immunosorbent Assay (ELISA)
Take 30 mg of cortical tissue from the left side of the rat brain, homogenize, centrifuge at 4,500 r/min for 15 min, and take the supernatant. Strictly refer to the ELISA kit to detect the levels of each inflammatory factor.
Immunofluorescence
Following three washes with physiological saline, the rat brain tissues were fixed with 4% paraformaldehyde for 2 h, dehydrated, rendered transparent, wax immersed, and embedded. Subsequently, the tissues were sliced into sections (5 µm), incubated in an incubator at 37°C for 30 min, and dewaxed afterward. Antigen retrieval solution (potential of hydrogen (pH) 9.0) was added, and the slices were heated on medium heat for 20 min. Afterward, the sections were left for 10 min and incubated with 50 g L−1 goat serum for 1 h and with rabbit anti-rat synaptophysin (1:100) overnight at 4°C. Following three washes with antigen retrieval solution, the sections were incubated with goat anti-mouse immunoglobulin G (IgG) secondary antibodies (1:150) at 37°C for 1 h. Finally, the slides were sealed for observation using fluorescence microscopy.
Determination of Malondialdehyde (MDA) Content and Superoxide Dismutase (SOD) Activity in Rat Brain Tissue
Hippocampal and occipital lobe tissues were separated from the rat brain, ground into a homogenate, and centrifuged at 4,500 r/min for 25 min. The clear liquid was mixed with thiobarbituric acid (TBA) reagent and then heated in a 95°C water bath for 40 min. After centrifugation at 3,500 r/min for 10 min, the supernatant was obtained. The absorbance value of each tube was measured at a wavelength of 532 nm using a spectrophotometer.
Hematoxylin and Eosin (HE) Staining
Brain tissue cells fixed in 4% paraformaldehyde were rinsed, dehydrated with graded ethanol, cleared with xylene, and embedded in paraffin. The resulting paraffins were sectioned into 3 µm thick sections and subsequently dewaxed. Complete staining with the hematoxylin-eosin staining solution was performed, followed by xylene clearing and mounting. The sections were observed through an optical microscope.
Neuronal Apoptosis Index (AI) Detection
The cerebral cortex was removed from sacrificed rats, fixed, dehydrated, embedded, and cut into 3 µm slices. Dewaxing was performed, followed by soaking in a 0.1% Triton X-100 solution for 8 min, rinsing with running water, and then with phosphate-buffered saline (PBS) buffer. The sections were treated with a labeling mixture, incubated at 37°C for 1 h, rinsed with PBS buffer, mounted, and the AI value was calculated using the image analysis system.
Statistical Analysis
The data were analyzed using statistical package for the social sciences (SPSS) 21.0 and GraphPad Prism software. The measurement data were tested for normality, and data consistent with a normal distribution were represented by mean ± standard deviation (SD). The comparison between the two groups was performed using an independent samples t-test. The F-test was used among multiple groups. Data that did not conform to a normal distribution were represented by the median (quartile), and the Mann–Whitney U test was used to compare groups. The classification data were expressed by percentage, and the Chi-square or Fisher’s exact test was used to compare the disordered data groups. The Mann–Whitney U test was used to compare ordered classification data groups. If there are no special requirements, p < .05 refers to a significant difference.
Results
Modeling Results
No deaths or adverse events occurred during the intervention period. Examination through HE staining revealed evident cell edema (indicated by the arrows), disrupted organelle arrangement, and cell apoptosis in rat brain tissue sections (Figure 1). While the scores of rats in each group appeared normal, those in the model group displayed abnormal involuntary movements, posture, muscle tone, back grasping angle, and gait, with a Bederson score exceeding 1 (Table 1). This affirms the successful construction of the CP rat model with a 100% success rate.
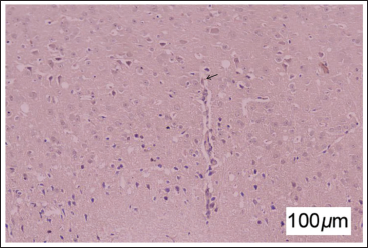
Behavior Scores of Rats in Each Group.
β-Sitosterol Improves Synaptic Plasticity in High-risk Children with CP
To investigate the impact of β-sitosterol on high-risk children with CP, we administered β-sitosterol to the rats in the animal experiment. Results indicated that the positioning navigation test’s latent period in the model group was significantly prolonged compared to the blank group. In the β-sitosterol group, the latency of the positioning navigation test was significantly shortened compared to the model group, though it remained longer than the blank group (Figure 2A). Various behavioral scores of rats indicated the protective effect of β-sitosterol. After 1 week of β-sitosterol intervention, rats exhibited increased suspension test scores, reduced involuntary action scores, and decreased Bederson scores (Figure 2B).
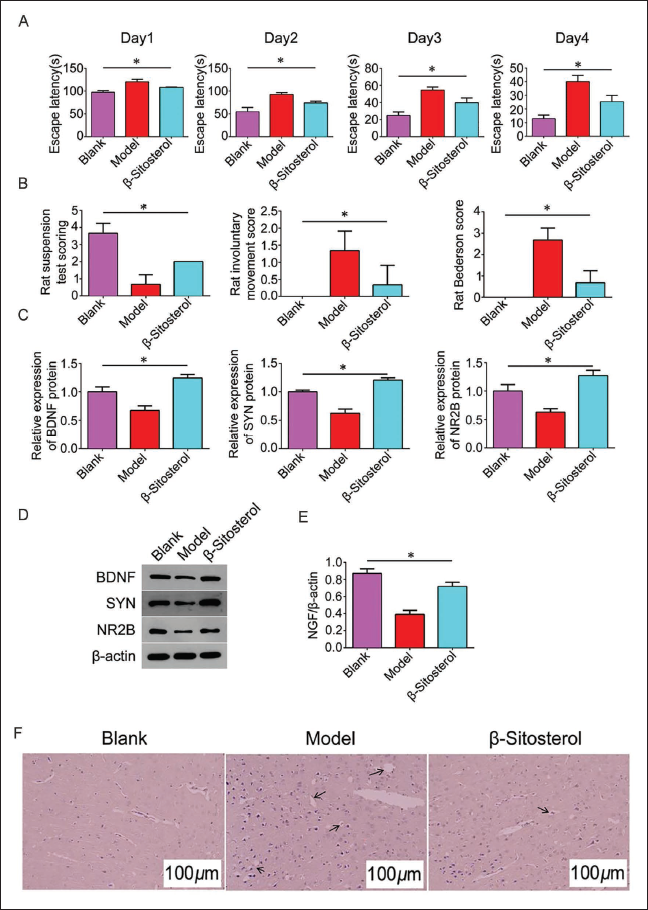
Compared with the blank group, the number of synapses in the hippocampal CA1 area of the model group decreased, and the anterior membrane activation zone and posterior membrane dense zone were narrowed, with fewer transmitter vesicles in the synaptic terminal and an unclear boundary. Examination of synaptic conditions and expression of synaptic receptors in the rat hippocampus revealed that the β-sitosterol group displayed an increase in synapses and improved structure compared to the model group, and the active zone became thicker and longer (Figure 2D). HE staining further corroborated these findings. Expression levels of synapse-related receptors (BDNF, SYN, NR2B, and NGF) in the β-sitosterol group increased significantly compared to the model group (Figure 2C–E), suggesting a potential association with NGF. With standard and complete nerve cell morphology and structure in the control group, the nerve cell structure in the model group was destroyed, with many vacuoles and necrotic nerve cells. However, in the β-sitosterol group, the number of vacuoles and necrotic nerve cells was reduced, and the ischemic and hypoxic changes were alleviated (Figure 2F). This suggests that β-sitosterol can improve synaptic plasticity in high-risk children with CP and may be related to NGF.
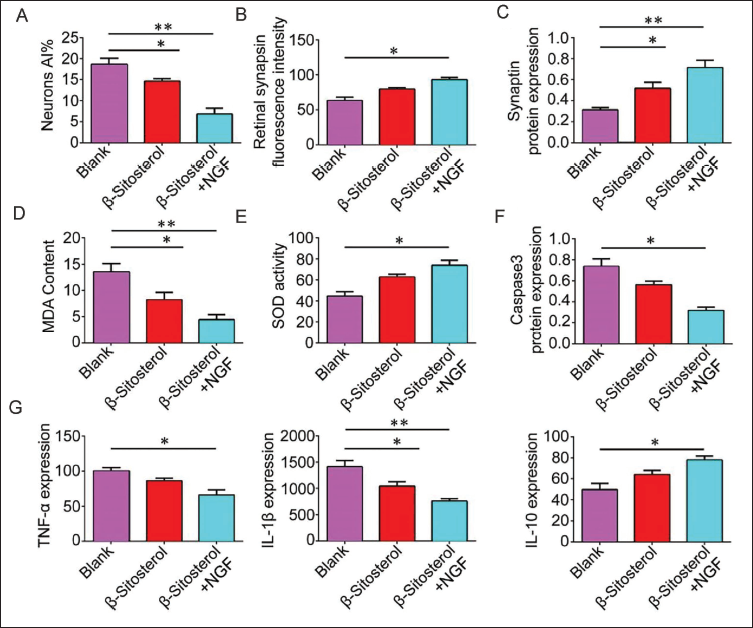
β-Sitosterol May Improve Inflammatory Status by Regulating NGF, Thereby Improving Synaptic Plasticity in CP
To delve into the mechanism of β-sitosterol in regulating NGF to enhance synaptic plasticity, NGF was added to β-sitosterol treatment. Synaptophysin immunofluorescence intensity in the β-sitosterol group surpassed that of the model group and further increased with injection of NGF (Figure 3B). The relative expression of synaptophysin and neuronal AI followed a similar trend, further increasing in the β-sitosterol + NGF group (Figure 3A, C). Exploring the impact on the inflammatory response, we measured the content of MDA and inflammatory factors, as well as the activity of SOD. MDA content significantly decreased in the β-sitosterol groups, and the addition of NGF intensified this reduction (Figure 3D–G). The contents of Caspase-3 protein, TNF-α, and IL-1β displayed a similar trend. Conversely, SOD activity and IL-10 expression increased significantly in the β-sitosterol groups, with further augmentation in the β-sitosterol + NGF group.
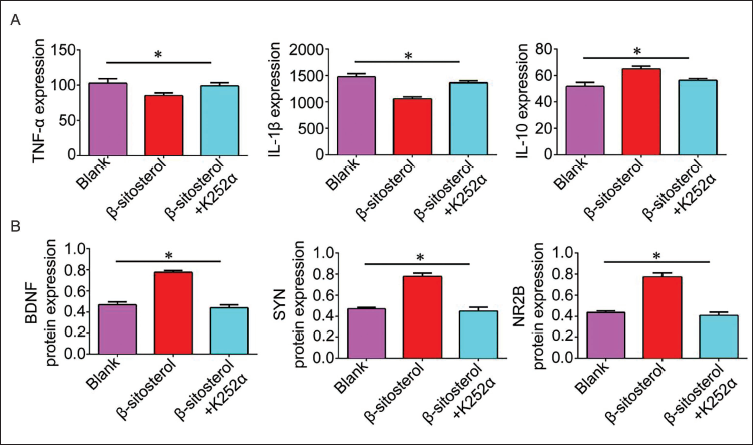
NGF Inhibitor K252α Promotes the Inflammatory Response and Decreases the Expression of Synaptic Receptors
To ensure experimental rigor, we introduced the NGF inhibitor K252α to β-sitosterol treatment, revealing that the β-sitosterol + K252α group had lower expression of synaptic receptors (BDNF, SYN, NR2B) than the β-sitosterol group. Inflammatory factors TNF-α and IL-1β showed an upward trend, while IL-10 exhibited a downward trend in the β-sitosterol + K252α group (Figure 4).
Discussion
CP mainly manifests as central movement disorders and abnormal posture (Friedman et al., 2022). At the molecular level, CP’s occurrence involves various mechanisms, including gene mutations, oxidative stress, inflammatory response, and apoptosis (Alpay Savasan et al., 2021). Although the management and rehabilitation of CP have improved with advances in modern medicine and rehabilitation treatments, there are still many children who face quality of life and long-term health problems due to impaired neurological function. These mechanisms can damage neurons and glial cells to a certain extent. Studies indicate that mutations in genes like GDNF and BDNF can impact neuron growth, differentiation, and synaptic plasticity, contributing to CP (Jin et al., 2020). Neurotrophic factors such as NGF play an important role in neuronal survival, development, and functional maintenance during neurodevelopment. By binding to high-affinity receptors, NGF promotes neuronal growth, survival, and synaptic plasticity, which profoundly impact the development and function of the nervous system. However, in children with CP, abnormal development of the nervous system and chronic inflammatory states may lead to abnormalities in the NGF signaling pathway, thus affecting the normal development and functional reconstruction of neurons.
β-Sitosterol can inhibit endoplasmic reticulum stress, apoptosis, and repair tissue (Khan et al., 2022). With anti-inflammatory activity, it can target the glucocorticoid receptor (GR) to improve airway inflammation and remodeling by inhibiting type II immune response and collagen deposition (Xu et al., 2023). In conditions like asthma, β-sitosterol reduces inflammatory factors and regulates dendritic cells to improve the pathological damage caused by asthma (Wang et al., 2022). Tang et al. noted that β-sitosterol not only reduces cerebral infarction volume and brain edema, as well as neuronal apoptosis, and alleviates neurological dysfunction (Tang et al., 2024). Meanwhile, β-sitosterol can increase the activity of hypoxic and glucose-deficient cerebral cortical neurons and reduce cell apoptosis, with high affinity to the key cholesterol transporter NPC1L1, through which β-sitosterol can inhibit cholesterol overload/plasma reticulum stress/apoptosis signaling pathway to improve ischemic stroke. β-Sitosterol’s activity extends to neurological conditions in Alzheimer’s disease, in which it enhances NGF activity and promotes neurite growth, penetrating the blood-brain barrier to improve the condition (Koga et al., 2020).
We initiated a study to assess the impact of β-sitosterol on high-risk children with CP by establishing a rat model of CP and implementing group interventions. In comparison to healthy rats, CP rats exhibited a significantly prolonged latency in the positioning navigation test. Remarkably, β-sitosterol intervention notably reduced test latency, increased suspension scores, and diminished involuntary action and Bederson scores. Concurrently, it augmented the number of synapses in the rat hippocampus, ameliorated synaptic structure, reduced vacuole formation, and decreased necrotic nerve cells, thereby alleviating ischemic and hypoxic changes. Furthermore, β-sitosterol significantly upregulated the expressions of BDNF, SYN, NR2B, and NGF, indicating its substantial enhancement of synaptic plasticity in CP, possibly through its interaction with NGF.
Studies have clearly shown that the administration of NGF can rescue basal forebrain cholinergic neurons (BFCS) and restore cognitive dysfunction (Mitra et al., 2021). Ciafrè et al. (2020) stated that NGF involves several physiological mechanisms leading to neurotrophic, metabolic, and/or immunotrophic processes.
Neuron-specific deletion of NGF prevents sympathetic innervation and white adipose tissue (WAT) browning, indicating its important role in the regulation of neurological diseases (Samario-Roman et al., 2023). In a model of paclitaxel-induced peripheral neuropathy, NGFR100W messenger ribonucleic acid (mRNA) enhances the recovery of intraepidermal nerve fibers (Yu et al., 2023).
NGF’s involvement in Alzheimer’s disease suggests its significance in counteracting atrophy and cholinergic function impairment (Reis et al., 2023). Additionally, NGF-based signaling has been shown to improve septal insulinotropic signaling in 3 × Tg-AD mice (Triaca et al., 2021) and play a vital role in various aspects of microbial stem cell function, contributing to survival, growth, differentiation, and angiogenesis in brain degenerative diseases and trauma (Zha et al., 2021).
To investigate NGF’s role in β-sitosterol-mediated improvement of synaptic plasticity in high-risk children with CP, we administered NGF to CP rats in conjunction with β-sitosterol intervention. Compared to CP rats, β-sitosterol intervention significantly decreased the neuronal apoptosis index, increased retinal synaptophysin fluorescence intensity, and elevated fluorescein’s relative expression. Additionally, it induced a significant decrease in MDA content, Caspase-3 protein expression, TNF-α, and IL-1β levels, while significantly increasing SOD content and IL-10 levels. The combined intervention of β-sitosterol and NGF further amplified these effects, underscoring β-sitosterol’s ability to mitigate the inflammatory response by elevating NGF levels and improving synaptic plasticity in high-risk children with CP. Compared to other studies, our study focused on the exploration of β-sitosterol, which improves synaptic plasticity in high-risk children with CP. In contrast, other studies have shown that NF-κB may be the reason for this increase, but in the study, it is possible that the effects of the studies taken on sex differ, leading to differences in study results. The sample size of our study is small due to funding problems, but the conclusion is relatively consistent. We will expand the sample size for in-depth research at a later stage.
To corroborate our experimental findings, we introduced the NGF inhibitor K252α into the experiment. Supplementing β-sitosterol intervention with K252α significantly reversed the beneficial effects of β-sitosterol on the inflammatory response. Simultaneously, it reduced the expression of the synaptic receptors BDNF, SYN, and NR2B. This suggests that inhibiting NGF expression markedly counteracts the ameliorative effects of β-sitosterol on high-risk children with CP.
Conclusion
In conclusion, our study elucidates the potential of β-sitosterol to improve NGF levels, regulate inflammatory homeostasis, and enhance synaptic plasticity in children at high risk of CP, providing a new perspective for an in-depth understanding of the pathogenesis of CP. By further studying the application of β-sitosterol in treating CP, we expect to provide new therapeutic strategies for clinical practice. It also contributes to improving the quality of life and long-term rehabilitation of children with CP and provides a reference for treating other neurological diseases. While the study has yielded promising results, the limitation of a small sample size underscores the need for further research to validate and expand upon our findings.
Footnotes
Abbreviations
BDNF: Brain-derived neurotrophic factor; CP: Cerebral palsy; MDA: Malondialdehyde; NGF: Nerve growth factor; NR2B: N-Methyl
Acknowledgments
We gratefully acknowledge the Qingdao Municipal Hospital for providing the necessary equipment for this study.
Authors Contribution
All authors participated in the completion of the manuscript.
Shaoyong Luan and Caixia Wang were responsible for drafting the manuscript.
Peipei Wang provided the overarching direction and guidance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the Ethics Committee of Qingdao Municipal Hospital.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
