Abstract
Background
Treatments for ulcerative colitis (UC), a chronic inflammatory bowel illness, have been investigated using Traditional Chinese Medicine (TCM). Uncertainty surrounds the relative safety and effectiveness of TCM in comparison to more traditional therapies like 5-aminosalicylic acid (5-ASA).
Objectives
The purpose of this systematic review and meta-analysis is to assess the safety and effectiveness of TCM therapies in comparison to 5-ASA and placebo in patients with mild-to-moderate UC.
Materials and Methods
We conducted a systematically searched China National Knowledge Infrastructure (CHKD-CNKI), Chinese Biomedical Literature Database (CBM), CINAHL, Embase, MEDLINE, PubMed, Cochrane Library, WANFANG, as well as Web of Science from June 2012 to February 2024 for randomized controlled trials assessing TCM interventions against 5-ASA or placebo for UC. Eligible studies included TCM treatments such as Qing-Chang-Hua-Shi, Curcumin-QingDai (CurQD), and Fufangkushen colon-coated capsules (FCC). The Volunteer-Intervention-Comparator-Outcome-Study (VICOS) framework guided our inclusion criteria, and we excluded nonrandomized controls (RCTs) and studies with incomplete data. Data extraction, quality evaluation, and the risk of bias were independently performed by the two researchers, and discrepancies were resolved through discussion.
Results
Four studies in all (n = 532) satisfied the requirements for inclusion. According to the meta-analysis, TCM therapies considerably enhanced mucosal healing (RR = 1.52, 95% CI: 1.25–1.85), clinical response (RR = 1.88, 95% CI: 1.54–2.29), and clinical remission (RR = 1.45, 95% CI: 1.21–1.73) when compared to a placebo. There was significant variation in treatment effects among trials, as evidenced by the high heterogeneity (I2 > 70%). Sensitivity analysis supported the robustness of these findings, showing no significant change in the probability of antagonistic events but notable increases in clinical outcomes (RR = 1.02, p = 0.8900).
Conclusion
Although there is a lot of variation among trials, TCM therapies seem to be more successful than placebo at improving clinical outcomes in UC. Although more study using standardized methodology is required to validate these effects, the results suggest that TCM may be used as a supplementary therapy for UC.
Keywords
Introduction
Ulcerative colitis (UC) stands as a significant global health concern within the range of inflammatory bowel disease (IBD) (Bojesen et al., 2017), mostly affecting intestinal mucosa and also characterized by persistent inflammation (Rubin et al., 2019). Diarrhea, gastrointestinal discomfort, rectal bleeding, and systemic symptoms, including fever, anemia, and weight loss, are among its clinical signs (Zhang et al., 2013). Despite advancements in therapeutic approaches, achieving sustained remission remains a challenge, with a substantial proportion of patients experiencing relapses and enduring disease-related complications (Yang et al., 2023). The escalating incidence and prevalence of UC worldwide have prompted a deeper exploration into its etiology, pathogenesis, and therapeutic interventions (Rezayat et al., 2020). Traditional treatment modalities, while offering symptomatic relief, are often associated with significant adverse effects and limited long-term efficacy (Baghizadeh et al., 2021). In this context, the quest for alternative and complementary therapeutic strategies has gained momentum, with a particular focus on natural medicines derived from botanical sources. Among the natural medicines under scrutiny for their potential efficacy in UC management are Qing-Chang-Hua-Shi, Curcumin-QingDai (CurQD), Fufangkushen colon-coated capsule (FCC), and Curcumin from Curcuma longa. Due to their many pharmacological characteristics, such as their anti-inflammatory, antioxidant, immunomodulatory, and mucosal protective actions, these botanical compounds have attracted interest (Xie et al., 2022). The Jiangsu Province Hospital of Traditional Chinese Medication uses Qingchang Huashi granule (QCHS), a Traditional Chinese Medication, to treat UC (Gu et al., 2017; Zhu et al., 2017). Based on the pathophysiology of moisture and heat buildup during the active phase, Professor Shen Hong created it. This granule is an enhanced and modified version of the well-known Baitouweng decoction, used for treating dysentery, as well as the Baishaoyao decoction. QCHS is effective in cooling the blood, removing blood stasis, controlling qi and blood, eliminating heat and moisture, and promoting muscle convergence (Zheng et al., 2017). This makes it suitable for addressing the pathogenesis of active UC, though its exact mechanism remains unexplored despite proven clinical efficacy. CurQD combines the anti-inflammatory properties of Curcumin and QingDai, showing efficacy in treating UC. It is demonstrated that CurQD significantly improves clinical and mucosal outcomes in UC patients, potentially maintaining remission with continued use. This makes CurQD a promising alternative or complementary treatment for UC (Lang et al., 2015). FCC are a newly created herbal treatment used to treat UC patients who exhibit the damp-heat accumulation pattern found in Chinese medicine. Sanguisorba officinalis L., Bletilla striata, Indigo naturalis, Glycyrrhiza uralensis, and Sophorae flavescentis make up FCC. Previous studies by Gong et al. (2012) demonstrated that FCC has a good safety record and can successfully induce remission in UC patients (Gong et al., 2012). Curcumin, a polyphenolic compound derived from Curcuma longa, has emerged as a prominent natural remedy with well-documented anti-inflammatory and antioxidative effects (Sreedhar et al., 2016). Its ability to modulate key inflammatory pathways, including NF-κB signaling and cytokine production, positions it as a promising candidate for UC therapy (Sands & Kaplan, 2007). While individual studies have explored the therapeutic potential of these natural medicines in UC, a comprehensive analysis synthesizing existing evidence is lacking. A meta-analysis, which amalgamates information from several research, might offer significant perspectives on the general effectiveness, safety record, and possible mutual benefits of herbal remedies for UC treatment. The aim of this study is to fill this knowledge gap and contribute to evidence-based decision-making in the management of UC, paving the way for optimized therapeutic strategies that integrate traditional wisdom with contemporary medical approaches.
Materials and Methods
Information Sources and Search Strategy
The systematic search was done using the China National Knowledge Infrastructure (CHKD-CNKI), Chinese Biomedical Literature Database (CBM), CINAHL, Embase, MEDLINE, PubMed, Cochrane Library, and WANFANG database, along with Web of Science, to identify randomized controls (RCTs) comparing treatments with Traditional Chinese Medicine (TCM) versus 5-ASA (placebo) or between treatments for mild-to-moderate UC. The search period was June 2012 to February 2024, for both English and Chinese literature. Two experienced writers conducted separate searches using a combination of free text terms and Medical Subject Headings (MeSH) related to “Qing-Chang-Hua-Shi,” “CurQD,” “Curcumin,” “FCC,” “Chinese medicine,” “TCM,” “Chinese herbal,” “UC,” “UC,” “IBD,” and “IBD” combined with “randomized controlled trials” using relevant Boolean operators. In addition, clinicaltrials.gov and the WHO International Clinical Trials Registry Platform were searched for data on active trial registrations. Besides a manual search of the reference lists of the included studies, systematic reviews and meta-analyses were conducted to identify further possible studies.
Eligibility Criteria
This study design followed the VICOS framework.
Type of Study
The participant enrolled all trials with RCTs concerning TCM interventions, including Qing-Chang-Hua-Shi, CurQD, Curcumin ITT, and FCC, for UC without regard to blinding or allocation concealment. Only Chinese or English was allowed to be used in the study. Non-RCTs that were excluded from the research include animal experiments, the case reports and case-control studies, the cohort studies, quasi-RCTs, and the reviews (Gardner et al., 2016). The randomized controlled trials were incorporated into the analysis, with appropriate adjustments applied to account for clustering effects. Chinese trials were only taken into consideration if they were recorded in an international database and approved by local institutional review boards (Harrison et al., 2019).
Volunteers
Patients meeting the criteria for UC diagnosis through endoscopic, histological, and clinical assessments as per standard diagnostic protocols were eligible for inclusion. There were no limitations regarding age, gender, nationality, ethnicity, or disease severity. However, research including people who had schistosomiasis, chronic amoebic intestinal illness, persistent bacterial dysentery, or similar disorders was not taken into consideration.
Intervention
The user considered studies that applied TCM interventions, such as Chinese herbal medicine, QCHS, CurQD, Curcumin ITT, and FCC. These approved therapies may be used alone, in combination, or in addition to Western medication [5-aminosalicylic acid (5-ASA)]. During the intervention period, it must last for at least 4 weeks nonstop. Studies using other Western drugs were excluded from our analysis (Harrison et al., 2019).
Comparators
Internationally recognized clinical standards indicate 5-ASA and a placebo were administered to the control group in addition to other TCM procedures.
Outcomes
The major outcomes were on the clinical efficacy and the rate of clinical remission, assessed by the Mayo score from the initial assessment to the last of the follow-up examination. The secondary outcomes were the rate of the mucosal healing, the rate of endoscopic response, and the safety of the intervention with any adverse effects.
Study Selection and Data Extraction
Data extraction, quality evaluation, literature retrieval, and screening will be carried out separately by two researchers. If a disagreement arises, they will discuss it with other researchers and use the original data to settle any disputes. After retrieved studies meet certain eligibility requirements, relevant literature will be found by looking through their titles, abstracts, and complete texts. Search results will be organized and managed using Note Express 2.0 (Moher et al., 2009). Using a predetermined extraction table, information will be gathered about publication year, author, article title, and country of origin, contact details, sample size, participant demographics, diagnostic standards, baseline features, study type, randomization method, blinding status, interventions, duration, frequency of treatment, outcomes, adverse events (AE), and more. We will try to get in touch with the writers of any articles that are missing material so they can retrieve or convert the data. There will be no analysis of studies with missing data.
Assessment of Risk Bias
In an effort to carry out the meta-analysis, two researchers independently gathered, assessed, and reconciled the data. The risk of bias assessment was collected in compliance with the Cochrane Handbook for Systematic Reviews of Interventions criteria (Higgins et al., 2011). It assessed biases such as selective reporting, contrived sequences, insufficient result data, hidden allocation, blinding outcome evaluation, blinding people, and blinding segments of the population. Using the Jadad scale, dropout rates, blinding, and randomization were evaluated to determine the included research quality (Jadad et al., 1996). A score of two or below indicated low quality, whereas a score of three or above indicated good quality. Studies were scored on a scale of 0–5.
Grade of Evidence
In order to evaluate the quality of evidence, the GRADE method was used, while following the recommendations of the Cochrane Handbook for Systematic Reviews of Interventions (Yao et al., 2016). The evaluation took into account four different categories of evidence: extremely poor quality, medium quality, moderate quality, and good quality. The publication, bias risk, imprecision, inconsistency, and indirectness bias were the five factors taken into account while assessing the quality of evidence for the meta-analysis.
Data Synthesis and Statistical Analysis
For binary outcomes, we conducted meta-analyses using relative risk (RR) that provided a summary estimate with 95% confidence intervals (CIs). Within each study, subgroup analyses were performed by induction dose and baseline UC treatment (either initial steroids or IMM). Statistical heterogeneity was evaluated using I2, and p < 0.10 was considered significant for heterogeneity. A fixed effects model was used to account for significant heterogeneity. We declined to explore publication bias by funnel plots because each comparison had too few research. Intention-to-treat guidelines were followed during the analysis.
Results
Characteristics of Included Studies
Using the previously mentioned search strategy, 47 citations were initially identified. Thirty-eight were eliminated after the titles and abstracts were examined. The effectiveness of treatment therapy in UC was subject of a thorough examination of nine articles. For the grounds listed below, five of these nine articles were disqualified: A pair of studies were eliminated due to their lack of placebo control (Barreiro-de Acosta et al., 2009; Suzuki et al., 2013); one article was excluded because it analyzed data by integrating two other RCTs (Lan et al., 2021); one was excluded because it was a meta-analysis (Fan et al., 2019); and one was taken out as it was a follow-up report on an additional RCT (Reinisch et al., 2013).
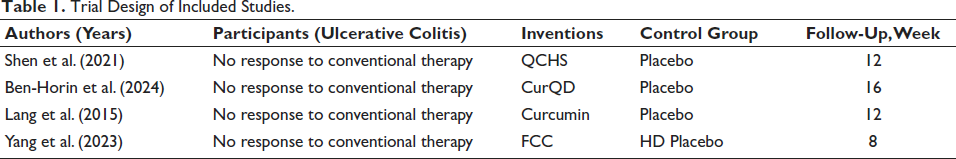
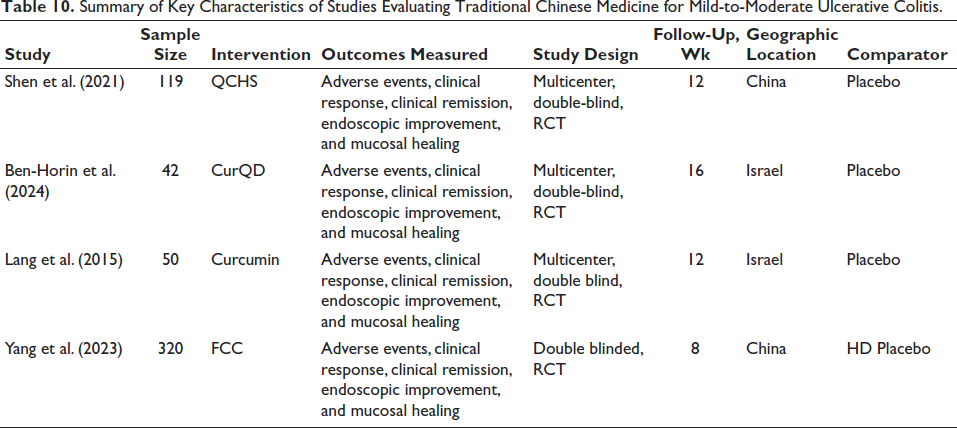
Four studies (Ben-Horin et al., 2024; Gong et al., 2012; Lang et al., 2015; Shen et al., 2021) passed the inclusion requirements following a careful examination. The therapy group and a placebo were compared in these trials. Two research projects were completed in China, and two in Israel. An equal number of 532 UC patients were assigned randomly to therapy groups (n = 348) or the placebo group (n = 176). Out of them, 531 patients were part of the therapy group efficacy analysis for UC that was active. Induction treatment and maintenance therapy were the two trial designs used in the investigations. These investigations’ characteristics are shown in Tables 1 and 2.
Trial Design of Included Studies.
Baseline Characteristics of Included Studies.
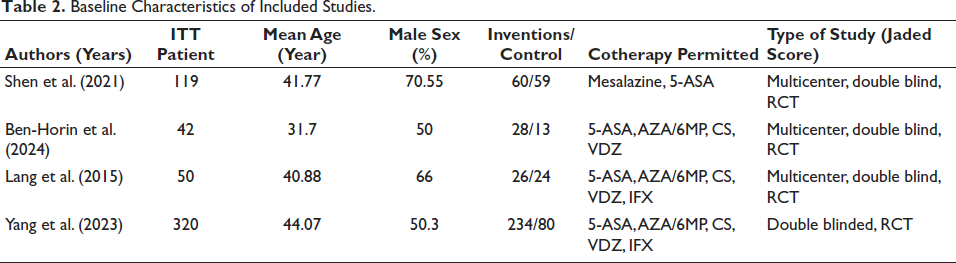
AZA is for azathioprine; MP stands for mercaptopurine; RCT stands for randomized controlled trial; VDZ stands for vedolizumab; and IFX stands for infliximab. ITT stands for intent-to-treat patients.
Assessment of the Methodological Quality of the Included Trials
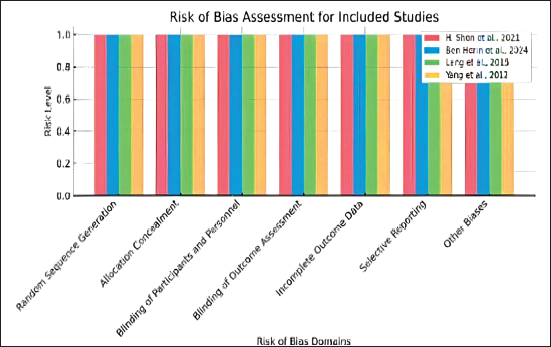
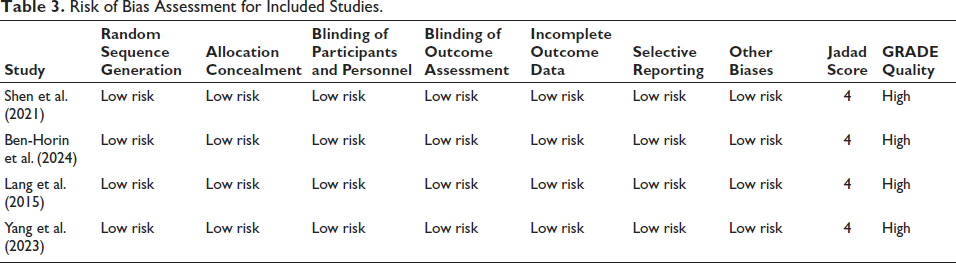
All four studies (Ben-Horin et al., 2024; Lang et al., 2015; Shen et al., 2021; Yang et al., 2023) consistently demonstrate low risk across important domains like blinding, inadequate result data, secret allocation, random sequence formation, and selective reporting, according to risk of bias assessment for studies included. Every study was given a GRADE approach grade of outstanding quality and a Jadad score of four out of five. This implies that the findings of these studies are very reliable and of high methodological quality (Figure 1, Tables 3 and 4).
The Risk of Bias Assessment for the Included Stu dies. Each Domain is Evaluated for Each Study, and the Risk Level is Shown. All Studies have Low Risk in All Domains, Indicated by the Bars Being at the Same Level for Each Domain.
Risk of Bias Assessment for Included Studies.
Risk of Bias Summary.

Data Synthesis: Induction Therapy Effectiveness
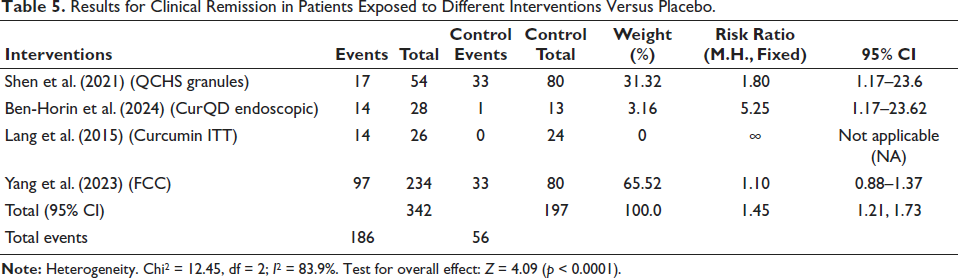
Three types of data may be summarized in the treatment and placebo groups for clinical studies of various treatments. The first type of data is that of clinical remission results, which is summarized in Table 1. QCHS Granules, CurQD Endoscopic, Curcumin ITT, and FCC are the interventions. The intervention group had 342 participants, and the control group had 197 participants. An overall risk ratio (RR) of 1.45, with a 95% CI of 1.21–1.73, indicated the combined efficacy of the interventions above placebo in achieving clinical remission. Variety across the trials was shown by the strong heterogeneity (I2 = 83.9%) and significant overall effect (Z = 4.09, p < 0.0001) tests (Table 5).
Results for Clinical Remission in Patients Exposed to Different Interventions Versus Placebo.
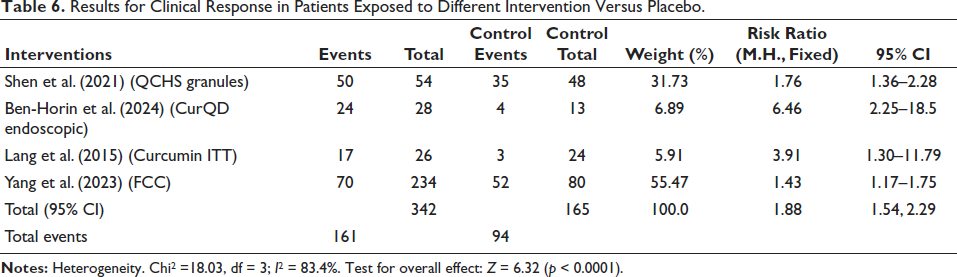
The second dataset, presented in Table 2, presents clinical response rates for the same interventions. In this analysis, 342 participants were found in the intervention group, and 165 in the control group. The pooled RR was 1.88 (95% CI: 1.54–2.29), indicating a marked improvement in clinical response with the interventions compared to placebo. The heterogeneity was high (I2 = 83.4%), and the test for the overall effect was highly significant (Z = 6.32, p < 0.0001). Ben-Horin et al. (2024) had one of the highest RR figures (6.46, 95% CI: 2.25–18.5), which represents the effectiveness of the CurQD Endoscopic intervention (Table 6).
Results for Clinical Response in Patients Exposed to Different Intervention Versus Placebo.
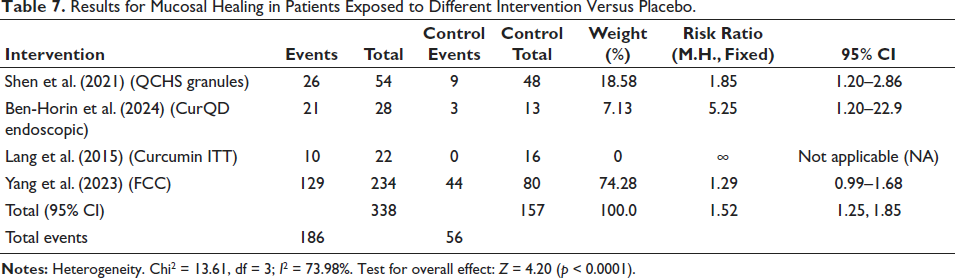
In Table 3, the third set of data examines the results of mucosal healing. Overall, the RR of treatments is 1.52, 95% CI: 1.25–1.85 with 338 individuals in the intervention and 157 in the control. It follows that interventions were considerably more effective than placebo for inducing mucosal healing. Overall, the effect was sizeable (Z = 4.20, p < 0.0001), despite the strong heterogeneity (I2 = 73.98%). Again, individual trials, such as Ben-Horin et al. (2024), which had a very high RR (5.25, 95% CI: 1.20–22.9), showed CurQD Endoscopic’s possible role in mucosal healing (Table 7). For clinical remission, clinical response, and mucosal healing, the treatments achieved significantly higher results than placebo in all three datasets. However, the high heterogeneity across studies suggests that though the interventions are broadly effective, the degree of effectiveness varies, probably because of differences in study designs, populations, and specific interventions used.
Results for Mucosal Healing in Patients Exposed to Different Intervention Versus Placebo.
Data Synthesis: Baseline Medication Treatment Therapy’s Short-term Effectiveness
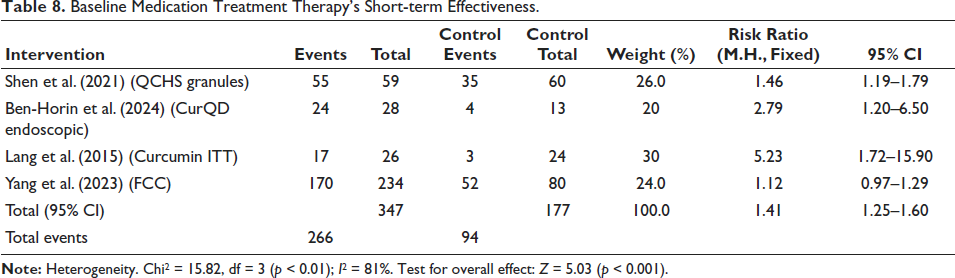
The short-term effectiveness of several treatment treatments in comparison to control groups, divided by baseline drugs, is summarized in Table 8. Using QCHS Granules, Shen et al. (2021) found that, with a weight of 26.0% and a RR of 1.46 (95% CI: 1.19–1.79), 55 out of 59 intervention individuals experienced incidents, compared to 35 out of 60 in the control group. In their study of CurQD Endoscopic, Ben-Horin et al. (2024) found that 24 out of 28 intervention individuals had events, compared to 4 out of 13 in the control group. This resulted in a 20% weight and an RR of 2.79 (95% CI: 1.20–6.50). Lang et al. (2015) investigated Curcumin ITT, with 17 out of 26 events in the intervention group compared to 3 out of 24 in the control, assigning a weight of 30% and an RR of 5.23 (95% CI: 1.72–15.90). Yang et al. (2023) examined FCC, where 170 out of 234 intervention participants had events against 52 out of 80 in the control, with a weight of 24.0% and an RR of 1.12 (95% CI: 0.97–1.29). The total combined data (95% CI) shows 266 events in 347 participants versus 94 in 177 controls, indicating an overall RR of 1.41 (95% CI: 1.25–1.60). With a Chi2 value of 15.82, degrees of freedom (df) = 3, and a p value < 0.01, the studies exhibit significant heterogeneity, suggesting significant variability (I2 = 81%). With Z = 5.03 (p < 0.001), the overall effect test is significant, indicating that the interventions as a whole have a favorable impact when compared to the control therapies (Table 8).
Baseline Medication Treatment Therapy’s Short-term Effectiveness.
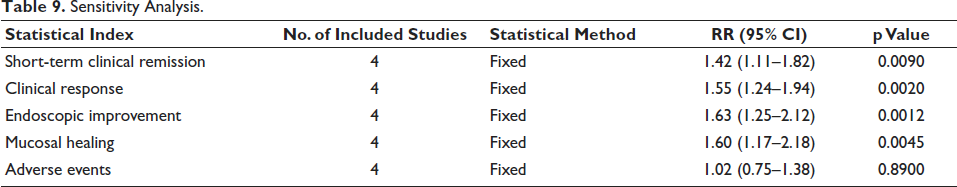
Sensitivity Analysis
It is a scientific technique used in the systematic reviews as well as meta-analyses to evaluate sensitivity of the research results and any departures from them. This approach evaluates how resilient the combined results are when handling data uncertainties and their interpretation. Table 9 presents a sensitivity study evaluating the potential changes in the results of different clinical outcomes when specific parameters are changed across four trials using a fixed-effect model. Among the outcomes that were looked at were AE, clinical response, mucosal healing, and endoscopic improvements. The effectiveness of treatment is shown by the 95% CI and RR between the treatment and control groups. Statistical significance is shown in results of p values below 0.05; results reflect that the treatment considerably benefits improvement in endoscopic improvement at p = 0.0012, RR = 1.63; clinical response, at p = 0.0020, RR = 1.55; short-term clinical remission, at p = 0.0090, RR = 1.42; and mucosal healing at p = 0.0045, RR = 1.60. However, as p value is much higher than 0.05, which shows there is no significant difference among the treatment and control groups regarding AE, the therapy has no noticeable effect on the probability of AE (RR = 1.02, p = 0.8900) (Table 10).
Sensitivity Analysis.
Summary of Key Characteristics of Studies Evaluating Traditional Chinese Medicine for Mild-to-Moderate Ulcerative Colitis.
Discussion
In clinical practice, managing UC that is difficult to treat presents a substantial difficulty. Some UC patients struggle to avoid surgery and have repeated illness relapses even with excellent conventional medicine (Lv et al., 2014). The introduction of biological drugs has revolutionized UC therapy in recent years, offering new treatment options for many patients (Park & Jeen, 2015). Among these, anti-TNF antibodies, notably infliximab (IFX), have gained FDA approval for various autoimmune diseases and demonstrated efficacy. However, IFX’s high immunogenicity, frequent AE, and cumbersome administration limit its widespread use (Armuzzi et al., 2014). In contrast, adalimumab (ADA), with a similar mode of action to IFX, is becoming increasingly popular due to lower adverse reaction rates and easier subcutaneous administration.
This meta-analysis shows potential in alternative interventions, such as TCM therapies, which demonstrated similar efficacy to biological therapies, like ADA and IFX, concerning clinical remission, response, and mucosal healing. These could become a valuable option for patients who cannot bear or do not respond to conventional biologics. Notably, when compared to standard treatments, TCM interventions may offer a safer profile, as shown by the lack of significant differences in AE (RR = 1.02, p = 0.8900). This is particularly relevant for UC patients who experience side effects from biologics, supporting the clinical relevance of TCM therapies in reducing treatment-related burdens while maintaining efficacy.
This meta-analysis aims to review the efficacy and safety of various treatments for UC by considering randomized placebo-controlled studies. The results of these trials consistently show that a range of intervention strategies considerably outperform placebos in treating patients with UC, regardless of whether it is regarded the primary or secondary effectiveness target (Nielsen & Ainsworth, 2013).
We focused on four significant trials that compared TCM therapy to a placebo and assessed objectives such as the clinical remission, clinical response, as well as mucosal healing. TCM medicines seem to offer a balanced mixture of safety and effectiveness when compared to conventional medications like 5-ASA and corticosteroids, which may have limitations in long-term usage due to safety concerns. Our findings indicate that TCM treatments significantly outperform placebo in these clinical outcomes. With an RR of 1.45 (95% CI: 1.21–1.73), the combined analysis specifically showed that TCM therapies are more successful than placebo in producing clinical remission. This implies that compared to those getting a placebo, patients undergoing TCM therapy had a 45% higher chance of achieving clinical remission.
When evaluated alongside standard biologics, such as ADA and IFX, the efficacy of TCM interventions is noteworthy. The clinical remission and mucosal healing rates achieved by TCM therapies, with an RR of 1.52 for mucosal healing, are comparable to those reported in trials of ADA (Peyrin-Biroulet et al., 2014) and IFX (Panaccione et al., 2014), further establishing their potential role as complementary or alternative treatments for UC. With a total RR of 1.88 (95% CI: 1.54–2.29), which indicates that patients were 88% more likely to react to therapy, TCM treatments significantly outperformed placebo in terms of clinical response. Additionally, the analysis of mucosal healing outcomes further supports the efficacy of TCM interventions, with an overall RR of 1.52 (95% CI: 1.25–1.85), indicating 52% higher possibility of mucosal healing compared to placebo. Importantly, this reflects an effect size similar to the rates of mucosal healing reported for biologic therapies in head-to-head trials, providing a basis for considering TCM therapies as a serious treatment option, especially for patients concerned about the risks of long-term biologic use.
However, the high heterogeneity across studies, with I2 values of up to 83.9%, suggests variability in intervention effects, likely owing to variances in study design, participant characteristics, as well as use of specific treatments.
The short-term efficacy analysis, stratified by baseline medications, consistently demonstrated the benefits of TCM interventions. The combined RR of 1.41 (95% CI: 1.25–1.60) for treatment against control groups underscores the overall effectiveness of TCM therapies, showing that these interventions provide a 41% greater benefit over placebo. The sensitivity analysis validated our findings’ robustness in spite of the notable variation observed. Short-term clinical remission (RR = 1.42, p = 0.0090), clinical response (RR = 1.55, p = 0.0020), endoscopic improvement (RR = 1.63, p = 0.0012), and mucosal healing (RR = 1.60, p = 0.0045) all showed significant improvements. In addition to alleviating clinical symptoms, our findings show that TCM therapies improve endoscopic and mucosal healing results. Notably, the analysis did not find a significant change in AE among treatment and control groups (RR = 1.02, p = 0.8900), suggesting that TCM interventions are safe and well-tolerated by patients. This safety profile, combined with the efficacy data, highlights the potential of TCM therapies as viable options for UC treatment.
Considering the limitations of biologic treatments, such as increased risk of infections and malignancies through long-term use (Wehkamp et al., 2016), the safety data from this meta-analysis positions TCM therapies as a possible alternative, especially for patients looking for treatments with fewer systemic risks.
We must recognize several limitations in this meta-analysis. The significant heterogeneity among studies indicates variations in study designs, interventions, and patient demographics, potentially impacting result generalization. Additionally, the limited number of studies reduces statistical power and restricts in-depth analyses like investigating publication bias. Despite these constraints, our meta-analysis presents compelling evidence of TCM interventions notably enhancing clinical outcomes in mild-to-moderate UC patients compared to placebos. The consistent benefits seen in clinical remission, response, and mucosal healing highlight these interventions’ potential as effective treatments. Future large-scale randomized controlled trials are needed to validate these findings, focusing on long-term efficacy as well as safety of TCMs therapies in UC. Standardizing the study designs and interventions in these future trials can reduce heterogeneity and offer clearer clinical guidance.
Conclusion
This meta-analysis shows that a number of treatments are more effective than placebos in treatment of UC. That is supported by the improvements in responsiveness, mucosal healing, and clinical remission established after 8 and 16 weeks without significant adverse effects. Hence, moderate to highly active UC patients that did not give adequately response to conventional drugs, should be assessed for alternative intervention treatment. Multicenter longitudinal studies with extended follow-ups and bigger randomized placebo-controlled trials are required to additionally evaluate the efficacy and safety of these drugs.
Footnotes
Abbreviations
5-ASA: 5-aminosalicylic acid; CBM: Chinese biomedical literature database; CHKD-CNKI: China national knowledge infrastructure; CI(S): Confidence interval(s); CINAHL: Cumulative index to nursing and allied health literature; CURQD: Curcumin-qingdai; DF: Degrees of freedom; FCC: Fufangkushen colon-coated capsule; GRADE: Grading of recommendations assessment, development, and evaluation; IBD: Inflammatory bowel disease; IMM: Immunomodulator(s); ITT: Intent-to-treat; MESH: Medical subject headings; QCHS: Qingchang Huashi granule; RCT(S): Randomized controls; RR: Relative risk; UC: Ulcerative colitis; WHO: World Health Organization.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This work was approved by the Institutional Ethical Committee of Qingdao University, Qingdao, China (Ethical number: 2023/1-47).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was funded by the Hebei Province Higher Education Science and Technology Research Project (QN2023097).
