Abstract
Background
Epilepsy is a complex neurological condition characterized by recurrent seizures due to abnormal neuronal activity.
Objectives
The major aim of this study is to examine the therapeutic activities of leonurine in a pentylenetetrazole (PTZ)-challenged epileptic seizure model in rats.
Materials and Methods
Epilepsy was initiated in the rats by administering 45 mg/kg of PTZ. Rats received pretreatment of leonurine (10 mg/kg) and the standard drug diazepam 30 min before the PTZ treatment. Seizure severity was evaluated in both the control and experimental rats. The levels of oxidative stress indicators, inflammatory cytokines, ion transports, and neurotransmitters were assessed utilizing the appropriate test kits. Molecular markers such as cytochrome-c (cyt-c), caspase-3, cyclooxygenase-2 (COX-2), toll-like receptor 4 (TLR-4), mammalian target of rapamycin (mTOR), and nuclear factor kappa B (NF-κB) concentrations were studied using respective kits. Histological studies were conducted on the brain tissues of the experimental rats.
Results
Leonurine treatment significantly diminished seizure severity and increased locomotor activity in the epileptic rats. Leonurine treatment also effectively regulated neurotransmitters and ion transport mechanisms, reduced oxidative stress, decreased TLR-4 and high mobility group box 1 (HMGB1), decreased inflammatory cytokine, regulated protein kinase B (AKT)/mTOR axis, and diminished caspase-3, cyt-c, COX-2, and NF-κB concentrations in both cortex and hippocampal regions of the rats with epilepsy. The findings of histopathological studies also proved the therapeutic effects of leonurine.
Conclusion
It may be concluded that leonurine offers a promising therapeutic candidate to treat epilepsy. Further studies are necessary to understand the underlying mechanisms of leonurine’s therapeutic benefits on epilepsy.
Introduction
Epilepsy is a chronic neurological condition characterized by recurrent seizures. This condition affects more than 50 million individuals worldwide annually (Shen et al., 2022). Epilepsy poses a remarkable burden on both individuals and healthcare systems. In many low-income countries, the treatment gap, where affected individuals do not receive consistent treatment, is estimated to be greater than 60%. The condition is connected with a risk of premature mortality. Furthermore, epilepsy is generally comorbid with mental health disorders, with a majority of adults with epilepsy experiencing depression (Ioannou et al., 2022). The pathophysiology of epilepsy is complex and involves various underlying mechanisms. Genetic factors, structural abnormalities, metabolic disturbances, and even infectious agents have been implicated in the development of epilepsy. The disorder is characterized by changes in gene expressions that regulate neurotransmitter signaling, ion channels, gliosis, and inflammation. These changes can lead to an imbalance between excitation and inhibition within the brain, ultimately resulting in the occurrence of seizures (Boleti et al., 2024).
Animal models play an essential role in studying its underlying processes and developing effective treatments. The pentylenetetrazole (PTZ)-induced epilepsy model in rats is an extensively utilized experimental approach that has contributed significantly to the advancement of epilepsy research (Ngoupaye et al., 2022). The PTZ model is a chemical-induced seizure model, where the administration of PTZ, a gamma-aminobutyric acid (GABA) antagonist, triggers a series of seizure-like events in rats. This model mimics certain aspects of human temporal lobe epilepsy, including the development of spontaneous recurrent seizures following an initial seizure-inducing event. The model has been instrumental in unraveling the pathophysiological changes associated with epilepsy, such as neuronal hyperexcitability, synaptic reorganization, and the development of pharmacoresistance (Singh et al., 2021). One of the major advantages of the PTZ model is its capacity to reliably induce seizures in a controlled manner, allowing for the systematic investigation of various anti-epileptic drugs and their mechanisms of action (Hoeller et al., 2017). Furthermore, the model has been utilized to study the role of neurotransmitter systems, ion channels, and signaling pathways in the pathogenesis of epilepsy, providing insights that can inform the development of novel therapeutic strategies (de Souza et al., 2019).
Despite the availability of several drugs, a substantial portion of epileptic patients still experience intractable seizures, highlighting the need for alternative treatment strategies (Ghosh et al., 2023). The current landscape of epilepsy treatment is oppressed with limitations. It is estimated that one-third of epileptic patients do not respond to pharmacological treatment. Moreover, many of the available anti-seizure drugs are connected with severe side effects, ranging from cognitive impairment to life-threatening complications. These limitations have fueled the quest for novel, more effective, and safer therapeutic approaches (Anand et al., 2024). The need for new therapeutic targets has led researchers to study the potential of plant-based natural compounds as a promising avenue for addressing the unmet needs in epilepsy management. Leonurine is a natural bioactive alkaloid compound isolated from Herba leonuri. Its broad spectrum of pharmacological properties has gained much interest, including anti-tumor, anti-inflammatory, and antioxidant activities (Huang et al., 2021). Furthermore, it has cardioprotective (Gao et al., 2010), anti-depressantlike effects (Jia et al., 2017), anti-neuroinflammatory (Jin et al., 2019), neuroprotective (Deng et al., 2022), anti-Alzheimer’s disease (Xie et al., 2023), and anti-aging (Chen et al., 2019) properties. There are no documented studies on the therapeutic effects of leonurine’s protective effects on epileptic seizures. The primary objective of this work is to assess the therapeutic activities of leonurine in a PTZ-induced epileptic seizure in rats.
Materials and Methods
Experimental Rats
Six to eight-week-aged Wistar rats weighing over 210–230 g were utilized for this work. Rats were caged in sterile polyacrylic enclosures inside a conventional laboratory setting, maintained at a constant temperature of 22°C ± 3°C, humidity levels of 55%–65%, and a 12-h dark/light series. The rats were granted free access to food and purified water throughout the study. The protocols for the animal experimentation were verified and approved by the Institutional Animal Ethics Committee.
Experimental Design
The rats were allocated into four groups, each comprising six rats (n = 6). Group I rats were controlled and only administered 0.9% saline. To initiate epileptic seizures in group II rats, PTZ (45 mg/kg), diluted in saline (0.9%), was delivered via intraperitoneal injection. Group III rats were administered 10 mg/kg of leonurine via the oral route 30 min prior to the PTZ challenge. Group IV rats were treated with diazepam (2 mg/kg) 30 min before the PTZ challenge. The rats subjected to PTZ treatment were meticulously monitored for 30 min to assess the convulsions, the duration of tonic/clonic seizures, and the incidence of fatalities. All acquired results were meticulously organized and tabulated.
Induction of Kindling
The subconvulsive concentration of PTZ (35 mg/kg) was administered (i.p.) to the rats 15 times over intermittent days. The rats were monitored meticulously for 30 min prior to the PTZ administration to identify the commencement of clonic–tonic seizures. The classification of seizures was studied using the scaling method (Schroder et al., 1993), wherein stage 0 indicates no response, stage 1 denotes mouth and facial jerks, stage 2 signifies waves of axial convulsion, stage 3 represents myoclonic jerks, stage 4 corresponds to clonic convulsions, and stage 5 refers to tonic–clonic seizures.
Following the termination of seizures, the locomotor response of all rats was evaluated using the actophotometer. Animals were individually positioned on the actophotometer, and their activity was monitored for 5 min, with findings expressed as counts per 5 min. Subsequently, the depressive behaviors of the rats were studied using forced swim test, wherein they were placed in a glass cylinder measuring 25 × 12 × 25 cm3, filled with water to a height of 15 cm for a duration of 5 min, during which the immobility period was recorded.
Preparation of Brain Tissue Homogenates
Subsequent to the assessment of behavioral alterations, the rats were sedated and sacrificed. Subsequently, brain tissues were extracted from all animals and processed with saline solution. Subsequently, brain tissue homogenates were produced utilizing 0.1 M saline. The tissue homogenate was subsequently centrifuged for 15 min at 5,000 rpm, and the supernatant was employed for further biochemical analyses.
Analysis of Dopamine, Glutamate, GABA, Calcium Adenosine Triphosphatase (Ca+-ATPase), and Sodium–Potassium Adenosine Triphosphatase (Na+K+-ATPase) Concentrations
To analyze the concentrations of glutamate, dopamine, and GABA in the brain tissue homogenates, the diagnostic kits obtained from Abcam, USA, were employed. The tests were performed using the manufacturer’s instructions. The concentrations of Na+K+-ATPase and Ca+-ATPase enzymes in the brain tissue homogenates were analyzed using test kits from Elabscience, USA.
Analysis of Oxidative Stress Marker Levels
The malondialdehyde (MDA), nitric oxide (NO), glutathione (GSH), and superoxide dismutase (SOD) concentrations in the brain tissue homogenates were quantified using commercial kits, with tests conducted in triplicate following the manufacturer’s specifications (Abcam, USA).
Analysis of Biochemical Marker Levels in the Brain Tissues
The concentrations of neuronal nitric oxide synthase (nNOS), inducible NOS (iNOS), interleukin (IL)-1β, interleukin-1 receptor type 1 (IL-1R1), IL-6, and tumor necrosis factor-alpha (TNF-α) in the brains of the experimental rats were assessed using respective diagnostic kits (Abcam, USA). The concentrations of toll-like receptor 4 (TLR-4) and high mobility group box 1 (HMBG1) in the brains were assessed using commercial diagnostic kits (Elabscience, USA). The concentrations of mammalian targets of rapamycin (mTOR) and protein kinase B (AKT) in both the cortex and hippocampus were assessed using commercial diagnostic kits (LSBio, USA).
Analysis of Apoptotic Marker Levels in the Brain Tissues
The concentrations of apoptotic biomarkers, including caspase-3, cytochrome-c (cyt-c), cyclooxygenase-2 (COX-2), and nuclear factor kappa B (NF-κB) in both the cortex and hippocampus portions of the experimental rats were studied using respective diagnostic kits (Abcam, USA).
Histopathological Analysis
Hippocampal tissues of rats were excised and preserved in 10% formalin for 24 h before histological examination. After dehydration, paraffin embedding, and slicing into 5 µm sections, the brain tissues were stained with eosin and hematoxylin and then studied under a microscope at 40× magnification to analyze the histopathological changes.
Statistical Analysis
The findings were illustrated as a Mean ± Standard Deviation (SD) of three replicate assays. A one-way analysis of variance (ANOVA) and Tukey’s post hoc test were performed to scrutinize statistical significance, with p < .05 as the significance level.
Results
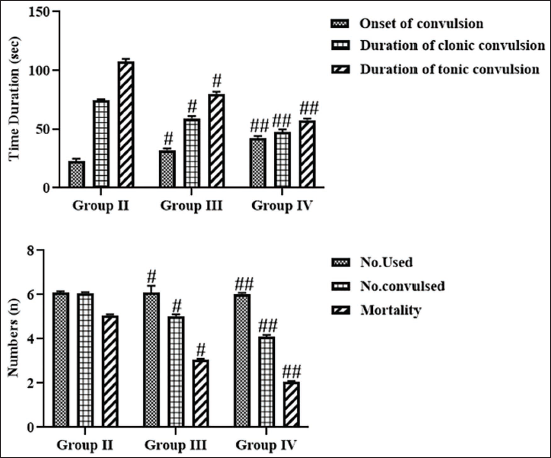
Effect of leonurine on the onset of convulsion and clonic/tonic convulsion duration, and mortality rates in experimental rats.
The results of leonurine on the onset and duration of convulsions and mortality rates in experimental rats were presented in Figure 1. The PTZ-triggered epileptic rats exhibited a higher incidence and duration of both convulsions. The death rates were also elevated in the PTZ-stimulated convulsed rats. Captivatingly, the treatment of 10 mg/kg of leonurine substantially decreased the duration of clonic/tonic convulsions in epileptic rats. Furthermore, the leonurine treatment also reduced the death rates in the PTZ-induced epileptic rats. Standard drug diazepam also diminished the duration of convulsions and reduced mortality in epileptic rats, which further supports the activity of leonurine.
Effect of Leonurine on the Onset of Convulsion, Clonic/Tonic Convulsion Duration, and Mortality Rates in the Experimental Rats. The Findings Are Depicted as a Mean ± Standard Deviation (SD) of Three Replicate Assays. Each Data Were Tested Using One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Test. An Asterisk “#” Specifies the Statistical Significance (p < .01) Level When Compared With the Control Group (Group I), While an Asterisk “##” Specifies the Statistical Significance (p < .05) Level When Compared with the Pentylenetetrazole (PTZ)-induced Group (Group II).
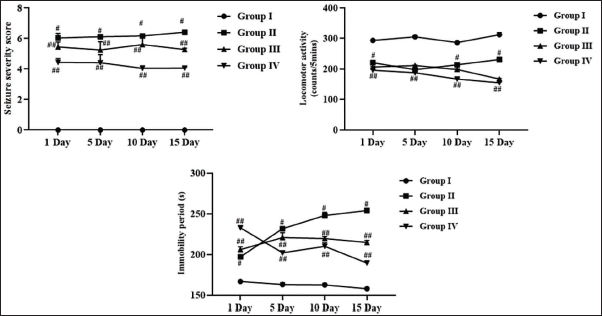
Effect of Leonurine on the Seizure Severity, Immobility, and Locomotor Activity in the Experimental Rats
The findings of leonurine on the locomotor activity, severity of seizure, and immobility in the rats are given in Figure 2. The rats with PTZ-induced epilepsy had reduced locomotor activity, prolonged immobility duration, and elevated seizure severity. Nonetheless, the 10 mg/kg of leonurine significantly enhanced locomotor activity, reduced immobility, and mitigated the severity of seizures in epileptic rats. The decreased seizure severity, reduced immobility duration, and heightened locomotor activity were also observed in epileptic rats treated with the standard drug diazepam.
Effect of Leonurine on the Seizure Severity, Immobility, and Locomotor Activity in the Experimental Rats. The Findings Are Depicted as a Mean ± Standard Deviation (SD) of Three Replicate Assays. Each Data Were Tested Using One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Test. An Asterisk “#” Specifies the Statistical Significance (p < .01) Level When Compared With the Control Group (Group I), While an Asterisk “##” Specifies the Statistical Significance (p < .05) Level When Compared With the Pentylenetetrazole (PTZ)-induced Group (Group II).
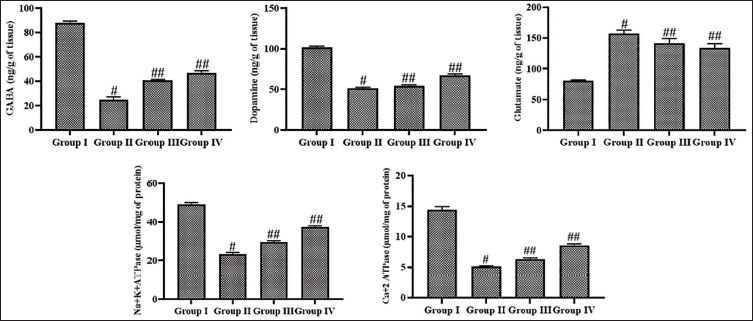
Effect of Leonurine on the Glutamate, Dopamine, GABA, Ca+-ATPase, and Na+K+-ATPase in the Experimental Rats
The results of leonurine on the concentrations of glutamate, dopamine, GABA, Ca+-ATPase, and Na+K+-ATPase in the experimental rats are given in Figure 3. The rats with PTZ-induced epilepsy exhibited increased glutamate levels, along with diminished concentrations of dopamine, GABA, Ca2+-ATPase, and Na⁺/K⁺-ATPase when compared with control. The epileptic rats treated with 10 mg/kg of leonurine demonstrated a considerable diminution in glutamate levels, alongside an enhancement in GABA and dopamine levels. The concentrations of Ca2+-ATPase and Na+K+-ATPase were enhanced by the leonurine treatment. The standard drug diazepam treatment also effectively regulated these biomarker levels in the epileptic rats.
Effect of Leonurine on the Glutamate, Dopamine, Gamma-aminobutyric Acid (GABA), Calcium Adenosine Triphosphatase (Ca+-ATPase), and Sodium–Potassium Adenosine Triphosphatase (Na+K+-ATPase) Levels in the Experimental Rats. The Findings Are Depicted as a Mean ± Standard Deviation (SD) of Three Replicate Assays. Each Data Were Tested Using One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Test. An Asterisk “#” Specifies the Statistical Significance (p < .01) Level When Compared With the Control Group (Group I), While an Asterisk “##” Specifies the Statistical Significance (p < .05) Level When Compared With the Pentylenetetrazole (PTZ)-induced Group (Group II).
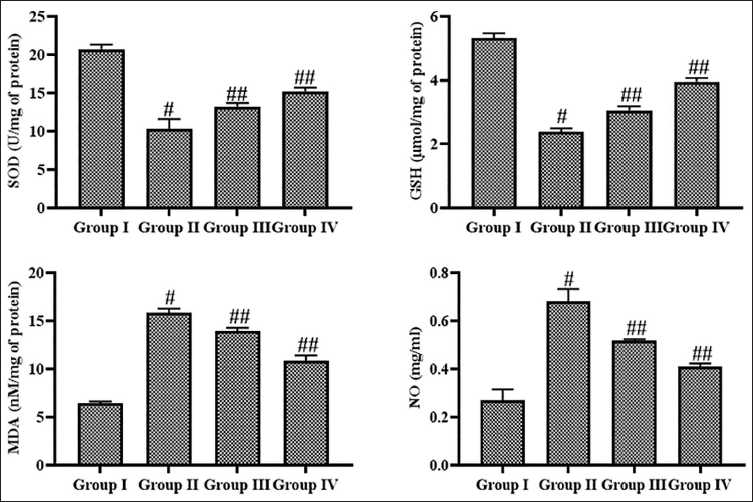
Effect of Leonurine on the Oxidative Stress Markers in the Experimental Rats
Figure 4 displays the results of leonurine on the oxidative stress markers in the brains of experimental rats. Elevated levels of NO and MDA, along with diminished GSH and SOD concentrations, were noted in the PTZ-induced epileptic rats. The 10 mg/kg of leonurine remarkably reduced the NO and MDA levels while enhancing SOD and GSH concentrations. The standard drug diazepam also reduced the NO and MDA levels while increasing antioxidant concentrations in the brain tissue of epileptic rats.
Effect of Leonurine on the Oxidative Stress Markers in the Experimental Rats. The Findings Are Depicted as a Mean ± Standard Deviation (SD) of Three Replicate Assays. Each Data Were Tested Using One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Test. An Asterisk “#” Specifies the Statistical Significance (p < .01) Level When Compared With the Control Group (Group I), While an Asterisk “##” Specifies the Statistical Significance (p < .05) Level When Compared With the Pentylenetetrazole (PTZ)-induced Group (Group II).
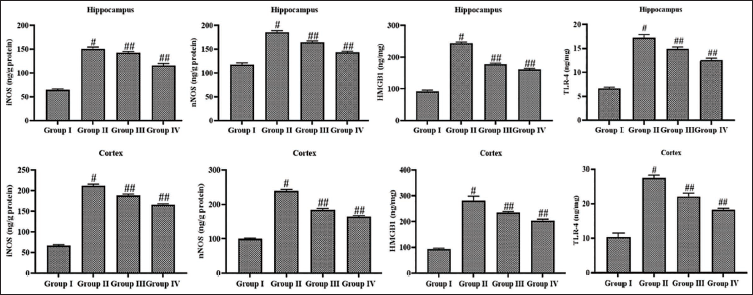
Effect of Leonurine on the nNOS/iNOS and TLR-4/HMBG1 Levels in the Brain Tissues
The results of leonurine treatment on the nNOS/iNOS and TLR-4/HMBG1 concentrations in cortical and hippocampal tissues are illustrated in Figure 5. The epileptic rats revealed a marked elevation in both TLR-4/HMBG1 and eNOS/iNOS concentrations in both cortical and hippocampus tissues when compared with the control group. Interestingly, the leonurine treatment at 10 mg/kg concentration successfully reduced the TLR-4/HMBG1 and nNOS/iNOS concentrations in the rats with PTZ-induced epileptic rats, which is similar to the effects observed with standard drug diazepam treatment (Figure 5).
Effect of Leonurine on the Neuronal Nitric Oxide Synthase (nNOS)/Inducible Nitric Oxide Synthase (iNOS) and Toll-like Receptor 4 (TLR-4)/High Mobility Group Box 1 (HMBG1) Levels in the Brain Tissues. The Findings Are Depicted as a Mean ± Standard Deviation (SD) of Three Replicate Assays. Each Data Were Tested Using One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Test. An Asterisk “#” Specifies the Statistical Significance (p < .01) Level When Compared With the Control Group (Group I), While an Asterisk “##” Specifies the Statistical Significance (p < .05) Level When Compared with the Pentylenetetrazole (PTZ)-induced Group (Group II).
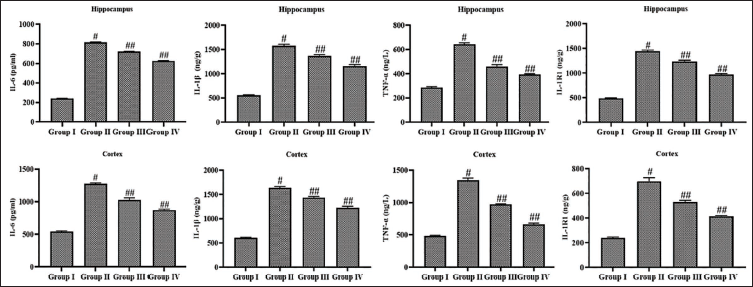
Effect of Leonurine on the Inflammatory Cytokines in the Brain Tissues of Experimental Rats
The effects of leonurine treatment on the inflammatory cytokines in both cortical and hippocampal tissues are presented in Figure 6. The rats with epilepsy exhibited a drastic elevation in the IL-1β, IL-1R1, IL-6, and TNF-α concentrations in both cortical and hippocampal tissues relative to the control. Captivatingly, the 10 mg/kg of leonurine successfully reduced these cytokine concentrations, which is similar to the findings of standard drug diazepam treatment (Figure 6).
Effect of Leonurine on the Inflammatory Cytokine Levels in the Brain Tissues of Experimental Rats. The Findings Are Depicted as a Mean ± Standard Deviation (SD) of Three Replicate Assays. Each Data Were Tested Using One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Test. An Asterisk “#” Specifies the Statistical Significance (p < .01) Level When Compared With the Control Group (Group I), While an Asterisk “##” Specifies the Statistical Significance (p < .05) Level When Compared With the Pentylenetetrazole (PTZ)-induced Group (Group II).
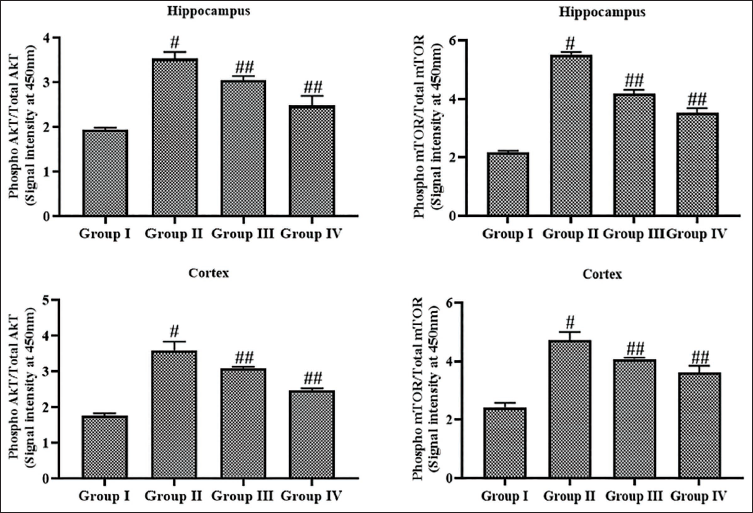
Effect of Leonurine on the mTOR and AKT Levels in the Brain Tissues of Experimental Rats
The concentrations of mTOR and AKT in the brain tissues of both control and treated epileptic rats are depicted in Figure 7. In comparison to control rats, the mTOR and AKT concentrations were progressively increased in the brain tissues of the epileptic rats. Nonetheless, leonurine treatment at a 10 mg/kg dosage remarkably reduced the mTOR and AKT concentrations in the epileptic rats. These findings were analogous to those observed in epileptic rats treated with the standard drug diazepam.
Effect of Leonurine on the Mammalian Target of Rapamycin (mTOR) and Protein Kinase B (AKT) Levels in the Brain Tissues of Experimental Rats. The Findings Are Depicted as a Mean ± Standard Deviation (SD) of Three Replicate Assays. Each Data Were Tested Using One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Test. An Asterisk “#” Specifies the Statistical Significance (p < .01) Level When Compared With the Control Group (Group I), While an Asterisk “##” Specifies the Statistical Significance (p < .05) Level When Compared With the Pentylenetetrazole (PTZ)-induced Group (Group II).
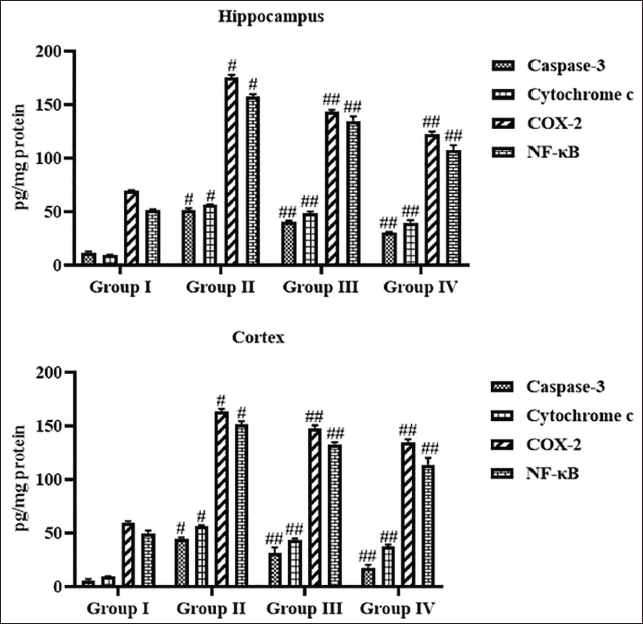
Effect of Leonurine on the Apoptotic Marker Levels in the Experimental Rats
The findings from an assay of the effects of leonurine treatment on the caspase-3, cyt-c, COX-2, and NF-κB levels in the experimental rats are illustrated in Figure 8. The PTZ-induced epileptic rats exhibit increased caspase-3, cyt-c, COX-2, and NF-κB concentrations. Captivatingly, the epileptic rats treated with 10 mg/kg of leonurine exhibited a substantial reduction in the caspase-3, cyt-c, COX-2, and NF-κB levels. The findings of leonurine treatment were similar to those of standard drug diazepam treatment in epileptic rats (Figure 8).
Effect of Leonurine on the Apoptotic Marker Levels in the Experimental Rats. The Findings Are Depicted as a Mean ± Standard Deviation (SD) of Three Replicate Assays. Each Data Were Tested Using One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Test. An Asterisk “#” Specifies the Statistical Significance (p < .01) Level When Compared With the Control Group (Group I), While an Asterisk “##” Specifies the Statistical Significance (p < .05) Level When Compared With the Pentylenetetrazole (PTZ)-induced Group (Group II).
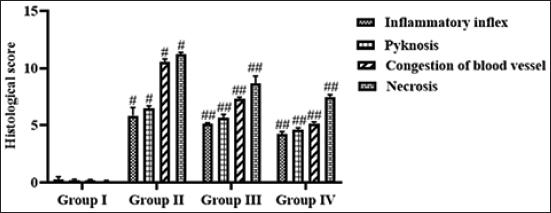
Effect of Leonurine on the Brain Histopathology of the Experimental Rats
Figure 9 represents the results of the histopathological study of the brain from the experimental rats. The untreated control rats exhibited normal brain histological structures without significant alterations, whereas the PTZ-induced epileptic rats exhibited various histological abnormalities in brain tissue, such as influx of inflammatory cells, pyknosis, blood vessel congestions, and neuronal necrosis when compared with the control. Interestingly, epileptic rats treated with leonurine at 10 mg/kg concentration exhibited a substantially decreased influx of inflammatory cells, pyknosis, blood vessel congestions, and neuronal necrosis in the brain tissues, which is similar to the findings observed with the standard drug diazepam treatment (Figure 9).
Effect of Leonurine on the Brain Histopathology of the Experimental Rats. The Findings Are Depicted as a Mean ± Standard Deviation (SD) of Three Replicate Assays. Each Data Were Tested Using One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Test. An Asterisk “#” Specifies the Statistical Significance (p < .01) Level When Compared With the Control Group (Group I), While an Asterisk “##” Specifies the Statistical Significance (p < .05) Level When Compared With the Pentylenetetrazole (PTZ)-induced Group (Group II).
Discussion
Epilepsy, a complex neurological condition, is characterized by recurrent seizures resulting from abnormal neuronal activity. The pathogenesis of epilepsy implicates a complex interplay of various neurotransmitters and ion transport mechanisms, which play an essential role in the regulation of neuronal excitability and the progression of seizures (Tandon et al., 2024). Neurotransmitters are the messengers that mediate communication between neurons in the brain. Numerous neurotransmitters, including dopamine, glutamate, and GABA, have participated in the pathogenesis of epilepsy. Dopamine, a key neurotransmitter involved in various brain functions, has been shown to have a complex and multifaceted role in epilepsy. Glutamate, the primary excitatory neurotransmitter in the brain, also participates in neuronal hyperexcitability and the generation of seizures (Teleanu et al., 2022). Conversely, GABA, the main inhibitory neurotransmitter, plays a critical role in regulating the equilibrium between excitation and inhibition in the brain (Perucca et al., 2023). Ion transport mechanisms, such as Ca2+-ATPase and Na+K+-ATPase, are pivotal for the maintenance of electrochemical gradients and the regulation of neuronal excitability. The Na+K+-ATPase, also known as the sodium–potassium pump, participates in numerous pathways and in the onset of epilepsy. Alterations in the activity or expression of these ion pumps can lead to disruptions in ion homeostasis, which can trigger the onset of epilepsy (Sun, Li, et al., 2022; Sun, Zheng, et al., 2022). In this work, the observed results clearly showed increased dopamine, GABA, Ca2+-ATPase, and Na+K+-ATPase concentrations and a subsequent reduction in glutamate concentration in the leonurine-treated rats with epilepsy. These findings evidenced that leonurine successfully regulated the neurotransmitters and ion transport mechanisms in the treatment of epilepsy.
It has already been suggested that oxidative stress serves a crucial role in the pathogenesis of epilepsy. Oxidative stress is characterized by an imbalance between reactive oxygen species (ROS) generation and its neutralization by the body’s antioxidant mechanisms, leading to cellular damage and impaired neuronal function (Yilgor & Demir, 2024). MDA, an indicator of lipid peroxidation, has been found to be elevated in the brain, blood, and cerebrospinal fluid of individuals with epilepsy. This indicates the presence of increased oxidative damage to lipids, which can disrupt membrane integrity and impair neuronal signaling (Sun, Li, et al., 2022; Sun, Zheng, et al., 2022). NO, a free radical that acts as a signaling molecule in the brain, also participates in the pathophysiology of epilepsy. Increased NO levels can result in excitotoxicity and contribute to seizure activity. NO is synthesized by three distinct isoforms of the enzyme NO synthase: nNOS, iNOS, and endothelial NOS. While nNOS is involved in physiological functions such as synaptic plasticity and neurotransmission, iNOS is upregulated in response to various inflammatory stimuli and can result in excessive NO production, contributing to excitotoxicity and neuronal damage. It has been demonstrated that the expression of iNOS and nNOS are augmented in epileptic brains, both in animal models and in patients with epilepsy (Sharma et al., 2019). On the other hand, antioxidants such as SOD and GSH play a crucial role in mitigating the harmful effects of oxidative stress. Decreased activity or expression of these antioxidants has been observed in individuals with epilepsy, suggesting a diminished capacity to counteract the adverse effects of ROS (de Melo et al., 2023). The present study has shown reduced MDA, NO, iNOS, and nNOS levels and increased SOD and GSH concentrations in the brain tissues of the leonurine-treated rats, which proves its antioxidant properties.
TLR-4 is a pattern recognition receptor that plays a critical role in the activation of innate immunity. Its involvement in epilepsy has been extensively studied, as it has been shown to participate in the onset of seizures. Activation of TLR-4 by endogenous ligands, such as HMGB-1, can initiate a cascade of inflammatory responses, resulting in the release of inflammatory cytokines and chemokines (Paudel et al., 2020). HMGB-1, on the other hand, is a damage-associated molecular pattern that is released by cells undergoing stress or injury. Increased HMGB1 levels have been noted in the brains of epilepsy patients, and it is believed to participate in the initiation of the disease. HMGB1 is a potent activator of TLR-4, and its interaction with this receptor can amplify inflammation, ultimately participating in the pathogenesis of epilepsy (Foiadelli et al., 2023). Furthermore, it has been suggested that HMGB1 may serve as an indicator of the early diagnosis of patients at high risk of developing epilepsy, and for monitoring disease progression and treatment response (Zhao et al., 2020). The present results have demonstrated reduced levels on both TLR-4 and HMGB1 in the cortex as well as hippocampal regions of the leonurine-treated epilepsy rats.
Neuroinflammation, a complex mechanism implicating the initiation of innate immunity, has been identified as a contributing factor to the seizure onset and the development of drug-resistant, focal-onset symptomatic epilepsies (Kwon & Koh, 2020). It has been suggested that the pathophysiology of epilepsy is connected with elevated levels of certain inflammatory cytokines, like TNF-α, IL-1β, IL-1R1, and IL-6. These cytokines play a crucial role in the modulation of learning, memory, and neuroplasticity, and their dysregulation has been connected with the onset of epilepsy (Soltani Khaboushan et al., 2022). TNF-α is a major cytokine that has been widely explored in the context of epilepsy. It has been shown to increase neuronal excitability by modulating the activity of various ion channels, thereby initiating the onset of seizures. Similarly, IL-1β has been found to enhance neuronal excitability and synaptic transmission, leading to the initiation and propagation of seizures (Wolinski et al., 2022). IL-1R1, the receptor for IL-1β, has also been participated in the pathophysiology of epilepsy. IL-6, another proinflammatory cytokine, has been found to play a complex role in the modulation of learning and memory processes. While some studies have highlighted that augmented IL-6 levels can impair cognitive function (Vishwakarma et al., 2022). The current results have evidenced the significant diminution in the TNF-α, IL-1β, IL-1R1, and IL-6 concentrations in both cortex and hippocampal regions of the brain tissues of leonurine-treated epilepsy rats, which highlights its anti-inflammatory properties.
The mTOR/AKT pathway is the key pathway that has participated in the onset of epilepsy. The mTOR signaling is a pivotal regulator of cellular mechanisms, like protein synthesis, cell growth, and metabolism. Dysregulation of this signaling has been observed in numerous neurological disorders, including epilepsy. The mTOR pathway is activated by the protein kinase B signaling cascade, which participates in the regulation of cellular survival, proliferation, and metabolism (Hodges & Lugo, 2020). In the context of epilepsy, the mTOR and AKT dysregulations have participated in the onset of the disease. Activating mutations in the phosphatidylinositol-3-kinase (PI3K) gene, which results in the activation of the AKT signaling, have been observed in some cases of epilepsy. Additionally, the inhibition of the mTOR pathway has been shown to have a therapeutic effect in animal models of epilepsy, suggesting the potential of targeting this pathway as a treatment strategy (Śmiałek et al., 2023). The results of this study have highlighted the decreased AKT/mTOR concentrations in both cortex and hippocampal regions of the brain tissues of leonurine-treated epilepsy rats.
Caspase-3 is a crucial executioner of apoptosis, and its activation has been implicated in the neuronal death associated with epilepsy (Unnisa et al., 2023). Cyt-c, a key molecule of the mitochondrial electron transport chain, can be released into the cytoplasm during apoptosis, activating caspase-3 and other downstream signaling cascades. COX-2 is an enzyme that plays a role in the inflammatory response and has been shown to be upregulated in the brain during seizures, contributing to the pathogenesis of epilepsy. NF-κB is a transcription factor that controls the expression of genes related to inflammation, cell survival, and other cellular processes (Sokolova et al., 2022). In this work, the rats with epilepsy demonstrated an increased caspase-3, cyt-c, COX-2, and NF-κB concentrations when compared with control. Interestingly, the leonurine treatment results in a substantial decrease in the caspase-3, cyt-c, COX-2, and NF-κB concentrations in both cortex and hippocampal regions of the rats with epilepsy.
Conclusion
The current findings indicate that leonurine pretreatment effectively mitigates the PTZ-induced seizures in rats. In epileptic rats, leonurine treatment successfully regulated the neurotransmitters and ion transport mechanisms, reduced oxidative stress, reduced the TLR-4 and HMGB1, decreased inflammatory cytokine levels, regulated AKT/mTOR axis, and diminished caspase-3, cyt-c, COX-2, and NF-κB concentrations in both cortex and hippocampal regions of the rats with epilepsy. Consequently, it may be concluded that leonurine may be a promising therapeutic candidate for treating epilepsy. Further studies are necessary to elucidate the underlying mechanisms of leonurine’s therapeutic benefits on epilepsy.
Footnotes
Abbreviations
AKT: Protein kinase B; ANOVA: Analysis of variance; Ca+-ATPase: Calcium adenosine triphosphatase; COX-2: Cyclooxygenase-2; cyt-c: Cytochrome-c; GABA: Gamma-aminobutyric acid; GSH: Glutathione; HMGB1: High mobility group box 1; IL-1R1: Interleukin-1 receptor type 1; IL-1β: Interleukin-1 beta; IL-6: Interleukin-6; iNOS: Inducible nitric oxide synthase; MDA: Malondialdehyde; mTOR: Mammalian target of rapamycin; Na+K+-ATPase: Sodium–potassium adenosine triphosphatase; NF-κB: Nuclear factor kappa B; nNOS: Neuronal nitric oxide synthase; NO: Nitric oxide; PI3K: Phosphatidylinositol-3-kinase; PTZ: Pentylenetetrazole; ROS: Reactive oxygen species; SD: Standard deviation; SOD: Superoxide dismutase; TCM: Traditional Chinese medicine; TLR-4: Toll-like receptor 4; TNF-α: Tumor necrosis factor-alpha.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This work has been approved by the Institutional Animal Ethical Committee of Xi’an TCM Hospital of Encephalopathy, Xi’an, Shaanxi, China.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
