Abstract
Flavonoids are bioactive plant secondary metabolites with various pharmacological activities. However, their application is limited by poor solubility, low bioavailability and instability under environmental conditions. Microparticle technology has emerged as a promising approach to overcome these limitations by enhancing flavonoid stability, solubility and controlled release. This narrative review summarises recent advances in the formulation and characterisation of herbal-based flavonoid microparticles, highlighting polymer selection, encapsulation techniques (e.g., solvent evaporation, ionotropic gelation, spray drying, freeze drying and spray chilling), physicochemical properties, and biological activities. The review emphasises that microparticle-based delivery systems may enhance flavonoid bioavailability and preserve bioactivity, offering a potential strategy for the development of novel therapeutic agents. These insights provide a comprehensive reference for future research on the pharmaceutical application of flavonoid microparticles.
Introduction
Flavonoids are a group of plant secondary metabolites that are widely found in medicinal plants, fruits and vegetables.
1
Several crude drugs listed in the Indonesian Herbal Pharmacopoeia 2nd Edition and its supplement, such as
Flavonoids are widely recognised for their diverse biological activities, including antioxidant, antitumor, antimicrobial, anti-inflammatory, antiallergic, antiviral and immunomodulatory effects.4,5 However, the clinical application of flavonoids still faces several challenges, particularly their low bioavailability. Contributing factors include their complex chemical structures, relatively high molecular weight, poor water solubility, limited absorption and metabolism in the gastrointestinal tract, as well as rapid elimination. Flavonoids are also highly susceptible to degradation due to exposure to oxygen, high temperatures, ultraviolet radiation and environmental pH changes. 6 Therefore, technological approaches are required to optimise the stability and bioavailability of flavonoids.
Microparticle technology has been developed as an effective strategy to overcome the limitations of flavonoids by enhancing their physicochemical stability and protecting active compounds from degradation. Therefore, microparticle-based delivery systems are considered promising for enhancing the therapeutic potential of flavonoid-rich herbal extracts.7,8 Various preparation methods and polymer types have been applied in the development of flavonoid-loaded microparticles, resulting in diverse physicochemical characteristics and biological activity profiles.
However, existing review articles on microencapsulation of natural products have predominantly emphasised encapsulation techniques and improvements in physical attributes such as stability, handling properties and shelf life. A focused and critical synthesis dedicated specifically to flavonoid-based systems is still lacking. In particular, the interrelationships between microencapsulation methods, polymer selection, resulting physicochemical properties, and their ultimate impact on biological activity have not yet been systematically analysed. This knowledge gap limits the rational design of flavonoid microparticle delivery systems. Therefore, this review aims to provide a comprehensive and critical discussion on microparticle formation techniques, polymer selection, physicochemical characterisation and current biological applications of flavonoid microparticles derived from both pure compounds and natural product extracts.
Methods
This narrative review was conducted by retrieving relevant literature from Google Scholar, PubMed and ScienceDirect. The search strategy employed combinations of keywords, including
The inclusion criteria comprised experimental studies (in vitro, in vivo, or formulation-based) involving microparticle or microencapsulation techniques applied to flavonoid-containing herbal extracts. Selected studies were required to report both physicochemical characterisation and biological activity. Only peer-reviewed articles published in Indonesian or English between 2015 and 2025 were included. Exclusion criteria included review articles, opinion papers, conference abstracts, patents and books. Studies not involving microparticle or microencapsulation approaches, not based on flavonoid-containing herbal extracts, or lacking relevant physicochemical and biological data were also excluded. Articles without accessible full texts were not considered.
The literature search initially identified 1,079 articles. After excluding non-open-access publications, 563 articles were subjected to preliminary screening. Following the title and abstract evaluation, 154 articles were selected for full-text review. Ultimately, 15 studies were included as the main references in this narrative review.
Characteristics of Flavonoids and Herbal Extracts
Flavonoids are secondary metabolites produced through the combination of two major biosynthetic routes: the polyketide pathway, which is responsible for the formation of ring A, and the shikimic acid (phenylpropanoid) pathway, which contributes to the formation of rings B and C. 1 As a result of this biosynthetic origin, flavonoids possess a characteristic polyphenolic structure consisting of 15 carbon atoms arranged in a basic C₆–C₃–C₆ skeleton, comprising two aromatic rings (C₆) connected by a three-carbon aliphatic chain.4,9 Based on the position of the carbon atom in the C-ring where the B-ring is attached, as well as the degree of unsaturation and oxidation of the C-ring, flavonoids can be classified into several subclasses (Figure 1). Isoflavones are flavonoids with the B-ring attached at the 3-position of the C-ring, while those with the B-ring attached at the 2-position are divided into subgroups including flavones, flavonols, flavanols, flavanones, anthocyanins and chalcones.
Classification of Flavonoids and Their Subclasses. (a–j) Are Apigenin, Luteolin, Kaempferol, Quercetin, Genistein, Daidzein, Cyanidin, Delphinidin, Phloretin and Chalconaringenin, Respectively.
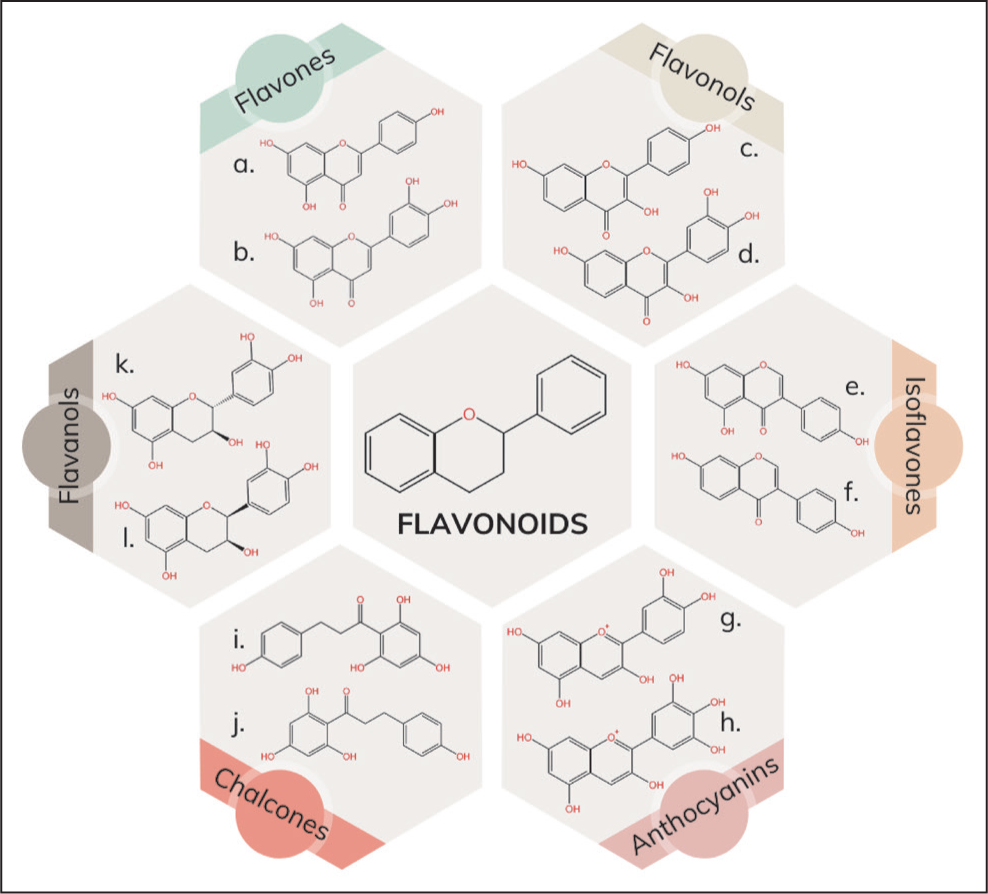
Flavonols are characterised by a hydroxyl group at the 3-position and generally possess a ketone group at the 4-position; they are also known as precursors of proanthocyanidins. Examples include kaempferol, quercetin, myricetin, rutin and fisetin. Another important subclass is flavanones, which are abundant in citrus fruits such as oranges, lemons and grapes; hesperetin, naringenin and eriodictyol belong to this group. 1 As members of the phenolic compound family, flavonoids share the same secondary plant metabolism pathways with other phenolic derivatives such as phenolic acids, tannins, lignans and coumarins. 10
Flavonoids in nature generally occur in two forms: aglycones (free form) and glycosides (sugar-conjugated form). 11 The aglycone form consists only of the basic flavonoid skeleton with hydroxyl groups, whereas glycosides are typically bound to carbohydrates at the C-3 or C-7 position, forming O-glycosides such as rhamnosides, rutinosides, galactosides and arabinosides. In addition, C-glycosides are also found, in which the sugar moiety is directly linked to the carbon atom of the flavonoid backbone. 12 In the isoflavone subclass, aglycones can bind to sugars to form daidzein and genistein, while in the flavonols subclass, glycosylation yields compounds such as rutin, myricetin glycosides and astragalin. 11 Flavones, one of the largest subclasses of flavonoids, possess a 4H-chromen-4-one backbone with a phenyl substituent at the C-2 position. These compounds are commonly present as 7-O-glycosides and are found in celery, parsley, red pepper, mint and ginkgo. Apigenin and luteolin are among the most abundant flavones. In nature, apigenin usually occurs in glycosylated forms, either as O-glycosides (sugars bound via hydroxyl groups) or C-glycosides (sugars directly bound to the carbon atom of the tricyclic core).13,14 In addition to free flavonoids and glycosides, several other derivatives are also present in nature, such as flavonoid sulfates and other conjugated forms, which contribute to their solubility, stability and bioactivity.
Flavonoids are a group of natural phenolic-based compounds that belong to secondary metabolites and can be found in both plant and animal products. In animals, for example, bees, flavonoids have been identified in propolis. Phytochemical analysis of red propolis revealed the presence of flavonoids, isoflavonoids, phenolics, luteolinidin, kaempferide, malvidin, poniciretin, hesperetin and liquiritigenin. 15 In plants, flavonoids are widely distributed across different organs such as fruits, leaves, seeds, bark, roots, stems and flowers. 16 Dietary sources rich in flavonoids include tea, red grapes, apples, onions and tomatoes. Flavonoids are present in almost all green plants and play a major role in producing yellow, red, orange, blue and purple pigments in fruits, flowers and leaves. 9
From a taxonomic perspective, several plant families have been identified as abundant sources of flavonoids. The Asteraceae family, for instance, is rich in flavonols and often used as taxonomic markers, with total flavonoid content ranging from 18.03 to 185.44 mg QE/L in species such as
Beyond taxonomic distribution, the presence of flavonoids has also been confirmed through the standards of the Indonesian Herbal Pharmacopoeia (IHP) and phytochemical studies. For example, extracts of
One of the main reasons flavonoids have been extensively studied is their broad spectrum of biological activities, particularly antioxidant and therapeutic effects. 25 As antioxidants, flavonoids can directly neutralise free radicals by donating hydrogen atoms, rendering them non-reactive, or indirectly by chelating metal ions involved in radical formation. 9 The in vitro antioxidant activity of flavonoids is strongly influenced by the arrangement of functional groups in their core structure, particularly the configuration and number of hydroxyl groups. Their protective mechanisms include free radical neutralisation, enhancement of endogenous defence enzymes, inhibition of oxidant formation and prevention of cell damage caused by metals and nitric oxide (NO). 13
In addition to antioxidant properties, flavonoids exhibit anti-inflammatory effects through multiple mechanisms. They can suppress the expression of pro-inflammatory cytokines such as TNF-α, IL-1, IL-6, IL-8 and monocyte chemoattractant protein-1 (MCP-1) in Jurkat T cells, peripheral blood mononuclear cells, and RAW macrophages. For example, apigenin can attenuate TNF-α-induced inflammation by reducing the prolonged production of pro-inflammatory mRNA during inflammatory processes. Apigenin also reduces the expression of intercellular adhesion molecule-1 (ICAM-1), E-selectin, and vascular cell adhesion molecule-1 (VCAM-1) in endothelial cells. Other flavonoids, such as quercetin and catechin, have been reported to increase IL-10 production by inhibiting combined TNF-α and IL-1 signalling, while flavonols such as morin, quercetin, kaempferol and myricetin are known to inhibit lipoxygenase activity. Quercetin also exerts protective effects by suppressing heat shock factor (HSF) activity, thereby reducing heat stress-induced damage. 13
Flavonoids also hold potential as antimicrobial and antiviral agents. Their antibacterial activity occurs through various mechanisms, including disruption of bacterial cell membranes, inhibition of biofilm formation, suppression of nucleic acid and ATP biosynthesis, and interference with the electron transport chain. Several flavonoids, including quercetin, apigenin, naringenin, chrysin, genistein and kaempferol, have been shown to inhibit biofilm formation, while quercetin and myricetin interfere with bacterial DNA replication. 13 On the other hand, flavonoids are also effective as antiviral agents by blocking viral attachment, preventing entry into host cells, and inhibiting replication, transcription, and translation of viral genomes. 26 These mechanisms include direct interactions with viral surface proteins, modulation of the immune system, and reduction of viral load. Both in vitro and in vivo studies have demonstrated that apigenin possesses antiviral activity against RNA and DNA viruses such as herpes simplex virus Types 1 and 2, hepatitis B virus and hepatitis C virus. 13 Luteolin has been reported to inhibit HIV-1 reactivation, while genistein suppresses HIV-1 infection in CD4+ T cells and macrophages. 27
Despite their wide-ranging biological potential, flavonoids possess unique characteristics that present both advantages and limitations in their applications. Flavonoids are polar compounds and are therefore readily soluble in polar solvents such as ethanol, methanol, butanol, acetone and dimethylformamide.9,28 Glycosylated flavonoids tend to be more polar and are better suited for dissolution in aqueous-organic solvent mixtures, whereas aglycone forms such as flavones, flavonols and flavanones are more soluble in nonpolar solvents like chloroform and ether. 9
Each flavonoid subclass also exhibits specific physicochemical properties. Quercetin, for example, has a molecular weight of 302.24 g/mol and a melting point of 316°C. 29 The log P values of flavonoid aglycones range between 0.4 and 3.2, while melting points vary from 141°C to 358°C. Flavanols have relatively low melting points (141°C–220°C), flavanones 227°C–251°C, while isoflavones, flavonols and flavones are generally above 250°C. In terms of acidity, isoflavones, flavonols and flavones have an approximate pKa of 6.5, though experimental data show considerable variation, for instance, baicalein with pKa1 5.30, apigenin 8.54 and quercetin 5.81–8.45. 11 The molecular weights of flavonoids also vary, with flavones and isoflavones averaging 222 a.m.u., flavanones and chalcones 224 a.m.u. and dihydroflavonols 240 a.m.u. 30
The diversity of flavonoid physicochemical properties poses challenges for their use as therapeutic agents. Flavonoids generally have low water solubility and are unstable due to susceptibility to degradation by oxidation, temperature, ultraviolet radiation, light and pH changes, all of which reduce their bioavailability.6,19,31,32 Structural changes due to oxidation or degradation may result in loss of biological function as active agents. To overcome these limitations, several strategies have been developed, including structural modification into flavonoid-glycosides through enzymatic transglycosylation using transferase enzymes, 32 as well as modern drug delivery systems. These include vesicular systems (liposomes, phytosomes, ethosomes, transfersomes) and particulate systems (microspheres, nanoparticles, microparticles). 33 Among these technologies, microparticles have been widely developed as they can improve the physicochemical properties of flavonoids while overcoming their limited bioavailability. 7
Microparticles Synthesis Methods
Micronisation is defined as the process of reducing particle size to less than ten microns through various mechanisms. Reducing particle size to the micron scale is usually accompanied by changes in structural, physicochemical and functional properties. Characteristics such as solubility, taste-masking ability, water absorption capacity, reaction rate and flavour release may be improved due to the increased surface area at the micron scale. 34 In addition, the term microparticles refers to particles possessing a polymer matrix that can protect the encapsulated active substance. 8 Structurally, microparticles range in size from 1 to 1,000 μm, while nanoparticles have a diameter of 1–100 nm. Based on their structure, microparticles are divided into two main types: microspheres, in which the active compounds and carriers are homogeneously dispersed or dissolved within the matrix, and microcapsules, which contain a membrane layer covering a solid core, thereby entrapping the active substance inside (Figure 2). 35 Microencapsulation is essentially a technique for packaging solid, liquid, or gaseous materials within a continuous film layer, protecting the functional material from environmental influences and forming particles ranging in size from a few nanometers to several millimetres. 36 The encapsulated substance is referred to as the core, fill, active ingredient, or internal phase, while the encapsulating substance is known as the coating, membrane, shell, capsule, carrier, external phase, or matrix. 34
Types of Microparticles: Microspheres (a) and Microcapsules (b). 35
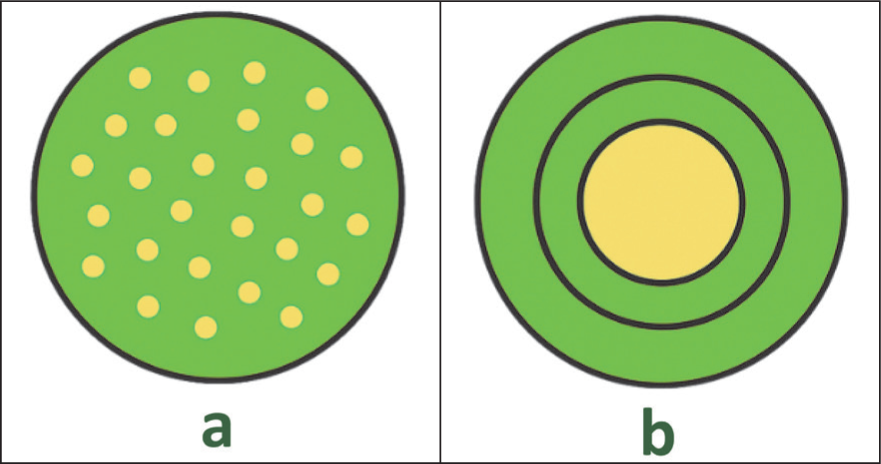
Microparticles are polymer-based drug delivery systems designed to protect active compounds from oxidation, temperature, light, humidity, microorganisms and degradation, thereby enhancing the stability and shelf life of bioactive compounds.36–39 In general, microparticles are solid matrices in which the active ingredients are dispersed or dissolved homogeneously. In contrast, microcapsules represent a specific type of microparticle characterised by a core of active material surrounded by a polymeric protective shell. The fundamental difference lies in their structure: microparticles exhibit a matrix dispersion, while microcapsules follow a core–shell configuration. Both systems can improve bioavailability by shielding sensitive compounds from enzymatic or pH-related degradation, while also enabling controlled release governed by the properties of the polymer. This controlled release mechanism offers additional therapeutic advantages, including enhanced patient compliance through reduced dosing frequency and minimised systemic toxicity. 40
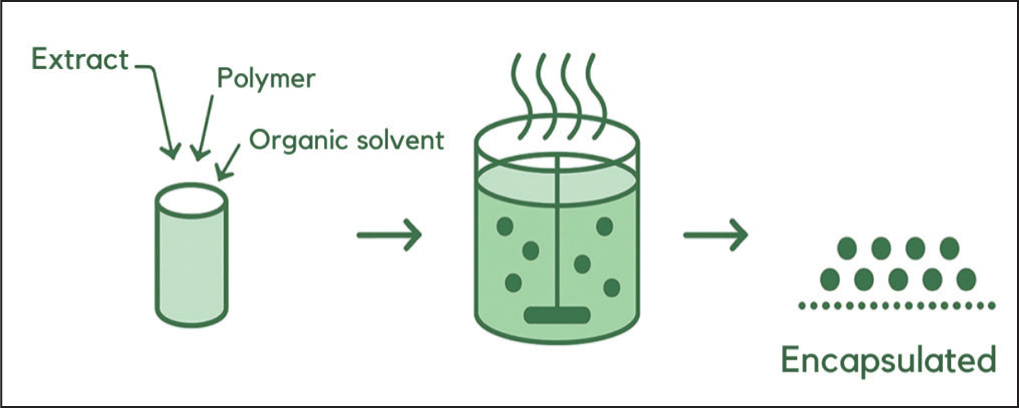
Several methods have been developed for microparticle fabrication, including solvent evaporation, ionic gelation, spray drying, freeze drying, spray chilling, coacervation and supercritical fluid extraction.7,37,41,42 Among these, solvent evaporation is one of the most widely applied techniques. This method is particularly suitable for encapsulating active compounds that are poorly water-soluble, sparingly soluble, insoluble in nonpolar solvents, or reactive, such as enzymes. The principle involves dissolving or dispersing the active ingredient into a polymer solution, followed by emulsification into an external phase such as water or oil. Microparticle formation occurs as the solvent diffuses or evaporates, leading to polymer precipitation and microparticle solidification, which are subsequently dried (Figure 3).43,44 This method is favoured due to its relative simplicity and low cost. However, it also presents limitations, including poor reproducibility, broad particle size distribution, challenges in morphology control, and suboptimal %EE. 45
Basic Process of Microparticle Preparation Using Solvent Evaporation. 46
In addition to the solvent evaporation method, another widely used technique is ionic gelation. This method involves the formation of cross-links between polyelectrolytes and multivalent ions. 47 For example, negatively charged polymer solutions such as alginate can react with positively charged ions like calcium. This mechanism is often described using the egg-box model, in which guluronic acid residues of alginate crosslink with calcium ions to form a gel network (Figure 4).7,48 Ionic gelation generally employs hydrophilic, biodegradable polymers such as sodium alginate, gelatin and chitosan, and is more suitable for encapsulating hydrophobic compounds to prevent them from mixing with the wall material. 48
Microparticle Formation Process by Ionic Gelation Method. 48
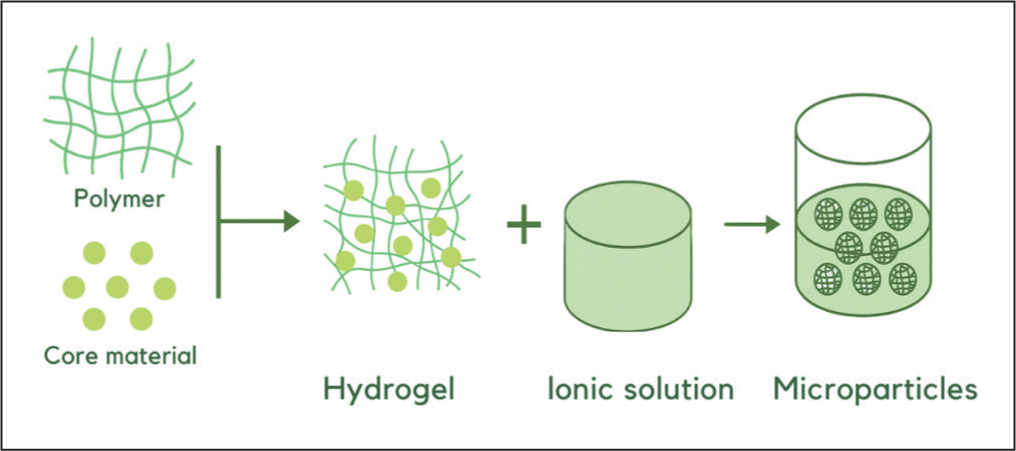
This method offers several advantages, including being relatively simple, fast and inexpensive; it does not require organic solvents, can be performed at low temperatures, and utilises safe natural polysaccharides. 49 Therefore, ionic gelation is highly suitable for thermolabile compounds, as microparticles can be formed without high heat. 50 However, its main drawback lies in the low mechanical stability of the microparticles. This limitation can be addressed through the formation of polyelectrolyte complexes, such as chitosan with other polymers (e.g., alginate, dextran, or chondroitin), or by modifying chitosan chains with specific functional groups. 51 Another drawback is its limited ability to control particle size. Microparticles obtained by ionic gelation should be stored in sealed containers because, if left exposed, the water content in the hydrogel may evaporate, leading to gel degradation and limiting storage stability. 48
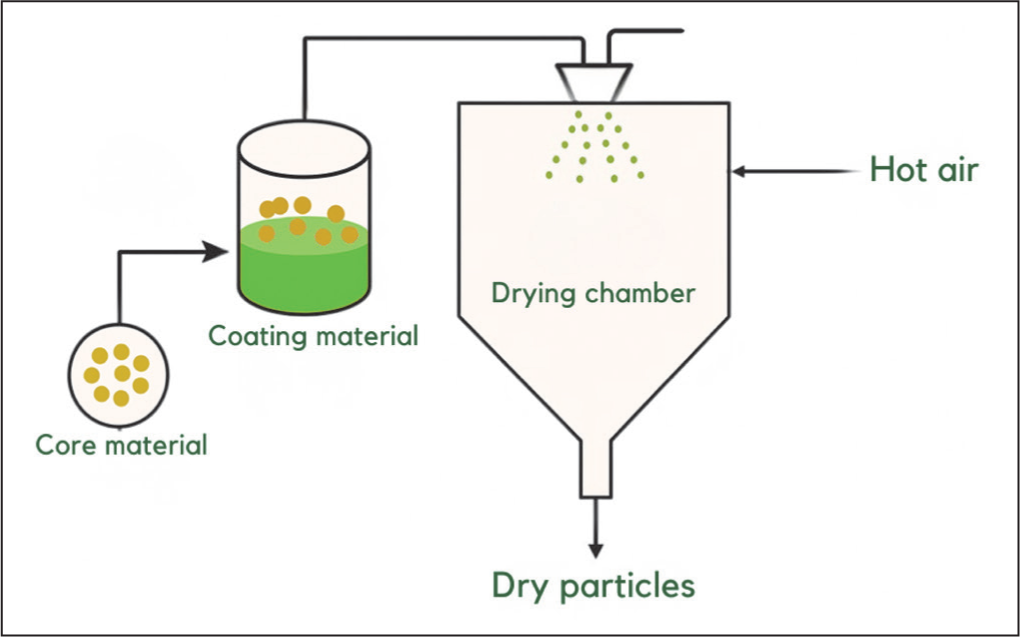
In contrast to ionic gelation, which is carried out at low temperatures, the spray drying method produces microparticles by converting a solution or suspension into a dry powder using hot air. The process begins with atomisation of the liquid into fine droplets, followed by rapid drying in a stream of hot air (Figure 5). 52 Spray drying is the most widely applied microencapsulation technique because it is relatively simple, economical and capable of producing products with good stability and long shelf life. 48 This method also offers high flexibility, a large encapsulation capacity, and suitability for continuous production processes. 52 Such advantages make spray drying effective for simultaneously drying and encapsulating bioactive compounds, including flavonoids, with the aid of appropriate polymers.53,54 The choice of polymer is critical, as it can improve solubility, reduce hygroscopicity, enhance emulsification, mask undesirable taste, form a protective film layer, and lower production costs. 55 Nevertheless, the major limitation of this method lies in the risk of degradation of heat-sensitive compounds, since the atomisation temperature typically ranges between 140°C and 180°C. 48
Formation Process of Microparticles by the Spray Drying Method. 48
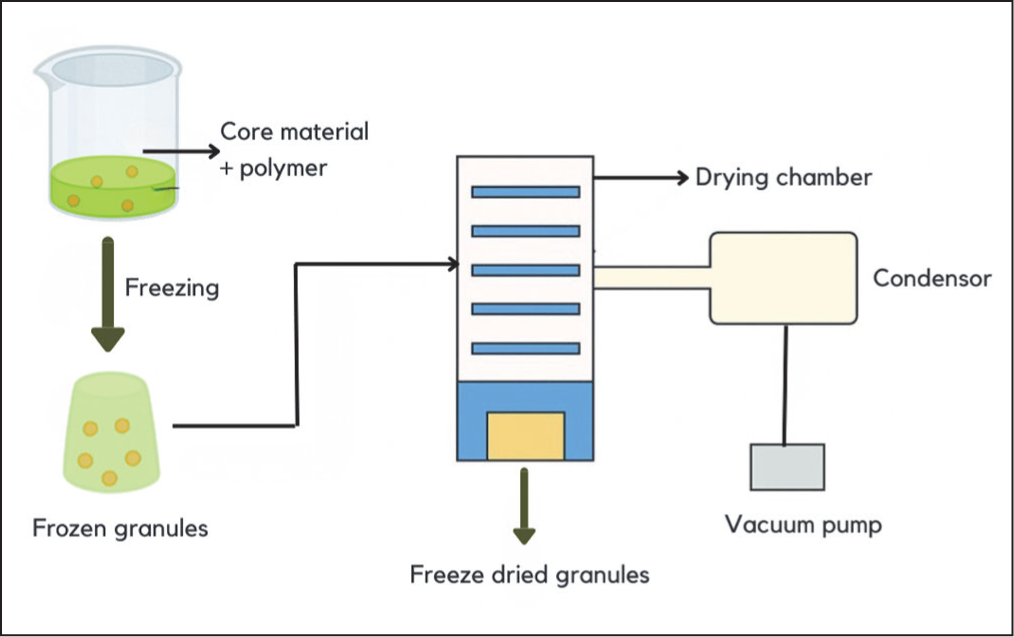
In contrast to spray drying, which involves high temperatures, freeze drying (lyophilisation) is carried out under low temperature conditions (<0°C) and pressures below the triple point, allowing the sublimation of ice into vapour without passing through the liquid phase. The process occurs in two stages, namely sublimation (primary drying) and desorption (secondary drying). 56 This technique is highly suitable for thermolabile compounds because the low temperature and pressure conditions help maintain stability, prevent oxidation and protect bioactive components that are sensitive to heat. 48 The preparation of microparticles by freeze drying is usually preceded by high-pressure homogenisation or microjet processing to mix the core material (active substance) with the wall material (polymer), forming an emulsion that is then dried under vacuum to obtain microparticles (Figure 6). This method is also beneficial for materials that are unstable in aqueous solution but become more stable once in a dry form. 56 However, freeze drying has certain limitations, particularly due to its longer drying time, high energy consumption, and costs that can be 30–50 times greater compared with spray drying. 48
Microparticle Formation Process by Freeze-drying Method.
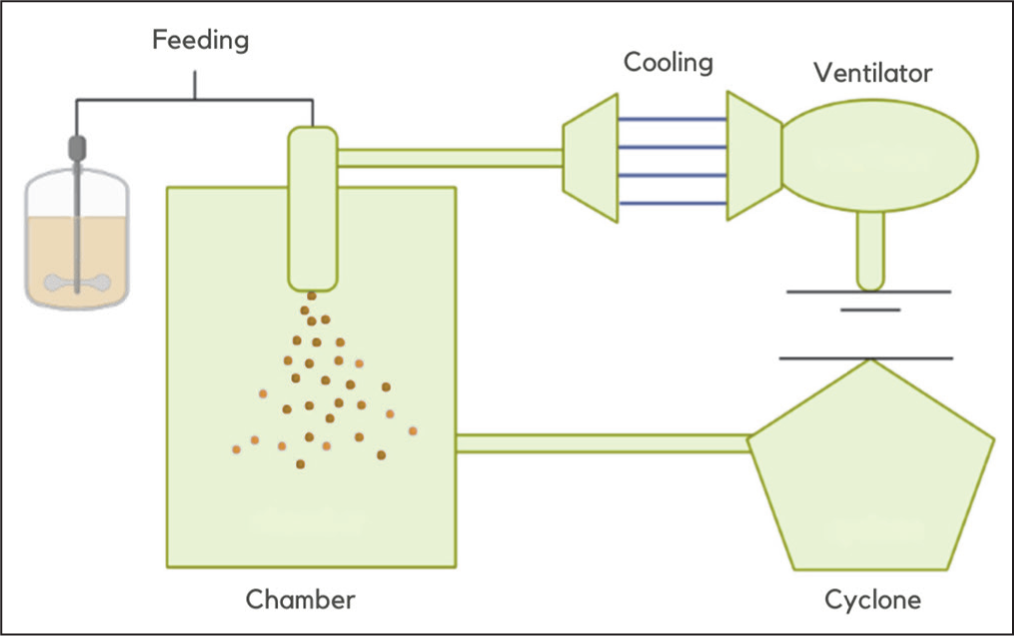
In addition to drying-based techniques such as spray drying and freeze drying, there is also the spray chilling method, which utilises lipid solidification as the coating matrix. In this method, the active ingredient is dispersed or dissolved in melted lipid, then pumped through a heated atomising nozzle and sprayed into a chamber maintained below the lipid’s melting point. Under these conditions, droplets are formed and immediately solidify into solid lipid microparticles (Figure 7).57,58 The main advantage of spray chilling is that it does not require organic solvents and can be performed at relatively low temperatures, thereby helping to prevent the degradation of bioactive compounds. 59 The resulting microparticles are usually solid with a homogeneous distribution of the active ingredient throughout the lipid matrix, making this method suitable for compounds with low to moderate water solubility.57,59 Furthermore, this method is considered simple, rapid and cost-effective. However, spray chilling also has limitations, including the potential release of active ingredients during storage due to lipid recrystallisation, as well as technical challenges when using lipids with high melting points (>70°C), which may cause nozzle clogging and thermal degradation of the active ingredient. 58
Microparticle Formation Process by Spray Chilling Method. 60
Polymers Used in Microparticle Synthesis
Polymers play an essential role in microparticle formation, functioning as coating agents that protect active compounds from external factors. Beyond enhancing stability, polymers can also mask undesirable tastes or odours and prevent the evaporation of volatile compounds. 61 Furthermore, polymers enable the controlled release of active compounds through various mechanisms, such as pH or temperature changes, diffusion, or vapour pressure. 62 As macromolecules composed of repeating monomeric units, polymers exhibit diverse characteristics depending on chain length, three-dimensional configuration, and degree of asymmetry. Thus, polymers not only serve as structural components but also play a decisive role in determining the functional properties of microparticles. The ideal polymer should be stable, inert, safe, biodegradable, biocompatible, and cost-effective. Based on their origin, polymers are generally divided into two broad categories: natural polymers and synthetic polymers. Both groups may be biodegradable, such as alginate, chitosan, and gelatin as examples of natural polymers, as well as polylactide (PLA) and polycaprolactone (PCL) as examples of synthetic polymers. 63
Natural polymers are biologically formed during the life cycle of organisms and are generally derived from plants or animals. These polymers offer several advantages, including accessibility, biocompatibility, water solubility, and lower toxicity compared to synthetic polymers. 63 Another advantage is their ability to mimic the structure of biological macromolecules, enhance specific cell adhesion, and degrade through enzymatic pathways without producing cytotoxic by-products. 64 In addition to being abundant in nature and relatively inexpensive, natural polymers can also be chemically modified for broader applications. 65 Broadly, natural polymers can be grouped into two main categories: polysaccharides and proteins. 66
One of the most widely used natural polymer groups is polysaccharides, carbohydrate polymers consisting of monosaccharide units linked by
In addition to polysaccharides, natural polymers can also be protein-based, such as gelatin, which has been widely applied as an encapsulating material through spray drying, ionic gelation, and coacervation methods. Protein-based polymers are generally chosen for the encapsulation of heat-sensitive lipophilic compounds and are often combined with freeze-drying techniques to maintain stability. 65
Other important natural polymers in microparticle systems include maltodextrin and gum Arabic. Maltodextrin is an enzymatically modified starch derivative that is water-soluble, functions as a film-forming polymer and protects active compounds from oxidation while reducing agglomeration during storage. 67 Meanwhile, gum Arabic is a complex heteropolysaccharide with low viscosity and excellent emulsifying properties, making it highly suitable as a primary coating material in spray drying techniques to produce high-quality microparticles. 68
In addition to natural polymers, synthetic polymers are also widely used in microparticle preparation due to their customisable properties. Examples of synthetic polymers include poly(lactic acid) (PLA), poly(glycolic acid) (PGA), poly(lactic-co-glycolic acid) (PLGA), poly(hydroxyalkanoates) (PHA), polycaprolactone (PCL) and various copolymers. These polymers are generally bioresorbable and naturally degrade through enzymatic and nonenzymatic mechanisms, producing biocompatible, non-toxic degradation products that can be eliminated through normal metabolic pathways. 69
PLA offers several advantages, including biodegradability and biocompatibility, making it one of the ideal candidates for controlled drug delivery systems. PLA-based microparticles can regulate drug release over extended periods, with relatively simple production processes such as the emulsion–solvent evaporation method. Their applications have been reported to enhance storage stability and show potential to improve bioavailability. 68 Beyond PLA, the combination of poly(lactic acid) and poly(glycolic acid) produces PLGA copolymers, widely regarded as one of the most promising synthetic polymers in drug delivery. PLGA-based microparticles have been shown to be effective as long-term drug delivery systems due to their ability to maintain morphology, particle size, and drug release profiles in both in vivo and in vitro systems. Another advantage of PLGA is its excellent biodegradability, high biocompatibility, and safety status, which has been recognised by the U.S. Food and Drug Administration (FDA) for use in drug delivery applications. 70
Physicochemical Characterisation of Microparticles
Characterisation is a crucial step in the development of microparticle-based delivery systems, as it determines the quality, stability and effectiveness of the encapsulated active compounds.33,71,72 This process not only ensures the success of the formulation but also explains the relationship between the physicochemical properties of microparticles and their biological performance. The main aspects commonly evaluated include particle size and size distribution, surface morphology, entrapment efficiency, moisture content, release profile and storage stability.73–75 In addition, chemical analysis and interaction studies between the polymer matrix and the active compound—such as through FTIR or DSC—can provide insights into system compatibility. 76 Thus, microparticle characterisation is not merely descriptive but also plays an essential role in ensuring product consistency and predicting the pharmacological performance or bioactivity of the formulated active compound.
The particle size of microparticles is generally measured using particle size analysers, laser diffraction, or dynamic light scattering (DLS).77,78 The resulting size is highly influenced by the preparation method. For instance, spray drying produces particles ranging from 5 to 5,000 µm, coacervation from 2 to 1,200 µm, and solvent evaporation from 0.5 to 1,000 µm. 41 Other factors, such as the core-to-polymer ratio, also affect particle size. 79 Particle size has significant implications for powder flow properties, dosage uniformity, stability of encapsulated compounds, sedimentation rate and patient acceptability.53,80 Particles that are too small may enter the bloodstream and penetrate cells, potentially causing DNA damage, while oversized particles may lead to vascular blockage.
Particle size distribution is commonly evaluated through the polydispersity index (PDI). A low PDI value indicates a narrow size distribution, more homogeneous particles, and a more stable formulation. Conversely, a high PDI value reflects heterogeneity, leading to flocculation. 70 A narrow size distribution also indicates a controlled particle formation process, while smaller particles generally increase surface area and accelerate the release of active compounds. 7
Another important parameter is zeta potential, the electrical charge at the surface of colloidal particles. High zeta potential values (≥±30 mV) indicate strong particle repulsion, preventing flocculation and maintaining dispersion stability. Measurement can be conducted using a particle size analyser.70,79
Particle morphology is typically analysed using Scanning Electron Microscopy (SEM) or Transmission Electron Microscopy (TEM). These techniques allow observation of particle shape (spherical, polygonal, irregular), surface structure (smooth, rough, porous) and detailed microparticle size.41,58,81
%EE describes the percentage of active compounds successfully entrapped within the polymer matrix. Factors such as polymer concentration, inlet air temperature, airflow rate, formulation composition, and preparation method significantly influence %EE.58,82 Higher %EE values indicate greater protective capacity of the polymer toward the active compound. Determination of %EE can be carried out using UV-Vis spectrophotometry, gas chromatography, or HPLC.83,84
Moreover, chemical interactions between the polymer and the active compound can be analysed using Fourier Transform Infrared Spectroscopy (FTIR), which identifies changes in functional groups. 80 Thermal properties can be examined through Differential Scanning Calorimetry (DSC). Shifts or reductions in melting points observed in DSC results indicate changes in thermal stability, reduced lattice energy, and potential improvements in solubility and bioavailability of the active compound. 85
Result and Discussion
Preparation of Flavonoid-loaded Microparticles
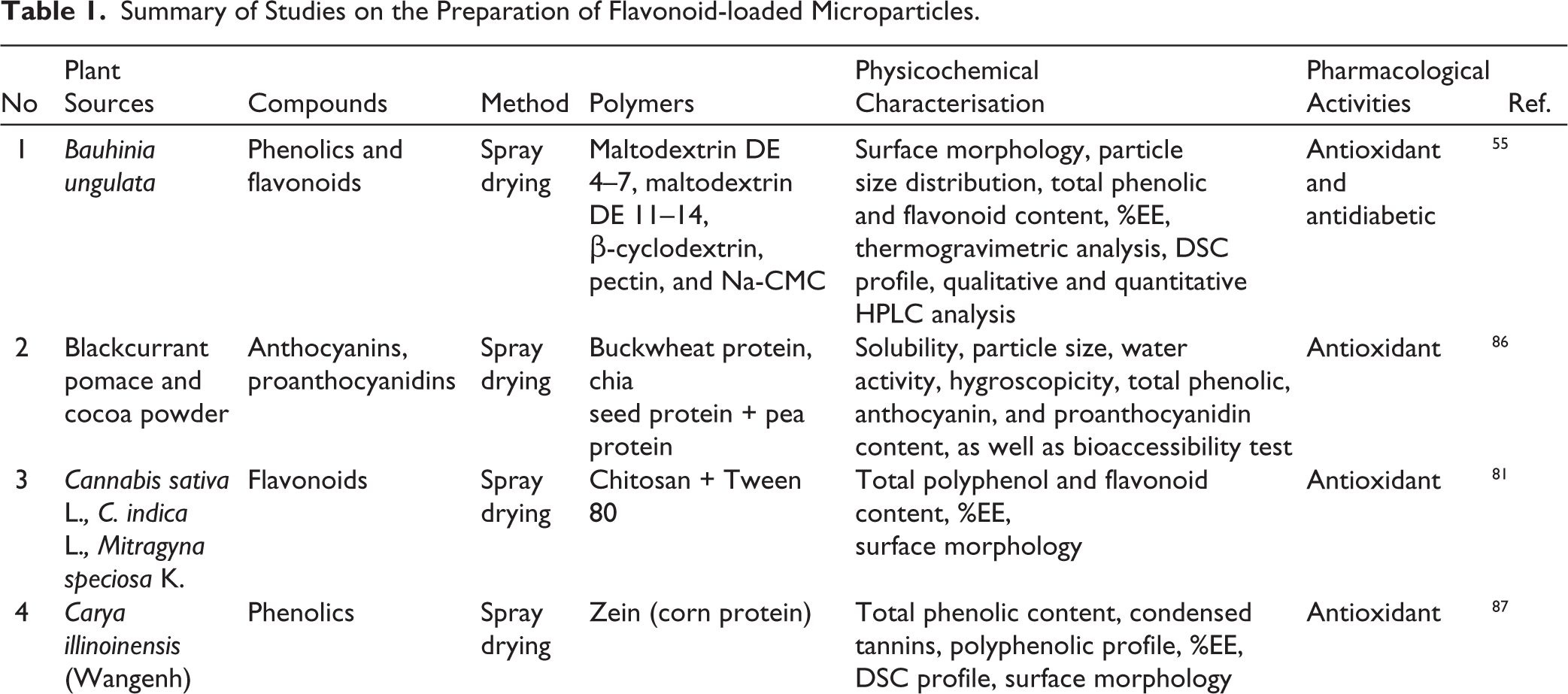
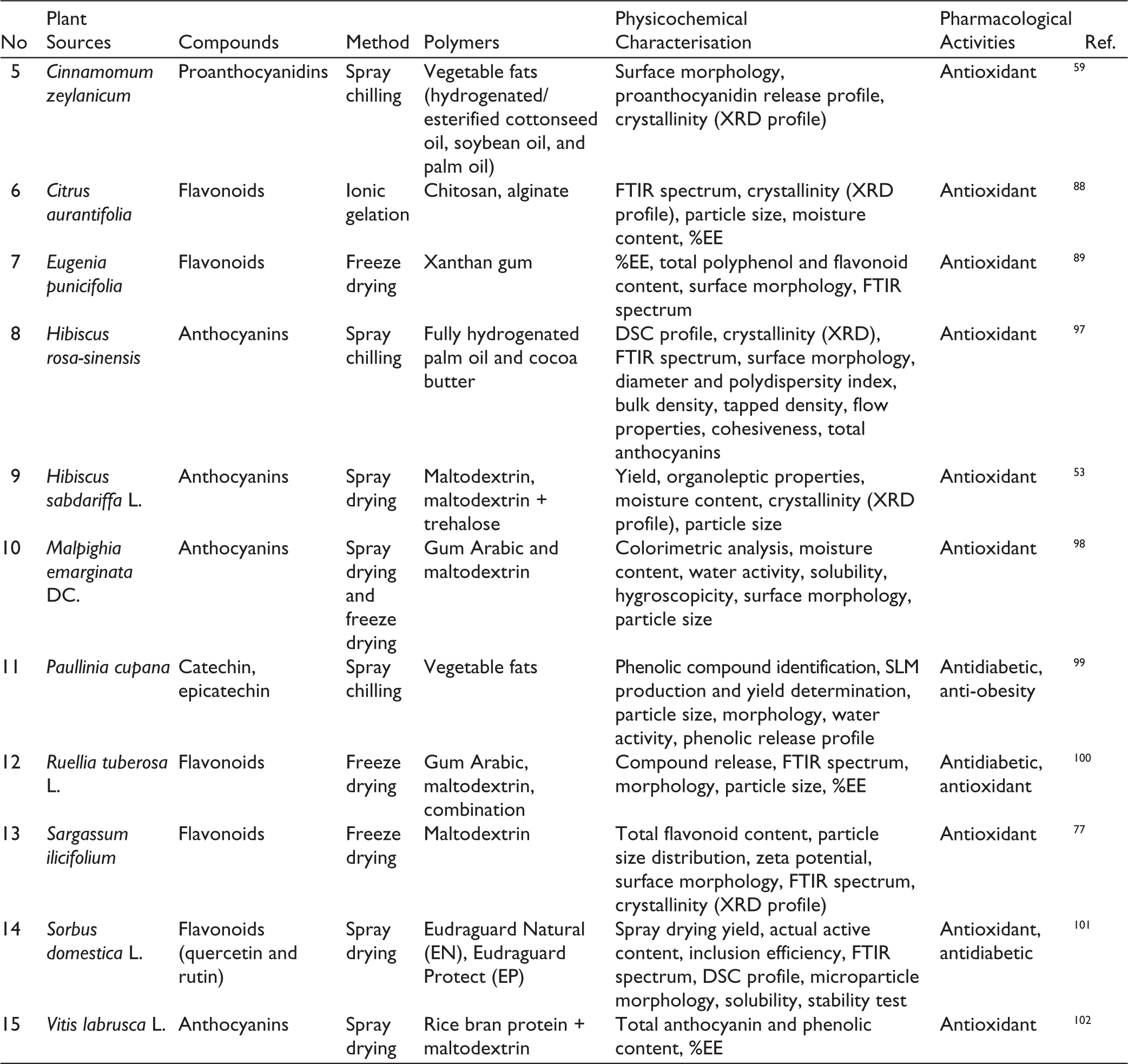
Various studies have explored the preparation of flavonoid-loaded microparticles from diverse plant sources using different methods and polymers. These approaches have been shown to improve key physicochemical properties, including %EE, particle morphology, and storage stability, while maintaining or even enhancing the biological activities of flavonoids, particularly their antioxidant and antidiabetic effects. A summary of representative studies is presented in Table 1, highlighting the potential of microparticle-based systems as an effective strategy for the delivery of flavonoid-rich extracts.
Summary of Studies on the Preparation of Flavonoid-loaded Microparticles.
Discussion
Several articles reviewed in this study demonstrate that microparticles of herbal extracts containing flavonoids can enhance chemical stability, modulate in vitro release profiles, and, in some cases, also improve biological activity. Differences among studies generally lie in the methods and types of polymers used to form the microparticles. Based on the data in Table 1, spray drying is the most widely applied technique for producing herbal extract microparticles containing flavonoids. This method is chosen because it can achieve a relatively high %EE, around 70%–85%. Studies on extracts of
In spray drying studies, the inlet temperature typically ranges between 150°C and 180°C with an outlet temperature of 75°C–80°C. These variations are adjusted according to the thermal properties of the active compounds and the type of polymer used. For example,
Freeze drying is also widely employed for microparticle production. In extracts of
Spray chilling is generally used for lipophilic extracts, such as
On the other hand, ionic gelation remains relatively less applied for herbal-based microparticles. For
Different microencapsulation techniques exhibit varying effectiveness in preserving flavonoid stability and bioactivity. 90 Spray drying is among the most commonly employed methods due to its scalability, cost-effectiveness, and ability to produce relatively uniform microparticles. Several studies have demonstrated that spray drying results in high %EE and satisfactory storage stability. However, the elevated processing temperatures involved may lead to partial degradation of heat-sensitive flavonoids. 91 In contrast, milder techniques such as ionic gelation and freeze drying, which operate at lower temperatures, are more suitable for preserving thermolabile compounds, although they often produce larger particle sizes or require longer processing times. 92 In general, the particle size of flavonoid microparticles varies considerably depending on the preparation method and polymer composition, which in turn influences dissolution behaviour and release profiles. 93 Overall, these findings indicate that the selection of a microencapsulation technique involves a trade-off between %EE, particle characteristics, and dissolution properties, ultimately impacting flavonoid bioactivity.
Overall, natural polymers are the primary choice for flavonoid microparticle production, including gum Arabic, maltodextrin, chitosan, alginate and plant proteins such as zein and chia seed protein. These polymers are selected for their biocompatibility and ability to protect active compounds from degradation. Gum Arabic and maltodextrin are the most frequently used combination, producing particles with high solubility, low hygroscopicity and spherical morphology with high %EE, as shown in
Meanwhile, chitosan and alginate are commonly employed in ionic gelation, as they can crosslink with multivalent ions to form a stable gel matrix. Their combination enhances flavonoid %EE while maintaining antioxidant activity. Lipid-based polymers, such as hydrogenated palm oil or vegetable fats, are also widely used in spray chilling, making them ideal for lipophilic compounds and crucial for ensuring microparticle stability during storage.
The type of polymer plays a critical role in determining the release behaviour and stability of encapsulated flavonoids. Hydrophilic polymers, such as maltodextrin and gum Arabic, enhance solubility and improve microparticle dispersion in aqueous environments. 94 Their high water solubility promotes rapid matrix hydration and swelling, which facilitates faster diffusion of flavonoids and consequently accelerates release, potentially enhancing in vitro antioxidant activity. 93 In contrast, lipid-based matrices commonly used in spray cooling provide stronger protection against oxidative degradation and enable a more sustained release profile, particularly for lipophilic compounds. 60 These matrices form solid lipid barriers that limit water penetration and slow down compound diffusion. Overall, the interplay between polymer properties, encapsulation techniques, and the physicochemical characteristics of the active compounds governs the resulting physicochemical properties and biological performance of flavonoid microparticles, which may be tailored according to the desired release kinetics and target application.
In summary, various microparticle synthesis methods—spray drying, freeze drying, spray chilling, and ionic gelation—have been extensively applied to herbal extracts containing flavonoids. Each technique has advantages and limitations in terms of %EE, chemical stability, release profile, and suitability for different active compounds. Careful selection of the appropriate method and polymer is critical to preserving bioactivity, improving bioavailability, and maintaining or even enhancing biological activity, particularly antioxidant and antidiabetic effects. However, despite numerous reports demonstrating improved physicochemical stability, controlled release profiles, and enhanced in vitro biological activity, evidence supporting enhanced bioavailability in in vivo studies remains limited. Most of the studies included in this review predominantly focus on formulation development and in vitro evaluations, such as antioxidant capacity and enzyme inhibition assays. Therefore, further in vivo studies, particularly those integrating pharmacokinetic and pharmacodynamic assessments, are required to establish whether improvements in physicochemical properties of flavonoid microparticles can translate into enhanced systemic bioavailability and therapeutic efficacy. 95
Nevertheless, several challenges remain, particularly in terms of process optimisation, formulation standardisation, and achieving a deeper understanding of the relationship between the physicochemical properties of microparticles and their biological performance. Future research should therefore focus on the development of novel, more biocompatible and sustainable polymers, as well as the integration of microparticle preparation techniques with green and environmentally friendly technologies. Overall, the development of flavonoid-loaded microparticles holds great promise for creating more effective, stable and applicable natural product delivery systems for pharmaceutical, functional food and herbal health product applications.
From an industrial perspective, scalability and regulatory compliance are critical considerations in the development of flavonoid microparticle systems. Techniques such as spray drying are particularly advantageous for large-scale production due to their continuous processing capabilities and relatively lower operational costs compared to freeze drying. 96 However, industrial-scale manufacturing must comply with Good Manufacturing Practice (GMP) standards to ensure product quality, safety, and batch-to-batch reproducibility. In addition, regulatory approval of phytopharmaceutical or herbal-based microparticle formulations requires comprehensive safety assessments, stability studies, and well-defined quality control parameters prior to commercialisation.
Conclusions
Flavonoids are bioactive compounds with various therapeutic activities, yet their clinical applications are limited due to low bioavailability and susceptibility to environmental degradation. Microparticle technology, including microspheres and microencapsulation based on natural or synthetic polymers, has been proven to enhance the stability, controlled release, and pharmacological effectiveness of flavonoids. Various formulation methods, such as solvent evaporation, ionic gelation, spray drying, freeze drying and spray chilling, can be tailored to the physicochemical properties of the compounds and the types of polymers used. Characterisation of microparticles—including size, distribution, morphology, %EE, zeta potential, as well as thermal and chemical analysis—is crucial to ensure the quality and consistency of the delivery system. Overall, the utilisation of flavonoid-based herbal microparticles offers an innovative strategy to improve bioavailability and preserve the biological activity of flavonoids, thus holding great potential for the development of drugs and health products derived from natural sources. Therefore, future research should focus on developing more sustainable and biocompatible polymer materials, optimising formulation parameters to improve large-scale production reproducibility, and further studying the relationship between the physicochemical properties of microparticles and their in vivo biological performance. Furthermore, further research is needed to ensure the long-term stability, safety, and clinical effectiveness of flavonoid microparticle systems to support their application in pharmaceutical products, functional foods and herbal medicines.
Footnotes
Authors’ Contribution
All authors contributed substantially to the study’s conception and design, data acquisition and/or data analysis and interpretation. They were involved in drafting the manuscript or critically revising it for significant intellectual content, approved its submission to the current journal and gave final approval of the version to be published. Furthermore, all authors agree to be accountable for all aspects of the work and meet the authorship criteria established by the International Committee of Medical Journal Editors (ICMJE).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Data Availability Statement
All the data is available with the authors and shall be provided upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Directorate of Research and Community Service (DPPM), Directorate General of Research and Development, Ministry of Higher Education, Science, and Technology, Indonesia, through the PUI-PT Research and Innovation Ecosystem Enhancement Grant (Contract No. 029/C3/DT.05.00/PL-PUIPT LANJUTAN/2026). The authors also thank Universitas Surabaya for funding through the Master’s Thesis Research Grant (Contract No. 040/SP-Lit/LPPM-01/FF/VIII/2025).
Informed Consent
Not applicable.
Use of Artificial Intelligence-assisted Tools
During the preparation of this work, the authors used ChatGPT (OpenAI) for language editing, paraphrasing, and translation purposes only. After using this tool, the authors reviewed and edited the content as needed and took full responsibility for the final content of the manuscript. No generative AI or AI-assisted technologies were used for idea generation, data analysis, interpretation, or development of scientific conclusions.
