Abstract
Background
Breast carcinoma is a deadly malignancy with a greater risk of morbidity and death, which has contributed significantly to the sharp rise in cancer cases. It has a recognized impartial effect on women. The need for a variety of treatment choices is prompted by the current therapy modules’ lack of effectiveness and adverse effects.
Objectives
In the present investigation, we examined the therapeutic value of Sanggenol L (San L) in human breast carcinoma cells. San L impacts on carcinoma of the breast cells are not fully understood in terms of its molecular and cellular pathways.
Materials and Methods
In order to assess the effectiveness of San L, this investigation employs a number of qualitative approaches, such as cytotoxicity analysis, sulforhodamine B (SRB), 4′,6-diamidino-2-phenylindole (DAPI), reactive oxygen species (ROS), and cell cycle assessment, which were conducted using breast cancer BT-474 cells. Additionally, cell death caused by San L through caspase-dependent and independent apoptosis has been investigated with western blot analysis.
Results and Conclusion
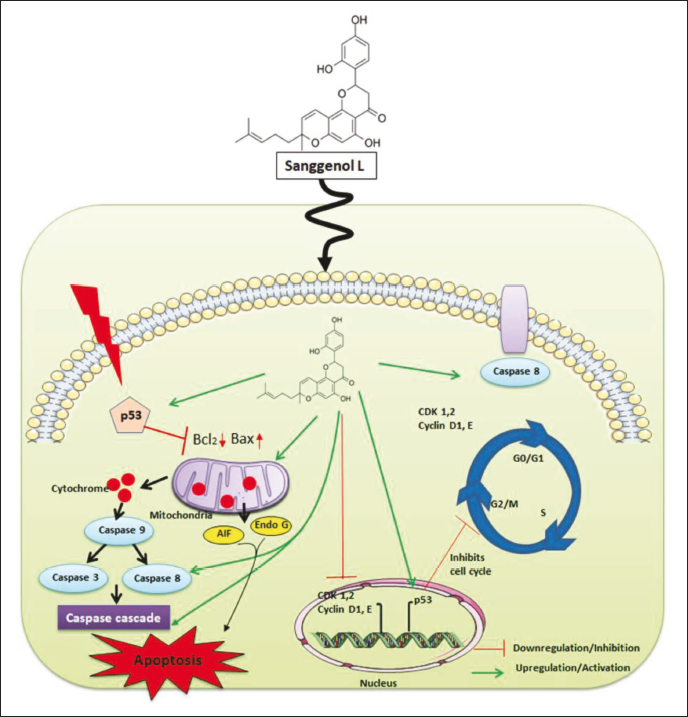
San L therapy significantly diminished the ability of BT-474 breast carcinoma cells to proliferate and impeded colony formation while having no impact on HBL-100 normal breast epithelial cells in a dose- and time-dependent way. San L treatment resulted in cell cycle arrest and cell death in breast carcinoma cells, which were characterized by acridine orange/ethidium bromide (AO/EtBr), DAPI, and flow cytometry at time-dependent way. The present study also explained that the half-maximal inhibitory concentration (IC50) concentration of (21 and 17.3 µM/mL) San L treatment induced caspase-dependent apoptosis [enhanced the expression of Bax and decreased the expression of B-cell lymphoma 2 (Bcl-2), procaspase-3, -8, and -9], stimulation of caspase-independent apoptosis [upregulation of apoptosis-inducing factor (AIF) and endonuclease G (Endo G) on cytosol], and suppression of cell cycle [downstairs regulation of cyclin-dependent kinase 1/2 (CDK1/2), cyclin D1, cyclin E, or upregulation of p53] in breast tumor cells. According to these findings, San L causes both caspase-dependent and independent apoptosis by triggering p53 in breast carcinoma cells.
Introduction
Globally, breast cancer happens to be one of the greatest common and fatal malignancies among women in both industrialized and developing nations (Wang et al., 2020). By 2040, 3,059,829 individuals are expected to be affected by breast carcinoma (Hashemi et al., 2019). About seven million of these instances result in death annually, or roughly 6.9% of all cancer-related deaths (Sung et al., 2021). Each year, over two million new cases are identified with the disease. Geographical location, as well as ethnicity and nationality, all influence the occurrence of breast carcinoma. Breast cancer patients may have a lower chance of survival due to the disease’s fast growth and infiltration of healthy tissues (Fahad Ullah, 2019). Many chemical medicines with serious adverse reactions, including kidney damage, neurotoxicity, and cardiovascular toxicity, in addition to being resistant to drugs, were originally created to combat the spread of breast carcinoma (Alkahtani et al., 2021; Muley et al., 2020; Najjary et al., 2020). Although the treatment of this illness has greatly improved thanks to new drugs, additional research is still required to find substances that may simultaneously target tumor cells and lower the chance of developing cancer. Substances with chemopreventive and chemotherapeutic qualities have traditionally been derived from natural extracts (Neophytou et al., 2020). Natural products are used in the therapy of carcinoma, alongside the goal of lowering drug resistance. They do this by working in concert with anticancer medications to reduce the level of drugs and, as a result, lessen the adverse effects of cancer therapy.
The benefits of accessible collection, lower toxicology, and fewer adverse reactions have made traditional Chinese medicine a popular choice in medical treatment. Plants produce flavonoids, which are known to protect those (Nabavi et al., 2020). Natural flavonoids attack cancer cells in various ways, including cell death, halting the cell cycle, destruction of deoxyribonucleic acid (DNA), and the mitochondrial-targeted. Its potent anticancer effects both in vitro and in vivo have garnered significant interest (Rodríguez-García et al., 2019). The leaves and fruit of Morus alba, commonly referred to as mulberries, have been utilized in traditional medicine for a variety of purposes. These include hyperglycemia and antihypertensive actions, as well as diuretic, antiedemic, hypolipidemic, and anticancer properties (Eo et al., 2014; Park et al., 2020). This is utilized in cooking and as a beverage in addition to being an herbal medicine. Beneficial ingredients within the root of M. alba, such as Sanggenol L, Q, and F, have been shown by current investigations to have anti-inflammatory, antitumor, and antidiabetic properties (Park & Park, 2022; Seo et al., 2015; Song et al., 2021). An earlier study demonstrated that in B-type RAF (BRAF)-mutant mice, the isoprene flavonoid morusin, which is isolated from Morus alba, enhanced the tumor-fighting properties of mitogen-activated protein kinase (MAPK) pathway blockers (Zhao et al., 2022). In reality, morusin has demonstrated potent anticancer properties in a range of cancer types by a number of methods. For example, morusin blocks the protein kinase B (AKT) circuit to prevent the development of human colorectal carcinoma (Zhou et al., 2021). By AMP-activated protein kinase (AMPK)-mediated G1 arrest and antiglycolysis, musolin also had an anticancer impact on liver cancer (Cho et al., 2021). However, research on the physiological and molecular bases associated with the beneficial components is still scarcely revealed for many of the molecules, like Sanggenol L (San L) in Morus alba. San L inhibits nuclear factor kappa B (NF-κB) signaling and activates caspases to provide cytotoxic and apoptotic effects on ovarian cancer (Nam et al., 2016). Prior research has shown that San L, derived from Morus alba, stimulated melanoma cell death (Won & Seo, 2020b). Apoptosis is a recognized typical molecular and cellular phenomenon associated with natural chemicals in a variety of malignancies (Cho et al., 2018). Caspases are frequently involved in apoptosis, which is another name for programmed cell death. Apoptotic cell death can be the ultimate outcome of an apoptotic-signaling pathway triggered by either external or internal stimuli, such as caspases (Obeng, 2020). Furthermore, the apoptosis-inducing factor (AIF) caspase-independent mechanism of apoptosis involves the dissolution of AIF from mitochondria. AIF is released as a result of mitochondrial permeation, which aids in the destruction of DNA following the process of apoptotic cell death (Apostolova, 2008). AIF has been linked to caspase-independent cell death (CICD) in a variety of cancer cell categories, according to earlier research (Won & Seo, 2020b). It remains to be investigated, yet, whether San L causes apoptotic in breast cancer cells through caspase-dependent or independent mechanisms. Furthermore, the San L apoptotic mechanism in human breast cells produced from primary malignant tumors has not been reported.
In the present study, we examined how caspase-dependent or caspase-independent mechanisms are used by San L to cause cytotoxicity and apoptosis in carcinoma cells. The current investigation is the first to demonstrate that San L can activate the tumor suppressor p53, thereby triggering apoptosis and cell cycle arrest in human breast cancer cells (Scheme 1). An additional understanding of San L mechanism’s effectiveness in breast carcinoma may open up new avenues for treatment and chemopreventive strategies.
Pictorial Representation Showing Antiapoptotic Effect of Sanggenol L’s in Breast Cancer Cells.
Materials and Methods
Cell Lines Culture and Maintain
Human breast carcinoma cells BT-474 and HBL-100, a human breast epithelial cell line, were obtained via the American Type Culture Collection (ATCC, Rockville, ND, USA) and cultivated in Roswell Park Memorial Institute (RPMI) 1,640 medium and Dulbecco’s modified eagle’s medium (DMEM) nourished with 10% fetal bovine serum (FBS), penicillin (100 IU/mL), and streptomycin (100 µg/mL). The incubator was maintained at 37°C in a humid environment with 5% CO2 in it.
Cellular Viability
After being treated with San L, cell viability was shown using Skehan et al.’s technique (1990). BT-474 cells and normal HBL-100 cells were introduced onto 48-well plates at a density of 5 × 104 cells/well, and they were exposed for 24 and 48 h with different doses of San L (0–35 µM). Following treatment, 10% trichloroacetic acid was added at 4°C after the medium was evacuated. The plate was cleaned with distilled H2O and allowed to air dry for 1 h. At ambient temperature, 0.4% (w/v) sulforhodamine B (SRB) was used to stain the cells, and 1% acetic acid was used to rinse them. 10 mM Tris (pH 10.5) was used to solubilize the bound SRB, and a microplate reader was used to detect the absorbance at 540 pixels. The half-maximal inhibitory concentration (IC50) of San L was further investigated using a concentration (21 and 17.3 µM/mL) and time (24 and 48 h) dependent manner.
The SRB test was also used to assess the impact of the caspase inhibitor carbobenzoxy-valyl-alanyl-aspartyl-[O-methyl]-fluoromethylketone (z-VAD-FMK) and the AIF inhibitor N-phenylmaleimide (N-PM) on cell survival. BT-474 cells were cultivated in RPMI 1,640 media for 24 h after being inserted at a density of 5 × 104 cells per well in a 48-well plate. Following a 3-hour preincubation period with 10 µM z-VAD-FMK or 5 µM N-PM, the cells were exposed to several doses of San L (0–35 µM) for 24 and 48 h. As mentioned above, SRB tests were carried out.
Assay for Colony Development In Vitro
Following the San L administration, colony formation was calculated using Franken et al.’s (2006) approach. 48-well plates were provided with BT-474 cells at a mass of 1 × 104 cells per well, and the cells were treated with the IC50 concentration of San L for 24 and 48 h. The medium was extracted and washed with phosphate-buffered saline (PBS) buffer after treatment. At ambient temperature, the combination of 0.5% crystal violet and 6.0% glutaraldehyde was then administered. The plates were subsequently air-dried and cleaned with distilled water after 30 min. The ImageJ software was used to quantify the capacity for colony creation.
Cellular Reactive Oxygen Species (ROS) Measurement
ROS within cells were enumerated by the fluorescent probe 2′,7′-dichlorofluorescein diacetate (DCFH-DA). BT-474 cells were seeded into a 24-well plate at a density of 1 × 105 cells per well. The cells were cultivated for 24 and 48 h, correspondingly, either with or without IC50 dosages of San L (21 and 17.3 µM/mL). After treatment had persisted for 24 and 48 h, the cells were permitted to incubate for 30 min at 37°C in the dark in a serum-free medium containing 10 µM DCFH-DA. After being trypsinized twice and placed back into the PBS solution, they were immediately subjected to a flow cytometric analysis. A 530-nm band-pass filter was used to extract fluorescence signals for 2′,7′-dichlorodihydrofluorescein (DCF). Every calculation depends on 10,000 cells’ worth of average luminescence intensity. The cells were cultured in the absence of DCFH-DA in bare controls (Liu et al., 2020).
Cell Apoptosis Assay by Flow Cytometry
The measurement of programmed cell death using Annexin V staining was carried out by following the manufacturer’s instructions and using Muse Annexin V and the dead cell reagent. In 6-well plates, BT-474 cells were placed at a count of 5 × 104 cells per well and cultivated for a duration of 24–48 h. Following a 24- and 48-hour San L treatment, the cells were washed twice with 0.001% FBS-PBS buffer. Each microcentrifuge tube was then filled with 0.1 mL of reagent and 0.01 mL of cell suspension, and the tubes were allowed to incubate for 20 min at ambient temperature. The Muse cell analyzer [Merck Kommanditgesellschaft auf Aktien (KGaA), Darmstadt, Germany] was then used to examine the cells. The overall average amount of fluorescence in every single one of the four quadrants was used to compute the cell percentages, and 5,000 events (gated cells) per experiment were used to collect flow cytometry data.
Assessing for Hoechst Staining
The specific morphological alterations associated with apoptosis were evaluated using fluorescence microscopy and the Hoechst staining technique. After being seeded at a concentration of 5 × 104 cells per well in 6-well plates, BT-474 cells were subjected to a 24 and 48-hour San L treatment. Following harvesting, cells were subjected to two rounds of washing in 0.001% FBS-PBS buffer before being stained for 10 min at ambient temperature using 200 µL of Hoechst 33,258 (5 µg/mL). After that, a cover slip was applied to 10 µL of this solution on a glass slide. Finally, nuclei disintegration and condensed chromatin were assessed in cells using a fluorescence microscope (Olympus Optical Co. Ltd. Japan).
Western Blot Analysis
Following a 24-hour growth period, 5 × 104 BT-474 cells were planted per well. For 24 and 48 h, cells were treated with San L at its IC50 concentration. Following a 30-minute ice-soaked lysis of the cells in a lysis solution, the protein attentiveness was ascertained using a bicinchoninic acid (BCA) protein kit (Pierce, Rockford, IL, USA). 8%–12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) was used to separate the samples for 90 min at a constant voltage of 100 V per slab. The membranes were blocked with 2.5% of bovine serum albumin (BSA) and then treated with the primary antibody for an entire overnight at 4°C. Lastly, secondary antibodies were applied to the membranes for 1 h at 4°C. An enhanced chemiluminescence (ECL) kit (Santa Cruz Biotechnology, Santa Cruz, CA) was used to show the presence of every protein (Das et al., 2020).
Evaluation of Statistics
As a proportion to control, the outcomes were presented. The values of the statistics were reported as the mean ± standard deviation of three separate measurements. In order to examine differences, Dunnett’s test or Student’s t-test was used in conjunction with one-way analysis of variance (ANOVA). Prism (GraphPad, La Jolla, CA, USA) was the program used for the analysis. **p < 0.01, ***p < 0.001, and *p < 0.05 indicate statistical implications.
Results
Impact of San L on Cell Proliferation of BT-474 Cells
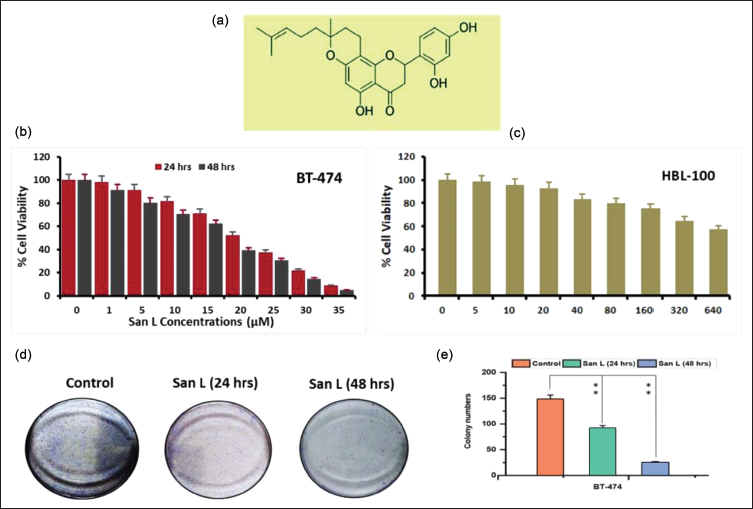
The chemical structure of San L is depicted in Figure 1a. A variety of San L doses (0–35 µM) were applied to human breast cancer cells for 24 and 48 h, after which they were subjected to an SRB test evaluation (Figure 1b). The growth of BT-474 was significantly suppressed by San L in a concentration- and time-based manner, as shown in Figure 1b. BT-474 breast tumor cells showed an IC50 value of San L of 21 and 17.3 µM/mL at 24 and 48 h, respectively. Yet, at lower concentrations, San L’s influence on the proliferation of normal cell lines (HBL-100) did not show any discernible cytotoxicity; however, at 320 and 640 µM/mL, cytotoxicity did somewhat rise (Figure 1c). Our findings unambiguously show that San L has a cytotoxic impact on cancer cell lines but not on normal cell lines. According to the results of the SRB test, San L reduced the vitality of BT-474 cells in a dose (21 and 17.3 µM/mL) and time-dependent manner at 24 and 48 h, respectively. It is evident from these results that the administration of San L has a significant suppressive effect on the proliferation of BT-474 breast carcinoma cells.
San L’s Atomic Structure (a) San L Prevents the Propagation of Human Breast Carcinoma (BT-474) Cell Line. (b) Following Pre-treatment with San L for 24 and 48 h, the Viability of BT-474 Cells were Assessed. (c) The SRB Test was Used to Evaluate Cell Viability of HBL-100 Normal Cell Line. (d) The Impact of San L on BT-474 Cells’ Ability to Form Colonies. (d) San L Prevented Breast Cancer Cells from Forming Colonies for a Period of Both 24 and 48 h Respectively. (e) Capabilities for Colony Development were Measured. The Mean ± SD of Three Separate Determinations was Used to Express the Data Values. One-way ANOVA was Used to Evaluate Significant Differences with the Control at **p < 0.05.
San L Effects on the Ability to Establish Colonies
A clonogenic test was used to assess BT-474 breast cancer cells after they were treated via IC50 concentration of San L for 24 and 48 h, accordingly. Our findings demonstrate that San L markedly reduced the BT-474 cells’ ability to form colonies (Figure 1d). In a time-dependent way and at their inhibitory dose, San L can dramatically lower the viability along with the proliferation of BT-474 cells. After the BT-474 cells under investigation, the IC50 concentrations (21 and 17.3 µM/mL) of San L were generally greatly lowered; after 24 and 48 h, respectively, cell viability decreased from 25% to 35% (Figure 1e). According to these findings, treatment with San L considerably abridged the capability of carcinoma cells from humans to form colonies.
San L Improved Intracellular ROS Levels
In the treatment of cancer, ROS can be a useful therapeutic tool due to the cellular harm that arises from its accumulation. To determine if San L performs a similar role in cancer cells, we searched for ROS alterations in breast cancer cells. The results were confirmed by flow cytometry, and increased ROS levels were detected using 2′,7′-dichlorodihydrofluorescein diacetate (DCFDA), a cell-permeable fluorescent probe.
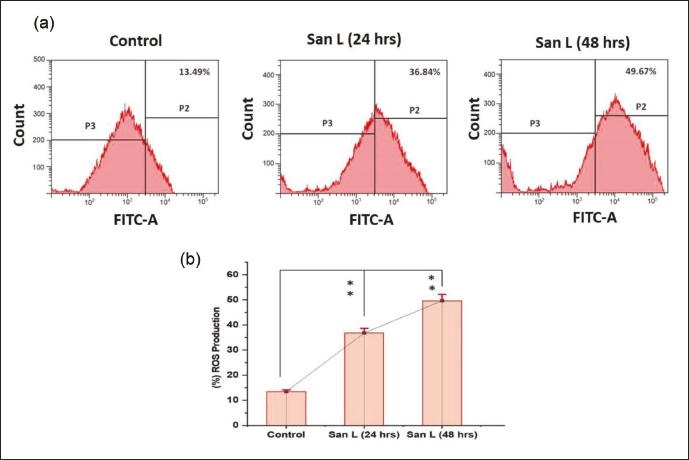
BT-474 cells were treated to San L at IC50 doses for 24 and 48 h afterward, with the goal of investigating the accumulation of internal ROS levels. Following DCFDA staining, the cells were examined using flow cytometry. The results of the flow cytometry demonstrated that San L (21 and 17.3 µM/mL) markedly raised the levels of ROS in an orderly dose and time-dependent manner. A discernible increase in ROS generation was seen when cells treated with San L were matched with untreated cells (Figure 2a). The flow cytometry histogram showed that the accumulation of ROS in the treated cells was much higher than in the control cells after 48 h of treatment (Figure 2b). According to these results, natural San L in BT-474 cells may cause intracellular ROS production.
(a) Measurement of Reactive Oxygen Species (ROS) Generation in Control Cells Using Flow Cytometry, and BT-474 Cells Treated with San L for 24, and 48 h after Treatment at an IC50 concentration. (b) A Sample 2′,7′-Dichlorodihydrofluorescein Diacetate (DCFDA) Staining Histogram of Cells Treated with San L. The Mean ± SD for the Three Experiments is Shown in Columns. **p < 0.05 Relative to the Control Group.
Impacts of San L on Apoptosis of BT-474 Cells
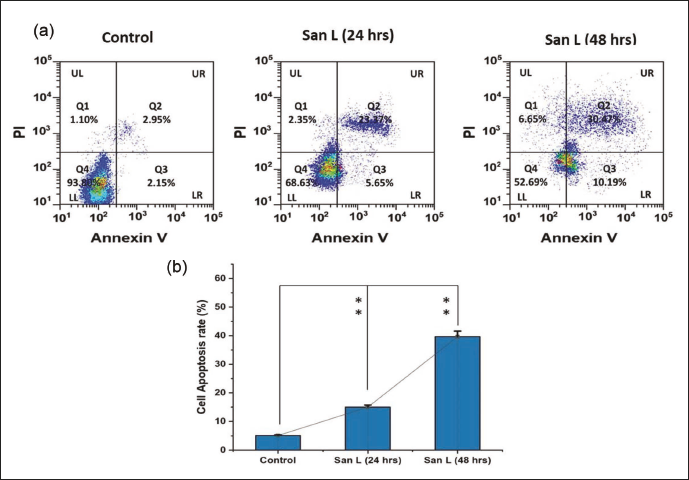
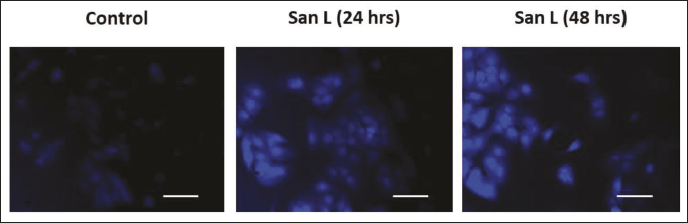
Using Annexin V and Hoechst 33,258 staining, we investigated if San L causes apoptosis in human breast carcinoma BT-474 cells (Figures 3 and 4). An IC50 value of San L was applied or not to BT-474 cells for a duration of 24 and 48 h. Figure 3a and b of the study report that the San L therapy markedly increased the accumulation of apoptotic cells. When the IC50 concentration of San L was added to BT-474 cells, the percentage of early apoptosis increased from 2.95% in the control (untreated cells) to 23.37% and 30.47% in the treated cells (p < 0.05) after 24 and 48 h of treatment, as shown in Figure 3. Furthermore, according to Figure 4, San L administration for 24 and 48 h elevated apoptotic biological markers such as cell death and nuclear condensation. These findings unequivocally demonstrate that San L causes BT-474 carcinoma cells to undergo apoptosis.
(a) Measurement of Reactive Oxygen Species (ROS) Generation in Control Cells Using Flow Cytometry, and BT-474 Cells Treated with San L for 24, and 48 h after Treatment at an IC50 concentration. (b) A Sample 2′,7′-Dichlorodihydrofluorescein Diacetate (DCFDA) Staining Histogram of Cells Treated with San L. The Mean ± SD for the Three Experiments is Shown in Columns. **p < 0.05 Relative to the Control Group.
The Hoechst Staining Strategy was Used to Identify Nuclear Shrinkage in Reactions Following Sanggenol L (San L) Administration.
Impact of San L with the Stimulation of BT-474 Cells’ Caspase-mediated Apoptosis
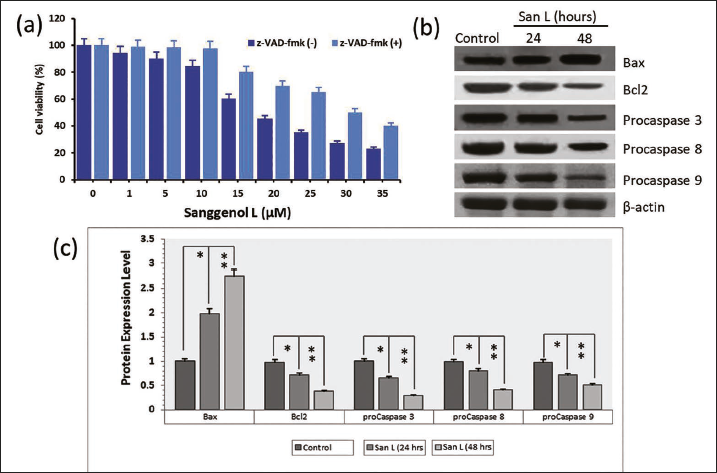
The present study investigated the potential correlation between San L administration and the caspase-mediated apoptosis pathway in BT-474 carcinoma cells (Figure 5). In order to confirm how San L triggers apoptosis through caspase-associated mechanisms, cells were exposed to the ubiquitous caspase inhibitor z-VAD-FMK at a dosage of 10 µM for 3 h, and subsequently, they were cultured with different San L doses (0–35 µM/mL) for 48 h, respectively (Figure 5a). The use of a caspase inhibitor substantially lowered the amount of cell death that San L caused in BT-474 cells. Additionally, the current investigation employed western blotting to investigate the expression of caspase-related protein molecules, which have been quantified (Figure 5b and c). As could be seen in Figure 5b and c, San L therapy considerably diminished the expression of procaspases-3, -8, and -9, along with B-cell lymphoma 2 (Bcl-2). Additionally, the Bax protein activity was elevated in BT-474 cells. The findings presented here indicate that San L promoted apoptosis through a caspase enzyme cascade-dependent system in BT-474 cells.
(a) Using the SRB Test, the Impact of the Caspase Inhibitor Carbobenzoxy-Valyl-Alanyl-Aspartyl-[OMethyl]-Fluoromethylketone (z-VAD-fmk) was Determined. (b) To Determine the Levels of Caspase-Dependent Apoptotic Protein Expression Using Western Blot Analysis. (c) The Amount of Whole Apoptotic Proteins was Measured, and the Data are Shown as Mean Value ± SE. Mean Significant Difference (*p < 0.05, **p < 0.01) Compared with the Control. In Western Blots, β-actin is Typically Utilized as an Internal Control.
San L Influence on AIF Expression in BT-474 Cells
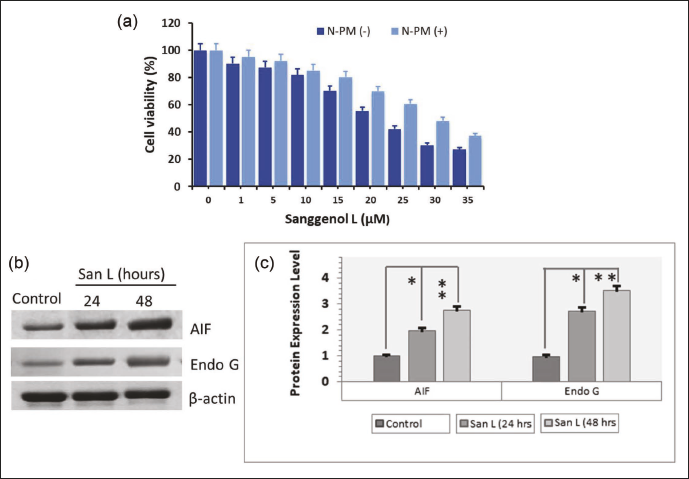
Two recognized effectors linked to the triggering of apoptosis through a caspase-independent mechanism are AIF and endonuclease G (Endo G). BT-474 cells were stimulated with or without a 5 µM N-PM inhibitor for 3 h, followed by being treated with different dosages (0–35 µM/mL) of San L for 48 h (Figure 6a) to examine the involvement of AIF and Endo G induction in San L persuaded a cell death. Treatment with San L produced apoptotic cell death, which was greatly inhibited by the N-PM inhibitor. Additionally, we used western blotting to ascertain whether San L could control the amounts of produced AIF and Endo G in BT-474 cells (Figure 6b). The software application ImageJ was used to quantify the quantities of AIF and Endo G proteins (Figure 6c). During San L therapy in BT-474 cells, AIF and Endo G levels rose considerably, as seen in Figure 6b and c. These results imply that in BT-474 cells, San L-triggered apoptotic cellular death is in addition associated with a caspase-independent mechanism.
(a) The SRB Test was Used to Quantify the Impact of N-PM, an AIF Inhibitor. Cells had a 3 h Pretreatment with 5 µM N-PM, Followed by a 24 and 48 h Incubation Period with San L. (b) The Amounts of Endo G and Apoptosis-inducing Factor (AIF) Proteins in Tumor Cells Bt-474. Western Blot Analysis was Employed to Assess the Altitudes of Caspase-independent Apoptotic Protein Expression in Whole Cell Lysates. Cumulative Apoptotic Proteins were Measured (c), and the Results were Presented as the Mean Standard Deviation of three Independent Measurements. The Data are Shown as Mean Value ± SE. Mean Significant Difference (*p < 0.05, **p < 0.01) Compared with the Control. In Western Blots, β-actin is Typically Utilized as an Internal Control.
San L Causes Cell Cycle Arrest in BT-474 Cells
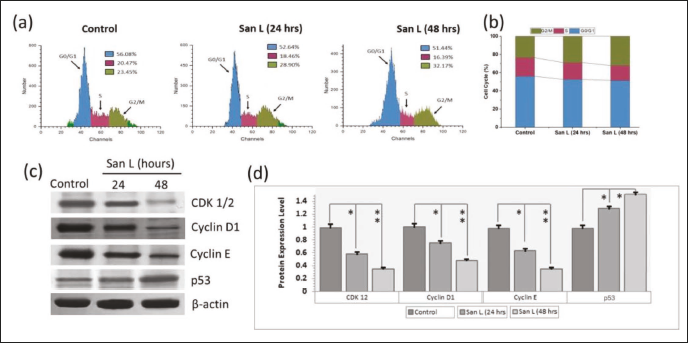
The purpose of this study was to determine how well San L inhibits the cell cycle. Using the Muse cell cycle kit, we investigated the suppression of cell cycle progress in BT-474 cells following San L administration in order to proceed to study whether San L promotes cell cycle arrest in breast carcinoma cells (Figure 7). San L halted BT-474 cells in the G2/M phase of the cell cycle, as seen in Figure 7a and b. While San L administration had no effect on the remaining phases (G0/G1 and S) of the cell cycle, it did boost the G2/M phase in BT-474 cells relative to control cells in a dosage- and influenced by time pattern (Figure 7a and b). Additionally, we assessed the amounts of proteins linked to the cell cycle in BT-474 cells after treating them for 24 and 48 h at the IC50 quantity (21 and 17.3 µM/mL) of San L. In response to San L treatment, p53 proteins were upregulated in a dose- and time-dependent mode while cyclin-dependent kinase 1/2 (CDK1/2), cyclin D1, and cyclin E, were downregulated (Figure 7c and d). These findings show that San L, particularly in the G2/M phase, can stop the BT-474 cells’ ability to proliferate by causing cell cycle arrest.
The Cell Cycle Arrest is Provoked in BT-474 Cells by San L. (a) The Growth of the Cell Cycle was Investigated after Treating Cells with San L at an IC50 Frequency for 24 h and 48 h, Respectively. (b) The Cell Distribution at Different Cell Cycle Stages was Enumerated and the Results were Reported as the Mean Standard Deviation of Triplicate Assays. (c and d) The Amounts of Tumor Suppress Proteins and the Proteins Linked to the Cell Cycle in BT-474 Cells Using Western Blot Analysis. The Housekeeping Gene β-Actin Served as a Denominator in Order to Measure the Relative Amounts of Gene Expression. The Data are Shown as Mean Value ± SE. Mean Significant Difference (*p < 0.05, **p < 0.01) Compared with the Control.
Discussion
It is necessary to research the anticancer properties of natural resources, particularly those obtained from natural sources, in order to develop effective therapies for cancer. Morus alba constituents include Albanol B, Mulberrofuran G, Sanggenol L, Sanggenol F, and Sanggenol Q, which exhibit antioxidant, antidiabetic, and antitumor properties (Sadaf et al., 2021; Won & Seo, 2020b; Zhu et al., 2017). Subsequent research revealed that Morus alba is rich in flavonoids that have anticancer abilities (Li et al., 2021; Park et al., 2021). Researchers discovered that morusinol, an isoprene flavonoid isolated from the bark of the Morus alba root, had a strong inhibitory impact on cancer cells based on earlier drug screening utilizing a Morus alba chemical dataset (Guo et al., 2023). On the other hand, nothing is known about the molecular and cellular processes of Morus alba compounds. Strong antiviral activity was demonstrated by leachianone G, which has a structure resembling San L (Wei et al., 2016). According to Jung et al. (2015), San L is one of the elements of Morus alba that has been shown to be efficient. The substance has been linked to medicinal properties, including cytotoxic and neuroprotective properties. It has also been observed that San L causes ovarian cancer cells to undergo apoptosis by activating caspase and inhibiting NF-κB signaling (Nam et al., 2016). San L (Figure 1a) ability to cause cell death in breast carcinoma cells is yet unknown, nevertheless. The current work demonstrated that via activating the caspase cascade and AIF, San L therapy had significant cell proliferation inhibitory impacts on BT-474 breast carcinoma cells (Figure 1b). Compared to BT-474, a normal breast cell line, San L treatment showed no influence on the proliferation of HBL-100 cells. It indicates that utilizing higher doses of San L did not harm HBL-100 cells (Figure 1c). Since its development, the colony-forming experiment continues to be employed to examine the viability of cells in vitro, including cancer and epithelial cells, as well as their ability to survive and grow (Franken et al., 2006). The BT-474 cells’ ability to form colonies was dramatically reduced by San L exposure based on the dose and time-dependent mechanism (Figure 1d and e).
ROS contributes to tumorigenesis in two ways (Huang et al., 2021). On the one hand, ROS can encourage protumorigenic indicating, which helps cancer cells proliferate, survive, and adapt to low oxygen levels. On the other hand, ROS can cause oxidative stress-induced tumor cell death and support antitumorigenic signaling. Because of their elevated metabolism and malfunctioning mitochondria, cancer cells are more susceptible to oxidative stress and have higher ROS levels than normal cells (Arfin et al., 2021). Therefore, oxidative stress-related cell death can occur when ROS gradually rises over an acceptable level that is unsuitable for cellular viability. It has been demonstrated that a number of natural compounds that raise cellular ROS levels focus on tumor cells. For instance, plant-based phytochemicals such as flavonoids, polyphenols, and stilbenes can cause autophagy and death in cancer cells, as well as decrease their proliferation (Luo et al., 2022; Mitra & Bhattacharya, 2020). For instance, piperine, the most prevalent alkaloid in Piper longum, induces cell cycle arrest at G2/M and death through the generation of ROS, which reduces tumor development both in vitro and in vivo (Benayad et al., 2023; Kung et al., 2021; Mitra & Bhattacharya, 2020). Through generating mitochondrial ROS generation, we were able to demonstrate in the current work that San L boosted ROS production (Figure 2a and b) and triggered both apoptosis and cell cycle arrest at G2/M, suggesting that San-L has an antiproliferative impact in human breast cancer cells.
Programmable cell death, or apoptosis, is characterized by certain morphological traits like chromatin condensation, DNA fragmentation, retention of cells beginning during the cell cycle’s sub-G1 stage, and the creation of apoptotic bodies. Subsequently, it is also linked to the control of caspase cascades (Elmore, 2007). Numerous natural compounds have been demonstrated to have anticancer characteristics according to their demonstrated ability to induce apoptosis in different categories of tumor cells (Cho et al., 2018; Won & Seo, 2020a). In this work, using Annexin V labeling, we further investigated whether San L causes apoptosis in human prostate cancer-3 (PC-3) cells. BT-474 cells treated with San L at an IC50 concentration for 24 and 48 h showed a significant rise in apoptotic cell accumulations (Figure 3a and b). Furthermore, treatment with San L enhanced the number of BT-474 cells’ apoptotic biological signatures, such as apoptotic bodies and nucleus shrinkage in the cells. The current results imply that San L dose- and time-dependently promotes apoptosis in BT-474 cells (Figure 4).
The caspase cascade process, which is primarily engaged in apoptotic cell death, is separated into initiator caspases (caspases-2, -8, -10, and intrinsic route; caspase-9) and effector caspases (caspases-3, -6, -7) (McIlwain et al., 2013). Apoptosis-related proteins, sometimes referred to as Bcl-2 members and caspase functions are responsible for controlling apoptosis (Singh et al., 2019). In the present work, San L therapy boosted the actions of Bax in BT-474 cells and drastically decreased the expression of procaspases and Bcl-2. Additionally, we demonstrated that z-VAD-FMK, a caspase inhibitor, significantly repressed San L provoked cell death in BT-474 as contrasted with cells that were not treated with z-VAD-FMK (Figure 5).
Both the caspase-independent and caspase-dependent pathways’ maintenance and apoptosis are mutually related. According to Sun et al. (2016), AIF and Endo G are constituents of the CICD pathway that are free from mitochondria into the cytosol. According to a number of additional investigations, mitochondrial membrane permeation following apoptotic cell death causes DNA destruction, the expulsion of AIF and Endo G, and a reduction in mitochondrial activity (Cho et al., 2018; Shelar et al., 2015). In this work, San L administration raised the levels of Endo G and AIF in BT-474 cells following both 24 and 48 h of treatment. Furthermore, in BT-474 cells, San L-stimulated apoptotic death of cells was significantly reduced by the AIF inhibitor “N-PM.” The current results imply that San L can cause BT-474 cells to undergo apoptosis via a caspase-independent mechanism (Figure 6) in a time-dependent and concentration-related approach.
Human malignancies frequently exhibit cell cycle dysregulation and cancer cells multiply because they lack a typical cell cycle regulatory mechanism (Malumbres & Barbacid, 2009). According to Peyressatre et al. (2015), cyclin-dependent kinase (CDK)/Cyclins are a family of kinases that play significant functions in the control of cell cycle development, along with erstwhile important biological functions and also state that these are regulators of distinct cell cycle parts.
In the current investigation, BT-474 cells’ G2/M phase was suppressed by San L. While the numbers of cells in other phases have been unaltered by San L therapies, G2/M phase aggregation in BT-474 cells increased in a dose-related and influenced by time mode (Figure 7a and b). Furthermore, suppression of CDK1/2, cyclin D1, and cyclin E occurred in a dosage- and intermittent manner following San L administration. Ultimately, the cell cycle-related proteins were reduced after treatment with San L (Figure 7c and d), even though the cell cycle evaluation only found G2/M phase arrest. Moreover, the administration of San L led to an increase in the expression of the p53 protein, which is linked to the cell cycle and tumor suppression. The primary role of the p53 protein is that of a transcription factor, which controls many different processes, including cell cycle arrest, DNA repair, cell death, autophagy, and metabolic processes (Feroz & Sheikh, 2020; Wang et al., 2023). Furthermore, CDKs and cyclin B, which are necessary for mitotic entry and are implicated in the G2/M phase block are repressed by p53 (Engeland, 2022; Fischer et al., 2016). According to Wang et al. (2023), p53 suppresses cell cycle protein-dependent kinase and promotes transcription of 14-3-3 sigma. These findings imply that San L can repress BT-474 cell development by initiating cell cycle arrest, particularly arrest in the G2/M phase and p53 activation. Our results agree with Merlin et al. (2021) and Won and Seo (2020b). These findings show that the IC50 dosages (21 and 17.3 µM/mL) of San L can stop the cell cycle, hence preventing BT-474 cells from proliferating. This is the first report to reveal that San L suppresses the growth of breast cancer cells and induces caspase-dependent and caspase-independent apoptosis and cell cycle arrest in the G2/M phase.
Conclusion
In summary, this investigation is the initial to show that the San L may promote cell death in breast carcinoma BT-474 cells by activating the p53, which in turn activates the caspase cascade pathway, the AIF signaling pathway, and halts the cell cycle in time and dose-based manner. Thus, we propose that San L-induced apoptosis of tumor cells may be an essential aspect of the cancer prevention approach. San L is a potentially effective chemotherapeutic drug that can be used to suppress breast carcinoma. Additional research is needed to determine how San L therapy is beneficial in preventing carcinoma of the breast, specifically in vivo animal experiments.
Abbreviations
AIF: Apoptosis-inducing factor; ANOVA: Analysis of variance; ATCC: American Type Culture Collection; BCA: Bicinchoninic acid; BSA: Bovine serum albumin; CICD: Caspase-independent cell death; CDK: Cyclin-dependent kinase; DCFH-DA: 2′,7′-Dichlorofluorescein diacetate; DMEM: Dulbecco’s modified eagle’s medium; DAPI: 4′,6-Diamidino-2-phenylindole; ECL: Enhanced chemiluminescence; Endo G: Endonuclease G; FBS: Fetal bovine serum; IC50: Half-maximal inhibitory concentration; N-PM: N-phenylmaleimide; PBS: Phosphate-buffered saline; PLGA: Poly lactic-co-glycolic acid; ROS: Reactive oxygen species; RPMI: Roswell Park Memorial Institute; SRB: Sulforhodamine B; z-VAD-FMK: Benzyloxycarbonyl-Val-Ala-Asp(O-Me)-fluoromethylketone.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
Science and Technology Department of Hubei Province(2022CFD065), Project name: Study of isomherin MTDH promoting breast cancer metastasis through epithelial stromal transformation. Huangshi City Central Hospital(ZX2023Q17), Project name: Epidemiological investigation and analysis of female breast cancer in Yellowstone area.
