Abstract
Objective
MicroRNAs (miRNAs) regulate prostate tumorigenesis and progression by involving different molecular pathways. In this study, we examined the role of miR-572 in prostate cancer (PCa).
Methods
The proliferation rates of LNCaP and PC-3 PCa cells were studied using MTT assays. Transwell migration and Matrigel invasion assays were performed to evaluate cell migration and invasion, respectively. Protein expression levels were examined using western blotting. Docetaxel-induced apoptosis was evaluated by Caspase-Glo3/7 assays. The putative miR-572 binding site in the phosphatase and tensin homolog (PTEN) 3ʹ untranslated region (3ʹ UTR) was assessed with dual-luciferase reporter assays. Additionally, miR-572 expression levels in human PCa tissues were examined by qRT-PCR assays.
Results
Upregulation of miR-572 promoted proliferation, migration, and invasion of PCa cells. Overexpression of miR-572 decreased sensitivity of PCa cells to docetaxel treatment by reducing docetaxel-induced apoptosis. MiR-572 can regulate migration and invasion in PCa cells. Furthermore, miR-572 could regulate expression of PTEN and p-AKT in PCa cells by directly binding to the PTEN 3ʹ UTR. MiR-572 expression levels were increased in human PCa tissues and associated with PCa stage.
Conclusions
miR-572 displayed essential roles in PCa tumor growth and its expression level may be used to predict docetaxel treatment in these tumors.
Keywords
Introduction
In the past decades, prostate cancer (PCa)-associated deaths have dropped dramatically because of early screening and advanced treatment. 1 However, PCa is still one of the most common cancers around the world. 2 There were approximately 191,930 new cases of PCa and 33,330 deaths from PCa in 2019. 3 PCa has become the second leading cause of cancer death in American men. Chemotherapy is important in PCa treatment. 4 Docetaxel has been used for metastatic castration-resistant PCa for many years. 5 Docetaxel inhibits microtubular depolymerization and increases apoptosis in cancer cells. However, resistance to docetaxel-based chemotherapy has been seen in patients with PCa. Multiple aspects are involved in docetaxel-based chemoresistance, including apoptosis, cell proliferation, and low intracellular drug concentration.6–8 Recently, more studies have been focused on identifying molecular targets that can overcome chemoresistance in the treatment of PCa.
MicroRNAs (miRNAs), small non-coding RNAs that are about 20 nucleotides long, can influence proliferation, apoptosis, differentiation, and other biological activities in a variety of diseases.10,11 Studies have demonstrated that miRNAs act as regulators in human malignancy. Some miRNAs have increased expression in cancer and support tumor proliferation, while other miRNAs display decreased expression in cancer and inhibit tumor proliferation. 12 MiRNA expression levels are associated with PCa development and prognosis.13–15 Recent miRNA profiling studies in PCa tissues have shown that some miRNA expression levels are different from those in adjacent normal prostate tissue. The aberrant expression patterns of these miRNAs are associated with prostate tumorigenesis and prognosis by interacting with different transcription factors in PCa. Hsu et al. 16 showed that miR-18a was upregulated in PCa tissues. Additionally, miR-18a levels in peripheral blood of PCa patients were higher than those in blood from benign prostate hyperplasia patients and healthy individuals. 17 Saini et al. 18 demonstrated that miR-203 plays important roles in the regulation of PCa progression and metastasis. These findings suggest that miRNAs may be able to serve as novel biomarkers for early diagnosis, as well as potential molecular targets for treatment. Studies have shown that miR-572 can function as an oncogene in different types of cancer. Wu et al. found higher levels of miR-572 in both ovarian cancer cells and human ovarian cancer tissues. 19 Further experiments showed that upregulation of miR-572 can promote the proliferation of ovarian cancer cells by targeting protein phosphatase 2 regulatory subunit Bgamma (PPP2R2C). 19 In addition, Zhang et al. 20 showed that increased miR-572 expression in ovarian cancer patients indicated poor survival. This miRNA also promoted the proliferation of ovarian cancer cells by inhibiting suppressor of cytokine signaling 1 (SOCS1) and p21.
Prior studies have shown that miR-572 can act as an oncomiR 12 in different types of cancer. However, the role of miR-572 on PCa remains unclear. In this study, we investigated the role of miR-572 in the growth and chemosensitivity of PCa cells. Our data demonstrated that miR-572 may regulate cell proliferation by targeting phosphatase and tensin homolog (PTEN).
Materials and methods
Prostate cancer (PCa) cell lines
LNCaP and PC3 PCa cell lines were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA), were grown in Roswell Park Memorial Institute (RPMI)-1640 medium (Thermo Fisher Scientific, Waltham, MA, USA). The complete culture medium was supplemented with 10% fetal bovine serum (FBS, Thermo Fisher Scientific), 100 units/mL penicillin and 100 mg/mL streptomycin (Thermo Fisher Scientific). Cells were grown at 37°C, 5% CO2.
miRNA transfections
LNCaP and PC3 cells were grown overnight in 24-well plates (20,000 cells/well) in complete culture medium. Then, either miR-572 mimic or miR-572 inhibitor (0.1 nmol) was transfected into PCa cells using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). After 6 hours, medium containing 10% FBS was used to terminate the transfection. Scrambled miRNA was transfected into PCa cells as a negative control.
RNA isolation and quantitative reverse transcription polymerase chain reaction (RT-qPCR)
miR-572 levels in PCa cells were detected by quantitative reverse transcription polymerase chain reaction (RT-qPCR). Briefly, total RNA was isolated from LNCaP and PC3 cells with TRIzol reagent (Invitrogen). High-Capacity cDNA Reverse Transcription Kit (Thermo Fisher Scientific) was used for reverse transcription reactions. PCRs were performed using a SYBR Green PCR Kit (Thermo Fisher Scientific) and the following conditions: 2 minutes at 94°C, and 38 cycles of 20 s at 94°C, 30 s at 60°C, and 1 minute at 72°C, followed by a dissociation stage. The primer sequences were as follows: miR-572 forward: 5′-GTCCGCTCGGCGGTGGCCCA-3′; reverse: 5′-CTACGTAAGCCTGACTCAT-3ʹ. U6 was included as the endogenous control.
Cell proliferation assay
MTT assays were performed to examine the effects of miR-572 on PCa cell growth. Briefly, transfected LNCaP and PC3 cells were placed in 96-well plates and cultured with medium containing docetaxel (0, 1, 5, and 10 nM) at 37°C. MTT reagent (Roche, Mannheim, Germany) was added into three wells at each concentration of docetaxel every 24 hours and incubated for 4 hours. Dimethyl sulfoxide (DMSO) was added to dissolve the crystals. A microplate reader (BioTek, Winooski, VT, USA) was used to detect the absorbance at 570 nm. To determine the combination of hyperbaric oxygen (HBO2) and docetaxel-induced inhibitory effects of miR-572 on PCa cell growth, cells were incubated at 37°C in a hyperbaric chamber containing 95% O2, 5% CO2 at 1 atmosphere absolute for 90 minutes.
Caspase 3/7 apoptosis assay
Docetaxel-induced apoptosis was evaluated using a Caspase-Glo assay (Promega, Madison, WI, USA) by detecting caspase-3/7 activity. Briefly, PCa cells were grown overnight in 24-well plates. Cells were then treated with different concentrations (0, 1, 3, and 5 nM) of docetaxel for 48 hours. Next, Caspase-Glo reagent (100 μL) was added to the cells, and they were incubated for 2 hours at room temperature. A plate-reading luminometer (Thermo Labsystems, Philadelphia, PA, USA) was used to measure the luminescence.
Transwell migration and invasion assays
Transfected LNCaP and PC3 cells (density: 1 × 105 cells/mL) in serum-free RPMI-1640 medium were seeded in upper chambers that were pre-coated with or without Matrigel. The upper chambers without Matrigel were used for migration assays. The bottom chambers were filled with RPMI-1640 with FBS. The PCa cells were grown overnight and Diff-Quik (Sigma-Aldrich, St. Louis, MO, USA) staining was performed to stain the invading cells, which were counted using a light microscope.
Luciferase assay
A candidate target of miR-572 was identified using the miRDB database. 21 The genetic sequence of the PTEN 3ʹ untranslated region (3ʹ UTR), including the putative miR-572 binding site, was synthesized from total DNA of primary prostate epithelial cells (HPrECs) (ATCC). Primer sequences: forward: 5′-TAGAGGAGCCGTCAAATCCA-3′ and reverse: 5′-TGGACATCTGATTGGGATGA-3′. The synthesized PTEN 3ʹ UTR was cloned into the Xbal site of the pmirGLO Dual-Luciferase miRNA Target Expression Vector (Promega). The wild type (WT) PTEN plasmid (WT-PTEN-3ʹ-UTR-pGL3) was used as a template to synthesize the mutant PTEN fragment (mu-PTEN-3ʹ-UTR-pGL3) by introducing three point mutations with a Phusion site-directed mutagenesis Kit (Thermo Fisher Scientific). LNCaP cells were co-transfected with Luc-PTEN and miR-572 (0.1 nmol, 0.2 nmol, and 0.5 nmol) using Lipofectamine RNAiMAX (Invitrogen). Luciferase signals were examined using the Dual-Luciferase Reporter Assay system (Promega).
Western blotting
Total protein was extracted from cells using radioimmunoprecipitation assay (RIPA) lysis buffer (Abcam, Cambridge, UK). Then, 30 μg of total protein was separated with sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) (10%). The proteins were transferred to a polyvinylidene difluoride (PVDF) membrane. After blocking with non-fat dried milk in phosphate-buffered saline (PBS), different primary antibodies (see Table 1) were added and incubated with the membrane overnight with gentle agitation. The membrane was washed with PBS and incubated with the secondary antibody for 1 hour at room temperature. Protein expression levels were normalized to protein loading control by densitometric analysis, as described previously. 22
Antibodies used in western blot analysis.
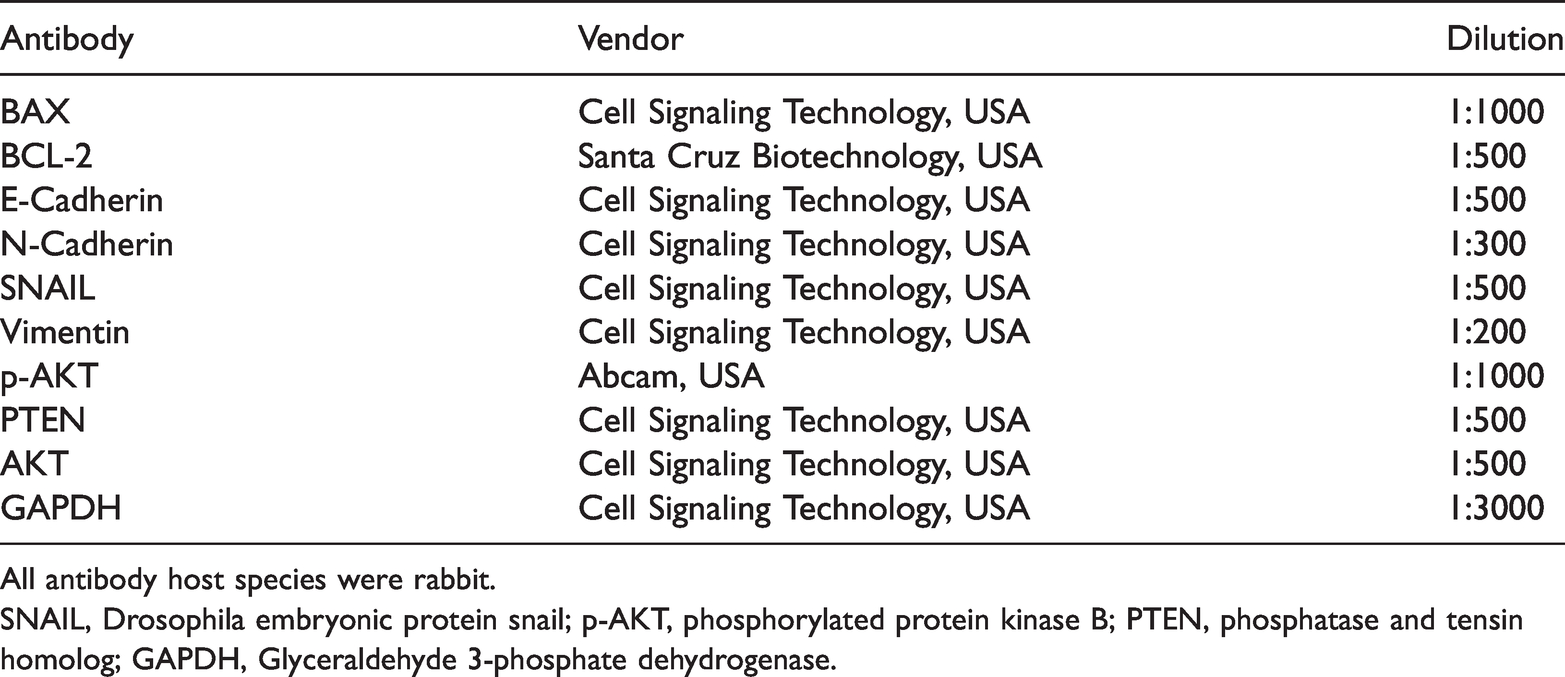
All antibody host species were rabbit.
SNAIL, Drosophila embryonic protein snail; p-AKT, phosphorylated protein kinase B; PTEN, phosphatase and tensin homolog; GAPDH, Glyceraldehyde 3-phosphate dehydrogenase.
miR-572 expression analysis in human PCa tissues
Formalin-fixed paraffin-embedded (FFPE) tissue was obtained from 43 PCa patients between 2015 and 2019. This study was approved by the ethical committee of Jilin University (Approval No. JLU20190516779). All patients provided written informed consent that the tissue could be used for research and molecular studies. The diagnosis was confirmed with hematoxylin and eosin (H&E) staining. The tissue was macrodissected into unstained FFPE sections. Total RNA extraction and RT-qPCR were performed following the abovementioned methods.
Immunohistochemical analysis
Immunohistochemical staining for PTEN and Ki-67 was performed on FFPE PCa tissue sections (5 μm). The normal prostate tissue adjacent to the tumor was used as a control. Immunohistochemistry was performed on an automated stainer (Bond rx, Leica, Wetzlar, Germany) using PTEN (Cell Signaling Technology, #9559, Danvers, MA, ISA 1:500) and Ki-67 (Cell Signaling Technology, #12202, 1:1000) antibodies following the manufacturer’s recommendations.
Statistical analysis
Statistical analysis was performed by one-way analysis of variance (ANOVA) coupled with Tukey correction and Student’s t test using SPSS 12.0 (SPSS Inc., Chicago, IL, USA). Differences were considered significant at
Results
miR-572 increased proliferation of LNCaP and PC3 cells and induced docetaxel resistance
As shown in Figure 1a and 1b, overexpression of miR-572 was detected in both LNCaP and PC3 cells after miR-572 mimic transfection. Increased expression of miR-572 improved growth of LNCaP (Figure 1c) and PC3 cells (Figure 1d) (

High levels of miR-572 promote growth and survival of LNCaP and PC3 cells. (a) miR-572 expression levels after LNCaP cells were transfected with miR-572 mimic. (b–e) Growth of LNCaP cells transfected with miR-572 mimic and treated with different doses of docetaxel (0, 1, 5, and 10 nM). (f) Growth of LNCaP cells with high levels of miR-572 in HBO. (g) miR-572 expression levels after PC3 cells were transfected with miR-572 mimic. (h–k) Growth of PC3 cells transfected with miR-572 mimic and treated with different doses of docetaxel (0, 1, 5, and 10 nM). (l) Growth of PC3 cells with high levels of miR-572 in HBO.

Low levels of miR-572 inhibit growth and survival of LNCaP and PC-3 cells. (a) miR-572 expression levels after LNCaP cells were transfected with miR-572 inhibitor. (b–e) Growth of LNCaP cells transfected with miR-572 inhibitor and treated with different doses of docetaxel (0, 1, 5, and 10 nM). (f) Growth of LNCAP cells with low levels of miR-572 in HBO. (g) miR-572 expression levels after PC3 cells were transfected with miR-572 inhibitor. (h–k) Growth of PC3 cells transfected with miR-572 inhibitor and treated with different doses of docetaxel (0, 1, 5, and 10 nM). (l) Growth of PC3 cells with low levels of miR-572 in HBO.
miR-572 inhibited docetaxel-induced apoptosis of LNCaP and PC3 cells
To examine the effects of miR-572 on docetaxel-induced apoptosis, LNCaP and PC3 cells were treated with docetaxel and the caspase 3/7 activities were measured. As shown in Figure 3, overexpressed miR-572 decreased caspase3/7 activities in LNCaP (Figure 3a) and PC3 cells (Figure 3b). Furthermore, upregulation of miR-572 was associated with decreased BAX protein expression and increased Bcl-2 protein expression in both LNCaP and PC-3 cells (Figure 3c and 3d).

miR-572 inhibits docetaxel-induced apoptosis in prostate cancer cells. (a) Effects of high levels of miR-572 on caspase 3/7 activity in LNCaP cells treated with docetaxel. (b) Effects of high levels of miR-572 on caspase 3/7 activity in PC3 cells treated with docetaxel. (c,d) Effects of miR-572 on BAX and BCL-2 expression in LNCaP and PC3 cells treated with docetaxel.
miR-572 altered invasion and migration of LNCaP and PC3 cells
As shown in Figure 4, overexpressed miR-572 increased both the migration and invasion rates of PCa cells. In contrast, decreased expression of miR-572 inhibited both the migration and invasion rates of PCa cells.
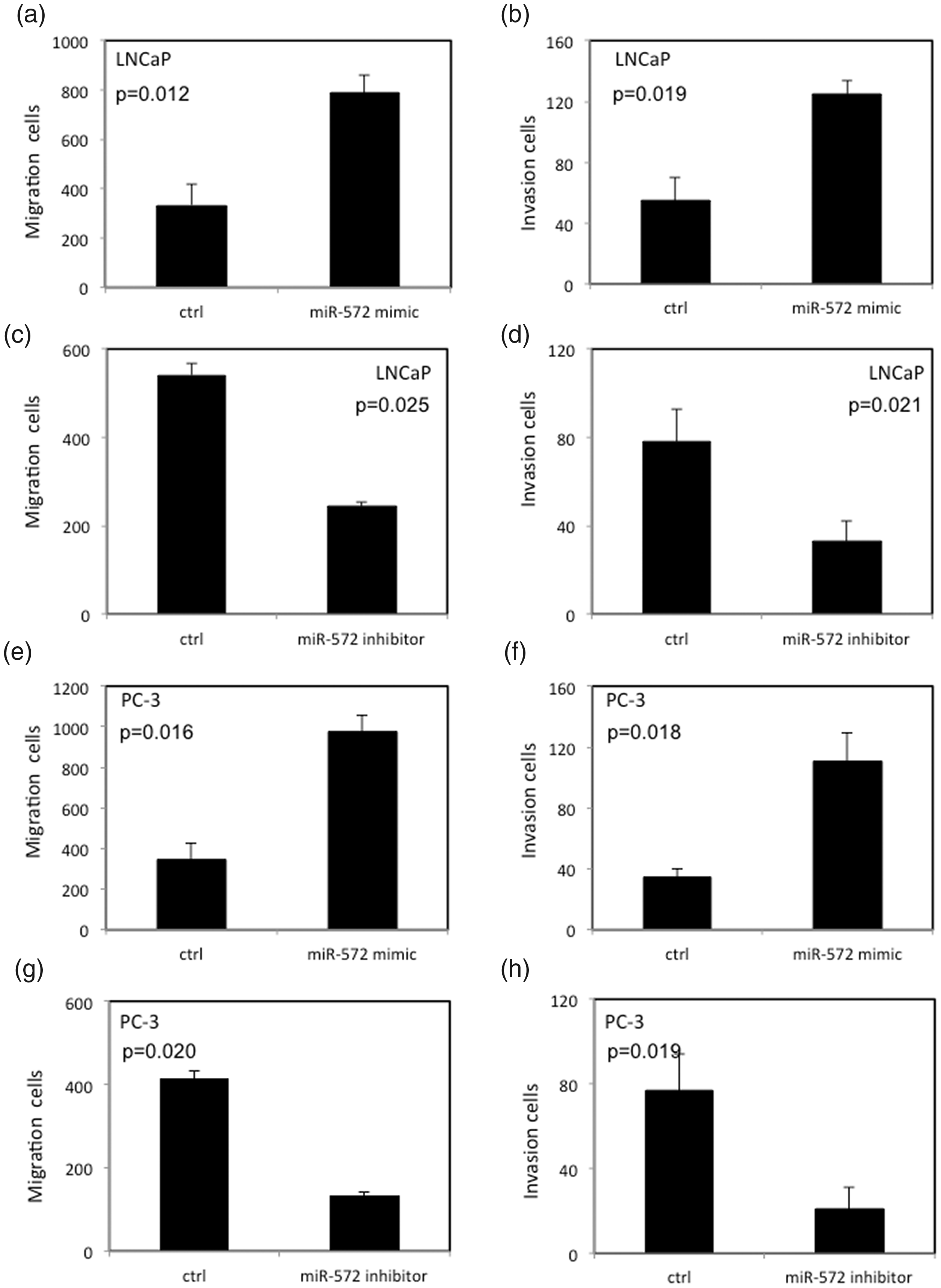
Invasion and migration of LNCaP and PC3 cells with aberrantly expressed miR-572. (a,b) High levels of miR-572 can increase the migration and invasion of LNCaP cells. (c,d) Low levels of miR-572 can decrease the migration and invasion of LNCaP cells. (e,f) High levels of miR-572 can increase the migration and invasion of PC3 cells. (g,h) Low levels of miR-572 can decrease the migration and invasion of PC3 cells.
miR-572 regulated epithelial–mesenchymal transition (EMT)-associated protein expression and impacted PTEN expression in PCa cells
To investigate the molecular mechanism of miR-572 in PCa cells, expression of different proteins involved in a variety of molecular signaling pathways were studied. We found that miR-572 overexpression inhibited E-cadherin protein levels, but enhanced protein levels of N-cadherin, Drosophila embryonic protein snail (SNAIL), and vimentin in PCa cells (Figure 5a–c). Similarly, low levels of miR-572 enhanced E-cadherin protein expression and inhibited protein levels of N-cadherin, SNAIL, and vimentin in PCa cells (Figure 5d–f). Additionally, miR-572 overexpression decreased PTEN protein levels, but enhanced phosphorylated protein kinase B (p-AKT) expression in PCa cells (Figure 6a–c); low levels of miR-572 enhanced PTEN protein expression, but decreased p-AKT expression in PCa cells (Figure 6d–f).

Effects of miR-572 on epithelial-mesenchymal transition (EMT)-related protein expression in LNCaP and PC3 cells. (a–c) Effects of high levels of miR-572 on expression of EMT-related proteins in LNCaP and PC3 cells. (d–f) Effects of low levels of miR-572 on expression of EMT-related proteins in LNCaP and PC3 cells.

Effects of miR-572 on PTEN/AKT protein expression in LNCaP and PC3 cells. (a–c) Effects of high levels of miR-572 on expression of p-AKT, AKT, and PTEN in LNCaP and PC3 cells. (d–f) Effects of low levels of miR-572 on expression of p-AKT, AKT, and PTEN in LNCaP and PC3 cells.
miR-572 can directly bind to the PTEN 3’ UTR in PCa cells
Using the MiRDB online database, we identified a putative miR-572 binding site in the PTEN mRNA 3ʹ UTR (Figure 7a). In our study, luciferase activity was not significantly changed in PCa cells co-transfected with miR-572 and mu-PTEN-3ʹ-UTR-pGL3 (Figure 7b). However, luciferase activity was significantly decreased in PCa cells co-transfected with miR-572 and WT-PTEN-3ʹ-UTR-pGL3 (
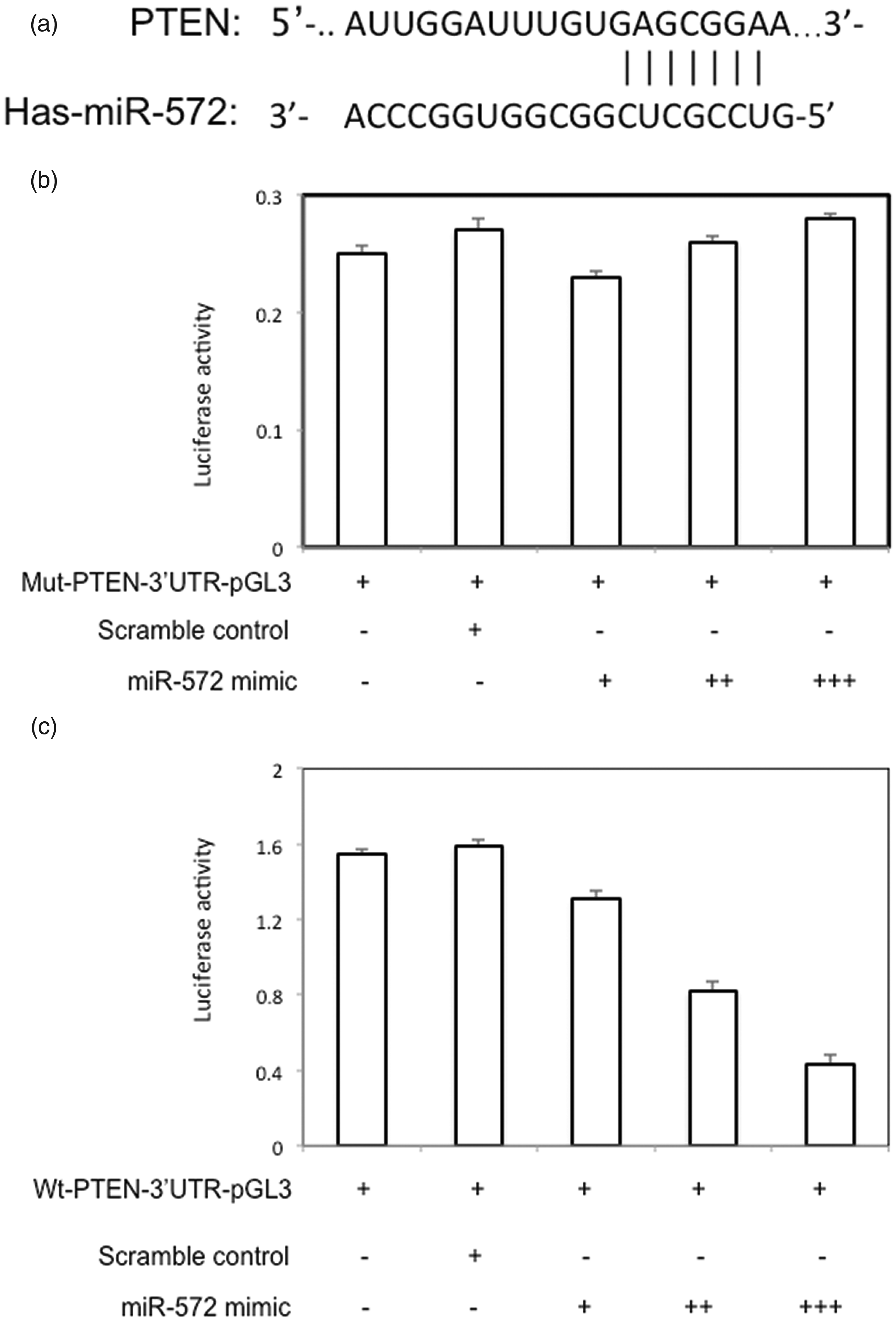
miR-572 can directly target PTEN. (a) A putative miR-572 binding site in the PTEN 3ʹ untranslated region (3ʹ UTR). (b) Luciferase activity of LNCaP cells co-transfected with miR-572 mimic and mut-PTEN-3ʹ-UTR-pGL3 (c) Luciferase activity of LNCaP cells co-transfected with miR-572 mimic and wt-PTEN-3ʹ-UTR-pGL3.
miR-572 expression in human PCa tissues
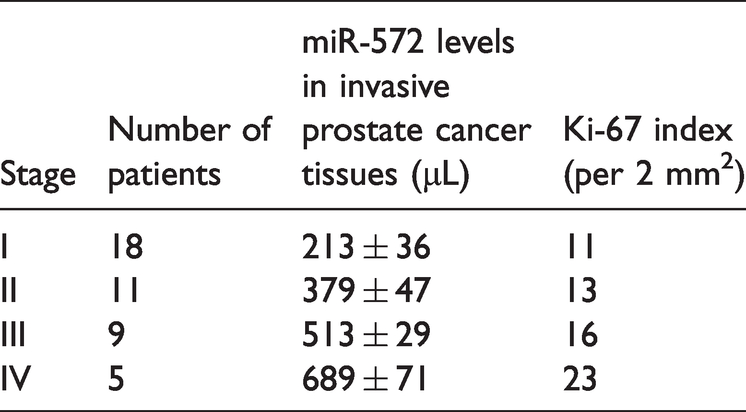
To examine miR-572 levels in human PCa tissues, FFPE tissues were collected and RT-qPCR was performed. As shown in Figure 8a, miR-572 levels were higher in PCa tissues compared with normal prostate tissues. miR-572 levels were associated with PCa stage (Table 2, Figure 8b). Later stage disease showed a higher miR-572 expression level. In PCa tissues with high miR-572 expression (Figure 8c), Ki-67 protein expression is increased (Figure 8d). PTEN protein expression was decreased in PCA tissues with high miR-572 expression compared with normal prostate tissue (Figure 8d).

miR-572 levels in human prostate cancer (PCa) tissues. (a) miR-572 levels in PCa tissues and normal prostate tissues. (b) miR-572 levels in PCa tissues of different stages. (c) miR-572 levels in different human prostate tissues. (d) Immunohistochemical studies for Ki-67 and PTEN in normal prostate tissues, PCa tissues with low levels of miR-572, and PCa tissues with high levels of miR-572.
miR-572 expression levels in different stages of prostate cancer.
Overall, miR-572 promoted proliferation, migration, and invasion of PCa cells. miR-572 decreased sensitivity of PCa cells to docetaxel treatment by reducing docetaxel-induced apoptosis. miR-572 could regulate expression of PTEN and p-AKT in PCa cells by directly binding to the PTEN 3ʹ UTR. miR-572 expression levels were increased in human PCa tissues and associated with PCa stage.
Discussion
Studies have shown that aberrant expression of miRNAs involves different signaling pathways in various cancers, including PCa cancer. Wang et al. 23 reported that elevated signal transducer and activator of transcription 3 (STAT3) expression increased proliferation, migration, and invasion of colorectal cancer cells via upregulation of miR-572. Moreover, increased miR-572 expression has been observed in renal cell carcinoma (RCC) tissues and cell lines. Overexpression of miR-572 increased proliferation and decreased apoptosis of RCC cells. 24 In our study, miR-572 overexpression enhanced growth of PCa cells, but downregulation of miR-572 decreased growth of these cells. Our findings demonstrate that aberrant expression of miR-572 is associated with growth of PCa cells.
Docetaxel has been used to treat different types of malignant neoplasms, including breast, non-small cell lung, head and neck, stomach, and prostate cancers. 25 Recent randomized trials have demonstrated that docetaxel combined with standard hormonal therapy markedly improved survival among patients with advanced PCa. 5 However, one significant clinical problem is tumor resistance to docetaxel resulting from different mechanisms such as limiting intracellular drug concentration, apoptosis escape, and tumor suppressor gene mutations.7,26 Shi et al. 27 reported that miR-21 was associated with docetaxel resistance in PC3 PCa cells by targeting programmed cell death protein 4 (PDCD4). Further study showed that elevated serum miR-21 levels correlated with prostate-specific antigen (PSA) levels in patients with androgen‐dependent PCa or hormone‐refractory PCa, and may indicate chemotherapy resistance in these patients. 28 Gao et al. 29 found that overexpression of miR-323 increased proliferation of PCa cells and promoted docetaxel resistance by repressing p73. In the current study, ectopic expression of miR-572 promoted survival of PCa cells in the presence of docetaxel. However, low levels of miR-572 decreased survival of PCa cells in the presence of docetaxel. Our study also shows that miR-572 can impact docetaxel-induced apoptosis in both PCa cell lines. These data suggest that miR-572 likely contributes to docetaxel resistance in PCa cells.
The EMT is responsible for the metastatic process in cancers, including PCa. 30 Recently, it has been demonstrated that the EMT is associated with PCa progression and treatment resistance.31,32 Ruan et al. 33 reported that Skp2-mediated Twist stabilization played important roles in acquisition of resistance to paclitaxel or doxorubicin in castration-resistant PCa. Another study showed that zinc finger E-box-binding homeobox 1 (ZEB1) could mediate chemoresistance by repression of E-cadherin in docetaxel-resistant PCa. 34 Further studies indicated that TGF-β not only induces vimentin expression and decreases E-cadherin expression in PCa, but also enhances expression of EMT transcription factors such as Snail 1/2 or ZEB1.35,36
Here, we provide evidence that miR-572 is involved in PCa progression by regulating EMT-related protein expression levels. Additionally, miR-572 could directly regulate expression of PTEN and p-AKT by binding to the PTEN 3ʹ UTR in PCa cells. PTEN, a tumor suppressor, 37 is involved in a variety of biological activities like proliferation and apoptosis. 38 PCa is a heterogeneous disease and is controlled by various mechanisms, molecular pathways, and genes, including PTEN. Aberrant expression of PTEN leads to poor prognosis in PCa patients. 39 Some miRNAs are involved in aberrant expression of PTEN in PCa. Nip and coworkers found that miRNA-4534 can decrease PTEN expression and act as an oncomiR in PCa. 40 Yang et al. 41 found that miR-21 regulates PTEN expression and can enhance the growth and invasion of PCa cells.
In the current study, we found that PCa tissues with higher miR-572 expression showed lower PTEN expression compared with normal prostate tissues with lower miR-572 levels. miR-572 expression was also associated with PCa stage in these tissues. Our data suggest that miR-572 acts as an oncomiR not only in PCa cell lines, but also in PCa tissues. Therefore, miR-572 is a potential biomarker in predicting chemotherapy response and prognosis in PCa patients. However, many molecular factors are involved in PCa development and progression. Further studies are required to explore how miR-572 correlates with other proteins and miRNAs in PCa.
Footnotes
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
MZ designed the project, performed the experiments, and wrote the manuscript; XG was involved with data analysis; MC designed the project and wrote the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
