Abstract
Background
Rheumatoid arthritis (RA) is a chronic inflammatory autoimmune disorder mainly affecting joints, yet the systemic inflammation can influence other organs and tissues. The objective of this study was to unravel the ameliorative capability of Ondansetron (O) or β-sitosterol (BS) against inflammatory reactions and oxidative stress that complicates Extra-articular manifestations (EAM) in liver, kidney, lung, and heart of arthritic and arthritic irradiated rats.
Methods
This was accomplished by exposing adjuvant-induced arthritis (AIA) rats to successive weekly fractions of total body γ-irradiation (2 Gray (Gy)/fraction once per week for four weeks, up to a total dose of 8 Gy). Arthritic and/or arthritic irradiated rats were either treated with BS (40 mg/kg b.wt. /day, orally) or O (2 mg/kg) was given ip) or were kept untreated as model groups.
Results
Body weight changes, paw circumference, oxidative stress indices, inflammatory response biomarkers, expression of Janus kinase-2 (JAK-2), Signal transducer and activator of transcription 3 (STAT3), high mobility group box1 (HMGB1), and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), as well as pro- and anti-inflammatory mediators in the target organs, besides histopathological examination of ankle joints and extra-articular tissues. Treatment of arthritic and/or arthritic irradiated rats with BS or O powerfully alleviated changes in body weight gain, paw swelling, oxidative stress, inflammatory reactions, and histopathological degenerative alterations in articular and non-articular tissues.
Conclusion
The obtained data imply that BS or O improved the articular and EAM by regulating oxidative and inflammatory indices in arthritic and arthritic irradiated rats.
Introduction
Arthritis is the inflammation of one or more joints, which can cause pain, discomfort, bone loss, and impairment. 1 Rheumatoid arthritis (RA), osteoarthritis (OA), and gouty arthritis (GA) are the three most prevalent forms of arthritis. 2 Chronic synovitis is a hallmark of RA, an inflammatory polyarticular autoimmune disease with several factors. 3 It is the most prevalent kind of inflammatory arthritis, primarily characterized by synovial hyperplasia, aberrant humoral and cellular immune responses, and inflammation of the synovial membrane, which can damage articular cartilage and juxta-articular bone. 4 Various inflammatory cells in the synovium facilitate RA initiation and development. 5 Furthermore, significant tissue damage linked to RA can happen in the heart 6 and the lungs, skin, eyes, kidneys, and blood vessels. 7 Adjuvant-induced arthritis (AIA) model has been developed extensively to simulate various human diseases, including arthritis. Amongst AIA models, CFA-induced arthritis seems the most reliable and justifiable in research . 8
CFA is extensively used to develop experimental models of autoimmune diseases, including rheumatoid arthritis (RA), gout, osteoarthritis, encephalomyelitis, neuritis, uveitis, thyroiditis, and orchitis, either in acute or chronic types. CFA-induced arthritis is more severe and systemic, 9 compared to antigen-free adjuvants such as incomplete Freund’s adjuvant (IFA) and pristine. 10 Its efficiency in stimulating antibody production is reported to be greater than other types of adjuvant. 10 It also produces nociceptive responses that may assist researchers in discovering new potential therapeutic drugs to combat arthritic pain. 11
Exposure to ionizing irradiation (IR) occurs during diagnostic or interventional radiology and radiotherapy. 12 IR is an environmental or human-made genotoxic agent that affects cells depending on their type, such as gamma and X-rays, alpha and beta particles, and neutrons. 13 Ionizing radiation can hurt living things directly by hitting them, or it can hurt them indirectly by breaking down water radioactively and making more reactive oxygen species, which can be too much for antioxidants to handle and cause oxidative stress. 14 Some free radicals are needed for cell communication. Still, too many reactive oxygen species (ROS), formed when cells are exposed to infrared radiation (IR), can damage the biological macromolecules in the joint if they are not adequately cleaned up. 15 Also, Lepetsos and Papavassiliou found that oxidative stress is connected to the development of arthritis. This is most likely because collagen-II in articular cartilage breaks down. 16
Low-dose radiation can attenuate An established inflammatory process, which changes the inflammatory state to an anti-inflammatory one. 17 Numerous studies have previously documented the modulatory effect of low-dose radiation to slow the course of arthritis in various animal models. 18 Previous trials using several doses (5/0.5 Gy) were used to report the modulatory effect of low-dose whole-body radiation on RA. 19 In a dosage-dependent way, whole-body irradiation (usually at a dose of >1 Gy) might harm the hematological, gastrointestinal, and neurovascular organ systems, which are vitally important organ systems. 20 Therefore, many studies aimed to maximize the therapeutic efficacy of low dose radiation while minimizing the radiation dose and number of exposures.
Beta-sitosterol (BS), one of the most abundant dietary phytosterols, resembles cholesterol in structure 21 and has various activities, including antioxidant, cholesterol-lowering, anti-hyperlipidemia, 22 anti-inflammation 23 and anti-tumor. 24 According to reports, the plant extract containing BS significantly reduces the activation of the NF-κB transcription factor in Phorbol-12-myristate-13-acetate (PMA)-stimulated macrophage cells with antiarthritic properties. 25 In addition, the expression of vascular endothelial growth factor (VEGF) was decreased by BS in renal tissue, and BS inhibited angiogenesis in renal cell carcinoma, 26 as the characteristics of synovial angiogenesis were similar to tumor angiogenesis. 27 Therefore, Qian et al. hypothesized that BS may inhibit synovial angiogenesis in RA by reducing joint swelling and destruction. 28
The long-term use of antiarthritic drugs may cause multiple side effects and limited therapeutic responses. 29 Therefore, novel treatments are in urgent demand. 5-hydroxytryptamine subtype 3 (5-HT3) receptor antagonists, including tropisetron and ondansetron, had anti-inflammatory properties. 30 Ondansetron (O), a 5-HT3 receptor antagonist, is commonly used to prevent and treat nausea and vomiting caused by radiation therapy, chemotherapy, gastroenteritis, and surgery, 31 as well as may also reduce hemodynamic changes caused by spinal anesthesia. 32 Moreover, preliminary data have shown that the intravenous injection of ondansetron has an analgesic effect on neuropathic pain. 33 It was proposed that ondansetron’s ability to reduce inflammation may be mediated by its impact on 5-HT3 receptor pathways. Adults with rheumatoid arthritis who had nausea from methotrexate (MTX) have also found success with ondansetron treatment. 34
Based on the promising biological efficacy of BS and O in various ailments, their anti-inflammatory and anti-arthritic activities are not fully elucidated, with particular emphasis when arthritic subjects are exposed to an enviable environmental stressor, such as γ-radiation. Therefore, the goal of this study is to (1) determine how Freund’s adjuvant, either with or without gamma radiation, affects oxidative stress and inflammatory manifestations in the liver, kidney, Lung, and heart and (2) determine how β-sitosterol and ondansetron affect any potential changes that Freund’s adjuvant, alone or in combination with gamma radiation. To confirm these objectives and assess the state of oxidative stress, we tracked the JAK-2/STAT-3 pathway by measuring lipid peroxidation and superoxide dismutase (SOD). Additionally, the levels of inflammatory mediators in rat groups were tested, including tumor necrosis factor-alpha (TNF-α), Toll-like receptor 4 (TLR4), High mobility group box one protein (HMGB), and NF-κB.
Materials and methods
Materials
Ondansetron (≥98% (HPLC), powder, Product Number: 3639, CAS-No.: 103639-04-9) and β-sitosterol (synthetic, ≥95%, Product Number: S1270, CAS-No.: 83-46-5); were purchased from Sigma-Aldrich Chemical Co in the United States. Reagents and chemicals utilized in this investigation were purchased from Sigma-Aldrich Chemical Co. in the United States.
Ethics approval statement
Experimental rats were handled by following the recommendations of the National Institute of Health (NIH No 85:23, revised 1996) for the care and use of laboratory animals and under the regulations of the Ethical Committee of the NCRRT, Atomic Energy Authority, Cairo, Egypt (Approval No 12A-21)
Induction of arthritis with freund’s adjuvant
Rats were injected subcutaneously with a single dose of 0.8 mL of Freund’s complete adjuvant (FCA) Part No.77140 (product of Thermo Scientific™, Catalog No: PI77140) containing 400 μg dry Mycobacterium tuberculosis (H37Ra, ATCC 25177) into the dorsal root of the tail under ether anesthesia. 35 Shortly after the FCA injection, classic symptoms of inflammation were observed and culminated on day 14, with the day of FCA immunization being considered day zero.
Radiation facility
Rats were exposed to whole-body gamma irradiation using Canadian γ-cell-40 (137Cs). Irradiation of the whole body of rats was performed at NCRRT (Cairo, Egypt) with a Canadian gamma cell. The dose rate during the execution of the experiment was 2 Gy/week for 4 weeks at a dose rate of 0.401 Gy/min at the time of the experiment.
Animals
The female adult Swiss albino rats weighing (120–150 g) used in this study were obtained from the experimental animal house lab of the National Center for Radiation Research and Technology.
The animal care was consistent with the guidelines of Ethics by the Public Health Guide for the Care and Use of Laboratory Animals (National Research Council, 1996) under the proper care and use of laboratory animals approved by the animal care committee of the National Center for Radiation Research and Technology, Cairo, Egypt.
Experimental design
70 Rats were divided into seven equal groups (10 rats/group): (1) Control group: normal rats just received physiological saline. (2) Group A: rats received Freund’s adjuvant (CFA) alone (a single intradermal injection of 0.1 mL) on day 1 to induce arthritis)zero time) (3) Group (A + R): rats received CFA and were also exposed to Radiation R (2 Gy/week) at day 1 (0 day) for 4 weeks up to a total dose of 8 Gy at a specific time point each week (beginning of the week), (4) Group (A + BS): rats received CFA at day one and then treated with β-sitosterol (BS) (40 mg/kg b.wt./day, orally); daily beginning from 14 till the end of experiment, 36 (5) Group (A+BS+R): rats received CFA at day 1, and exposed to R as in group 3 and then treated with BS as in group 4 from day 14, (6) Group (A + O): rats received CFA at day one and from day 14, treated with ondansetron O ((2 mg/kg) was given ip, daily till the end of the experiment 37 ), (7) Group (A + O + R): rats received CFA at day 1, and exposed to R as in group 3, then treated with O as group 6. At the end of the 4th week (day 28), rats were fasted overnight, anesthetized, and then sacrificed. The target tissues (liver, kidney, Lung, and heart) and Sera were collected and prepared to estimate biochemical and histopathological investigations.
Body weight changes
The rats’ body weight was measured 1 day before the CFA injection. The initial body weight at week zero was referred to as this. Nonetheless, the weight determined in the ensuing weeks has been referred to as the terminal body weight. For 5 weeks, starting from week zero, the body weight of the rats was monitored weekly to track any variations in the body weight of the rats in each experimental group.
Analysis of limb diameter (paw size)
Using a Vernier caliper, each rat’s right hind paw volume (mL) was measured on day zero before CFA injection 38 and observed every 3 days till the 28th day.
Western blot analysis
The protein expression of NF-κB, JAK, STAT, and HMGB in the heart, Lung, liver, and kidney were performed according to Omar et al. 39 using a homogenization lysis buffer (Sigma-Aldrich, St Louis, MD, USA). Tissue lysate was collected after centrifugation for 20 min (at 8678 g, at 4°C), and the protein concentration was measured via a BCA-protein kit (Catalog number: 23225 Thermo Fisher Scientific). Then, 7.5 μg of protein was denatured from each sample and loaded onto 8% SDS–PAGE (sodium dodecyl sulfate-polyacrylamide gel electrophoresis). After that, it was transferred to a nitrocellulose membrane (Amersham Bioscience-Piscataway-New Jersey-USA) throughout a semidry transfer apparatus (Bio-Rad, Hercules, Calif). The obtained membranes were incubated overnight at 4°C with milk-blocking buffer (at 5%) containing the following: Tris-HCl (10 mmol/L at pH = 7.4), NaCl (150 mmol/L) and TBST (Tris-buffered saline with Tween-20, 0.05%). Then, the membranes were washed with TBST and incubated with a dilution of anti- NFkB (NFkB p65 Polyclonal Antibody Cat #PA5-27617- thermo fisher scientific), anti-JAK (JAK2 Monoclonal phosphorylated Antibody (691R5) Catalog # AHO1352- thermo fisher scientific), anti-STAT (STAT3 Monoclonal phosphorylated Antibody (9D8)-Cat #MA1-13042 thermo fisher scientific) and anti-HMGB (HMGB1 Monoclonal phosphorylated Antibody (GT348)-Cat #MA5-17278 thermo fisher scientific) antibodies overnight at four °C on a roller shaker.
The appropriate horseradish peroxidase (HRP) conjugated goat immunoglobulin (Amersham Biosciences) was used after the relevant primary antibody to complete the immunoblotting. The Amersham detection kit’s manufacturer’s instructions were followed for the chemiluminescence detection, which was subsequently exposed to X-ray film. Next, the protein level in each sample was quantified using a scanning laser densitometer (Biomed Instruments) to perform a densitometric analysis of the auto-radiograms. Lastly, the collected records will be assayed and normalized to the expression of the housekeeping protein, β-actin protein. 40
Real-time PCR
RNA isolation and reverse transcription
RNA was extracted from liver, kidney, heart, and lung tissue using the RNase plus mini kit (Qiagen, Venlo, The Netherlands), according to the manufacturer’s instructions. Genomic DNA was removed using the included DNase-on-Column treatment method. The NanoDrop ND-1000 spectrophotometer measured the RNA concentration spectrophotometrically at 260 nm. (Thermo Fisher Scientific, Waltham, USA), and RNA purity was checked using the absorbance ratio at 260/280 nm. RNA integrity was assessed by electrophoresis on 2% agarose gels. RNA (1 μg) was used in the subsequent cDNA synthesis reaction, which was performed using the Reverse Transcription System (Promega, Leiden, The Netherlands). Total RNA was incubated at 70°C for 10 min to prevent secondary structures. The RNA was supplemented with MgCl2 (25 mM), RTase buffer (10X), dNTP mixture (10 mM), oligod (t) primers, RNase inhibitor (20 U), and AMV reverse transcriptase (20 U/μl). This mixture was incubated at 42°C for 1 hour.
Quantitative real-time PCR
Primers used for QRT-PCR. FW forward primer; RW reverse primer.

Biochemical Assay
Oxidative stress markers
Lipid peroxidation product (malonaldehyde (MDA)) was measured using the Malondialdehyde ELISA Kit (ab118970). The antioxidant enzyme (SOD) activity was measured by (Superoxide Dismutase 1 (SOD1) (Rat) ELISA Kit ab65354). In the heart, Lung, kidney, liver, and Serum, levels of (IL-10 (Rat IL-10 ELISA Kit (ab214566)), IL-6 (IL-6 (rat) ELISA Kit-(ab100772), and TLR-4 (Rat Toll-Like Receptor 4 (TLR4) ELISA Kit- Catalog Number. CSB-E15822r)) and ALT (ab234579), AST (ab263883), UREA (ab83362)), CREATININE (ab65340) and TN-1 (ab246529), in Serum only, were measured by enzyme-linked immunosorbent assays according to the manufacturer’s instructions via ELISA microplate reader (DV990 BV 416; Gio.DE VITA and CO., Rome, Italy).
Histopathological examination
Animals of each group were sacrificed via pentobarbital overdose (100 mg/kg). Then, the bone, kidney, heart, liver, and lung were removed, fixed in 10% neutral buffered formalin for 48 h, and decalcified in 10% ethylene diamine tetra-acetic acid (EDTA) for 30 days. The tissue was processed using graded ethanol, xylol, and paraffin, which included dehydration, clearing, impregnation, and embedding. After preparing histological slices with a thickness of 6 μm from every defect with an intact bone border, the samples underwent regular staining with hematoxylin and eosin. 41
Histological scoring parameters of joint.

Histological grading of Renal, myocardial, hepatic, and lung lesions.

Each tissue section of the Lung was given a score from 0 to 4 based on the amount of area affected by interstitial inflammation, alveolar wall thickening, peribronchial inflammation, and interstitial edema, 46 as shown in Table 3.
Immunostaining of Cyclooxygenase 2 (COX2), Inducible NOS (iNOS), Myeloperoxidase (MPO), and Nuclear factor kappa- β (NFKB) in ankle joints
Immunohistochemical detection of COX2, iNOS, MPO, and NF-κB expression was performed on 4 µm thick demineralized ankle joint sections. All sections were dewaxed by xylene, dehydrated, and thoroughly washed three times in PBS for 5 min before incubating with 3% H2O2 for 10 min at 37°C. They were then rinsed three times in PBS before being blocked with goat serum blocking solution. Excess fluid was removed, diluted primary anti COX2 (Cyclooxygenase 2 antibody (ab15191)), anti-iNOS (Anti-iNOS (phospho Y151) antibody (ab182640), anti-MPO (Anti-Myeloperoxidase antibody [EPR20257] – Rat IgG2a (Chimeric) – BSA and Azide free (ab300651)), and anti-NFκB (Recombinant Anti-NF-κB p105/p50 antibody [E381] (ab32360).), were placed drop by drop. Sections were preserved in a humidified cabinet at 4°C for 24 h. The slices were stabilized and shaken at 37°C for 20 min. Afterward, they were cleaned and treated with a secondary antibody labeled streptavidin-horseradish peroxidase for 20 min. The expression of biomarkers was examined using hematoxylin counterstaining and yellow 3, 3′-diaminobenzidine (DAB) staining. Saline buffered with phosphate (PBS) was used as the negative control. Immunohistochemical analysis was performed with Image-Pro Plus 6.0 software to determine immunostaining.
Statistical analysis
Statistical analysis was done using one-way analysis of variance (ANOVA) followed by a post hoc multiple comparisons test using Graph Pad Prism® version 8.00 (San Diego, CA, USA) software. Data of body weight changes and paw circumference were indicated using two-way analysis of variance (ANOVA) for two variables comparisons, the group effect and time intervals. Sample size (n) calculations were performed to imply the study power (β) at a significance level
(α) = 0.05. Data were presented as mean ± SEM (standard error of the mean).
Results
Body weight
Compared to normal control rats, arthritic rats displayed a weekly rise in body weight (Figure 1). Compared to arthritic rats, the body weight of AR rats (rats that received Freund’s adjuvant and were subjected to R increased until week 2, dropping until week 4. After week 4, the rats in the ABs group had lost weight (p < .001). After week 4, the rats in the ABsR group likewise displayed a decrease in body weight (p < .001) (Figure 1). The rats in the AO group exhibited a rise in weight during weeks 1 and 2, which was followed by a decrease in body weight until week 4 (p < .001). Figure 1 illustrates a drop in body weight until week 4 in group AOR (p < .001). Effect of Ondansetron or Beta-sitosterol on body weight, Paw size of irradiated arthritic rats. C: normal rats, A: rats received Freund’s adjuvant alone, AR: rats received Freund’s adjuvant and also exposed to γ-radiation R (2 Gy/week), ABs: rats received Freund’s adjuvant at day one and then treated with β-sitosterol (BS) (40 mg/kg b.wt./day, orally), AB + R: rats received Freund’s adjuvant at day 1, and exposed to R as in group 3 and then treated with BS, and AO: rats received Freund’s adjuvant at day one and from day 14, treated with ondansetron ((2 mg/kg) and AOR: rats received Freund’s adjuvant at day 1, and exposed to Ras in group AR, then treated with ondansetron (O).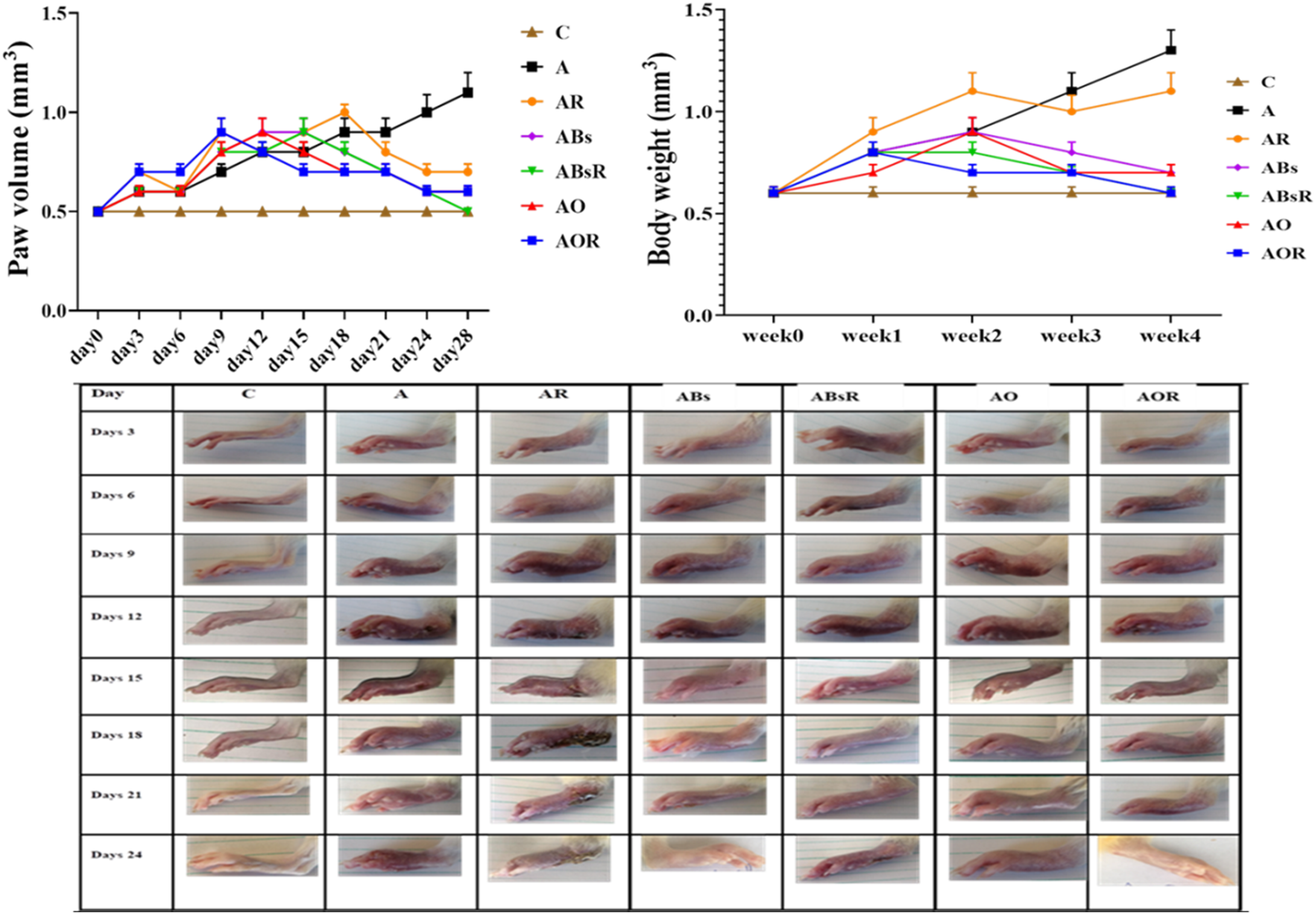
Paw thickness measurement
Paw swelling was confirmed as a sign of arthritis 8 days after the arthritis was produced, and by day 15, the disease model had fully developed. Ondansetron and beta-sitosterol were administered to treat rats suffering from arthritis caused by CFA. Paw thickness was measured on days 3, 6, 9, 12, 15, 18, 21, 24, and 28 (Figure 1). The limb joint scores of the A group showed that arthritis’s progression was very time-dependent, with the third week exhibiting the highest arthritis score and being significantly different from the other time points (p < .05; Figure 1). The limbs of the AR group showed decreased paw edema and a slower rate of illness progression from the 1st injection to day 18. Subsequently, a reduction in bow size was noted in contrast to the A group. Rats in group ABs treated with CFA and BS alone or in combination with γ-irradiation in the ABsR showed reduced paw edema. They slowed disease progression starting on day 12. Compared to the A group, whose paw size decreased beginning on day 8 and remained until the experiment’s end, the AO and AOR groups’ paws were substantially thinner.
Effect of Ondansetron or beta-sitosterol on serum of irradiated arthritic rats
In Figure 2, rats with arthritis treated with O or Bs and/or exposed to γ-irradiation had drastically changed oxidative status. Different groups’ changes in MDA levels and SOD activity to varied degrees served as evidence of this. The MDA concentration in the A group showed substantial increases (p < .05), whereas the SOD activity in the group showed a significant decrease compared to the control group’s comparable value. Compared to A group, γ-radiation treatment of arthritis-prone rats has resulted in a considerable drop in MDA and a significant increase (p > .05) in SOD activity. In addition, compared to the A group, the A + Bs group showed much higher SOD activity and significantly lower MDA concentrations. In contrast to the A + R group, the Bs and γ-radiation combination have appeared to modify the oxidative status in A + Bs + R by raising SOD activity and lowering MDA levels (Figure 2). Compared to the A and A + O groups, arthritic rats treated with O alone or with γ-irradiation showed a significant increase in SOD and a considerable drop in MDA levels. Effect of Ondansetron or beta-sitosterol on Serum of irradiated arthritic rats. Each value represents Mean ± SEM (n = 6). Columns denoted with ˝a˝ significant from C, ˝b˝: significant from A, c˝ significant from AR, ˝d˝ significant from ABs, ˝e˝ significant from A + Bs + R and ˝f˝ significant from AR + O, at (p < .05).groups as in Figure 1.
The inflammatory state biomarkers are shown in Figure 2 alongside TLR4. These include pro-inflammatory mediators (TNF-α) and anti-inflammatory cytokines (IL-10 and IL-6). The A group’s serum levels of IL-10 were significantly lower (p ˂ 0.05) than those of the control group, whereas those of IL-6, TNF-α, and TLR4 were substantially higher. The results showed that the A + R, A + Bs, and A + O groups had considerably higher IL-10 levels than the A group. The A + Bs + R and A + O + R groups had higher levels than the A + R group. The arthritic rats treated with BS or O after radiation showed significantly higher levels of TLR4, TNF-α, and IL-6 than the rats in the A group.
Compared to the control group, the arthritic group’s levels of ALT, AST, TN-1, creatinine, and urea levels; increased considerably. Unlike the A group, the A + R group’s exposure to γ-irradiation dramatically raised their levels. ALT, AST, TN-1, creatinine, and urea levels were markedly elevated by injection of either BS or O, either alone or in combination with γ-irradiation, in the A + Bs, A + O, and A + Bs + R, A + O + R groups relative to the A and A + R groups, respectively.
Effect of Ondansetron or beta-sitosterol on JAK, STAT, NFKB, and HMGB in the kidney, lung, heart and liver of irradiated arthritic rats
The JAK, STAT, NFKB, and HMGB protein expressions were detected in kidney and heart tissues using western blotting of the A group. The results showed a substantial increase (p < .05) in comparison to the normal control (Figure 3). This finding was in line with the mRNA expression of JAK, STAT, NFKB, and HMGB. In comparison to A group, γ-radiation of arthritic rats A + R in both tissues, were observed to dramatically reduce the protein expression of JAK, STAT, NFKB, and HMGB. The results for JAK, STAT, NFKB, and HMGB mRNA expression were consistent with the findings for protein expression of these genes in kidney and heart tissues, which were found to be significantly lower in A + R group of arthritic rats after they were exposed to radiation, either alone or in combination with beta-sitosterol A + Bs or ondansetron A + O (p < .05; Figure 3). Effect of ondansetron or beta-sitosterol on JAK, STAT, NFKB, and HMGB in the kidney, Lung, heart, and liver of Irradiated arthritic Rats. Each value represents fold of difference between the quantification of JAK, STAT, NFKB, and HMGB, from normal control group. Columns denoted with ˝a˝ significant from C, ˝b˝: significant from A, c˝ significant from AR, ˝d˝ significant from ABs, ˝e˝ significant from A + Bs + R and ˝f˝ significant from AR + O, at (p < .05). Legends as in Figure 1.
While in lung tissue, the mRNA expression of JAK, STAT, NFKB, and HMGB was consistent with the observed significant increase in lung tissue of the A group (p < .05) when compared to the control group based on Western blotting-based detection of these protein expressions (Figure 3). In comparison to A group, γ-radiation of arthritic rats A + R was observed to drastically reduce the protein expression of JAK, STAT, NFKB, and HMGB in lung tissue. The protein expression of JAK, STAT, NFKB, and HMGB in lung tissue was found to be significantly decreased by BS (A + Bs) or O (A + O), either alone or in combination in the A + Bs + R and A + O + R groups (p < .05; Figure 3), which was consistent with the results for JAK, STAT, NFKB, and HMGB mRNA expressions.
For protein expressions of JAK, STAT, NFKB, and HMGB in liver tissue of A group, significant increase compared to normal control (p < .05; Figure 3), which was consistent with the mRNA expression of JAK, STAT, NFKB, and HMGB. Compared to A group, γ-radiation of arthritic rats A + R markedly reduced the protein expression of JAK, STAT, and NFKB, raising the level of HMGB in liver tissue. The protein expression of JAK, STAT, NFKB, and HMGB in liver tissue was found to significantly reduced by BS (A + B) or O (A + O), either alone or in combination in the A + Bs + R and A + O + R groups (p < .05; Figure 3), which was consistent with the results for JAK, STAT, NFKB, and HMGB mRNA expression.
Effect of Ondansetron or beta-sitosterol on IKB, 7NACH, and TNF-α in the kidney, lung, heart, and liver of irradiated arthritic rats
According to the RTqPCR results, the kidney, lung and heart’s IKB gene, TNF-α and 7-NACH mRNA expression varied. The arthritic group A’s 7NACH mRNA expression levels were considerably lower than those of the normal control group (p < .05; Figure 4). Rats in the control group did not exhibit substantially more significant levels of IKB mRNA expression in their kidney, lung and heart tissues compared to the A group (p < .05). When compared to the A group and A + R, respectively, it was discovered that irradiating arthritic rats A + R, BS (A + Bs), or O (A + O), either separately or in combination in A + Bs + R and A + O + R groups, significantly decreased IKB gene expression and increased 7NACH mRNA expression (p < .05). The kidney, lung and heart’s TNF-α gene expression were significantly higher (p˂0.05) in the A group’s data compared to control group. When arthritic rats were exposed to γ-radiation, their TNF-α gene expression significantly increased. After being injected with O or BS, arthritic rats showed significantly lower TNF-α gene expression levels than the A group. In the meantime, it was discovered that γ-radiation treatment of arthritic rats, in conjunction with O or BS injection, increased TNF-α gene expression. This finding was in line with the results for TNF-α gene expression when compared to the A group. Effect of ondansetron or beta-sitosterol on IKB, 7NACH, and TNF-α in the kidney, Lung, heart, and liver of Irradiated arthritic Rats. Each value represents fold of difference between the quantification of IKB, 7NACH, and TNF-α, from normal control group. Columns denoted with ˝a˝ significant from C, ˝b˝: significant from A, c˝ significant from AR, ˝d˝ significant from ABs, ˝e˝ significant from A + Bs + R and ˝f˝ significant from AR + O, at (p < .05). Groups as in Figure 1.
The results of RTqPCR demonstrated that the expression of the IKB gene and 7NACH mRNA varied in liver, with arthritic group A exhibiting significantly lower levels of IKB gene expression and 7NACH mRNA expression as compared to the normal control group (p < .05; Figure 4). When compared to A group or A + R group, it was found that irradiating arthritic rats A + R, beta-sitosterol A + Bs, or ondansetron A + O alone or in combination in A + Bs + R and A + O + R groups increased IKB gene expression and 7NACH mRNA expression (p < .05).
In contrast, TNF-α gene expression was significantly higher. Rats with arthritis exposed to radiation showed an increased liver TNF-α gene expression. In arthritic rats injected with O or BS, TNF-α gene expression levels were observed to drop when compared to A group. On the other hand, TNF-α gene expression was shown to rise in arthritic rats that were injected with either BS or O. The effect which was in line with results of TNF-α gene expression compared to the A group.
Effect of ondansetron or beta-sitosterol on SOD, MDA, IL-10, IL-6, and TLR-4 in the kidney, lung, heart, and liver of irradiated arthritic rats
In this study, statistical analysis of data from kidney, lung, heart and liver; of arthritic rats treated with beta-sitosterol or ondansetron and/or exposed to γ-irradiation revealed significant changes in oxidative status, which were reflected in varying degrees in MDA concentration and SOD activity among different groups. The data indicated a substantial rise (p < .05) in MDA levels in the A groups. On the other hand, in contrast to control group, the activity of SOD in the kidney, lung, heart and liver tissues; were much lower. Rats with arthritis that were exposed to γ-radiation showed a considerable rise (p > .05) in MDA and a significant decrease (p > .05) in SOD activity. In addition, compared to A group, the A + Bs and A + O groups, showed significantly higher SOD activity and lower MDA concentrations in different tissues under study. Compared to A + R group, combining γ-radiation with Bs in A + Bs + R or O in A + O + R; decreased the MDA level and increased SOD activity in kidney, lung, heart and liver tissues (Figure 5). Effect of ondansetron or beta-sitosterol on SOD, MDA, IL-10, IL-6, and TLR-4 in the kidney, Lung, heart, and liver of Irradiated arthritic Rats. Each value represents fold of difference between the quantification of SOD, MDA, IL-10, IL-6, and TLR-4,from normal control group. Columns denoted with ˝a˝ significant from C, ˝b˝: significant from A, c˝ significant from AR, ˝d˝ significant from ABs, ˝e˝ significant from A + Bs + R and ˝f˝ significant from AR + O, at (p < .05). Groups as in Figure 1.
The kidney, lung, heart and liver 's IL-10 levels were significantly lower (p˂0.05) in the A group’s data compared to control group, while TNF-α gene expression, TLR4 expression, and IL-6 levels were significantly higher. When arthritic rats were exposed to γ-radiation, their IL-10 levels in kidney, lung, heart and liver tissues; were significantly decreased while their IL-6 levels and TLR4 levels in the 4 tissues; were significantly increased (p˂0.05). It was found that IL-10 levels in the A + Bs and A + O groups significantly raised compared to the A group, whereas IL-10 levels in the A + Bs + R and A + O + R groups did not considerably alter compared to the A + R group. After being injected with ondansetron or beta-sitosterol, arthritic rats showed significantly lower IL-6 and TLR4 levels than the A group; in kidney, lung, heart and liver tissues. In the meantime, it was found that; in γ-irradiation of arthritic rats, in conjunction with ondansetron or beta-sitosterol injection, there was no discernible difference in IL-6 or TLR4 levels compared to the A group; for kidney, lung, heart and liver tissues.
Histopathological examination of the joint, kidney, heart, liver and lung tissues
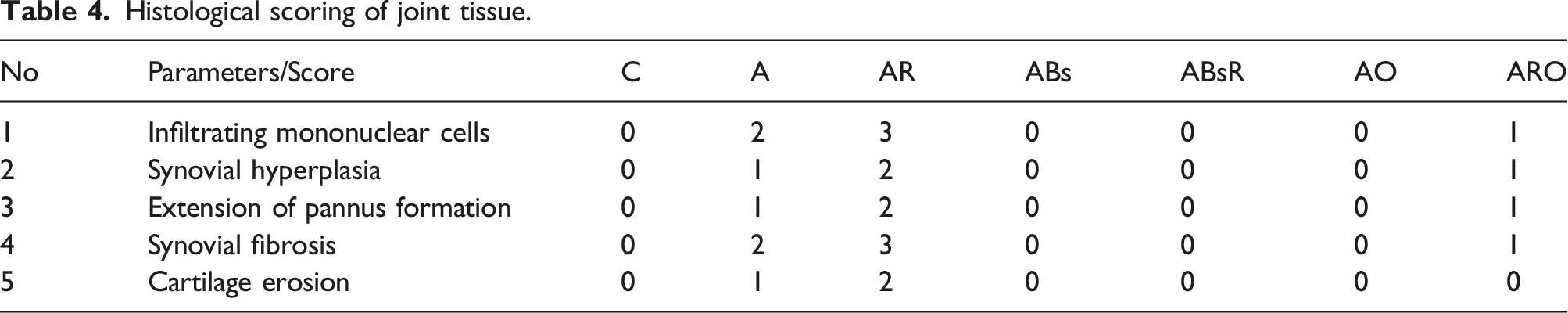
Histological scoring of joint tissue.
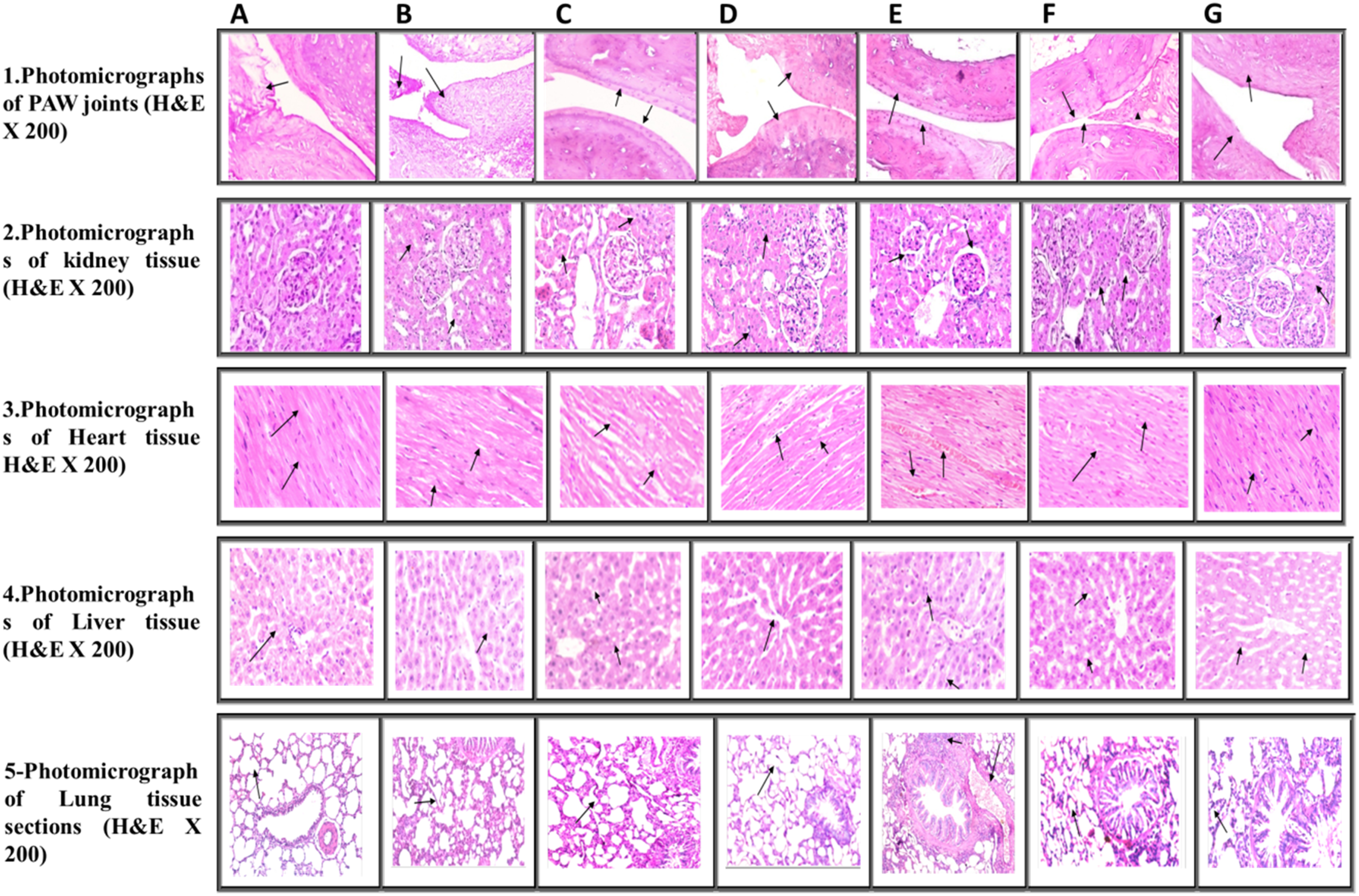
Photomicrographs of joint, kidney, heart, liver and lung tissues, as compared to those of normal rats (H&EX200): A. Photomicrograph of control (C) group, B. Photomicrograph of A group, C. Photomicrograph of AO group, D. Photomicrograph of ABs group, E. Photomicrograph of AR group, F. Photomicrograph of ARO group, and G. Photomicrograph of ABsR group tissues.
Degree of damage in kidney, heart, and liver in each animal group.

In Figure 6(3), the cardiac muscle in groups (C) & (ARO) &(AO) revealed a typical histological structure of heart muscle of control group. The muscle fibers appeared short-branched, joined each other forming a network. Cross striation and oval prominent central single nuclei were seen. Myocardial fibers were arranged regularly with clear striations, and no apparent degeneration or necrosis was observed. The degree of heart muscle damage revealed [(0) No change] (Table 5, Figure 6 (3:A, G,F)). Conversely, the heart muscle of group (AB) showed edema of heart muscle bundles with few mononuclear cell infiltration. The degree of myocardial muscle damage revealed [(1) Mild-focal myocyte damage or minor multifocal degeneration with a slight degree of inflammation] (Table 5, Figure 6 (3:D)). Heart muscle of the (AR) & (A) animals group showed moderate myofibrillar degeneration. A few mononuclear cell infiltrations, edemas between muscle fibers, and congestion of blood capillaries were noticed. The degree of heart muscle damage revealed [(2) Moderate-extensive myofibrillar degeneration] (Table 5, Figure 6 (3:C,B)). ARB group showed the same pathological picture with degree of heart muscle damage revealed [(3) Severe-Necrosis with diffuse inflammatory process] Figure 6 (3:E).
While, the liver tissue section showed a typical histological structure of (C), (ABs), (AO) & (AOR) groups, showing a standard hepatic lobules and organization of hepatic cords with a prominent central hepatic vein. Polygonal hepatic cells were joined in anastomosing plates, with clear borders facing either the sinusoids or adjacent hepatocytes grade 0 (Table 5, (Figure 6 (4:A- D-F-G)). The microscopic examination of the liver of (AR) group revealed degenerative changes in the hepatocytes characterized by ballooning of hepatocytes and narrowing of hepatic sinusoids hyperplasia of Kupffer cells with few mononuclear cells infiltration mainly lymphocytes and macrophages grade III (Table 5, Figure 6 (4:C)).
Liver tissue from group (A) showed low-grade cellular swelling and hyperplasia of Kupffer cells grade II (Table 5, Figure 6 (4:B)). While, the liver tissue section of group (ARO) revealed degeneration of hepatocytes characterized by vacuolation of hepatocytes with intracellular lipid droplets. Narrowing of hepatic sinusoids and hyperplasia of Kupffer cells in grade II (Table 5, Figure 6. (4:G)).
Also, lung sections in groups (C) & (ABs) & (AO) & (AOR) showed typical lung architecture. The airspaces were separated by refined, delicate inter-alveolar septa, normal vasculature with scant perivascular connective tissue, folded columnar epithelial cells of bronchiole, and normal fibrous tissue distribution. The alveoli appeared inflated with thin inter-alveolar septa. Squamous type I pneumocytes primarily lined them, and a few sizeable cuboidal type II pneumocytes scored 0 in Figure 6 (5:A, D, F, G). On the other hand, the lung tissue section of (ARB) groups revealed a moderate inflammatory response whith few mononuclear infiltrations, mainly lymphocytes and macrophages in peribronchial and perivascular spaces. Congestion and edema of peribronchial blood vessels were also detected. The bronchial and alveolar walls showed mild thickening score 2 (Table 5, Figure 6 (5:E)). The microscopic examination of the Lung obtained from (A)&(AR) treated rats groups revealed mild alveolar wall thickening with mononuclear infiltration, mainly lymphocytes and macrophages. Benign hyperplasia of bronchial epithelial lining with excessive eosinophilic mucous in its lumen was noticed in score 3 (Table 5, Figure 6 (5:B&C)).
Immunostaining of COX-2, NOS, MPO and NFKB in the joint tissues
The rats from the control group showed adverse COX-2 reactions, indicating no inflammatory reaction (COX-2 X100). (Lesion Score: 0) (Figure 7(1)). The joint in group (A) showed induces multi-focal positive brown color COX-2 reaction, indicating moderate inflammatory reaction (COX-2 X100). (Lesion Score: ++). Irradiation of arthritic rats defuse marked positive brown color COX-2 reaction, indicating an inflammatory severe response (COX-2 X200) (Lesion Score: +++). ABs group showed a multi-focal positive brown COX-2 reaction, indicating a mild inflammatory response with less severity than that of group (A) (COX-2 X200). (Lesion Score: ++). The joint of group (A + R + B) showed a focal faint positive brown colour COX-2 reaction, indicating a less severe mild inflammatory response than that of group (A + R), (COX-2 X100). (Lesion Score: +). Ondansetron induces multi-focal positive brown color COX-2 reaction indicating moderate inflammatory reaction less severity than that of group (A), in group (A + O), (COX-2X100). (Lesion Score:++). Meanwhile, A + R + O showed focal faint positive brown color COX-2 reaction indicating a less severe mild inflammatory response than in group (A + R), (COX-2 X100). (Lesion Score: +). Representative images of immunostaining of COX-2, NOS, MPO and NFKB in joint tissues of rats from different groups (H&Ex200). A. Photomicrograph of joint tissue of control (C) group (H&Ex200), B. Photomicrograph of joint tissue of A group, C. Photomicrograph of joint tissue of AO group (H&Ex200), D. Photomicrograph of joint tissue of ABs group (H&Ex200), E. Photomicrograph of joint tissue of AR, F. Photomicrograph of joint tissue of ARO group and G. Photomicrograph of joint tissue of ABsR group.
Figure 7(2) represents the immunostaining of i-NOS in joint of rats from different groups. The joint of control group showed a negative iNOS reaction, indicating no inflammatory reaction (iNOS X100). (Lesion Score: 0). While joints of group (A) showed multi-focal positive brown color iNOS reaction indicating severe inflammatory reaction (iNOS X100). (Lesion Score: ++). Group (A + R) showed multi-focal positive brown color iNOS reaction, indicating moderate inflammatory reaction (iNOS X200). (Lesion Score: ++). The joint of group (A + B) showed a focal positive brown color iNOS reaction, indicating a less severe inflammatory reaction than group (A) (iNOS X200). (Lesion Score: +). The joint of group (A + R + B) showed a focal faint positive brown color iNOS reaction, indicating a less severe mild inflammatory reaction than group (A + R) (iNOS X200). (Lesion Score: +). Treatment with ondansetron (A + O) induced focal positive brown color iNOS reaction indicating mild inflammatory reaction less severe than group (A), (iNOS X200). (Lesion Score: +). Group A + R + O showed focal faint positive brown color iNOS reaction indicating mild inflammatory reaction less severe than group (A + R), (iNOS X100). (Lesion Score: +).
Figure 7(3) represents the immunostaining of MPO in joint of rats from different groups. The joint of rats from control group showed adverse MPO reaction, indicating no inflammatory reaction (MPO X100). (Lesion Score: 0). The induction of arthritis in group (A) showed multi-focal positive brown color MPO reaction, indicating moderate inflammatory reaction (MPO X100). (Lesion Score:++). Group (A + R) showed multi-focal positive brown color MPO reaction, indicating moderate inflammatory reaction (MPO X200). (Lesion Score:++). While group (A + B) showing focal positive brown color MPO reaction indicating mild inflammatory reaction less severe than group (A), (MPO X200) (Lesion Score:+). The joint of rats from group (A + R + B) showed a focal faint positive brown color MPO reaction, indicating a mild inflammatory reaction less severe than group (A + R) (MPO X100). (Lesion Score: +). A + O group induced focal positive brown color MPO reaction indicating mild inflammatory reaction less severe than group A, (MPO X200). (Lesion Score:+). γ-Irradiation along with ondansetron (A + R + O) caused the appearance of a focal faint positive brown color MPO reaction, indicating a mild inflammatory reaction less severe than group (A + R) (MPO X100). (Lesion Score: +).
Joint of control group showed an adverse NF-kB reaction, indicating no inflammatory reaction (NF-kB X100) (Figure 7(4)). (Lesion Score:0). Arthritis in group (A) shows a multi-focal positive brown color NF-kB reaction, indicating moderate inflammatory reaction (NF-kB X100). (Lesion Score:++). A + R group showed defuse marked positive brown color NF-kB reaction indicating inflammatory severe response (NF-kB X200). (Lesion Score:+++). A + B group showed multi-focal positive brown color NF-kB reaction, indicating moderate inflammatory reaction less severe than group (A), (NF-kB X100). (Lesion Score:++). Also, joints of group (A + R + B) showed focal faint positive brown color NF-kB reaction indicating mild inflammatory response less severe than group (A + R) (NF-kB X100). (Lesion Score:+). In A + O group, multi-focal positive brown color NF-kB reaction noticed; indicating moderate inflammatory reaction less severe than group (A), (NF-kB X200). (Lesion Score:++). A + R + O group showed focal faint positive brown color NF-kB reaction indicating mild inflammatory reaction less severe than group (A + R), (NF-kB X100). (Lesion Score:+)
Discussion
Rheumatoid arthritis (RA) is an autoimmune systemic inflammatory disease distinguished by pannus formation, chronic synovitis, and irreversible degradation of cartilage and bone. 47 Accordingly, the present study evaluated the potential regulatory effect of Bs or O that might impact the liver, kidney, Lung, and heart in rats challenged with CFA and exposed to a fractionated dose gamma irradiation (IR; 2 Gy X 4) through assessing the concerted regulatory interaction between α-7nAchR/NF-kB signaling pathway and HMGB1/TLR4/NF-kB axis. Besides, the modulatory effect of Bs or O and IR on the Jak/STAT3 pathway has also been examined. Our findings demonstrated that CFA-induced arthritic rodents experienced a substantial reduction in body weight. As a result of rheumatoid arthritis, cachexia is distinguished by a loss of muscle mass and frailty. The loss of body weight in the Arthritis group rats could be due to an increase in leptin production caused by the CFA challenge, which may then contribute to a decrease in food intake, a lack of appetite, and ultimately, weight loss. 48 Weight loss in arthritic rats is primarily attributed to prolonged joint inflammation caused by local or general implications of pro-inflammatory cytokines generated by monocytes and macrophages, which could elicit muscle degeneration. 49
Paw size was noticed to increase progressively in CFA-induced arthritic rats throughout the experiment, whereas it was reduced after 14 days in the Bs-treated and O-treated groups. The alteration in paw size could be due to monocyte stimulation which could generate a range of pro-inflammatory cytokines, such as IL-6 and TNF-α, which initiate the synthesis of matrix metalloproteinases and acute phase proteins. Such reactions could elicit distinct humoral and cellular immune reactions. 50 An increased concentration of inflammatory cytokines was detected in experimentally induced arthritic rats. 51
Among the conditions that can cause oxidative stress is RA. The present study found that rats with arthritis treated with O or Bs and/or exposed to γ-irradiation had drastically changed oxidative status, the effect which was evidenced with different groups’ changes in MDA levels and SOD activity to varied degrees. A discrepancy between the production and inactivation of reactive oxygen species (ROS) results in cellular dysfunction and the development of undesirable inflammatory diseases like arthritis. 52 Free radicals are vital in cellular signaling; nevertheless, an overabundance of reactive oxygen species (ROS) can damage cellular genetic material, proteins, and lipids, as observed after IR exposure. 53 Persistent free radical production and chronic inflammation following radiotherapy or a radiation accident can impair organ function and lead to the development of a variety of diseases. 54
Based on the data of the current study, The inflammatory state biomarkers (pro-inflammatory mediators (TNF-α) and anti-inflammatory cytokines (IL-10 and IL-6)) alongside TLR4, were increased as shown in Figure 2. The finding which is in agreement with that TNF-α increases in serum of rheumatoid arthritis patients and induces local joint tissue deterioration and additional clinical symptoms. 55 IL-6 and other pro-inflammatory cytokines, including IL-10 and TNF-α, collaborate synergistically to augment inflammatory responses. Increased concentrations of chemokines and pro-inflammatory cytokines (IL-10, IL-6, TNF-α), are secreted by synovial tissue. 56 Our findings are in agreement with Abo-Zaid et al. who found that serum concentrations of transaminases (ALT and AST), were all diminished by Bs. 57 It was also noticed that TLRs was increased and significantly expressed in synovial fibroblasts even in the early phases of RA, 54 the finding which is in support to our finding. Toll-like receptors (TLRs) have recently been implicated in RA inflammation, playing a pivotal role in identifying molecular patterns associated with pathogens, initiating an innate immune response, and regenerating damaged tissues. 58 The activation of NF-κB and many other inflammatory pathways occurs during the progression of inflammation due to the stimulation of TLR4 on the cell membrane. 59 The role of NF-kB in arthritis is to play a pivotal role in the cartilage destruction and inflammation associated with RA through regulation of synthesis and secretion of inflammatory cytokines such as TNF-α. 54 TNF-α facilitates inflammatory cell infiltration in the joints by promoting adhesion of neutrophils and lymphocytes to endothelial cells. 60 IL-6 synergistically augments the inflammatory actions of IL-1β in synovial cells. 61 IL-10 is a potent anti-inflammatory cytokine that inhibit the progression and cartilage destruction in RA. The expression of these inflammatory cytokines is under the control of NF-kB, suggesting that NF-kB could be one of the master regulators of inflammatory cytokine production in RA. 62 The transcriptional factor NF-KB plays an vital role in arthritis by transcribing genes of cytokines contributing to the inflammation, and the elevated level of NF-KB seems to be reported in arthritis patients. 63
The JAK, STAT, NFKB, and HMGB protein expressions were detected in kidney, liver, lung and heart tissues using western blotting. As in rheumatoid arthritis, there is a correlation between the degree of inflammation and an increased risk of cardiovascular, pulmonary, hepatic, and skeletal diseases. 64 The intracellular JAK-STAT signalling regulates the cell proliferation, apoptosis and inflammation-like immune responses which seem to be involved in pathological conditions related to inflammation like arthritis. 65 IL-6 binds to the type-1 cytokine receptor activating JAK1 and JAK2 phosphorylating STAT3, and the STAT3 dimmers translocated to the nucleus stimulating the transcription of genes involved in acute-phase immune reactions causing inflammation. 66
Prior research indicates that IL-6 cytokines activate STAT-3, and the suppressive effect of Bs may be attributed to its anti-inflammatory properties. 48 Our results are in agreement with the previous results found that CFA-treated rats showed increased expression of STAT-3, and Bs significantly reduced the expression of STAT-3. 67 Moreover, Wang et al. 68 revealed that by stimulating the inflammatory NF-κB signaling pathway, IR induces disruption of cellular junctions. This is evident through the augmentation of oxidative stress and the elevation of cytokine concentrations, such as IL-6, IL-10 and TNF-α found within our research. In particular, HMGB1 interacts with TLR4, inducing cytokine production and inflammation. 69 HMGB1-stimulated peripheral blood monocytes release TNF-α, IL-10 and IL-6. 70 Supporting to our finding of increasing levels of HMGB, HMGB1 had been implicated in many inflammatory responses and autoimmunity, 71 where it could induce innate immune activation and inflammation amplification through its interactions with multiple receptors, including Toll-like receptors (TLR2 and TLR4) . 70
According to the RTqPCR results in kidney, heart, lung and liver; the IKB gene, TNF-α and 7-NACH mRNA expression varied compared to normal control group (p < .05; Figure 4). In our results, the decrease in α-7nAChR in arithritic group could be associated with a reduction in IL-10 level, as Gowayed et al. 72 manifested. Along with the well-established cholinergic anti-inflammatory pathway, this accelerates protein citrullination and the progression of rheumatoid arthritis. 47 Betasitosterol increase α-7nAChR compared to arithritic group (Figure 4), which may be due to the susceptibility of the α7nAChR pathway to rapid disruption in pro-inflammatory environmental conditions.
The present study found that exposing arthritic rats to fractionated whole body γ-irradiation (2 Gy/fraction for four consecutive weeks; A + R group) led to marked deterioration in the clinical arthritic signs, biochemical indices of oxidative stress and inflammation in liver, kidney, lung, and heart tissues (Figure 5), in addition to degenerative lesions in the ankle joints which are in line with the study of Thabet et al. 73 The observed results may be ascribed to an excessive generation of reactive oxygen species (ROS). This generates oxidative damage to biomolecules, linked to the synthesis of pro-inflammatory mediators. Meanwhile, when exposed to IR, reactive oxygen species (ROS) impair cellular antioxidant defense mechanisms by reducing glutathione levels and antioxidant enzyme activity. This is accompanied by an increase in detrimental oxidative molecules, including malondialdehyde (MDA), a prominent marker of lipid peroxidation. 74
In this study, the data of the CFA group revealed joint, liver, lung, kidney, and heart damage as manifested by decreased body weight, elevated hepatotoxicity and renal biomarkers, extensive histopathological degenerative lesions, excessive levels of TLR4, NF-kB, IL-6, provoked systemic inflammatory response, increased lipid peroxidation marker (MDA) associated with a notable reduction in antioxidant system (SOD) and IL-10. According to our findings, CFA significantly increases ROS and MDA concentrations while decreasing antioxidant SOD, α7nACHR, and IL-10 by a more pronounced margin (Figure 5). As a result of the harmful properties of these ROS, the body has developed an abundance of antioxidant mechanisms to prevent the damage it causes. According to research, reactive oxygen species (ROS) are a stimulatory signal to initiate the NF-kB pathway, which subsequently induces modifications in various pro-inflammatory cytokines and transcription mediators. The present experimental investigation observed that adjuvant-treated arthritic rodents were immunized with reduced levels of endogenous antioxidant SOD.
Recently, researchers have also examined the impact of Bs on additional facets of RA. BS decreased ankle edema, cytokine levels, and collagen-specific antibody concentrations in CIA mice by inducing macrophage polarization toward an anti-inflammatory phenotype. 48 In agreement to our results, Bs significantly increased body weight via an increase in the absorption of leucine and glucose via the intestine. 75
We investigate the function of inflammatory cytokines and lymphocytes in CFA-induced arthritis and the underlying molecular and cellular mechanisms in the present experimental investigation. Bs significantly diminished the inflammatory response in the synovial cavity of CFA-induced RA rodents. They decreased the arthritic grading of synovitis by lowering the level of IL-10, IL-6, and TNF-α in the Serum of experimental rats, as indicated before, according to Zhang et al. 75 In comparison to arthritic rodents, Bs significantly increased the level of endogenous antioxidant SOD. The elevated levels of TLR4 expression and NF-κB activation linked to inflammatory responses were substantially suppressed by Bs. 76 The expression of inflammatory mediators (interleukin-6 (IL-6), inducible nitric oxide (iNOS), tumor necrosis factor-α (TNF-α), and cyclooxygenase-2 (COX-2)) that LPS induces can be diminished through the administration of BS (Figure 6).
The treated groups with ondansteron showed a significant decrease in studied parameters in the present study. These results follow Silverman et al.77, who showed that O effectively decreases TNF-α levels significantly. These results suggest that O may reduce pro-inflammatory cytokines by inhibiting serotonin receptors on intestinal macrophages. 78 Our study found that in the O injection group, there was a significant decrease in interleukin (IL)-6, along with a reduction in the inflammatory damage to different tissues under study. It was previously found that therapeutic effect of O in patients with acute kidney injury may be elicited by regulating the NF-KB and JAK-STAT pathways. 79
Elevation of the studied biochemical markers is also reflected in histological changes in joints (Figure 6). Control animals show the transtibial joints with no infiltration and bone erosion. However, in CFA-induced arthritic rats, there was an increase in synovial cell linings, a decrease in synovial space, the creation of pauses, and the formation of new blood vessels. These transtibial joint histological alterations are similar to a previous research84. Bs or O treatment retards inflammatory cell infiltration and synovial hyperplasia. Thus, less bone destruction and joint space narrowing were observed than in CFA-induced arthritic rats. Earlier studies reported diminishing joint destruction by Bs and O treatment in arthritic rats. Bs significantly reduced the humoral immune reaction due to the suppression of acute inflammation via down-regulating the vascular permeability and inhibiting the pro-inflammatory cytokines. 67 Adjuvant develops the secondary response after 2 weeks and is significantly reduced by Bs. CFA-induced secondary lesions were exposed to delayed hypersensitivity reactions, and Bs also had an apparent effect on this, showing its antiarthritic effect. 28 Of note, this study did have some limitations, the first limitation is the extensive range of biochemical parameters and indicators that offer comprehensive mechanistic insights and detail on how BS and O may intervene with inflammatory events and oxidative stress in arthritic disorders, these cannot be investigated completely in our research because of the restricted sample size permitted by the ethical regulations of the animal ethics committee, which are adhered to by this study and all animal research (the more factors to be studied, the more samples that we needed). Hopefully, in further investigations, we will conduct more comprehensive detectors and indicators that accomplish these objectives. Another limitation of this study is lack of earlier researchs on the impact of ionizing radiation on arthritis and the investigated research avenue. Also, this study also has a drawback in that X-ray of the bone had to be assessed and will be examined further in subsequent investigations. Finally, to fully understand the mechanism of ondansetron (O) or β-sitosterol (BS) against inflammatory reactions and oxidative stress that complicate arithritis inflammation in the liver, kidney, lung, and heart of arthritic and arthritic irradiated rats, further mechanism research is required, there are undoubtedly still a lot of information gaps that need to be filled and studied.
Conclusion
Taken together, the obtained data imply that Beta-sitosterol (BS) or Ondansteron (O) were able to mitigate the detrimental impacts induced by gamma irradiation in arthritic irradiated rats and might efficiently improve the inflammatory response and oxidative stress in articular and extra-articular organs through their potencies as antioxidant and anti-inflammatory potential therapies. Thus, it might be recommended to include them in therapeutic countermeasures with exacerbated inflammatory responses, such as arthritic disorders inevitably exposed to a second stressor, such as gamma radiation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
