Abstract
Background
Stroke is a primary global health concern, resulting in substantial mortality and morbidity, with a significant proportion of cases attributed to ischemic stroke. While reperfusion therapies, including thrombus dissolution and removal of thrombus mechanically, are essential in the critical management of ischemic stroke, they can also lead to a paradoxical increase in neuronal damage due to reperfusion injury.
Objectives
This phenomenon has a significant impact on elevating disability rates and mortality linked to stroke, with survivors often experiencing persistent cognitive, motor, and psychological impairments that significantly impact their quality of life. Furthermore, the long-term consequences of cerebral ischemic–reperfusion injury place a substantial burden on healthcare systems, caregivers, and families, highlighting the need for effective strategies to mitigate this devastating condition. Therefore, in the present investigation, we explored the efficacy of phytoalkaloid dictamnine potency in ameliorating neuronal damage caused by cerebral ischemic–reperfusion.
Materials and Methods
In vivo condition, middle cerebral artery occlusion (MCAO) was performed in C57BL/6J mice and treated with different concentrations of dictamnine. The experimental animals were subjected to neurological scoring, and the tissue and blood samples were collected after the treatment period. Cerebral damage was assessed by analyzing the brain edema, infarct volume, and Evans blue stain leakage assay. Oxidative stress markers and antioxidants were quantified to investigate the oxidative damage.
Results
Nitrates matrix metalloproteinase-2 (MMP2) and matrix metalloproteinase-9 (MMP9) were measured to examine the vasodilation and blood–brain barrier (BBB) disruption induced by MCAO. Nuclear factor kappa B (NF-κB), cyclooxygenase-2 (COX-2), prostaglandin E2 (PGE2), and cytokines were measured to assess the neuroinflammation. The efficacy of dictamnine in attenuating NOD-like receptor family, pyrin domain-containing protein 3 (NLRP3) inflammasome, and toll-like receptor 4 (TLR4) signaling was evaluated to confirm the pharmaceutical potency of dictamnine in ameliorating cerebral ischemic–reperfusion. In vitro, SH-SY5Y cells were subjected to oxygen and glucose deprivation/reperfusion (OGD/R) and subsequently incubated with varied concentrations of dictamnine. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), lactate dehydrogenase (LDH) assay, and 4′,6-diamidino-2-phenylindole (DAPI) staining were performed with dictamnine-treated cells to confirm the inhibitory potency of dictamnine against the neuronal cell apoptosis induced by OGD/R. Dictamnine treatment effectively scavenged oxidative stress and inhibited the NLP3 and TLR4 signaling, thereby preventing neuroinflammation induced by MCAO. The results of our in vitro study also confirm the ameliorative potency of dictamnine against cerebral ischemic–reperfusion-induced neuronal damage.
Conclusion
Altogether, our research findings prove dictamnine is a potent bioactive compound that can be further analyzed to be formulated as a drug to treat cerebral ischemia.
Keywords
Introduction
Cerebral ischemia/reperfusion (I/R) injury is a diverse pathophysiology that takes place when cerebral vascularization is obstructed (ischemia) and subsequently restored (reperfusion). This sequence can cause substantial brain damage and is a primary factor in the morbidity and mortality linked to stroke and other cerebrovascular conditions (DeSai & Hays Shapshak, 2023). The global population is aging, with projections indicating that by 2050, approximately 2 billion people will be 60 years or older (World Health Organization, 2022). This demographic trend is expected to result in a rise in morbidity and death associated with I/R injury. Factors, specifically urbanization and lifestyle changes, including diet and physical activity, are also contributing to the heightened risk of I/R-related conditions (Tzoulaki et al., 2016; Wang et al., 2021). Over the past 30 years, 80% of developing countries have experienced a shift in disease patterns from contagious to noninfectious diseases.
Among these conditions, stroke has emerged as one of the leading causes of debility. It ranks as the second major cause of death and the third significant contributor to disability-adjusted life years (Murray et al., 2012). Each year, an estimated 13.7 million individuals worldwide experience a stroke, resulting in 5.5 million deaths and 16.9 million disability-adjusted life years lost (GBD 2016 Neurology Collaborators, 2019). Ischemic stroke (IS) represents 70% of all stroke cases and carries a likelihood risk of long-term recurrence. IS was responsible for 3.29 million deaths, which constituted 50.3% of all stroke-related fatalities and 17.7% of deaths related to cardiovascular diseases by the year 2019. This underscores the critical importance of preventing IS (Benjamin et al., 2019; Ding et al., 2022; Phipps & Cronin, 2020). The economic impact of I/R-related disorders is significant, with annual costs estimated to range between $134 billion and $229 billion (Strilciuc et al., 2021).
At the time of ischemic attack, the deprivation of oxygen and glucose forces a shift from aerobic to anaerobic metabolism, causing the buildup of lactate and other metabolic byproducts. When reperfusion occurs, it initiates a series of episodes comprising oxidative stress, inflammation, and excitotoxicity, aggravating the initial injury (Kuriakose & Xiao, 2020). NOD-like receptor family, pyrin domain-containing protein 3 (NLRP3) inflammasome is implicated in the development of cerebral I/R injury. Research has shown that mice lacking NLRP3 have reduced infarction and improved neurological outcomes (Bai et al., 2021). Similarly, toll-like receptor 4 (TLR4) is crucial in triggering inflammation following cerebral I/R. Research confirmed that TLR4-deficient mice experience reduced inflammation and enhanced functional recovery (Yang et al., 2011). Targeting the NLRP3 inflammasome and TLR4 signaling pathways could provide innovative therapeutic strategies to reduce inflammation and enhance outcomes subsequent to cerebral I/R injury (Zhang et al., 2023).
Swift reinstatement of vascular flow is critical for minimizing tissue damage. However, reperfusion itself can also contribute to injury, highlighting the need for adjunctive therapies to mitigate this effect (Mokin et al., 2018). Current stroke medications aim to slow the progression of ischemia, facilitate reperfusion, and restore brain tissue function. Furthermore, these therapies aim to minimize neuronal cell injury and mortality resulting from ischemia and the metabolic cascade that follows sudden reperfusion (Hurd et al., 2021). Dictamnine, a furoquinoline alkaloid isolated from the root barks of Dictamnus sp. in the family Rutaceae (Lv et al., 2015) and is extensively used in Chinese medicine to treat various ailments such as eczema, jaundice, rheumatic arthritis, atherosclerosis, and so on (Wang et al., 2013). The present investigation aimed to assess the potency of dictamnine to ameliorate the neuronal damage induced by cerebral ischemic–reperfusion via attenuating NLRP3 inflammasome signaling.
Materials and Methods
Chemicals
The dictamnine (purity: 95%) and other chemicals were purchased from Sigma–Aldrich, USA. The assay kits for biochemical parameters were acquired from LifeSpan BioSciences (LSBio), Antibody Cambridge (Abcam), and Thermo Fisher Scientific, USA.
Animals and Grouping
C57BL/6J mice, aged 2-month-old weighing 22–25 g, were obtained from an institutional animal facility and maintained in a lab that was adopted with international standards prescribed for rearing animals. The protocols for the animal experimentations were verified and approved by the institutional animal ethics committee. Seven days of acclimatization were done before the initiation of the experiment, and after that, mice were grouped in groups I–IV, with six mice in each group (n = 6). Group I served as the control group, consisting of mice that underwent sham surgery. Group II was subjected to an I/R model, which involved inducing cerebral arterial insufficiency followed by revascularization. Groups III and IV received pretreatment with dictamnine at doses of 25 and 50 mg/kg body weight for seven successive days before being subjected to the same I/R procedure. After 24 h post-revascularization, the mice were sacrificed by cervical dislocation under anesthesia, and blood samples were collected from the retro-orbital sinus. At the same time, the brains were isolated for further analysis.
Cerebral Ischemia Induction
Cerebral ischemia was induced in C57BL/6J mice, which were acclimatized to handling and the surgical environment, followed by a 2-h fasting period. Anesthesia was induced using isoflurane and maintained via inhalation. The surgical area, typically the neck, was shaved and disinfected. For transient focal ischemia, the right common and external carotid arteries were uncovered, and a 6-0 nylon suture was done into the external carotid artery, advancing it into the middle cerebral artery (MCA) for 30–60 min to induce occlusion. After the designated ischemic period, the blood flow was restored by removing the suture. Postoperatively, the animals were monitored until fully awake and provided with a warm recovery environment, and analgesics were administered as needed (Zhao et al., 2021). Neurological function was assessed using standardized scoring methods, and tissues were collected for further analysis. The experiment was concluded with humane euthanasia after the treatment period. The protocol adhered to institutional ethical guidelines for animal research.
Scoring of Neurologic Deficit
The assessment of behavioral deficits in mice was conducted using the Longa score (Longa et al., 1989), which ranges from 0 to 4 points. A score of 0 indicated no noticeable neurological dysfunction, while 1 point represented debility and an incapability to fully extend the left forelimb. A score of 2 was assigned for trunk rotation to the opposite side during movement, 3 points indicated a tipping of the trunk to the opposite side while walking, and a score of 4 signified an inability to walk or a comatose state.
Assessment of Cerebral Infarct
Experimental animals were euthanized with an injection of 2% pentobarbital sodium, and the brains were quickly harvested following an assessment of neurological deficits. The brains were then sliced into four coronal sections and stained with a 2% triphenyltetrazolium chloride solution. All resulting images were captured and investigated with the ImageJ software [National Institutes of Health (NIH), USA], following the established protocol (Li et al., 2020).
Assessment of Cerebral Edema in Dictamnine-treated Cerebral Ischemia-stimulated Mice
The brain tissues were excised from euthanized mice, followed by weighing the fresh tissue to obtain the wet weight. The samples are dried in an oven at 105°C for 24 h to determine the dry weight. The percentage of water content was evaluated by calculating the difference between the wet and dry brain weights and the cerebral edema was calculated using the formula: [(wet weight – dry weight)/wet weight] × 100, which provides a reliable indicator of edema (Tang et al., 2020).
Analysis of Evans Blue Leakage in Dictamnine-treated Cerebral Ischemia-stimulated Mice
A 2% solution of Evans blue dye was administered intravenously via the tail vein. After a 3-h infusion, the mice were euthanized and underwent transcardiac perfusion with phosphate-buffered saline (PBS). The brain was subsequently extracted, weighed, and homogenized in 1 mL of PBS, followed by sonication and centrifugation for 30 min at 9,424 g at 4°C. The resulting supernatant was then collected for analysis. The concentration of Evans blue stain was quantified using spectrophotometry at a wavelength of 620 nm, with results calculated based on a standard curve.
Assessment of Oxidative Stress in Dictamnine-treated Cerebral Ischemia-stimulated Mice
Malondialdehyde (MDA) levels were measured in experimental mice as a biomarker of lipid peroxidation. Brain tissue samples were homogenized in a mixture of trichloroacetic acid and thiobarbituric acid, and then incubated at 95°C for 1 h to facilitate the reaction between MDA and thiobarbituric acid. The mixture was centrifuged, and the supernatant was collected for spectrophotometric measurement at 532 nm (Ohkawa et al., 1979).
The brain homogenates of control and experimental mice were subjected to analysis of antioxidants. Superoxide dismutase (SOD) activity was measured spectrophotometrically at 560 nm, as described by Kakkar et al. (1984), while catalase (CAT) activity is evaluated by monitoring the decomposition of hydrogen peroxide at 240 nm, as outlined by Aebi (1984). Glutathione peroxidase (GSH-Px) activity is assessed using a coupled enzyme reaction that consumes glutathione (GSH), as described by Flohé and Günzler (1984), and GSH levels are quantified using a colorimetric assay at 412 nm, as reported by Ellman (1959).
8-Hydroxy-2′-deoxyguanosine (8-OHdG) levels that signify the severity of oxidative stress were quantified using the enzyme-linked immunosorbent assay (ELISA) kit procured from LSBio. The assay was done as per the protocol prescribed by the manufacturer.
Assessment of Vasodilation and Blood–Brain Barrier (BBB) Disruption in Dictamnine-treated Cerebral Ischemia-stimulated Mice
Nitrate levels were estimated in mouse plasma and tissues using the Griess reaction. Plasma or tissue samples were deproteinized with zinc sulfate and centrifuged to remove precipitated proteins. The supernatant was then mixed with Griess reagent, which converted nitrates to nitrites, and the resulting mixture was incubated for 10 min at 37°C. The absorbance was measured at 540 nm using a spectrophotometer, and the nitrate concentration was calculated using a standard curve prepared with known concentrations of sodium nitrate (Green et al., 1982).
Matrix metalloproteinase-2 (MMP-2) and matrix metalloproteinase-9 (MMP-9) levels were estimated in experimental mice’s cortex and striatum tissues using an ELISA kit purchased from Abcam. First, the tissues were homogenized in a lysis buffer and centrifuged to obtain clear supernatants. The tissue supernatants and standards were then added to a precoated MMP-2 and MMP-9 ELISA plate. The samples and standards were incubated with a prediluted antibody for 2 h, and secondary antibody conjugated to an enzyme was added. After thorough washing, a substrate solution was added, and the enzymatic reaction was allowed to develop. The absorbance was quantified at 450 nm using a plate reader, and the concentrations of MMP-2 and MMP-9 in the samples were quantified by comparing the absorbance values to the standard curve.
Assessment of Nuclear Factor Kappa B (NF-κB)– Cyclooxygenase-2 (COX-2)–Prostaglandin E2 (PGE2) Signaling Pathway in Dictamnine-treated Cerebral Ischemia-stimulated Mice
NF-κB–COX-2–PGE2 signaling pathway mediates inflammatory responses and cell death during cerebral ischemia, highlighting potential therapeutic targets for mitigating ischemic brain injury. NF-κB, COX-2, and PGE2 levels were measured using the ELISA kits acquired from MyBiosource.
Assessment of Inflammatory Signaling in Dictamnine-treated Cerebral Ischemia-stimulated Mice
The inflammatory signaling proteins tumor necrosis factor-alpha (TNF-α), interleukin 1 beta (IL-1β), interleukin 6 (IL-6), and interleukin 10 (IL-10) are in the brain tissue of the experimental mice. ELISA kits purchased from Abcam quantified TNF-α, IL-1β, IL-6, and IL-10 levels. The assay was performed as per the manufacturer’s instructions.
Assessment of Cerebral Ischemic Signaling in Dictamnine-treated Cerebral Ischemia-stimulated Mice
NLRP3 and TLR4 play critical roles in the inflammatory response associated with cerebral ischemia. NLRP3 and TLR4 were quantified in the brain tissues of experimental mice with the ELISA technique. The ELISA kits were purchased from LSBio. The test was conducted following the protocol prescribed by the manufacturer.
Culturing of SH-SY5Y Neuroblastoma Cells
SH-SY5Y neuroblastoma cells were purchased from ATCC, USA, and maintained in a growth medium made up of Dulbecco’s Modified Eagle Medium (DMEM), supplemented with 10% fetal bovine serum (FBS), and 1% penicillin-streptomycin. The cells were incubated in a humidified environment at 37°C with a 5% CO2 concentration. Upon reaching around 80% confluence, the cells were detached using trypsin-ethylenediaminetetraacetic acid (EDTA) and transferred to fresh culture flasks with a new medium to support further growth. The cells’ morphology and confluence levels were regularly monitored, and the medium was refreshed every 2–3 days to maintain optimal growth conditions. Additionally, the cells were passaged every 4–5 days to prevent overgrowth and sustain healthy cultures.
I/R Induction and Dictamnine Treatment
OGD/R was induced in SH-SY5Y neuroblastoma cells; the cells were initially cultured in DMEM supplemented with 10% FBS and 1% penicillin-streptomycin until they reached 80% confluence. Subsequently, the culture medium was replaced with glucose-free DMEM at 1% O2, 5% CO2, and 37°C for 6 h to simulate ischemic conditions following the method described by Morán et al. (2017). After this I/R, the cells were returned to normoxic conditions maintained at 37°C in a humidified atmosphere containing 5% CO2 for 24 h to allow for reperfusion. Different concentrations of dictamnine were introduced to the SH-SY5Y cells concurrently with reoxygenation, and the cells were then cultured for an additional 24 h.
MTT Assay
I/R-induced untreated and dictamnine-treated SH-SY5Y cells were replaced with fresh medium containing 0.5 mg/mL MTT and incubated for 4 h at 37°C until purple formazan crystals formed. The crystals were then dissolved in dimethyl sulfoxide (DMSO), and the absorbance was measured at 570 nm using a microplate reader.
LDH Assay
The culture medium was collected from I/R-induced untreated and dictamnine treated SH-SY5Y cells, and LDH levels were measured using a commercially available kit (Thermo Fischer Scientific), following the manufacturer’s instructions. The substrate reaction was incubated for 30 min, and the absorbance was measured at 490 nm.
DAPI Staining
DAPI staining was utilized to visualize cell nuclei in the cultured cells. Both untreated and dictamnine-treated SH-SY5Y cells, subjected to I/R, were fixed with 4% paraformaldehyde for 15 min at room temperature to preserve the cellular structure. After fixation, the cells were rinsed three times with PBS to remove any excess fixative. The nuclei were then stained with a DAPI solution (1 µg/mL in PBS) for 10 min in the dark at room temperature. Following staining, the cells were washed again with PBS to remove unbound DAPI. The stained cells were mounted and examined under a fluorescence microscope using a 358 nm filter to detect DAPI fluorescence. Images were captured, and nuclear morphology and density were analyzed using ImageJ software.
Statistics
Data analysis was conducted using SPSS version 19.0 and expressed as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) was performed to compare differences among multiple groups. For post hoc comparisons, Tamhane’s T2 test was utilized. A p value of less than 0.05 was considered to signify statistical significance.
Results
Phytoalkaloid Dictamnine Attenuates Cerebral Damage in Post-cerebral I/R Injury
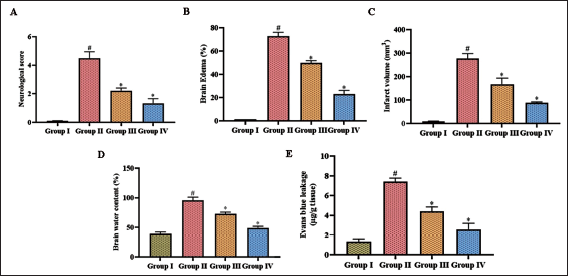
Figure 1 depicts the cerebral damage induced by middle cerebral artery occlusion (MCAO) and the ameliorative potency of dictamnine against it. The neurological scoring of MCAO-stimulated was considerably increased to 4.7 ± 0.0008, whereas it was decreased to 1.9 ± 0.0004 and 1.1 ± 0.0006, respectively, with 25 and 50 mg/kg dictamnine treatment. The water content in the brain region of MCAO-stimulated mice was increased, as evidenced by the brain edema results. MCAO-stimulated untreated mice showed increased levels of brain edema at 74 ± 0.007%, which was reduced to 52 ± 0.003% and 22 ± 0.008%, respectively with 25 and 50 mg/kg dictamnine treatment. Dictamnine treated mice exhibited considerably decreased levels of infarct volume 162 ± 4.2 mm3 (25 mg/kg), 89 ± 0.9 mm3 (50 mg/kg) and also decreased the Evans blue leakage to 4.1 ± 0.007 µg/g tissue (25 mg/kg), 2.3 ± 0.009 µg/g tissue (50 mg/kg) compared to the MCAO stimulated untreated mice which exhibited 285 ± 2.5 mm3 of cerebral infarct volume and 1.7 ± 0.0004 µg/g tissue of Evans blue leakage.
Phytoalkaloid Dictamnine Scavenges Oxidative Stress in Post-cerebral I/R Injury
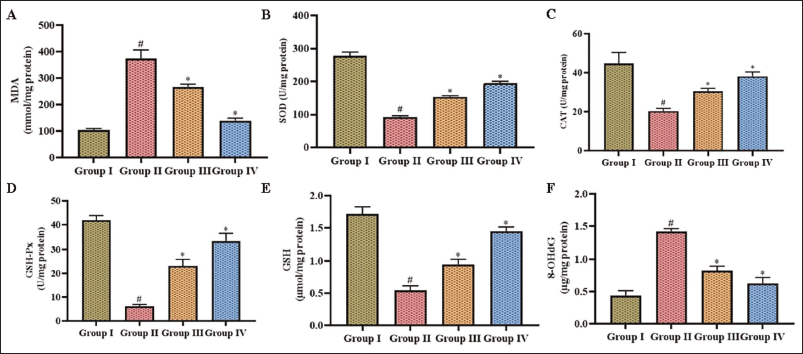
Oxidative damage induced by MCAO in experimental mice and the scavenging activity of dictamnine against it were assessed, and the results are illustrated in Figure 2. 8-OHdG is a prominent marker of oxidative deoxyribonucleic acid (DNA) damage, formed when reactive oxygen species attack deoxyguanosine in DNA, leading to potential mutagenic changes and malondialdehyde, a byproduct of lipid peroxidation generated when free radicals damage polyunsaturated fatty acids in cell membranes. Both 8HdG and MDA were elevated in the MCAO-stimulated untreated mice compared to the other groups. Dictamnine treatment considerably reduced 8-OHdG MDA levels and enhanced antioxidant levels in a dose-dependent manner. The levels of antioxidants SOD, CAT, GSH-Px, and GSH were substantially reduced in the MCAO-induced untreated mice compared to the control and dictamnine treated mice.
Phytoalkaloid Dictamnine Ameliorates Vasodilation and BBB Disruption in Post-cerebral I/R Injury
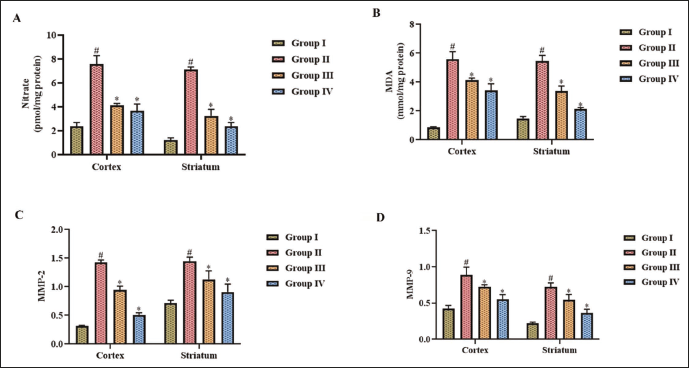
Vasodilation and BBB disruption are critical events that occur during cerebral ischemia, significantly impacting the pathophysiology of the condition. In MCAO-induced untreated mice, the nitrate levels were enhanced considerably, depicting the occurrence of vasodilation, and MMP-2 and MMP-9 levels were increased in the MCAO untreated mice, causing BBB disruption. An increase in nitrate and MMP2, MMP9 levels was detected in both cortex and striatum brain regions of MCAO untreated mice. Dictamnine treatment decreased the levels of nitrate, MMP2, and 9 in the cortex and striatum of the MCAO-induced mice. Then, the 25 mg/kg dictamnine treatment and the 50 mg/kg dictamnine treatment significantly attenuated the levels of nitrate and MMP2, MMP9 in MCAO-induced mice. The cortex and striatum MDA levels, an oxidative stress marker, were substantially enhanced in the MCAO-induced mice than in the dictamnine-treated mice (Figure 3).
Phytoalkaloid Dictamnine Regulates the NF-κB–COX-2–PGE2 Signaling Pathway in Post-cerebral I/R Injury
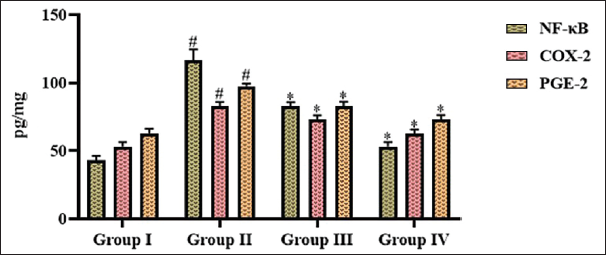
MCAO induction significantly enhanced the NF-κB and PGE2 levels, whereas treatment with dictamnine decreased NF-kB and PGE2 levels. Dictamnine treatment substantially increased the COX-2 expression in the MCAO-induced mice (Figure 4). The decrease in NF-kB, PGE2, and enhanced COX-2 levels were observed in a dose–responsive manner in the dictamnine manner.
Phytoalkaloid Dictamnine Attenuates Inflammatory Signaling in Post-cerebral I/R Injury
Figure 5 illustrates proinflammatory cytokines TNF-α, IL-1β, IL-6, and inflammatory attenuating cytokine IL-10 levels in the MCAO-induced untreated and dictamnine treated mice. MCAO induction had substantially increased the TNF-α, IL-1β, and IL-6 levels. It decreased the IL-10, whereas dictamnine treatment considerably enhanced IL-10 expression and attenuated the synthesis of TNF-α, IL-1β, and IL-6 in MCAO-induced mice. Compared to IL-1β and IL-6, the levels of TNF-α were considerably decreased with dictamnine treatment.
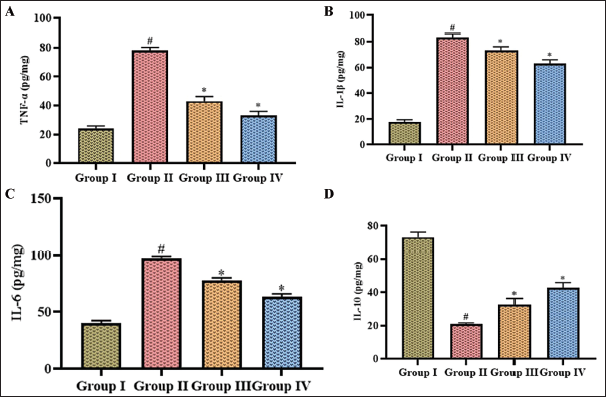
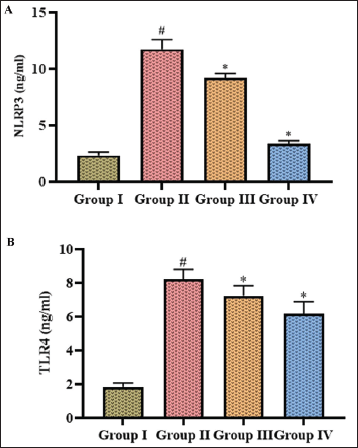
Phytoalkaloid Dictamnine Inhibits Cerebral Ischemic Signaling in Post-cerebral I/R Injury
Both NLRP3 and TLR4 are viewed as possible therapeutic targets for minimizing inflammation and neuronal damage in stroke patients; hence, these levels were quantified in the MCAO-induced untreated and dictamnine treated mice, depicted in Figure 6. Dictamnine treatment decreased the levels of NLRP3 to 9.1 ± 0.0004 ng/mL (25 mg/kg treatment). 3.9 ± 0.0003 ng/mL (50 mg/kg treatment) and TLR4 levels to 7.1 ± 0.0007 ng/mL (25 mg/kg treatment), 6.5 ± 0.0009 ng/mL (50 mg/kg treatment) in the MCAO induced mice. The MCAO-induced mice exhibited significantly enhanced levels of 12.3 ± 0.0008 ng/mL of NLRP3 and 7.8 ± 0.0008 ng/mL of TLR4. The levels of NLRP3 and TLR4 were 2.4 ± 0.0003 and 1.8 ± 0.0003 ng/mL respectively.
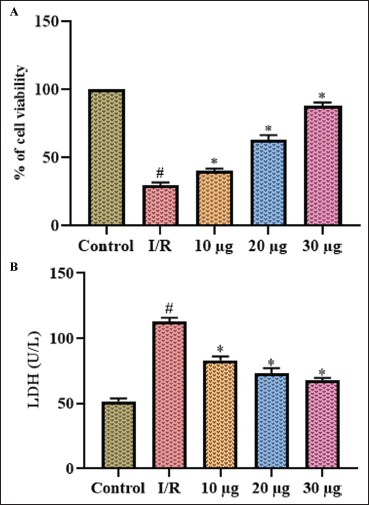
Phytoalkaloid Dictamnine Attenuates Apoptosis in I/R-stimulated SH-SY5Y Cells
Figure 7 illustrates the apoptosis induced by OGD/R in the neuroblastoma cell line, which was assessed using MTT and LDH assay. OGD/R stimulation enhanced the apoptosis induction in the neuroblastoma cells, which was evidenced by 123 ± 0.05 U/L of LDH levels and 22 ± 0.07% of live cells compared to the control cells, which exhibited 50 ± 0.03 U/L of LDH levels. Dictamnine treatment significantly decreased the levels of LDH to 78 ± 0.06 U/L, 65 ± 0.09 U/L, 59 ± 0.01 U/L with 10, 20, and 30 µg treatment and increased the percentage of live cells to 34 ± 0.05, 68 ± 0.09, and 88 ± 0.06 respectively.
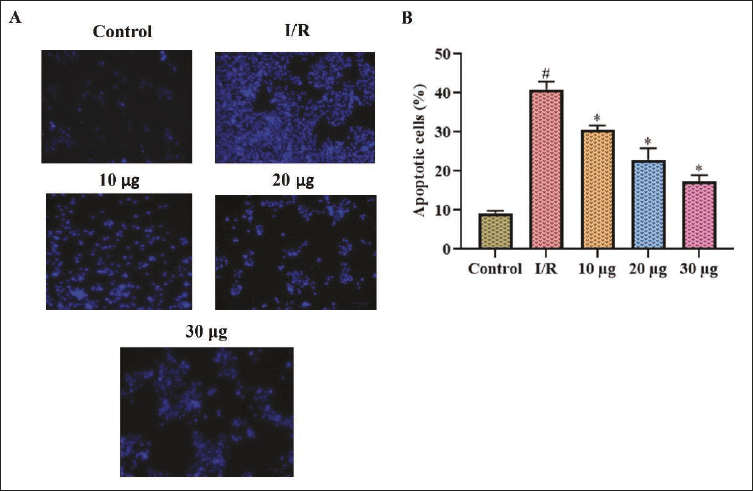
Phytoalkaloid Dictamnine Ameliorates Nuclear Damage in I/R-stimulated SH-SY5Y Cells
The ameliorative potency of dictamnine against I/R stimulated apoptosis in SH-SY5Y cells was assessed with DAPI staining. A bright blue fluorescence was observed in I/R-stimulated untreated cells, which was measured to exhibit 39 ± 0.009% of dead cells, whereas the treatment with dictamnine decreased the apoptosis and exhibited 27 ± 0.005%, 23 ± 0.01%, and 17.6 ± 0.007% of apoptotic cells. The blue fluorescence exhibited by the SH-SY5Y cells was considerably reduced in the dictamnine treatment in a dose–responsive manner. The control SH-SY5Y cells exhibited minimal fluorescence intensity with 9.8 ± 0.004% of dead cells (Figure 8).
Discussion
Worldwide, stroke is a significant public health issue, standing as the second foremost cause of mortality, with approximately 5.5 million fatalities annually. The aftermath of a stroke can be just as devastating, with nearly half of survivors experiencing permanent disability (Donkor, 2018). A multitude of causative factors stimulates stroke progression, including advanced age, hypertension, obesity, hypocholesteremia, hyperglycemia, smoking, and excessive alcohol consumption. Since aging is predominant among global inhabitants, the incidence of stroke is projected to escalate, potentially leading to a staggering mortality rate of over 12% by 2030 (Xing et al., 2012). Consequently, stroke poses a formidable challenge, placing an enormous economic and social strain on individuals, families, and communities worldwide.
The IS injuries occur in two phases. The foremost is ischemic injury, followed by reperfusion injury (Kunz et al., 2010; Wu et al., 2018). Treatment strategies for IS can be broadly categorized into two main approaches: restoring blood flow and safeguarding neuronal health. The former involves using thrombolytic agents or mechanical devices to rapidly reestablish vascularization, typically in a narrow window following the onset of IS. Additionally, antiplatelet and anticoagulant therapies can be employed to prevent subsequent stroke events. In contrast, neuroprotective strategies focus on shielding neurons from the complex cascade of pathological events triggered by IS, aiming to mitigate damage and promote recovery. This dual-pronged approach seeks to address both the immediate and long-term consequences of IS (Green, 2008; Minnerup et al., 2012).
Phytochemicals have garnered significant attention for their potent antioxidant and anti-inflammatory properties, enabling them to simultaneously modulate multiple cellular pathways (Kim et al., 2016). This multifaceted action allows phytochemicals to target various aspects of cellular function, making them promising candidates for therapeutic applications. A recent concept, the “one-drug-multitarget” approach, suggests that compounds capable of interacting with multiple targets may be more effective in treating complex conditions like IS (Chen et al., 2017). One such compound is dictamnine, an alkaloid which is a predominant component present in Cortex dictamni possessing anticancer, antibacterial, and antioxidant properties (Huang et al., 2008; Shi et al., 2019; Wang et al., 2018). Cortex dictamni is prescribed in traditional Chinese medicine to cure inflammatory diseases, jaundice, and others (Chang et al., 2016). Therefore, we examined the impact of dictamnine on cerebral arterial insufficiency followed by revascularization injury in animal and in vitro models.
The ischemic cascade represents an intricate network of interconnected molecular and cellular processes that unfold in response to reduced blood flow to the brain. This complex series of events involves a multitude of deleterious mechanisms, including excessive neuronal excitation, oxidative and nitrative stress, inflammation, and programmed cell death (Pan et al., 2022). These mechanisms converge to cause widespread damage to different cell types, including neurons, glial cells, and endothelial cells, ultimately contributing to the devastating consequences of IS (Iadecola & Anrather, 2011; Xu et al., 2010). Deprivation of oxygen and glucose during ischemic conditions attenuates adenosine triphosphate (ATP) synthesis, increasing intracellular calcium levels (Hu & Song, 2017) and enhancing glutamate signaling, causing excitotoxicity (Xu et al., 2010). It also triggers reactive oxygen species (ROS) synthesis (Bakaeva et al., 2022) and mitochondrial depolarization, eventually leading to neuronal cell death (Baev et al., 2022). Dictamnine treatment substantially increased the antioxidant levels and attenuated the oxidative stress in the MCAO animal model, subsequently preventing neuronal death, as evidenced by the reduced cerebral infarct volume. The attenuation of neuronal apoptosis during OGD/R by dictamnine was also confirmed with our in vitro analysis.
A surge in intracellular sodium levels triggers a series of signaling, including the influx of water into cells through the process of osmosis, causing BBB disruption and the exacerbation of cerebral edema (Wu & Anderson, 2022). The reperfusion-induced brain damage is further compounded by the release of a plethora of proinflammatory mediators, including tumor necrosis factor (TNF), interleukin-1 (IL-1), interleukin-6 (IL-6), interleukin-12 (IL-12), and interleukin-18 (IL-18), by immune cells. Additionally, these immune cells also produce matrix metalloproteinases, particularly MMP-2 and MMP-9, which play a vital role in exacerbating the damage (He et al., 2022). The activation of inducible pathways, including inducible nitric oxide synthase (iNOS) and COX-2, by these cytotoxic agents leads to the impairment of endothelial cells’ BBB disruption. The upregulation of MMPs following IS is a key event that contributes to the breakdown of the BBB, resulting in brain edema, hemorrhagic transformation, and ultimately, the demise of neuronal and glial cells (Yang et al., 2019). Dictamnine treatment significantly attenuated the synthesis of nitrates, MMP2, and MMP9, and it also inhibited the synthesis of inflammatory stimulators NF-κB, COX-2, and PGE2. Dictamnine ultimately prevented BBB disruption and vasodilation in the MCAO animal model, which was manifested with the decrease in cerebral edema percentage of the dictamnine-treated mice.
Therapeutic drugs targeting the NLRP3 and TLR4 signaling pathways have shown promise in treating cerebral ischemic–reperfusion injury by modulating neuroinflammatory responses. Studies have demonstrated that the activation of NLRP3 inflammasomes leads to the release of proinflammatory cytokines, exacerbating neuronal damage during ischemia and reperfusion (Alishahi et al., 2019). Inhibitors of NLRP3, such as MCC950, have been shown to reduce neuronal apoptosis and enhance neurological activity following ischemic events by attenuating inflammatory responses (Chen et al., 2022). Similarly, blockade of TLR4 signaling has been associated with decreased inflammation and oxidative stress, which are critical factors in cerebral ischemia (Kim et al., 2023; Suzuki et al., 2012). By targeting these pathways, therapeutic agents mitigate acute inflammatory damage and promote neuroprotection and recovery, highlighting the potential of NLRP3 and TLR4 as crucial therapeutic targets in cerebral ischemic conditions (Feng et al., 2020; Yang et al., 2020). Therefore, we analyzed the alleviating potency of dictamnine on NLRP3 and TLR4 proteins in the MCAO animal model. Dictamnine profoundly attenuated the NLRP3 and TLR4 signaling and prevented neuroinflammation-induced cerebral ischemia in MCAO animal model. Apart from these findings, the present study has some limitations. For example, the present work failed to disclose the underlying molecular mechanisms of dictamnine’s neuroprotective properties, which have to be addressed in future studies.
Conclusion
In conclusion, dictamnine demonstrates significant therapeutic potential in ameliorating cerebral ischemia by effectively attenuating the NLRP3 inflammasome signaling pathway. The findings suggest that dictamnine’s ability to inhibit NLRP3 activation and subsequent inflammatory cytokine release may protect neuronal cells from ischemic damage and enhance post-ischemic recovery. By reducing neuroinflammation and promoting neuroprotection, dictamnine could serve as a promising candidate for developing novel treatments targeting neuroinflammatory mechanisms associated with cerebral ischemia. Prospective investigations should emphasize discovering the specific molecular mechanisms underlying dictamnine’s potency and evaluating its efficacy in clinical settings, ultimately contributing to improved interventions for patients suffering from IS.
Footnotes
Abbreviations
BBB: Blood–brain barrier; Cerebral I/R: Cerebral ischemia/reperfusion; MCAO: Middle cerebral artery occlusion; MMP: Matrix metalloproteinases; NLRP3: NOD-like receptor family, pyrin domain-containing protein 3; OGD/R: Oxygen and glucose deprivation/reperfusion; TLR4: Toll-like receptor 4.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This work was approved by the institutional Ethical Committee at Yichun People’s Hospital, Jiangxi 336000, China.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
