Abstract
Endometrial hyperplasia (EH) is a medical condition that affects many females as it increases their uterine carcinogenic potential. EH results from entangling hormonal imbalance and inflammatory response. The study examined the role of a xanthine oxidase inhibitor, febuxostat, in a rat model of EH. Adult female Wistar albino rats were subjected to estradiol valerate (EV) 2 mg/kg for 10 days to induce EH. Another group was treated concomitantly with febuxostat 10 mg/kg for the same period. The uterine malondialdehyde, reduced glutathione (GSH), and superoxide dismutase (SOD) were assessed by chemical methods. Gene expressions of phosphatidylinositol-3-kinase (PI3K), Akt, and hypoxia-inducible factor 1 alpha were assessed by the quantitative real-time polymerase chain reaction. Moreover, the vascular endothelial growth factor (VEGF) and interleukin-6 (IL-6) were measured by enzyme-linked immunosorbent assay. Histopathology and immunohistochemical techniques were used for the detection of phosphatase and tensin homolog (PTEN). The results revealed that EV administration induced complex EH with focal atypia and loss of PTEN expression by the histological examination. Uteri of the EV group showed a significant drop in GSH content and SOD activity and rise in the expressions of PI3K, Akt, VEGF, and IL-6. Febuxostat administration significantly improved the uterine GSH and SOD levels. It decreased the expressions of PI3K, Akt, VEGF, and IL-6. The endometrium showed a regression of the proliferative epithelium with the restoration of PTEN expression and the absence of the atypical features. In conclusion, febuxostat protected the endometrium against estrogen-induced EH and may be beneficial in the management along with the hormonal therapy.
Keywords
Introduction
Endometrial hyperplasia (EH) is a common pathological condition of endometrial lining characterized by noninvasive nonproportional proliferation of its glandular and stromal components. 1 It affects women of different ages but peaks around the age of 50 to reach 140 cases per 100,000 women/year. 2 Endometrial tissue is regulated by sex hormones; the imbalance between estrogen and progesterone is the underlying cause of EH. The excessive and continuous unopposed estrogen stimulation of the endometrium results in different forms of hyperplasia with or without atypia. 3 Despite the fact that EH is a benign pathology, its progression to endometrial carcinoma draws attention for its management. 4 The current management depends mainly on progesterone treatment to regain hormonal balance.
On the other hand, inflammatory cytokines were reported to be involved in the pathogenesis of atypical EH under a state of local endometrial hyperestrogenia. 5 The proliferative property of estrogen was reported to be based on the activation of phosphatidylinositol-3-kinase (PI3K) that stimulates Akt. 6 PI3K/Akt is a signaling pathway that plays an essential role in cell proliferation and cancer progression. Its involvement in the initiation of endometrial carcinoma was demonstrated by Osaki et al. 7 Also, xanthine oxidase is an enzyme that catalyzes purine catabolism to uric acid. Furthermore, it liberates reactive oxygen and nitrogen species implicated in inflammation, proliferation, and mutagenicity. 8 –10 The expression of xanthine oxidase was reported in the glandular epithelia of the endometrium. Its overexpression is implicated in the pathogenesis of endometrial disorder as adenomyosis. 11 However, the role of xanthine oxidase in the pathogenesis of EH has not been explored. Febuxostat, 2-[3-cyano-4-(2-methylpropoxy)phenyl]-4-methyl-thiazole-5-carboxylic acid, is a selective and powerful inhibitor of xanthine oxidase enzyme. Febuxostat exerted anti-inflammatory and antioxidant effects in the model of vascular inflammation and myocardial ischemia. 12,13 To our best knowledge, no study examined the effect of xanthine oxidase inhibitors on EH. Therefore, the current study aimed at investigating the role of febuxostat in EH induced by estradiol valerate (EV) in rats as well as exploring the possible underlying mechanisms for the emerging effect.
Material and methods
Drugs, chemicals, and antibodies
Febuxostat was obtained from Global Napi Pharmaceuticals, Egypt, and EV (Cyclo-Progynova tablets, 2 mg/tablet) from Bayer Weimar GmbH and Co. KG (Weimar, Germany). Enzyme-linked immunosorbent assay (ELISA) kits for interleukin-6 (IL-6) and vascular endothelial growth factor (VEGF) were purchased from Invitrogen Thermo Fisher Scientific (Waltham, Massachusetts, USA; LOT 192587043) and Elabscience Biotech Co. Ltd. (Wuhan, China; Catalog No: E-EL-R260396T), respectively. Protein phosphatase and tensin homolog (PTEN) antibodies were purchased from Lab Vision (Fremont, California, USA). Other chemicals were obtained commercially.
Animals
Adult female Wistar albino rats of body weight 190 ± 30 g, aged 8–10 weeks, were obtained from National Research Centre, Giza, Egypt. Animals were left for 2 weeks for proper acclimatization to the animal house conditions (12-h lighting cycle, 25 ± 2°C temperature) before the start of experiments. Animals were arranged in polypropylene autoclavable rat cage (300 L × 200 H × 180 W mm3) with a stainless steel cover lid and built-in food feeder. Animals had free access to standard pellet food and tap water was given ad libitum and checked daily.
Twenty-four rats were arranged randomly into four groups (six rats/group) under uniform standard conditions. All animal procedures were in accordance with the Institutional Animal Ethics, Use, and Care Committee. Prior approval was obtained from The Ethical Committee of Faculty of Medicine; NO: 86-11/2018, Minia University, Egypt.
Study design
After 1 week of acclimatization, animal groups were administrated the following treatment orally for 10 days: Normal control groups received 1% carboxymethyl cellulose as a vehicle, 14 febuxostat control received 10-mg/kg/day of febuxostat suspended in the vehicle, EV group received 2-mg/kg/day of EV in the same vehicle and EV+ febuxostat group. The doses were selected according to our preliminary data and the previous published studies, respectively. 15,16 At the end of the experimental period, the animals were sacrificed by decapitation after anesthetized underuse of intraperitoneal injection of urethane hydrochloride (1 g/kg) anesthesia 17 and the uterus was gently dissected and cleaned with cold saline. Parts of the uterus were put in liquid nitrogen and then kept at −80°C for biochemical analysis. The other uterine parts were fixed in 10% formalin for 24 h, then embedded in paraffin for histological and immunohistochemical examinations. Uterine samples were homogenized in ice-cold phosphate buffer (0.01 M, pH 7.4; 20% weight/volume) then centrifuged to obtain the supernatant.
Measurements
Biochemical analysis
Uterine supernatant was used for the assessment of uterine products of lipid peroxidation as the reactive aldehydes, malondialdehyde (MDA), biochemically as previously described. 18 The uterine content of reduced glutathione (GSH) was determined by the estimation of color intensity produced after the reaction of uterine thiol groups with Elman’s reagent. 19 Enzymatic activity of superoxide dismutase (SOD) was determined by the method described previously and based on the fact that the autoxidation of pyrogallol was inhibited by SOD. 20 The stable oxidation end products of nitric oxide, nitrite, and nitrate were used as indicators of nitric oxide production. They were estimated after the reduction of nitrate to nitrite by copperized cadmium according to the method described by Sastry et al. 21 All results were calculated from the preformed standard curves and expressed as gram tissue.
Histological and immunohistochemical examination
Uterine sections of 4–5 µm thickness were prepared by paraffin processing and then were cut by Leitz 1512 Microtome (Charleston, South Carolina, USA). Sections were stained by hematoxylin and eosin stain. Histological findings of the uterus were assessed by a histologist that was blinded to study treatment. Olympus light microscope (Olympus, Shinjuko, Tokyo, Japan) was used for examining and capturing images for the histological and immunohistochemical sections. Slides were photographed using an Olympus digital camera. Images were saved as jpg. Sections were evaluated according to the World Health Organization classification system of EH. 4
For immunohistochemical staining, 5-µm thick deparaffinized sections were hydrated and then washed. Endogenous peroxidases were blocked by peroxidase blocking solution followed by washing in tris buffered saline (TBS). Nonspecific connection of immunoglobulin G was blocked using normal goat serum, diluted 1:50 in 0.1% bovine serum albumin with TBS for 30 min. The sections were incubated with the diluted (1:200) primary antibody for 45 min. Sections then washed three times each for 5 min in a buffer and incubated for further 10 min with biotinylated goat anti-rabbit secondary antibodies diluted 1:1000, followed by washing. Following a further 10-min incubation with Vectastain avidin, biotinylated horseradish peroxidase complex kits and washing for 10 min, the substrate, diaminobenzidine tetrahydrochloride in distilled water was applied for 5–10 min. All the slides were immersed in Mayer’s hematoxylin for counterstaining, then dehydrated by passing through ascending concentrations of alcohol, and then cleared by xylol. Finally, the sections were covered by coverslips using aqueous mounting medium.
The immunopositive cells for PTEN were scored as the average of three sections from each rat according to the following: 0 for negative staining, 1 for staining lower than normal tissue, 2 for staining similar to normal tissue, and 3 for staining higher than normal tissue. 22
Quantitative real-time polymerase chain reaction (qRT-PCR)
Total RNAs were isolated from homogenized uterine tissues using TRIzol reagent according to the manufacturer’s instructions (Invitrogen Life Technology, Carlsbad, San Diego County, California, USA). The expression of PI3K, Akt, and hypoxia-inducible factor 1 alpha (HIF-1α) genes were quantified using Verso SYBR Green quantitative real-time polymerase chain reaction (qRT-PCR) kit (Thermo Fischer Scientific, USA). The sets of primers used were given in Table 1. 23 –25 The qRT-PCR results were analyzed using the ΔΔCt method with normalization to the beta-actin as internal control in order to adjust the samples and avoid difference in sample loading. 26
Primers used for real-time PCR analysis.

PCR: polymerase chain reaction; PI3K: phosphatidylinositol-3-kinase; HIF-1α: hypoxia-inducible factor 1 alpha; β-actin: beta-actin.
Enzyme-linked immunosorbent assay (ELISA)
Protein assessment of uterine VEGF and IL-6 by ELISA kits was performed according to the manufacturer’s instructions. The absorbance values were translated into concentrations by the obtained standard curve and expressed as pg/ml.
Statistical analysis
Results were expressed as means (standard error of the mean [SEM]) and analyzed by one-way analysis of variance followed by Tukey’s test. p Values < 0.05 were considered significant. Graph Pad Prism software was used (version 5.01 for Windows, GraphPad Software, San Diego, California, USA, www.graphpad.com).
Results
Effect of febuxostat on oxidative stress parameters
Oxidative stress plays a role in the pathogenesis of EH. Thus, we explored the effect of treatments on the uterine lipid peroxidation, GSH content, SOD activity, and nitric oxide (NO) level. Table 2 revealed significant MDA rise, GSH drop, SOD decline, and NO elaboration in the uteri of EV group as compared to control groups. However, febuxostat administration improved the previous parameters significantly as compared to the EV group (Table 2).
Effect of febuxostat on uterine MDA, GSH, SOD, and NO (as nitrites/nitrates) on EV-induced EH in rats.a

MDA: malondialdehyde; GSH: glutathione; SOD: superoxide dismutase; NO: nitric oxide; EV: estradiol valerate.
a All parameters are expressed as means ± SEM (n = 5–6).
b Significantly different from the control group at p < 0.05.
c Significantly different from the EV group at p < 0.05.
Effect of febuxostat on uterine histology and PTEN expression
Control rats showed endometrium with a normal proportion of its glandular and stromal components (Figure 1). A significant change was observed in the EV group. There was a great degree of glandular proliferation, crowding out the intervening stroma and often eventuating in the presence of glands that are markedly variable in size and shape, some with totally anarchic side buds and outpunching. There was a marked increase in vascularity and inflammatory cell infiltration. There were features of atypia; nuclear rounding (as compared with the uniform columnar nuclei of hyperplasia without atypia), nucleomegaly, and nuclear pleomorphism. These findings completely regressed with coadministration of febuxostat. To confirm our microscopical findings, we examined PTEN expression immunohistochemically. Strong PTEN expression was observed in normal control groups. However, the EV group showed very few positive signals as compared to control rats. Febuxostat significantly increased PTEN expression in the endometrium of treated rats (Figure 2).

Gross pictures of the uteri of the experimental groups. (a) Carboxylmethyl cellulose control, (b) febuxostat control, (c) EV treated, and (d) EV + febuxostat. Control groups show average sizes uteri. Enlarged uteri were observed in EH and treated groups. Photomicrographs of endometrial tissue with hematoxylin and eosin stain at 20× magnification. (e) Carboxylmethyl cellulose control, (f) febuxostat control, (g) EV treated, and (h) EV + febuxostat. Control groups show normal histological appearance, endometrial glands (arrows), and abundant stroma in between. The EH group shows dilated crowded irregularly shaped glands. An obvious increase in the gland to stroma ratio and little intervening stroma were seen. Dilated congested vessels and ciliated endometrial epithelium could be seen (arrows). Treated group shows the nearly normal histological appearance of endometrial glands (arrows) and abundant stroma in between. Photomicrographs of endometrial tissue: (i) a photomicrograph of endometrial tissue with hematoxylin and eosin stain at 40× magnification for the EV group. It shows a voluminous gland with multiple intraluminal microlumens (cribriform pattern) and pleomorphic and round nuclei and (j) a photomicrograph of endometrial tissue with hematoxylin and eosin stain at 40× magnification for the EV group. It shows massive inflammatory cell infiltration mainly eosinophils and neutrophils (rectangle) that invade (oval) the abnormal epithelium (arrows). EV: estradiol valerate; EH: endometrial hyperplasia
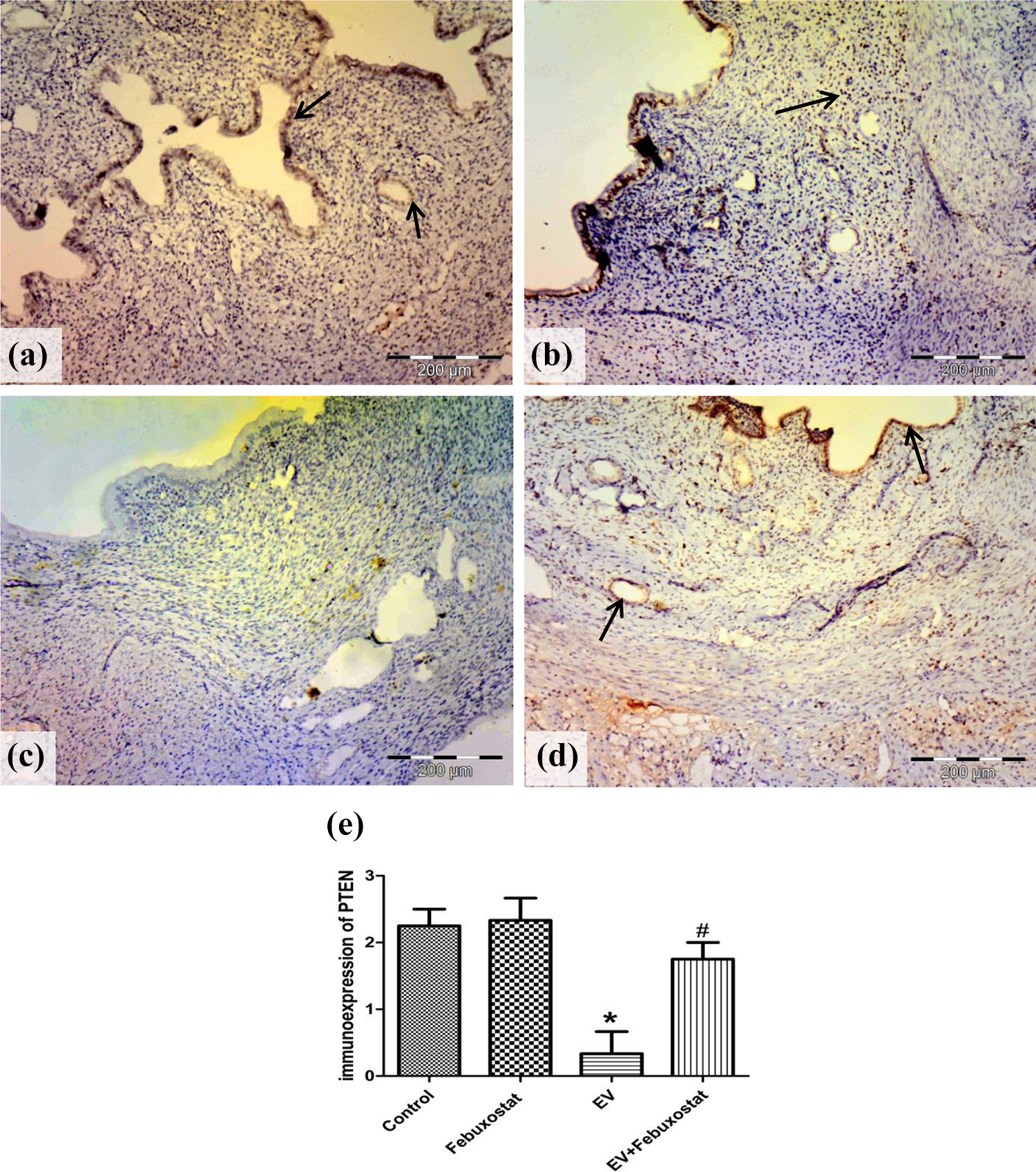
Photomicrographs of endometrial tissue for PTEN immunostaining at 40× magnification. (a) Carboxylmethyl cellulose control, (b) febuxostat control, (c) EV treated, and (d) EV + febuxostat. The control groups show strong immunoreactivity for PTEN (arrows) in the epithelial, the glandular, and the stromal cells. The EV group shows negative PTEN expression. The treated group shows strong PTEN expression (arrows). (e) The semiquantitative scoring of PTEN. PTEN: phosphatase and tensin homolog; EV: estradiol valerate. *Significantly different from the control group at p < 0.05. #Significantly different from the EV group at p < 0.05.
Effect of febuxostat on gene expression of uterine PI3K, Akt, and HIF-1α
EV showed a significant increase in expression of the three genes by qRT-PCR. Febuxostat coadministration with EV at a dose of 10 mg/kg nearly restored the normal expressions of the three genes (Figure 3(a), (b), and (c)). However, the febuxostat control group showed a significant decrease in the gene expression of Akt as compared to the normal control group (Figure 3).

Real-time PCR for quantification of mRNA in the endometrial tissue. Relative expression of (a) PI3K, (b) Akt, and (c) HIF-1α. Data are presented as mean ± SEM (n = 6) *Significantly different from the control group at p < 0.05. #Significantly different from the EV group at p < 0.05. PCR: polymerase chain reaction; mRNA: messenger RNA; EV: estradiol valerate; PI3K: phosphatidylinositol-3-kinase; HIF-1α: hypoxia-inducible factor 1 alpha.
Effect of febuxostat on protein expression of uterine VEGF-A and IL-6
The level of the angiogenic factor VEGF-A displayed a significant increase in the uterine tissue treated by EV from control values. Febuxostat-treated group showed a reduction in VEGF-A values significantly to EV group (Figure 4(a)). The reflection of increased VEGF on endometrial tissue of EV group was demonstrated in Figure 5. Uteri of EV group also displayed marked upregulation of protein expression of the cytokine IL-6 as compared to the control group. IL-6 significantly decreased with febuxostat treatment as compared to the EV group (Figure 4(b)).

ELISA analysis for protein expressions in the endometrial tissue. (a) VEGF-A and (b) IL-6. Data are presented as mean ± SEM (n = 6). *Significantly different from the control group at p < 0.05. #Significantly different from the EV group at p < 0.05. ELISA: enzyme-linked immunosorbent assay; EV: estradiol valerate; VEGF: vascular endothelial growth factor; IL-6: interleukin-6.

Photomicrographs of endometrial tissue with hematoxylin and eosin stain at 40× magnification. (a) Carboxylmethyl cellulose control, (b) febuxostat control, (c) EV treated, and (d) EV + febuxostat. The EV group shows marked angiogenesis in the stroma (arrows). The stroma of control and treated groups shows normal vascularity without new blood vessels formation. EV = estradiol valerate..
Discussion
The present study revealed a protective role of the xanthine oxidase inhibitor, febuxostat, in EH induced by EV. EH is a precancerous condition that is encountered among the most common and irritant female gynecological pathologies with bleeding comorbidity. 4 In the current study, EV administration disturbed uterine oxidative indices of rats, which were improved by febuxostat treatment. It was found that decreased SOD activity and increased lipid hydroperoxides in the endometrial tissue of EH patients were reflections of reactive oxygen species (ROS) overproduction. 27 Excess estrogen can induce ROS and oxidative damage from its intermediate metabolites, 4-hydroxyestradiol semiquinone and quinone, and specific and local formation of 4-hydroxyestradiol is central for endometrial carcinogenesis. 28,29 On the other hand, the role of xanthine oxidase in the formation of semiquinone free radicals from estrogen was reported. 30 Xanthine oxidase is expressed in uterine tissue and linked to the pathogenesis of different uterine diseases such as endometriosis and adenomyosis. 11 Based on the previously mentioned data, it seems that febuxostat ameliorated the bad effect of EV on the oxidative parameters via xanthine oxidase inhibition in agreement with Heikal et al. 31 and Jacob et al. 32 They conveyed the antioxidant effect of febuxostat to its inhibition of xanthine oxidase-induced ROS in models of hyperlipidemia and burn.
Protein PTEN is a tumor suppressor protein; its loss in the endometrial tissue indicates cellular atypia in EH and endometrial carcinoma. 33 Here, PTEN expression was nearly lost in the uteri of the EV group while epithelial atypia was observed and vascularity increased; this is in line with Abdelzaher et al. 16 The necessary role of PTEN in the development of complex atypical hyperplasia was assessed in our study by determining its downstream pathway PI3K/Akt. The current results revealed the upregulation of PI3K and Akt genes expression in the uteri of the EV group in line with Gao et al. and their drop with febuxostat treatment. Protein PTEN acts as a negative regulator that can dephosphorylate the PI3K pathway and inhibit the activation of Akt and its downstream targets. 34 Accordingly, loss of PTEN permits activation of the PI3K/Akt pathway that regulates several downstream pathways sufficient for complex atypical hyperplasia to develop. 35 These data strongly support the findings of the current study. On the other hand, febuxostat coadministration prevented the atypical proliferative changes and restored the ratio between the endometrial stroma to the glandular tissue along with the increase of PTEN expression and normalization of PI3K/Akt. In agreement with the current result, febuxostat conferred antioxidant and anti-inflammatory effects on lung tissue by inhibiting xanthine oxidase-induced ROS that is based on the reduction of the PI3K/Akt pathway. 36 It was proved that ROS was able to stimulate PI3K/Akt via reversible inactivation of PTEN within the cell. 37,38 This PTEN inhibition is essential for ROS-induced growth factors to act and for a tumor to develop. 39 Based on the previous data, it was supposed that febuxostat prevents PTEN inactivation and its downstream activation of PI3K/Akt through its antioxidant effect as a xanthine oxidase inhibitor. Moreover, loss of PTEN and subsequent activation of Akt was implicated in HIF-1α stabilization and VEGF production during tumorigenesis. 40
Hypoxia-inducible factor 1 alpha is an essential adaptive factor to hypoxia that was found elevated in endometrial tissue during EH and endometrial carcinoma. 41 Besides, HIF-1α plays an important role in cell proliferation and tumor angiogenesis by regulating VEGF. 42 The present study revealed an increase of HIF-1α gene and VEGF protein expressions and angiogenesis in the endometrium of EV rats that was reduced with febuxostat treatment. In accordance with our findings, estrogen was demonstrated to induce VEGF-A signaling that initiates endometrial growth leading to hyperplasia and cancer. 43 Furthermore, estrogen-induced activation of HIF-1α and VEGF expression in the uterus is mediated by the PI3K/Akt pathway. 44 Thus, the ameliorative effect of febuxostat on PI3K/Akt may explain the reduction of the HIF-1α/VEGF pathway and the decreased endometrial angiogenesis.
The putative role of estrogen in the pathogenesis of EH broadens to involve an inflammatory response in the endometrium. Pro-inflammatory environment may initiate a neoplastic transformation. 45 This was clear in the current study through the upregulation of the pro-inflammatory cytokine IL-6 protein expression in the uteri of the EV group. It was found that IL-6 with other cytokines involved in the progression of EH from simple through complex and atypical EH to endometrial carcinoma. 5,46 However, febuxostat decreased cytokines such as IL-6 due to its anti-inflammatory effect as previously reported in mice with atherosclerosis and patients with gout. 47,48 It is worth noting that IL-6 is a multifunctional cytokine. It could regulate VEGF to increase the angiogenesis and the invasiveness of gastric carcinoma in rats, 49 thus, IL-6 reduction by febuxostat may help in its ameliorative effect on VEGF at the level of protein expression and the histopathological picture. The limitations of our study were the lack of facility to investigate xanthine oxidase expression in the endometrium. However, the current study revealed a valuable protective role for a specific xanthine oxidase inhibitor against estrogen-induced EH; this may offer a new indication for febuxostat therapy.
Conclusion
Febuxostat offered a protection against estrogen-induced hyperplasia in rats through antioxidant, anti-inflammatory, anti-proliferative, and via modulation of PTEN expression.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
