Abstract
Osteosarcoma (OS) is a predominant malignancy among children and adolescents, ranked third in frequency after lymphoma and leukemia. It presents a significant risk of metastasis, particularly to the lungs and distal bones, leading to severe disease progression and increased mortality. Current treatment modalities, primarily surgery and chemotherapy, are associated with considerable side effects and variable prognoses, underscoring the urgent need for novel, more effective, and less harmful therapeutic options. Flavonoids, naturally occurring polyphenolic compounds, have shown promising potential in the management of OS. This article evaluates the molecular mechanisms through which flavonoids exert therapeutic effects on OS and explores their potential in developing innovative treatments. The review provides insights into their roles in inducing apoptosis, inhibiting cell proliferation, and modulating key signaling pathways in OS cells by examining the anticancer properties of specific flavonoids such as apigenin, quercetin, and baicalein. The comprehensive analysis aims to contribute to advancing flavonoid-based therapies, opening new avenues for practical and safer OS treatments.
Introduction
The distal femur, proximal tibia, and proximal humerus are among the weight-bearing long bones where osteosarcoma (OS), the oldest known human malignancy, is most frequently detected (Cho et al., 2010). After lymphoma and leukemia, OS is the third most prevalent kind of cancer and has a high prevalence in children and adolescents (Li et al., 2023). Children have an annual incidence rate of 5.6 per million (Abate et al., 2010; Kager et al., 2003). Sufferers with localized tumors have a 60% 5-year relative survival rate, but if metastasis develops in OS, the survival rate sharply declines to fewer than 20%, posing a significant risk to the lives and health of patients (Gorlick et al., 2013). OS is a primary mesenchymal malignant tumor that develops from tumor cells into immature bone-like cells (Belayneh et al., 2021). Current treatments for OS are mainly surgical resection of the cancer or stereotactic radiotherapy interventions. Although surgical resection improves OS survival rate, it is also prone to recurrence, and there are many postoperative complications, such as phantom limb pain and motor dysfunction. Moreover, OS in specific sites, such as bone marrow, cannot be surgically removed. Clinical data suggest that chemotherapy does not significantly develop patients’ survival rate due to rapid OS progression and the susceptibility of patients to chemoresistance, which is accompanied by serious side effects such as infection and neutropenia (Chen et al., 2021; Li et al., 2023). To prevent OS from developing in the first place and to create more potent therapies for OS, safe and efficient therapeutic drugs must be used. Pharmacological interventions may restrict OS to the microenvironment to inhibit its metastasis, which may be an important way to treat OS and enhance OS sufferers’ prospects going forward.
Flavonoids, polyphenolic compounds abundant in dietary plants and herbs like citrus fruits, grapes, green tea, eggplant, and parsley, constitute a diverse family of over 10,000 compounds with distinct structures, categorized into subfamilies such as flavanols, flavanones, flavonols, flavonoids, and anthocyanins (Liu et al., 2017 ; Patel & Alhakeem, 2021; Xia et al., 2023). Research has consistently demonstrated the multifaceted benefits of flavonoids, including anti-inflammatory, antioxidant, blood pressure-lowering, antithrombotic, and antitumor properties (Liu et al., 2017). Particularly noteworthy are their anticancer effects, which have been extensively studied across various tumors, including liver, breast, colorectal, prostate, lung, and leukemia. In the context of OS, flavonoids have emerged as promising therapeutic agents. Studies have revealed their potential to inhibit OS development through mechanisms such as mitochondrial apoptosis, iron-induced cell death, autophagy modulation, and attenuation of oxidative stress pathways. Consequently, flavonoids represent compelling candidates for future clinical interventions against OS (Koh et al., 2020; Luo et al., 2021). This article comprehensively reviews the current research progress on flavonoids in OS treatment, delineating their molecular mechanisms, exploring their potential synergistic effects in combination therapies (Nakatani et al., 2018), and investigating novel nanomaterial-based approaches leveraging flavonoids for OS treatment. By synthesizing these findings, we aim to contribute additional insights and theoretical foundations to further the clinical application of flavonoids in treating OS.
Flavonoids for the Treatment of OS
Apigenin
Strong anticancer effects are exhibited by apigenin (4′,5,7-trihydroxyflavone), a naturally occurring phenolic chemical that is mostly present in vegetables, herbs, and fruits. Apigenin targets the phosphorylation of the PI3K signaling pathway, which significantly reduces the Warburg effect and stem cell-like features in SOSP-9607 cells (Wang et al., 2019), according to research. Moreover, apigenin causes apoptosis in a time- and dose-dependent manner while exhibiting a notable suppression of OS cell (U2OS, MG63) proliferation (Lin et al., 2012; Liu et al., 2015; Ruan et al., 2018). The inhibition of the Wnt/β-catenin pathway, which results in cell cycle arrest during the G0/G1 phase, is thought to be the cause of these effects. In summary, apigenin mainly inhibits cell growth and regulates the cell cycle to produce its anticancer effects.
Quercetin
Quercetin is derived from
Baicalein
Baicalein is derived from
Noncoding RNAs (ncRNAs), such as miRNA and lncRNA, regulate several biological processes, including chromatin remodeling, transcription, post-transcriptional modification, and signaling. Notably, baicalein upregulated lncRNA-NEF and consequently suppressed miRNA-21, preventing cancer cell migration and invasion in OS cells. Baicalein also inhibited OS growth by regulating miR-25 and altering the expression of genes involved in the Wnt/β-catenin pathway. Another study showed that wogonin-induced activation of miR-183 led to apoptosis in OS cells, reducing their ability to proliferate, migrate, and invade. These findings highlight ncRNAs’ potential as targets for further research into baicalein’s therapeutic effects on OS.
Wogonin, the primary bioactive component of
Catechins
Among the many flavonoids, catechins have been widely studied by researchers for their powerful antioxidant capacity. The antitumor activity of catechins has been widely demonstrated as a cancer blocker, intricately involved in the proliferation of cells, inflammation, differentiation, and metastasis (Fujiki et al., 2017; Jiang et al., 2016; Khan et al., 2019). Modifications to catechins were found to augment the anticancer action of Se-HAp nanoparticles by promoting apoptosis in OS cell lines and generating excessive ROS to activate the caspase-3 cascade; however, they had no discernible impact on the development of normal human bone marrow cells (Khan et al., 2019). According to Dong et al. (2022), epigallocatechin-3-gallate (EGCG) is a powerful anticancer agent that may decrease OS cell proliferation and lung metastasis in a dose-dependent manner by modulating the Wnt/β-catenin signaling pathway in both
Summary of Flavonoids for in vivo Treatment of Osteosarcoma (OS).
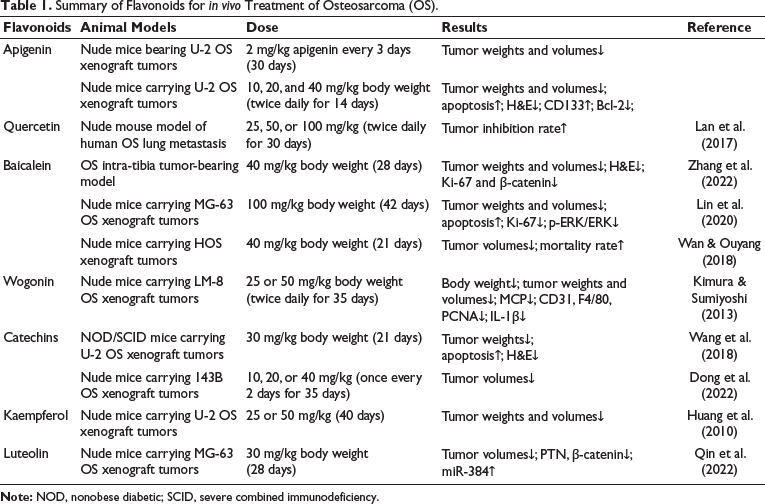
Kaempferol
A natural flavonoid, kaempferol, has been demonstrated to induce apoptosis in many cancer cell types. Using network pharmacological analysis and molecular docking, researchers discovered that kaempferol is one of the active constituents in Qingfei Gujin Decoction, which is used to treat organ failure. Additionally, kaempferol diminished the capacity of the activator protein-1 (AP-1) to bind deoxyribonucleic acid (DNA) and inhibited the phosphorylation of the MAPK pathway, which includes ERK, JNK, and p38. These actions downregulated the expression and enzymatic activity of MMP-2, MMP-9, and uPA, ultimately impeding the growth and metastasis of OS cells (Chen et al., 2013). Furthermore, kaempferol was shown to induce apoptosis and DNA damage in U-2 OS cells by dramatically increasing Ca2⁺ levels and lowering mitochondrial membrane potential (Huang et al., 2010). Possible treatment mechanisms of kaempferol for OS need further investigation.
Luteolin
Luteolin, found in medicinal plants and vegetables, has anti-OS properties (Table 2). Canine OS cell lines (D17 and DSN) are less likely to proliferate when luteolin and cisplatin are combined. Moreover, luteolin increased chemosensitivity and decreased OS chemoresistance to doxorubicin (DOX) and cisplatin by suppressing the PTN/β-catenin/MDR1 signaling axis through upregulation of miR-384 (Schrader & Kaufmann, 2011; Sergio & Matar, 2021). Luteolin induces autophagy in U-2 OS cells and acts as an enhancer to sensitize the adriamycin-mediated autophagy signaling pathway, demonstrating a synergistic effect with azithromycin. In short, luteolin may be a potential medication for OS management.
Flavonoids and Sources.
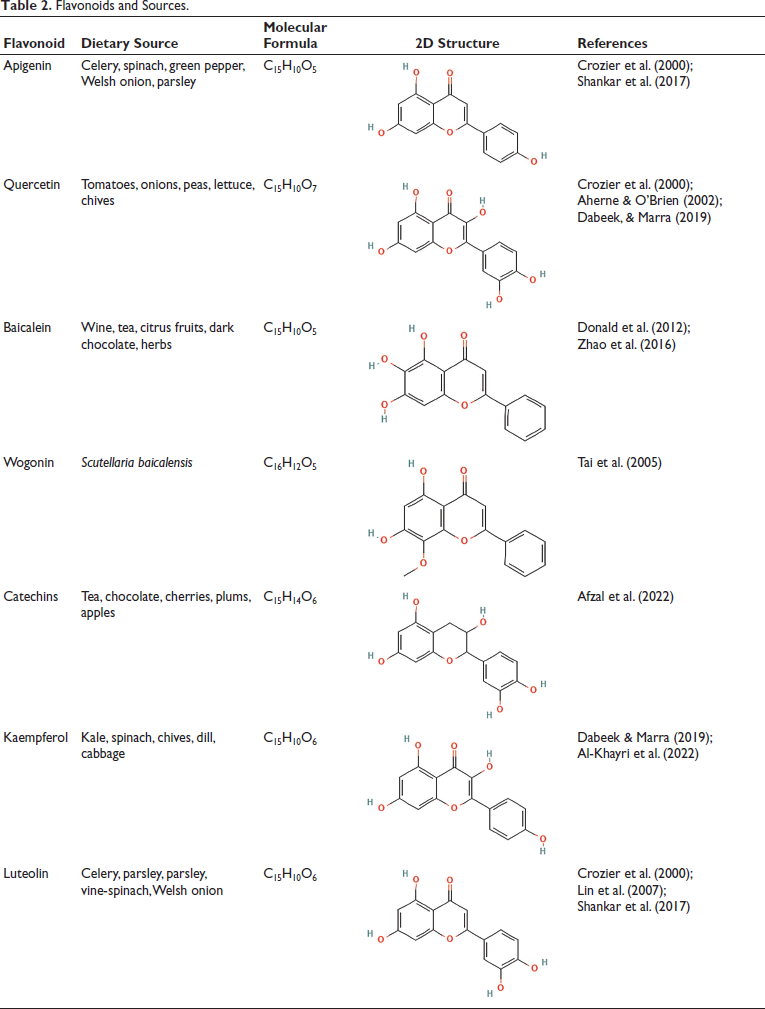
Molecular Mechanism of Flavonoid Therapy for OS
Cell Cycle Regulation
Genomic instability and uncontrolled cell cycle progression are common hallmarks of cancer (Hanahan & Weinberg, 2011). The process known as the cell cycle starts after one cell division and continues until the start of the subsequent one. There are four phases: mitosis (M), gap 2 (G2), synthesis (S), and gap 1 (G1). In the G2 phase, the cell prepares for cytokinesis, while DNA replication occurs in the S phase. A cell divides into two during the M phase, and the genetic material is evenly distributed across the daughter cells. G1/S and G2/M are two distinct checkpoints used in cell division to monitor the entire process. In an investigation, it was discovered that administering the flavonol glycoside Icarisid II to OS cells caused cell cycle arrest in the G2/M phase and decreased the number of cells in the G0/G1 phase. Furthermore, they found that Cyclin D1 is a major regulator of G1/S, and P21 can negatively regulate the activity of specific CDK (cyclin-cyclin-dependent kinase) complexes that drive the cell cycle and participate in the G1/S and G2/M transitions. Cyclin B1 and CDK1 complexes are regulated by the upstream gene Cdc25C, showing a significant function in the checkpoint G2/M. Icarisid II upregulates P21 and downregulates cyclin D1 in the G1/S phase to inhibit U2OS cell proliferation. Additionally, Icarisid II reduced the expression levels of CDC2 and P-Cdc25C and increased the expression of Cyclin B1, a protein linked to the G2/M cell cycle. These changes ultimately caused apoptosis in human OS cells in a dosage- and time-dependent manner.
Evaluating the antitumor capacity of 3′,4′-dihydroxyflavonol (DiOHF) in another type of OS cell (MG-63), the investigators found that DiOHF similarly reduced the expression of Cyclin B2, a protein critical for the exit of the G2 phase and the onset of mitosis, in MG-63 cells. Additionally, DiOHF decreased CDK1 expression, leading to more cell arrest in the S phase (Ferreira de Oliveira et al., 2021). A similar view was obtained in another study where fisetin similarly arrested OS MG-63 cells in the G2/M phase, preventing the cells from proliferating. Baicalein similarly induced G1 cell cycle arrest by decreasing cyclin D1 and CDK4 while activating the mitochondrial apoptotic pathway to induce apoptosis. Quercetin treatment decreased cyclin D1 and cyclin B1 expression, causing cellular G1/S phase alterations and affecting G2/M phase progression (Catanzaro et al., 2015). Consistent with the above findings, the addition of pelargonidin, flavokawain B, neohesperidin, and other flavonoids was able to arrest the G2/M phase of OS cells, reduce cyclin B1, Cdc2, and Cdc25C expression, and induce apoptosis and autophagy in a dose-dependent manner (Chen et al., 2018; Ji et al., 2013). Interestingly, Ampelopsin, on the other hand, induced cell cycle arrest in the G0/G1 phase, increasing P21 expression while inhibiting cyclin A and CDK2 expression, leading to apoptosis in MG-63 cells (Lu et al., 2015). It is evident that flavonoid compounds can effectively induce OS cell cycle arrest, preventing further mitosis and proliferation, thus exerting powerful antitumor activity, which provides more ideas for the future treatment of OS.
Oxidative Stress
ROS are a class of byproducts produced during aerobic metabolism that can act as signaling molecules involved in the regulation of cellular functions. ROS are beneficial to cell signaling at normal levels, but excessive ROS can cause oxidative stress, leading to DNA and ribonucleic acid (RNA) damage, oxidation of proteins and organelles, and even cell death (Dias et al., 2021). Numerous internal and environmental variables, including exposure to toxins and ultraviolet (UV) radiation, influence ROS levels. In cancer cells, ROS levels are usually high, contributing to tumor growth; however, an excess of ROS induces apoptosis. Therefore, balancing ROS levels is essential for the development of cancer and other diseases.
Plant secondary metabolites classified as flavonoids have a variety of biological effects, but their ability to scavenge free radicals and act as antioxidants is undoubtedly the most relevant. Flavonoid compounds play a significant role in maintaining redox homeostasis in living organisms. Generally, flavonoids occur in the form of glycosides, and their antioxidant properties are influenced by the number and position of sugar group linkages. They exist in diverse structures, particularly in the type and arrangement of functional groups (Moussaieff et al., 2021). These structural differences directly affect their antioxidant capacity, that is, the ability to absorb free radicals (Kumar & Pandey, 2013). Because flavonoids are antioxidants, they can scavenge ROS, prevent oxidative damage, and potentially inhibit tumor cell growth by inducing ROS production. The antioxidant mechanisms of flavonoid compounds mainly include (a) direct scavenging of ROS, (b) chelation of trace elements to prevent ROS formation, (c) inhibition of enzymes related to free radical generation [e.g., glutathione (GSH)
Iron death is a novel regulatory cell death mode mediated by intracellular iron that is closely linked to cellular ROS levels. Recent studies have shown that bavachin increases ROS accumulation in MG63 and HOS OS cell lines, leading to GSH reduction and malondialdehyde overexpression, which induced iron death in OS cells. The flavonoid compound wogonin was shown to downregulate PRX5 expression and induce ROS-mediated cell death in CD133+ Cal72 cells
Cell Signaling Pathway
PI3K/AKT
One of the key mechanisms thought to be involved in the creation of several malignancies is the PI3K/AKT signaling system. Numerous studies have demonstrated that the PI3K/AKT pathway may have an impact on many activities, such as cell replication, migration, proliferation, invasion, and apoptosis, that are involved in the pathological development of OS. U2OS cells showed a decrease in the ability of cell invasion and migration after exposure to different concentrations of morusin, and it was further found that PI3K, P-AKT, MMP-2, MMP-9, and Bax were all diminished in a concentration-dependent way; thus, PI3K/AKT has been suggested as a prospective mechanism for OS alleviation by morusin. Phellamurin, a flavonoid with the structure of a flavonoid glycoside, inhibited tumor growth by suppressing the PI3K/AKT/mTOR cascade in OS cells, and the PI3K inhibitor LY294002 induced apoptosis based on inhibition of the PI3K/AKT cascade; in dissimilarity, PI3K activators abrogated the effect of phellamurin on apoptosis. It offers more proof of the critical role played by the PI3K/AKT/mTOR signaling pathway in phellamurin resistance to OS. Another flavonoid glycoside, poncirin was obtained from the immature fruit of cloverleaf. Poncirin was discovered to disrupt the PI3K/AKT pathway and modulate the downregulation of the expression of adenosine triphosphate (ATP)-binding transporter proteins, MDR1 and MRP1, which in turn reduced the resistance of OS cells to cisplatin, enhanced their susceptibility to cisplatin, and increased OS cell apoptosis (Zhao et al., 2020). Licochalcone B was shown by Huang et al. (2022) to induce autophagy and death in OS cells (MG-63 and U2OS) via dose-dependently inhibiting the pathway consisting of PI3K/AKT/mTOR, which has a potential anticancer effect (Rao et al., 2017; Wang & Huang, 2022). Proanthocyanidin B2, a naturally occurring flavonoid was shown to block the PI3K/AKT signaling cascade to cause OS tumor development and death in both
JNK
JNK is a subgroup of the standard MAPK signal transduction pathway, often referred to as stress-activated protein kinase. Because JNK is extremely sensitive to stimulation, JNK protein kinases quickly activate JNK proteins in response to bacterial and viral infections, growth hormones, UV light, inflammatory cytokines, and other stimuli. After activation, JNK phosphorylates downstream target proteins such as activating transcription factor (ATF) and transcription factor activating protein-1 (AP-1) family proteins, thus affecting a variety of intracellular biological processes (Hammouda et al., 2020). Mitogen-activated protein kinases, or MAPKs, are well recognized as crucial apoptosis regulators, especially JNKs. JNK is closely associated with natural flavonoids, anticancer drugs, and various stress-induced apoptosis processes. JNK-mediated apoptosis is mainly involved in two pathways: (a) Fas ligand expression upregulation and (b) involvement of the mitochondrial pathway, which is mostly responsible for downregulating the antiapoptotic Bcl-2 protein and upregulating proteins that promote apoptosis, Bak and Bax (Hammouda et al., 2020). Indeed, there is mounting evidence that suggests the JNK signaling pathway may play a role in the antitumor potential of flavonoids. Human OS cells (SJSA and HOS) were unable to proliferate; instead, acetone caused apoptosis. Following administration of the JNK inhibitor SP600125, acetin’s proapoptotic impact was diminished (Wang et al., 2020). It is worth mentioning that this proapoptotic effect of JNK may be attributed to the upstream ROS outbreak. Another flavonoid, cardamonin, has inhibitory effects on a variety of tumors, including OS. Cardamonin significantly inhibited tumor growth of OS in mouse models and increased phosphorylation levels of P38 and JNK. JNK phosphorylates the BH3 protein as a pathway target to initiate a second apoptotic pathway (mitochondrial BAX-dependent apoptotic pathway). P38 inhibitors and JNK inhibitors neutralized the inhibitory effect of cardamonin to a certain extent and restored cell proliferation and migration. Nobiletin is a polymethoxy-flavonoid that has anticancer properties. Research has demonstrated that nobiletin inhibits the migration, invasion, and metastasis of HOS and U2OS cells by preventing the production of MMPs mediated by JNK. Cedrin, a polymethoxy-flavonoid, has the ability to suppress JNK expression, decrease MMP-2 and MMP-9 expression in U2OS and HOS cells, and lessen U2OS and HOS cell migration. These results clearly imply the therapeutic potential of JNK in flavonoid compounds for the treatment of OS. These results imply that the antitumor actions of different flavonoids may include JNK-mediated pathways for cell survival and death, which provides new research ideas and directions for the treatment of OS.
P38 MAPK
The following can activate P38: inflammatory cytokines, pathogen-associated molecular patterns (PAMPs), danger-associated molecular patterns (DAMPs), genotoxic and DNA-damaging agents (e.g., DOX, cisplatin, UV, and γ-radiation), heat shock, osmotic pressure changes, and oxidative stress. Through its interactions with a diverse range of substrates, P38 can control apoptosis, cell migration, differentiation, stress responses, proliferation, migration, and survival. According to reports, the expression of p38 has a strong protumorigenic ability because it regulates the production of cytokines such as interleukin-6 (IL-6), tumor necrosis factor (TNF), and other cytokines. On the other hand, p38 also has a proapoptotic ability and thus is also capable of inhibiting tumor development. Some chemotherapeutic drugs often rely on p38 activation for their antitumor effects. 7,8-dihydroflavone (7,8-DHF) reduced AKT, Bad, and p38MAPK and their phosphorylation levels, promoted apoptosis, and inhibited migration of OS cells (Zhao et al., 2020), and suppressed the tumor promoter function of p38. Delphinidin management led to noteworthy changes in the phosphorylated forms of ERK and p38; phosphorylated forms of p38 were significantly reduced, showing an anti-OS tumor effect. In contrast, cardamonin increased the phosphorylation level of p38, which implies that cardamonin activates the ability of p38 as a tumor suppressor that can promote OS apoptosis. Similar phenomena were observed in other tumor development processes; for example, TGFβRII activation in metastatic prostate cancer cells facilitated RB phosphorylation of p38, preventing bone metastases. In line with the above results, another study found that calycosin was also able to induce 143B cell apoptosis and inhibit tumor growth by inducing p38 MAPK phosphorylation. In canine OS (D-17 and DSN) cell lines, quercetin enhanced programmed cell death and phosphorylation of P38, c-Jun N-terminal kinase, and P90RSK proteins (Wang et al., 2023; Wang & Zhao, 2023). Because p38 MAPK has the dual function of being a tumor promoter and a tumor suppressor, its actions in flavonoid treatment for OS are not constant, or perhaps the reverse. In conclusion, more research is required to determine the exact mechanism behind p38’s involvement in the therapy of OS with various flavonoid drugs.
Signal Transduction and Transcriptional Activator 3 (STAT3)
As a crucial transcription factor implicated in inflammation and tumor growth, STAT3 is a member of the STAT family. STAT3 is frequently activated in numerous malignancies, such as OS, where it fosters cell proliferation, invasion, and antiapoptotic processes, as well as inhibiting cell death, promoting cell cycle advancement, and stimulating angiogenesis. For instance, the flavonoid quercetin inhibited STAT3 phosphorylation, diminished the expression of c-MYC, Bcl-2, and COX-2, and induced apoptosis in OS cells. Similarly, apigenin triggered apoptosis in OS cells by inhibiting the STAT3 pathway. Luteolin’s antitumor effects on OS cells may involve a mechanism by which STAT3 activity is inhibited. The evidence suggests that flavonoid-mediated modulation of the STAT3 signaling pathway may be an effective strategy for treating OS by targeting STAT3.
Wnt/β-catenin
Wnt/β-catenin signaling is an evolutionarily conserved signal that regulates organogenesis, tissue homeostasis, cell fate determination, and tumors/cancerous diseases. Numerous features of cancer incidence, development, and malignant transformation have been discovered to be strongly correlated with the Wnt/β-catenin signal. Studies have also demonstrated that overexpression or aberrant activation of the Wnt/β-catenin signal has a noninsignificant effect on mortality from cancer. Tumor formation requires the highly active Wnt/β-catenin pathway as a necessary precursor. According to recent research, EGCG dramatically increases apoptosis and drastically blocks 143B cell invasion and migration. This impact is directly correlated with the Wnt/β-catenin pathway. High-dose EGCG has a strong apoptotic impact, but low-dose EGCG only has a few apoptotic effects. While research indicates that baicalein controls Wnt/β-catenin via miR-25, it also decreases β-catenin, Axin2, and GSK-3β gene expression and stops Saos-2 cell growth. Research has shown that baicalein regulates the Wnt/β-catenin pathway, with β-catenin subsequently acting on the downstream effector protein c-MYC to trigger the death of MG-63 cells following baicalein’s interaction with the Wnt/β-catenin pathway. A comparable result was also observed with apigenin, which blocked human OS cell migration, invasion, and proliferation while also deactivating the Wnt/β-catenin signal in OS cells, which in turn prevented OS cell tumor growth. In conclusion, there may be hope for the therapy of human OS with the discovery of novel flavonoids that target Wnt/β-catenin targets.
Nuclear Factor Kappa-B (NF-κB)
The immune system, cell proliferation and transformation, apoptosis, and tumor growth are all significantly impacted by NF-κB, a well-known modulator of cellular inflammatory factors, stress signaling, and pathogen-induced gene expression. In several malignancies, aberrant expression of NF-κB has been noted. There has been a lot of discussion about flavonoids’ ability to suppress NF-κB. Prior research has demonstrated that in U-2 OS osteosarcoma xenografted mice, astragalus mentoflavone suppresses both the activation of NF-κB p65 (Ser536) and the development of tumors. Dihydromyricetin inhibits promoter binding to urokinase prothrombin activator (uPA) via NF-κB, which gives it anti-OS tumor properties. Amentoflavone inhibited ERK and NF-κB activation in U2OS cells, which decreased the potential for tumor metastasis, according to another study. Consequently, it appears that one of the key mechanisms by which flavonoids have antitumor effects is the reduction of NF-κB expression. Not only that, but reduced NF-κB expression can also make OS cells more susceptible to certain conventional chemotherapy medications. Amentoflavone made OS cells more susceptible to Sorafenib by suppressing NF-κB expression both
Novel Treatment of OS with Flavonoids
Chemotherapy Combination Therapy
Currently, cisplatin chemotherapy remains an important treatment for OS and can improve patient survival. However, cisplatin monotherapy often causes various toxic effects in OS patients, including ototoxicity, neurotoxicity, nephrotoxicity, and hepatotoxicity. In addition, resistance to chemotherapy also leads to poor responsiveness to conventional chemotherapy in some patients with aggressive OS. Therefore, the search for molecularly targeted drugs with tumor specificity is a current research hotspot, and solving this problem is crucial to improving the efficacy of chemotherapy. Quercetin is one of the flavonoids that gives optimism for the current situation. Research has demonstrated that the coadministration of flavonoids and chemotherapeutic medicines can enhance the susceptibility of OS cells to these agents and magnify their therapeutic impact. The combination of quercetin and cisplatin significantly increased the cytotoxicity of the drug and promoted tumor cell death. Quercetin, when combined with cisplatin, was found to enhance cytotoxicity in 143B cells, increase sensitivity to cisplatin, and improve the effectiveness of OS chemotherapy. The miR-217-KRAS signaling axis may be involved in this process. When quercetin and methotrexate were given together to Saos-2 cells, the MTX IC50 value decreased from 13.7 to 8.45 ng/mL. Additionally, this combination therapy increased the expression of tumor suppressor genes, reduced the expression of antiapoptotic genes, and increased the susceptibility of Saos-2 cells to MTX chemotherapeutic agents. Furthermore, the combination of quercetin and cisplatin accelerated the DNA damage and death of Saos-2 cells, disrupted the DNA repair process, and encouraged the downregulation of miR-22 and DNA damage response components such as P53, ATM, and ATR. Genistein flavonoids are nontoxic flavonoids. Notably, genistein flavonoids also appear to have the same effect as quercetin. Genistein increased the chemosensitivity of OS to gemcitabine. Additionally, the concomitant administration of gemcitabine and genistein flavonoids increased the drug’s inhibitory impact on tumor development, activated the AKT pathway, and downregulated NF-κB activity. Additionally, it has been proposed that genistein increases the induction of apoptosis by reversing the OS cells’ resistance to gemcitabine (Sung & Aggarwal, 2022). Furthermore, lignans and cisplatin work in concert, and combination treatment stops the growth of OS cells and causes them to die by changing signaling and causing mitochondrial dysfunction. When used alongside the anticancer medication etoposide, another flavonoid called fisetin raises the proportion of OS cells in the G2 and S phases and lowers the proportion of cells in the G1 phase, resulting in cell cycle arrest and more potent antiproliferative effects. The combined use of hesperidin and the chemotherapeutic drug etoposide showed a synergistic effect of inhibiting cell growth, inducing G2 phase arrest in U2OS cell lines with higher cytotoxicity, and amplifying the chemotherapeutic effect of etoposide. Alopecurone B was shown to have a considerable inhibitory effect on resistance-associated proteins, increasing the susceptibility of MG-63/DOX cells to other standard chemotherapeutic drugs. The flavonoid glycoside poncirin exhibits antitumor activity and has also been shown to reverse cellular resistance to cisplatin and enhance cisplatin sensitivity in OS cells. DiOHF binds to DOX, resulting in a reduction in the DOX dose, synergistically altering cell cycle dynamics, and co-operatively exerting antiproliferative effects in U2OS cells. In conclusion, the combination of flavonoids and chemotherapeutic agents delivers hope to OS patients, and this combination therapy seems to be a rational, novel, and promising new strategy for OS treatment.
Nanotherapy of Flavonoids
In recent years, nanomaterials have gradually become a hot research topic in various industries. Nanomaterials are gradually becoming suitable carriers for optimizing conventional drugs due to their good biocompatibility, degradability, and release responsiveness. Currently, polymer nanoparticles, carbon-based nanoparticles, and metallic nanomaterials are involved in cancer treatment. The clinical application of quercetin is limited due to its low water solubility and oral availability. Additionally, the hydrogen bonding between quercetin and albumin causes low absorption and rapid metabolism of quercetin in the body, further limiting its effects. Despite its potent antitumor activity, researchers have attempted to improve the target delivery efficiency of quercetin by liposome encapsulation. It was shown that liposome-encapsulated quercetin exhibited better antitumor effects, as reflected by the reduction of tumor volume
Potential Toxicity or Adverse Effects of Flavonoids in the Treatment of OS
Flavonoids exert inhibitory effects on OS by inhibiting tumor cell proliferation, inducing apoptosis, and regulating the cell cycle. However, the anticancer effects of these compounds may be accompanied by adverse reactions under certain conditions. For example, dihydromyricetin has potentially toxic effects on the liver and kidney at high concentrations. Studies have shown that although these compounds exhibit significant anticancer activity
Conclusion
This review lists several widely studied flavonoid compounds for the management of OS and focuses on the key molecular mechanisms involved in the treatment of OS by flavonoids. Apart from this, we also discussed the combination of flavonoids and chemotherapy. Flavonoids can decrease tolerance, increase the effectiveness of chemotherapy, and increase the sensitivity of OS cells to chemotherapy medicines. Although flavonoids show great potential in the areas of anti-inflammatory, antioxidant, anticancer, and neuroprotection, their translation from the laboratory to clinical practice faces many challenges. First, flavonoids have poor bioavailability and pharmacokinetics, with limited absorption after oral administration, rapid metabolism, and easy elimination from the body. In addition, the dose and efficacy of flavonoids are not easy to determine, and the biological activity varies widely among different flavonoid compounds, making it difficult to establish an optimal dosage in clinical practice. At high doses, their safety and toxicity issues also need to be resolved, and long-term use may cause hepatotoxicity, nephrotoxicity, and other side effects. Meanwhile, there are difficulties in the standardization and quality control of flavonoid extracts, with different sources and extraction processes leading to large fluctuations in chemical composition. In addition, regulatory and intellectual property issues for flavonoids have hindered their large-scale production and clinical application. Therefore, future research must focus on developing new formulations to improve bioavailability, optimize treatment protocols, and establish quality control standards for flavonoid-based therapies. By addressing these challenges, flavonoids have the potential to become an important part of OS treatment in the future.
Footnotes
Abbreviations
APGAR: Appearance, pulse, grimace, activity, and respiration (score for newborns); BPB: Balloon pressure band; CI: Confidence interval; GA: Gestational age; Hb: Hemoglobin; IV: Intravenous; NBAs: Neonatal asphyxia; OR: Odds ratio; PH: Postpartum hemorrhage; RCT: Randomized controlled trial; RR: Relative risk; SD: Standard deviation, SPSS: Statistical Package for the Social Sciences; US: Ultrasound; WHO: World Health Organization.
Authors’ Contributions
Zhenhai Cai and Zhengwei Zhou conceived, designed, and supervised the research. Zhenhai Cai, Jieen Pan, Xiaojun He, and Wenmin Wang collected the related literature. Zhenhai Cai and Zhengwei Zhou wrote the manuscript. The final draft of the work has been read and approved by all writers.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
