Abstract
Background
Malignant tumors represented by breast cancer still have limitations in clinical treatment methods. Matrine is a compound with anticancer activity, but its inhibitory effect on breast cancer and its mechanism for regulating miR-188 are still unclear.
Objectives
Therefore, it is necessary to explore more effective breast cancer treatment methods and understand the intervention mechanism of matrine and its effect on miR-188.
Materials and Methods
A nude mouse model of breast cancer was in this study constructed and divided into a model group, 4 mg/kg group, 8 mg/kg group, drug group, and miR-188 mimic group. Different concentrations of matrine were administered intragastrically, and cell proliferation and apoptosis were observed. At the same time, a specific mechanism for miR-188 in the anti-breast cancer effect of matrine was explored by overexpressing miR-188.
Results
It was found that matrine inhibited the proliferation of breast cancer cells and caused cancer cell apoptosis. In addition, it inhibited cell migration in a dose-dependent manner. At the same time, miR-188 was lowly expressed in breast cancer and significantly increased after matrine treatment. Overexpression of miR-188 enhanced the antitumor effect of matrine. Phosphatase and tensin homolog (PTEN) was also activated in the surface by matrine, and expressions of matrix metalloproteinase-9 (MMP-9) and matrix metalloproteinase-2 (MMP-2) were inhibited. This study shows that matrine has a significant inhibitory effect on breast cancer, and its mechanism is related to upregulation of miR-188, activation of PTEN, and inhibition of MMP-9 and MMP-2 expressions.
Conclusion
miR-188 is involved in the anti-breast cancer process, which can be used as the theoretical basis of matrine potential drugs for breast cancer treatment. Meanwhile, miR-188 and matrine will also have great potential in developing new breast cancer treatment methods, which can provide ideas and a basis for further development of related studies.
Introduction
Breast cancer is one of the most common malignant tumors in women worldwide, and its incidence is increasing year by year. It has become the largest cancer in the world, and it is also one of the major challenges facing the public health field. Although existing therapeutic methods such as surgery, chemotherapy, radiotherapy, and targeted therapy have achieved certain efficacy, problems such as drug tolerance, side effects, and recurrence rate still plague clinicians and patients. Therefore, the identification of new and effective therapeutic targets for breast cancer is currently the focus of national research. In recent years, with the rise of bioinformation analysis technology, researchers have found that there is a large difference in gene expression in cancer during the process of exploring the mechanism of malignant tumor, which also provides an innovative theoretical basis for the mechanism study of breast cancer.
MicroRNA (miRNA), as an endogenous non-coding RNA molecule, plays a crucial role in regulating gene expression. It affects protein synthesis by binding to the 3′UTR region of the target mRNA, resulting in mRNA degradation or translation inhibition (Li et al., 2019; Yang & Wang, 2019). Studies have shown that miR-188 can reduce phosphatase and tensin homolog (PTEN) expression and further increase GSK3β phosphorylation at Ser9 to activate Wnt/β-catenin signaling in gastric cancer, thereby enhancing gastric cancer cell activities and tumor metastasis in vivo. This is consistent with the results by Wang et al., who studied the relationship between miR-188 and gastric cancer (Wang et al., 2019). The levels of miR-188 are increased in colorectal cancer. Wu et al. (2021) first determined that FOXL1 is a direct target for miR-188, which can upregulate Wnt signaling in colorectal cancer cells by knocking down FOXL1, thereby promoting cancer development. It has been reported that inhibition of miR-188 appeared to reverse this process. In addition, miR-188 in pancreatic cancer cells showed upregulation, and inhibition of miR-188-5p can inhibit LINC00491, thereby reducing ZFP91 expression and inhibiting pancreatic cancer development (Shi et al., 2019). It has been seen that miR-188 is related to a variety of cancers, but there are currently few reports on its role in breast cancer, so further research is still needed.
Matrine is mainly an alkaloid extracted from dried roots and plants of Sophora flavescens Aiton. It can effectively reduce the automaticity of cardiomyocytes and regulate the body’s immune function, and it also has a certain anti-inflammatory effect (Shi et al., 2019). In addition, matrine can inhibit the growth of tumor cells (Figure 1). Relevant studies have shown that it can cause mitochondrial dysfunction by regulating Bcl-2, Bax, p53, and Caspase, thereby releasing cytochrome C into the cytoplasm and further activating Caspase-9 and Caspase-3 to induce apoptosis (You et al., 2019). At the same time, matrine can increase the level of autophagy activity by downregulating the expression of autophagy-related genes such as PI3KIII, Beclin1, Atg5, and Bax. It can also further accelerate the autophagy process for liver cancer cells (Chi et al., 2019). It can also significantly upregulate p38MAPK, ERK, and JNK, and increase the levels of Caspase-8 and Fas to significantly inhibit tumor growth (Ma et al., 2017). At the same time, previous studies have found that matrine can also destroy the cellular metabolic process by inducing oxidative stress in the body and promoting the apoptosis of tumor cells to a certain extent (Cao et al., 2019; Mo et al., 2018). However, matrine has a wide range of mechanisms of action in the body, and its effects on different diseases are different. Although it has a relatively powerful antitumor effect, its potential mechanism of action in regulating miR-188 and its effect on human breast cancer is still unclear, so further research is needed.
Matrine Extraction Process and Research Ideas Diagram.
Materials and Methods
Experimental Materials
Cell Culture Material
Human breast cancer cell SKBR-3 (Wuhan Shangen Biotechnology, product number: SNL-058), MCF-12A (Shanghai Yingwan Biotechnology, C1751), MCF-10 (Wuhan Shangen Biotechnology, product number: SNL-225); normal human breast cells were purchased from Shanghai Kanglang Biotechnology Ltd.; trypsin-EDTA digestion solution (Beijing Solebao Ltd.); fetal bovine serum (FBS) (GIBCO, USA); Petri dish (Corning Company, USA); phosphate-buffered saline (PBS) buffer (Shanghai Xiaopeng Biotechnology Ltd.); cell culture flask (American Corning Company).
Antibody Material
Anti-matrix metalloproteinase-9 (MMP-9)/matrix metalloproteinase-2 (MMP-2) antibody (Shanghai Zeye Biotechnology) Technology Ltd.); anti-PTEN antibody (Abcam, UK); anti-GAPDH antibody (ab9485) (Abcam, UK); pERK1/2 antibody were purchased from the American Cell Signaling Company; SDS-polyacrylamide gel electrophoresis (SDS-PAGE) gel preparation kit (Shanghai Beyotime Reagent Company).
Experimental Intervention Material
Matrine (Shaanxi Haochen Biotechnology Ltd., CAS: 519-02-8) is extracted from the dried roots of S. flavescens. It can not only clear away heat and remove dampness but also has certain antitumor and antiallergic pharmacological properties. Tamoxifen (Shanghai Haoyuan Biomedical Technology Ltd., CAS: 10540-29-1); miR-188 mimic (Guangzhou Ribo Biotech); crystal violet dye (Foshan Nanhai Jiangshun Chemical Products Factory; CAS: 603-48-5); formaldehyde (Guangzhou Aiju Environmental Technology Ltd.); xylene (Jinan Shengquan Group Co., Ltd.); 1% eosin (alcohol-soluble) (DH0045) (Beijing Leigen Biotech Ltd.); hematoxylin dye (G1140) (Beijing Solebao Ltd.); neutral gum G8590 (Beijing Solebao Ltd.); horseradish peroxidase (Hubei Maoerwo, CAS: 9003-99-0).
Material Examination
Synthesis and construction of dual luciferase reporter vector (Hunan Fenghui Biotechnology Ltd.); Dual Luciferase Reporter Gene Assay Kit (Shanghai Yiji Industrial Ltd.); Methyl thiazolyl tetrazolium (MTT) (Shanghai Micro-Sensitive Automation Technology Ltd.); Western Protein Marker I (Wuhan Sevier). All kit tests are conducted in strict accordance with the instructions.
Construction of Nude Mouse Model of Breast Cancer
Twenty-five female specific pathogen-free (SPF) grade BALB/c-Nude mice were selected (Changzhou Cavins Laboratory Animal Co., Ltd.) and inoculated with SKBR-3 cells on the right chest wall of the nude mice under sterile conditions at 0.3 mL/mouse dose until the tumor grew to 0.8–1.0 cm. Moreover, it will be possible to touch the tumor to generate a hint of modeling success. Nude mice were randomly divided into the model group, 4 mg/kg group, 8 mg/kg group, drug group, and miR-188 mimic group. Mice in the 4 mg/kg group were intragastrically administered with matrine 4 mg/kg; while mice in the 8 mg/kg group were given matrine 8 mg/kg by gavage. Moreover, mice in the drug group were given tamoxifen single drug 8 mg/kg by gavage, while the miR-188 mimic group was given a tail vein injection of 4 mL/miR-188 mimic kg; the model group nude mice were given 0.9% sodium chloride solution in the same volume. The frequency of intervention was once a day for all mice, and the intervention time was all after the mice ate in the morning. The mice were continuously subjected to intervention treatment for 3 weeks, and their body weight was measured every 3 days. The Animal Ethics Committee of our hospital has approved this animal experiment.
Cell Culture
Breast cancer tissues were taken from nude mice from each group and immediately added with 10 mL of pre-warmed culture medium. Quickly, the tube was thawed in a water bath at 38°C, and then the tissue samples were transferred to a new culture vessel. We waited for the tissue to adhere before adding a new culture medium. The tissue samples were incubated for 2 days in an incubator at 37°C until tissue adhesion and growth were stable before washing and enzymatic digestion. When the cell density was between 70% and 80%, the cells were washed and enzymatically digested into single cells, and the sample cells were cultured.
Tumor Data Monitoring
The mass of each tumor was weighed, and the tumor inhibition rate and incidence rate were calculated. Tumor inhibition rate = (tumor weight of model group – tumor weight of administration group)/tumor weight of model group × 100%.
Transwell Detection of the Number of Tumor Cell Migration
After digestion with digestive juice, an FBS medium was added to terminate the digestion, and the Transwell experiment was performed. The cells were counted and placed in an EP tube, followed by a centrifuge to remove the supernatant, and the serum-free medium was added and resuspended. Then, Gibco serum was added for 24 hours. Further, the samples were fixed, stained with crystal violet, cleaned and fixed on a glass slide, observed, and counted through a microscope with a random field of view.
Hematoxylin and Eosin (HE) Staining to Observe Tumor Cell Morphology
After the tumor cells were fixed in paraformaldehyde, they were washed, dehydrated in ethanol, cleared in xylene, embedded in paraffin, sectioned, and then stained with HE and placed on glass slides, and the morphologies of tumor cells were observed through an optical microscope.
Scratch Test to Detect Tumor Cell Migration Ability
When the cell densities of each group were close, they were washed three times with PBS and then added to a serum-free medium for cell culture. The cells were then sampled and photographed at different periods.
MTT Assay was Used to Detect Tumor Cell Viability
The cells of each group were inoculated on 96-well plates and 3,000 cells/well were inoculated respectively, and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) was added to the incubator at 37°C and 5% CO2 for 4 hours. After the incubation, 100 µL dimethyl sulfoxide (DMSO) was added and shaken evenly to fully dissolve the formazan. Finally, the absorbance value of each hole was measured in the microplate reader.
Western Blot
Total protein was extracted with TriZol reagent, and its concentration was detected. The electrophoresed total protein was heated, electrophoresed using SDS-polyacrylamide gel (120 V, 100 minutes), and transferred to a polyvinylidene fluoride (PVDF) membrane for blocking, and primary antibodies were added with miR-188 (1:1,000), β-actin (ab8226, 1:1,000), and secondary antibodies (goat anti-rabbit 1:2,000), followed by incubation at 4°C. After further repetition of incubation three times washing of the membrane and addition of horseradish peroxidase, the reaction changes were observed and recorded.
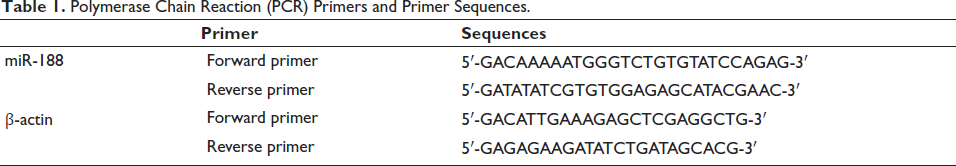
Polymerase Chain Reaction (PCR) Detection of miR-188 Expression
Total mRNA was extracted from breast cancer tissue by grinding and homogenizing it with liquid nitrogen and was transcribed into cDNA. Analysis was performed by real-time PCR system analysis using primers shown in Table 1, with β-actin as a reference. Relative levels were estimated using the 2−∇∇Ct method.
Polymerase Chain Reaction (PCR) Primers and Primer Sequences.
Dual Luciferase Assay to Detect Relative Luciferase Activity
FBS culture medium was added to cells in each group to terminate digestion, and transfection liquid was added to a 96-well plate for secondary cell culture. The samples were washed with PBS, added with cell lysis buffer, and incubated for 5 minutes. Then, the luciferase reaction solution and Renilla luciferase reaction solution were added, their activity was detected, and the time was controlled within 30 minutes. R. luciferase was used as the internal control, and the activation degree for the reporter gene between different samples was compared based on the measured data values.
Statistical Method
The data were analyzed using SPSS21.0 and GraphPad Prism software. p < 0.05 was used as the test standard for significant results.
Results
Establishment and Validation of a Nude Mouse Model of Breast Cancer
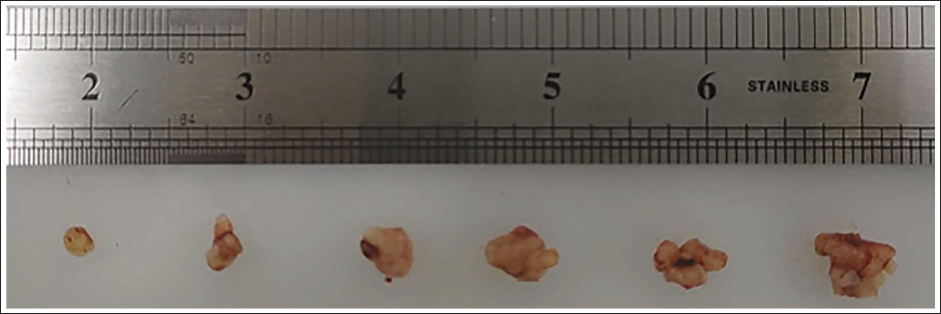
To observe whether the modeling of nude mice with breast cancer was successful, the breast cancer tumor tissues of nude mice were measured every 3 days, and the mass of all mice was (1.65 ± 0.21) g, and the volume was (1.21 ± 0.11) cm3. After observation, it was found that the breast cancer tumor tissues of nude mice became larger and larger as time went by, indicating that the nude mouse model of breast cancer was successfully constructed. No mice died during the modeling process, and the modeling success rate was 100% (Figure 2).
Tumor Size of Nude Mice in the Model Group was Measured Every 3 Days.
Matrine Inhibits Breast Cancer Cells Growth
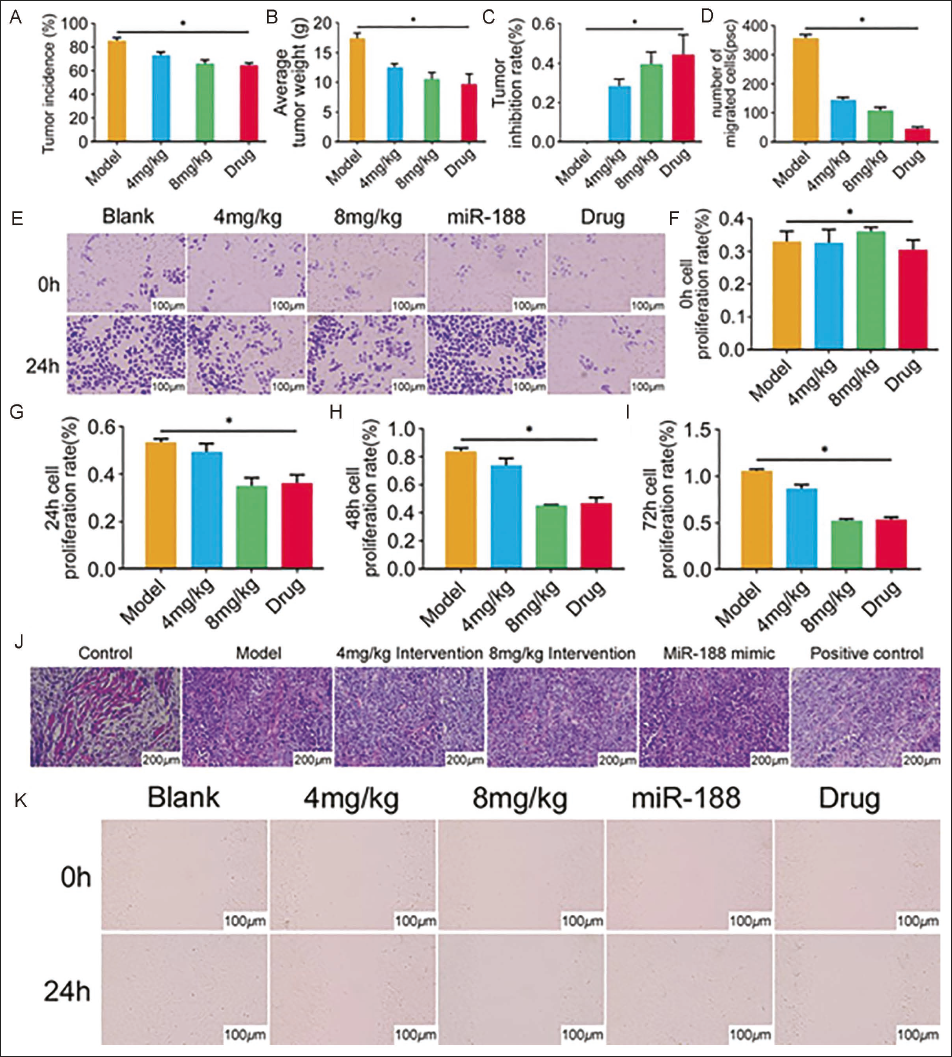
We found that the tumor incidence rate and average tumor mass were significantly lower than the model group after adding matrine at various doses, but were still higher than the drug group. 8 mg/kg group was significantly lower than the 4 mg/kg group (Figure 3A and B). There was almost no tumor inhibition rate in the model group, while the matrine group increased in a dose-dependent manner at each dose, but was still lower than the drug group (all p < 0.05, Figure 3C). Further cell counting showed that the number of cell migrations in the 8 mg/kg group was (100.65 ± 16.32), significantly less than that in the 4 mg/kg group (127.39 ± 15.47) (t = 2.659, p = 0.029, Figure 3D). The Transwell test results showed that the cell migration number of matrine group was significantly lower than that of the model group but higher than that of the drug group (Figure 3E). The results from cell viability detection by MTT showed that there was not much difference in the cell proliferation rate between groups at 0 hours (Figure 3F). The proliferation rate of precursor cells increased after 24 hours, but the cell proliferation rate group decreased for each dose of matrine (vs. model group), especially the 8 mg/kg group, which had the most obvious decrease, which was even lower than that of the drug group (all p < 0.05, Figure 3G). Although the overall cell proliferation rate was still increasing after 48 hours, the trend among each group was still the same as that at 24 hours (all p < 0.05, Figure 3H). The same trend was observed at 72 hours (all p < 0.05, Figure 3I). The results showed that the cell proliferation rate was positively correlated with time but negatively correlated with matrine concentration. Moreover, results from the HE staining showed that the staining color was relieved, and cell nuclei were shrunk after adding various doses of matrine, but it was still worse than that in the drug group. Further careful observation also revealed that the staining situation in the 8 mg/kg group was reduced compared with the 4 mg/kg group (Figure 3J). Scratch test results showed that there was no difference in cell migration among all groups at 0 hours, and cells migrated after 24 hours. The migration ability of matrine group was significantly lower than that of the model group but higher than that of the drug group. Further observation found that the migration ability of matrine 8 mg/kg group was reduced compared with the 4 mg/kg (Figure 3K). Therefore, matrine had a specific effect on breast cancer cell growth.
Matrine can Inhibit the Growth of Breast Cancer Cells. (A, B, C) Tumor Data Monitoring; (D, E) Transwell Detection of Cell Migration Number; (F, G, H, I) 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) Detection of Cell Viability; (J) Hematoxylin and Eosin (HE) Staining to Observe Cell Morphology; (K) Scratch Test to Detect Cell Migration Ability (p < 0.05).
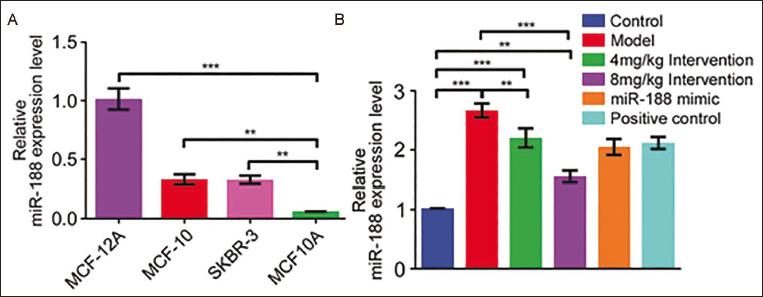
The mechanism by which matrine inhibits the growth of breast cancer cells is related to miR-188. It was found that human breast cancer cells MCF-12A, MCF-10, and model SKBR-3 significantly differed from human normal breast cells MCF10A. Compared with miR-188, the expression level decreased significantly (all p < 0.05, Figure 4A). The expression level of miR-188 increased simultaneously when we added various doses of matrine, compared with the model group (all p < 0.05, Figure 4B).
Matrine’s Inhibition of Breast Cancer Cell Growth is Related to miR-188. (A, B) Polymerase Chain Reaction (PCR) Detection of miR-188 Expression Level (p < 0.05).
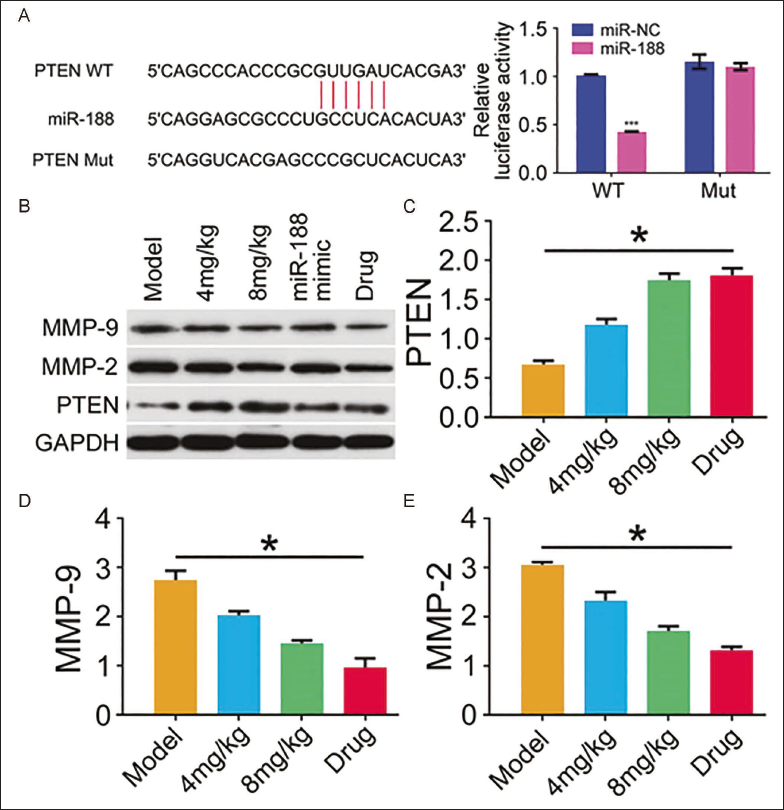
miR-188 can target and activate PTEN to inhibit MMP-9 and MMP-2 expression, thereby inhibiting the growth of breast cancer cells. The relative luciferase activity of cells transfected with PTEN-WT with miR-188 mimic was reduced, and there was no difference in cells transfected with PTEN-Mut (Figure 5A). In the Western blot experiment, we found that PTEN expression increased after adding matrine, while MMP-9 and MMP-2 expressions decreased in a dose-dependent manner (Figure 5B–D). Therefore, matrine targets and activates PTEN through miR-188 to inhibit MMP-9 and MMP-2 expression, thereby inhibiting the growth of breast cancer cells.
miR-188 can Target and Activate Phosphatase and Tensin Homolog (PTEN). (A) Dual-luciferase Experiment Detects Relative Luciferase Activity; (B, C, D, E) Western Blot Detects Matrix Metalloproteinase-2 (MMP-2), Matrix Metalloproteinase-9 (MMP-9), and PTEN Protein Expression (p < 0.05).
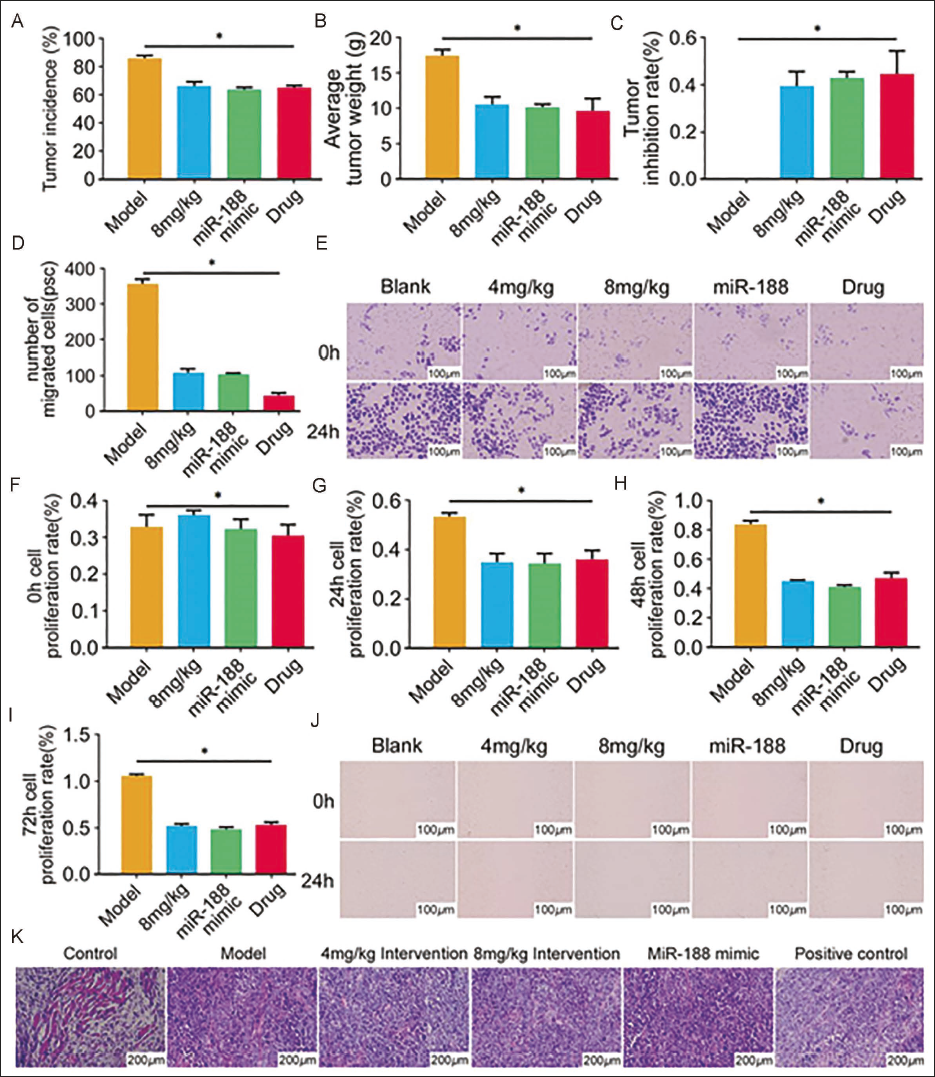
Verification using miR-188 mimics shows that the growth of breast cancer cells is related to miR-188, and matrine can inhibit breast cancer cell growth by downregulating miR-188. In order to ensure the accuracy of the experimental results, we added a miR-188 mimic group to verify that miR-188 inhibits the growth of breast cancer cells. The following test results showed that after adding miR-188 mimic to the model group, the incidence of tumor (61.25 ± 1.36) % and average mass (9.85 ± 1.66) g were significantly decreased in the model group (t = 16.662, 4.194, p = 0.000, 0.003, respectively) (Figure 6A and B). The tumor inhibition rate of miR-188 mimic group was (0.45± 0.19) %, which was significantly higher than that of the model group (Figure 6D). The mobility of miR-188 mimic group was also significantly lower than that of the model group (t = 116.866, p = 0.000), and even lower than that of the matrine 8 mg/kg group (t = 3.228, p = 0.012) (Figure 6D and E). The cell proliferation rate was detected at different times, and it was found that the cell proliferation rate of the miR-188 mimic group decreased significantly and tended to be lower than that of the matrine 8 mg/kg and drug groups, suggesting that the increased miR-188 inhibits proliferation of tumor cells (Figure 6F–I). In addition, scratch test and HE staining observation results showed that the migration of cells and pathological morphology of cells in the miR-188 mimic group were improved compared with other groups (Figure 6J and K).
Validation Using miR-188 Mimics. (A, B, C) Tumor Data Monitoring; (D, E) Transwell Detection of Cell Migration Number; (F, G, H, I) 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) Detection of Cell Viability; (J) Scratch Test Detection of Cells Migration Ability; (K) Hematoxylin and Eosin (HE) Staining to Observe Cell Morphology (p < 0.05).
Discussion
Matrine has the effects of clearing heat, diuresis, and killing insects, and also plays a certain role in regulating the immune system, anti-pathogen, and antitumor (Shi et al., 2019). According to relevant studies, matrine can inhibit the activation of the Wnt/β-catenin pathway by downregulating AGRN in colorectal cancer cells, showing an anti-colon cancer effect. At the same time, it can also downregulate the expression of miR-10b-5p and upregulate the level of PTEN protein, thus inhibiting the biological process of colon cancer cells. For cervical cancer, Jin et al. found through mouse experiments that matrine could induce iron apoptosis by activating Piezo1 and reducing the level of GPX4 protein, thus promoting SiHa cell death and playing a protective role against cervical cancer. Another study showed that matrine increased the mRNA expression of TLR7, TLR8, MyD88, TRAF-6, and IKK by activating the toll-like receptor (TLR) pathway, upregulated the levels of IL-12, IL-6, and TNF-α, and increased the expression of MHC-II, CD54, and CD80. It exerted an antitumor effect on dendritic cells. In addition, matrine not only promotes autophagy in cancer cells by inhibiting AKT/mTOR pathway, but also has anti-breast cancer activity. It can also significantly inhibit the expression level of vascular endothelial growth factor (VEGF) and reduce the activity of AKT, thereby reducing the activity of nuclear factor nuclear factor kappa B (NF-κB) p65, inhibiting the activity of matrix metalloproteinases MMP-9 and MMP2, which affect invasion, and thus inhibiting the metastasis of breast cancer.
In recent years, studies have found that tamoxifen, which is composed of the skeleton structure of triphenylethylene, can make it bind to the estrogen receptor α and inhibit its function, thus playing a role in the treatment of breast cancer. Therefore, it was used as a negative control group for drug intervention in this study. In order to further clarify the therapeutic effect of matrine in breast cancer, this study used matrine gavage to intervene in breast cancer mice. In this study, in order to ensure the repeatability and comparability of the research results and confirm that the mouse tumor has reached the trend of stable growth and can be measured by naked eye or palpation, we conducted intervention on the nude mouse model of breast cancer with tumor size of 0.8–1.0 cm. At the same time, different doses of matrine were administered intragastric treatment. We took breast cancer tissues from each group of nude mice. We found through cell experiments that the migration ability, cell proliferation rate, and cell viability of tumor cells were all the same. Gradually reduced, even under the intervention of high-dose matrine, the tumor proliferation rate was lower than that of the drug group, showing that the higher the dosage of matrine was, the more obvious was the inhibitory effect on breast cancer. At the same time, PCR detection found that the miR-188 expression also increased in a dose-dependent manner after tumor growth was inhibited. Therefore, matrine’s ability to inhibit proliferation of breast cancer cells is related to a certain extent by promotion of miR-188 expression.
Some studies have suggested that miR-188 is closely related to the growth of breast cancer cells. Various human breast cancer cells have significantly lower levels of miR-188 (Wu et al., 2014). miR-188-5p was downregulated in breast cancer and several breast cancer cell lines (Hamam et al., 2016). According to experimental results from other studies, it can be seen that miR-188-5p overexpression can inhibit ZFP91 to a certain extent and reduce the expression of NF-κB p65 (Rel), thereby exerting an inhibitory effect on breast cancer. The overexpression of miR-188 can not only increase PARP1, and Bax, but also inhibit Bcl-2 to a certain extent, thereby further promoting cancer cell apoptosis (Zhu et al., 2020). In our study, it was found that increased miR-188 level after matrine intervention, so we believe that matrine can promote miR-188 expression, thereby affecting the growth of breast cancer cells. In addition, according to research reports, the increased invasive ability of advanced breast cancer cells is due to increased activity or increased expression of the MMPs protein family (Radisky & Radisky, 2015). Luteolin can downregulate the expression of MMP9 by reducing the level of AKT/mTOR that promotes H3K27Ac and H3K56A in the MMP9 promoter region, thereby hindering the biological process for triple-negative breast cancer cells (Yang et al., 2012). Zhu et al. found that the protein levels of MMP-2, MMP-9, PI3K, and p-Akt in KF transfected with miR-188-5p mimics were inhibited.
Based on the above findings, to explore how miR-188 affects the occurrence and development of breast cancer, the miR-188 mimic was used. Through further study of the influence mechanism of miR-188, it was found that while the level of miR-188 was upregulated, the MMP-9 and MMP-2 expressions decreased, while the expression of PTEN increased, resulting in decreased cell viability. At the same time, we used a dual-luciferase assay and found that miR-188 can specifically bind to PTEN. Therefore, we believe that matrine can promote the targeted binding of miR-188 to PTEN and reduce MMP-9 and MMP-2. To ensure the rigor of the experiment, we also added miR-188 mimic to verify whether this mechanism is correct and found that various levels of tumor cell migration ability, tumor cell viability, and tumor cell proliferation rate were significantly different from those of tumor cells under miR-188 mimic intervention. There was no difference when compared with matrine 8 mg/kg, and in most cases, it was lower than matrine 8 mg/kg. Therefore, it can be shown that matrine can promote specific binding of miR-188 to PTEN and reduce MMP-9 and MMP-2, thereby inhibiting the proliferation and metastasis of breast cancer cells.
Conclusion
In summary, matrine can promote the targeted binding of miR-188 and PTEN to inhibit the expression of MMPs protease family related to cell invasion, thus slowing down the proliferation and spread of breast cancer cells. This not only deepens the understanding of the biological mechanisms of breast cancer but also reveals the multifaceted nature of matrine as a potential anticancer drug. The results show that matrine can affect the life activities of breast cancer cells by regulating miR-188, which indicates that miR-188 plays a key role in the anticancer effect mediated by matrine, which also provides ideas for the development of breast cancer treatment drugs and also provides a new target for future cancer treatment. However, whether matrine can use other miRNAs to regulate other genes to play a role in alleviating cancer is still unclear, and further investigation is needed.
Abbreviations
MMP-2: Matrix metalloproteinase-2; MMP-9: Matrix metalloproteinase-9; PTEN: Phosphatase and tensin homolog.
Footnotes
Acknowledgments
None.
Authors’ Contribution
Zhao Liu: Conceptualization, Methodology, Software, Investigation, Formal analysis, Writing – Original draft.
Jian Xu: Data curation, Writing – Original draft, Visualization, Investigation.
Heng Xia: Resources, Supervision, Software.
Miaofeng Wang: Validation, Visualization, Writing – Review & editing.
Qianlan Xu: Conceptualization, Funding Acquisition, Resources, Supervision, Writing – Review & editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The study has obtained the ethical approval from the Animal Ethics Committee.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
