Abstract
Background
Hepatocellular carcinoma (HCC) is the third principal reason for tumor-linked fatalities globally. There is no effective cure for HCC due to rapid progression and poor prognosis. Rosmanol (RM) is a phenolic diterpene antioxidant that exhibits anti-inflammatory and anticancer properties.
Objectives
This research aims to examine the chemopreventive efficacy of RM on the PI3K/Akt pathway as a potential mechanism for its protective effect against N-diethylnitrosamine (DEN)-induced HCC.
Materials and Methods
Wistar albino male rats were separated into four sets: normal control (NC); DEN-induced HCC model; DEN+RM (40 mg/kg bw); and RM (40 mg/kg bw) alone. To evaluate the chemopreventive mechanism, we assessed the change in body weight, liver weight, hepatotoxicity markers, antioxidant enzymes, lipid peroxidation, proliferation proteins, cytokines, histopathology, and western blot.
Results and Conclusion
Our data revealed that RM could reduce the loss of body weight, liver weight, toxicity enzymes, oxidative stress, inflammation, and restructuring of the hepatocellular architecture against DEN-stimulated HCC rats. RM-alone treated rats exhibited comparable activity with NC. Furthermore, RM attenuates PCNA and cyclin-D1 protein expression by the regulation of the PI3K/Akt pathway. The possible chemopreventive activities of RM may be owing to its antioxidative, anti-inflammatory, and antiproliferative effects. Thus, RM may aid as an innovative remedial agent for HCC treatment.
Introduction
Liver cancer (LC) is the third most lethal malignancy worldwide (Sung et al., 2021). LC is hepatocellular carcinoma (HCC), an incidence of 500,000–1,000,000 fresh cases each year, and is related to 600,000 direct deaths annually. Numerous risk factors involved in HCC development include excessive alcohol consumption, smoking, chronic infection with hepatitis B or C virus, type 2 diabetes, obesity, immune-related disorders, aflatoxin-contaminated foods, and exposure to environmental carcinogens like nitrosamines (Plummer et al., 2016; Vogel et al., 2018; Zhou et al., 2016). Prolonged inflammation, along with oxidative stress (OS), is the typical cause of HCC (Fu & Chung, 2018), and it is mostly found in cirrhotic livers (Seitz & Stickel, 2006). Chronic inflammation from persistent damage to the liver leads to cirrhosis and fibrosis that eventually leads to the growth of carcinomas in the liver (Crusz & Balkwill, 2015).
Diethylnitrosamine (DEN) originates in tobacco smoke, gasoline, processed foods, cosmetics, and agricultural chemicals (Tolba et al., 2015; Zhou et al., 2016). DEN is accredited to its direct DNA alkylating impact and generation of reactive oxygen species (ROS) after its bio-activation in the liver by the hepatic cytochrome P450 enzymes (Heindryckx et al., 2009). Furthermore, persistent ROS formation has been described to trigger cell-survival signaling cascades of the PI3K/Akt signaling (Ding et al., 2017; Jo et al., 2011). Notably, unique reasons for hepatic carcinoma induction caused by DEN are the elevated expression of cyclin-dependent kinases involved in the G1/S-phase cell-cycle regulatory proteins.
Phenolic diterpene rosmanol (RM) is taken out from abundant herbal plants, principally from Rosmarinus officinalis L. (Rosemary). Several researchers have documented that rosemary employs antiproliferative, anticancer, antioxidant, and anti-inflammatory effects (Altinier et al., 2007; Cheung & Tai, 2007). Accumulating evidence reported that RM exerts a strong antioxidant; it averts superoxide anion generation, lipid peroxidation (LPO), and free radicals rummaging activity (Altinier et al., 2007; Del Baño et al., 2003; Doolaege et al., 2007). However, the antitumor efficacy of RM on HCC has not been elucidated yet. Given the efficiency of RM as a powerful antitumor antioxidant, this investigation was intended to acquire the hepatoprotective influence on DEN-prompted HCC mode in rats through antiproliferative, antioxidative, antilipid peroxidative, anti-inflammatory, and PI3K/Akt signaling pathways.
Materials and Methods
Reagents and Biochemicals
RM, DEN, and phosphate buffer were obtained from Ruicong Ltd. (Shanghai, China). Enzyme-linked immunosorbent assay (ELISA) assay kits were acquired from Ese-Bio (Shanghai, China). For the western blot assay, the antibodies were procured from Elabscience Biotechnology (Wuhan, China). Biochemicals and solvents analytical grades were used.
Animals
Wistar albino rats (8–10 weeks old), male, weighing 150–180 g were used for the HCC experiment. They were acclimatized for a week before the experiment and kept in sterile polypropylene cages under standard conditions. All animals were fed with pellet diet with distilled water and a 12-hour light–dark phase. The trial procedure was sanctioned by the Qian County People’s Hospital Animal Care and Ethical Committee approval number: 2024-811.
HCC Induction and Experimental Protocol
Induction of HCC was followed according to the previously reported method (Singh et al., 2018). A total of 24 rats were arbitrarily separated into 4 sets, each containing 6 rats. They are normal control (NC), DEN-induced HCC model, DEN+RM (40 mg/kg bw), and RM (40 mg/kg bw) alone. NC received oral gavage of normal saline for 16 weeks; DEN-induced HCC model rats received the first day of the trials in a solo injection of DEN (200 mg/kg bw) intraperitoneally (i.p.). The DEN+RM group was solo injected with DEN (200 mg/kg bw) on the first trial day subsequently oral gavage of RM (40 mg/kg bw) for 16 weeks, and RM-alone rats were treated with oral gavage of RM (40 mg/kg bw). The body weight of all rats was measured during the trials. Animals were sacrificed by cervical dislocation after the experimental period using ketamine-xylazine anesthetic injection. Then separated and cleaned the hepatic tissues were blotted with filter paper and weighed up. Collected the blood samples and kept them as serum for further biochemical estimations. Liver slices were kept in 10% formaldehyde for histopathological studies. Tumor volume was measured using the formula V = ¼ύ (D1/2) (D2/2) (D3/2), Where D1, D2, and D3 are the three diameters (mm) of the tumors. Tumor burden was calculated by multiplying tumor volume and the number of tumors/animals.
Tissue Homogenate Preparation
The separated hepatic tissues were made uniformly with phosphate buffer (pH 7.4) using a protease inhibitor cocktail and centrifuged for 20 minutes at 12,000 g to attain supernatants for biochemical assays.
Assay of Hepatic Toxicity Biomarkers
The hepatic enzyme markers such as lactate dehydrogenase (LDH), gamma-glutamyl transpeptidase (GGT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and alanine aminotransferase (ALT) were estimated by using the ELISA assay kits (Ese-Bio, Shanghai, China) permitting to the company’s guidelines.
Analysis of Antioxidant Enzymes
The antioxidant enzymes activity of glutathione peroxidase (GPx), catalase (CAT), glutathione reductase (GR), superoxide dismutase (SOD), and glutathione-S-transferase (GST) in the hepatic homogenate was measured with assay kits delivered by Biomed (Badr City, Egypt) following the manufacturer’s guidelines.
Estimation of LPO
The LPO markers of TBARS in serum and liver, as well as hepatic LOOH, were estimated with assay kits provided by Biomed (Badr City, Egypt) following the manufacturer’s guidelines.
Histopathological Evaluation
Histopathological analysis was performed in the rat’s liver tissue. The excised hepatic tissues were preserved with formalin (10%), fixed with paraffin, cut into sections (5 µm), and hematoxylin & eosin (H&E) staining. Lastly, the stained sections were imaged below light microscopy.
Determination of Inflammatory Mediators in Serum
The intensities of inflammatory mediators comprising NF-κB, TNF-α, and IL-6 were estimated by using an ELISA kit (Ese-Bio, Shanghai, China) as per the company’s procedures. The concentration was stated as pg/mL.
Western Blot Study
Entire proteins were isolated from the hepatic tissue by an ice-cold lysis buffer. When SDS-PAGE was performed, the gel was moved to PVDF film. This was blocked with 5% skimmed milk and the added diluents of the primary antibodies and kept throughout the night at 4°C. The primary antibodies (1:1,000 dilutions) were administered. Then, PBS was washed and preserved with HRP-conjugated secondary antibodies for 1 hour. The protein bands were stained and imagined for protein identification. Quantified the protein bands through densitometry with software ImageJ and homogeneous to β-actin expression.
Statistical Analysis
Data were denoted as mean ± SD, and the statistical comparisons were accompanied by GraphPad Prism software version 8.0.1, which was employed to perform an analysis of variance (ANOVA) and, subsequently, Duncan’s test. Student t-test has been conducted and p < 0.05 was deliberated significant.
Results
Influence of RM on Body and Liver Weight
A significant body weight reduction (p < 0.05) was noticed in the DEN-stimulated HCC model group versus NC. RM (40 mg/kg bw) administered to rats considerably decreased (p < 0.05) the DEN-stimulated body weight loss. There is no substantial variation observed in the body weight of NC and RM (40 mg/kg bw) alone treated rats. However, RM administration to the DEN-stimulated rats ameliorated the body weight expressively in contrast to DEN-prompted model rats (Figure 1).
Rosmanol (RM) Alleviates Loss of Body and Liver Weight in Diethylnitrosamine (DEN)-induced Hepatocellular Carcinoma (HCC) Rats. (A) Body Weight; (B) Liver Weight. Results were Displayed as Mean ± SD of Six Observations and the Significance is Considered as *p < 0.05 versus Normal Control (NC), #p < 0.05 versus the DEN-stimulated.
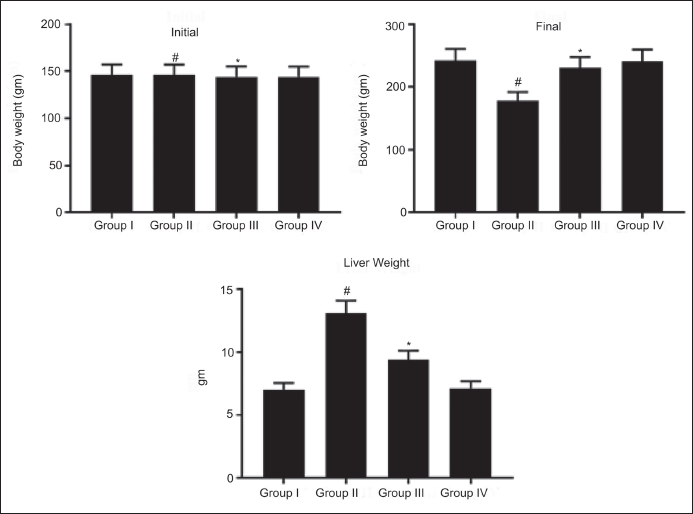
DEN-stimulated rats revealed substantially increased (p < 0.05) liver weight counter to NC. RM (40 mg/kg bw) administration to the DEN-stimulated rats displayed a momentous (p < 0.05) reduction in liver weight. There was no considerable change in the liver weight of NC and RM (40 mg/kg) alone treated rats. These findings are a sign of the chemopreventive activity of RM against DEN-induced HCC in rats (Figure 1).
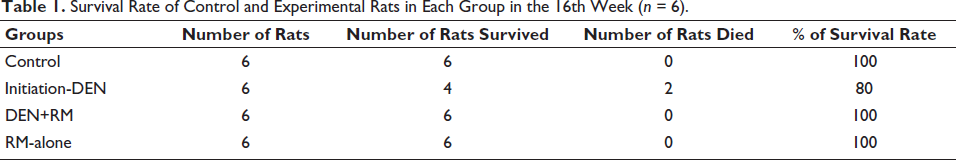
The survival rate of the rats was determined, and two rats died in group II; no deaths were found in other groups I, III, and IV (Table 1). RM decreased the liver incidence and the number of tumor induced by DEN in animals. Table 2 shows the total number of nodules and number of nodules in liver incidence-bearing animals. The RM-treated group III showed a significant decrease in liver incidence and number of nodules compared with DEN-induced animals (group II).
Survival Rate of Control and Experimental Rats in Each Group in the 16th Week (n = 6).
Effect of Rosmanol (RM) on the Incidence of Cancer Nodules on Experimental Rats (n = 6).

Influence of RM on Hepatic Toxicity Marker Enzymes in Serum
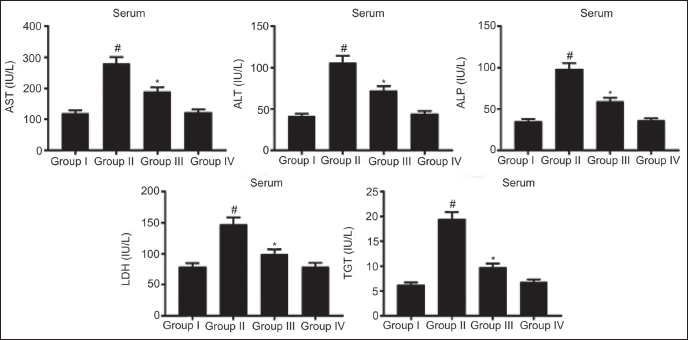
Liver-damaged marker enzymes, including LDH, AST, ALP, ALT, and γ-GT, were pointedly elevated (p < 0.05) in DEN-stimulated rats in contrast to NC. Treatment with RM (40 mg/kg bw) considerably attenuated (p < 0.05) these toxicity enzymes activity versus DEN-alone induced rats. Similar hepatic enzyme activity was noticed in NC and RM-alone administered rats. These data indicate the hepatoprotective activity exerted by RM treatment against DEN-prompted HCC in rats (Figure 2).
Rosmanol (RM) Reduces Liver Toxicity Enzymes in Diethylnitrosamine (DEN)-induced Hepatocellular Carcinoma (HCC) Rats. Results were Displayed as Mean ± SD of Six Observations and the Significance is Considered as *p < 0.05 versus Normal Control (NC), #p < 0.05 versus the DEN-stimulated.
Effect of RM on Hepatic Antioxidant Enzyme Status
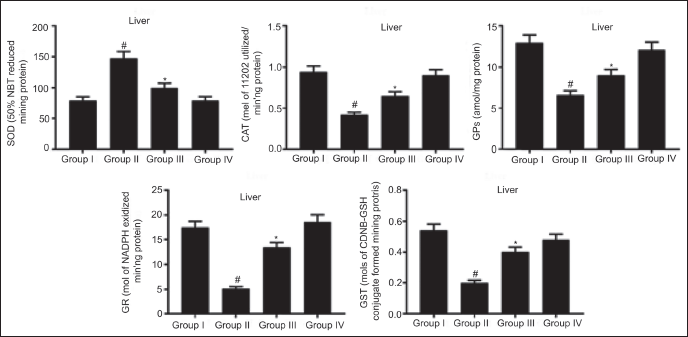
Hepatic antioxidant enzymes including CAT, GPx, GR, SOD, and GST concentration declined (p < 0.05) in the DEN-stimulated rats versus NC. Administration of RM (40 mg/kg bw) significantly elevated (p < 0.05) these enzyme levels counter to DEN-model rats. Comparable hepatic antioxidant enzyme activity was observed in NC and RM-alone administered rats. These outcomes unveil RM could enhance the antioxidant enzyme activity against DEN-prompted HCC in rats (Figure 3).
Rosmanol (RM) Diminishes Serum TBARS, Hepatic TBARS, and LOOH in Diethylnitrosamine (DEN)-induced Hepatocellular carcinoma (HCC) Rats. Results were Displayed as Mean ± SD of Six Observations and the Significance is Considered as *p < 0.05 versus Normal Control (NC), #p < 0.05 versus the Diethylnitrosamine (DEN)-model.
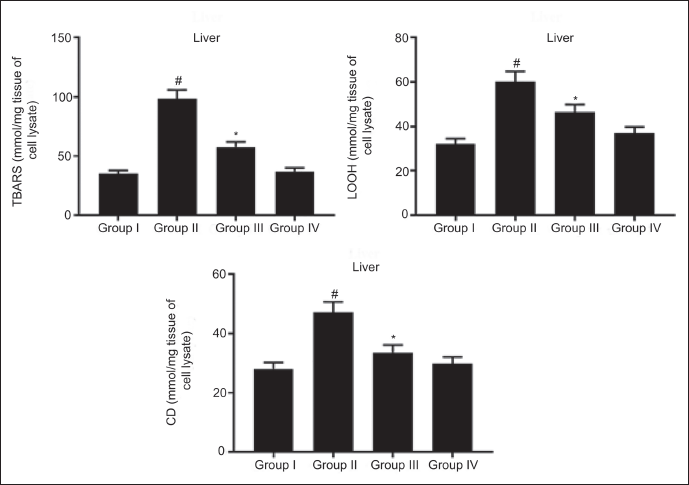
Response of RM on LPO of Control and Experimental Rats
LPO product TBARS in serum and liver, as well as hepatic LOOH and CD, were pointedly augmented (p < 0.05) in the DEN-stimulated rats against NC. Treatment with RM (40 mg/kg bw) considerably depleted (p < 0.05) these levels against the DEN-model group. A similar LPO level was observed in NC and RM-alone administered rats. These data mentioned that DEN-triggered high level of LPO that indicates OS, while RM could reduce LPO, thereby alleviating DEN-stimulated OS in rats (Figure 4).
Rosmanol (RM) Diminishes Serum TBARS, Hepatic TBARS, and LOOH in Diethylnitrosamine (DEN)-induced Hepatocellular Carcinoma (HCC) Rats. Results were Displayed as Mean ± SD of Six Observations, and the Significance is Considered as *p < 0.05 versus Normal Control (NC), #p < 0.05 versus the Diethylnitrosamine (DEN)-model.
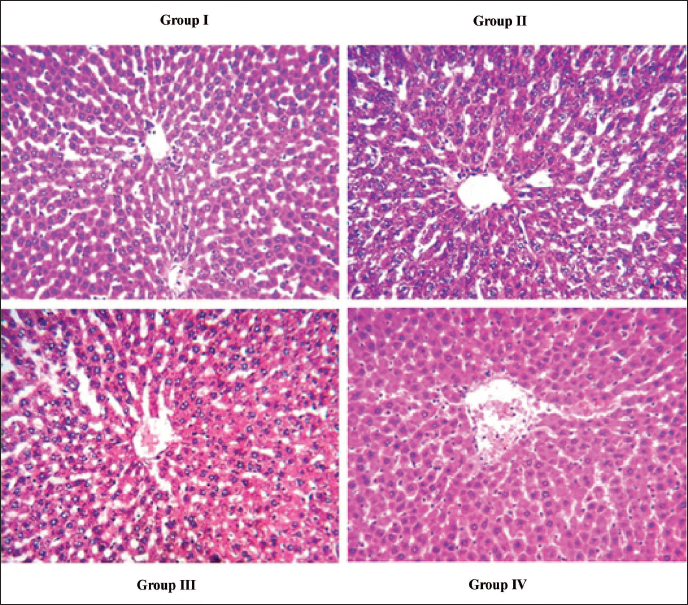
Histopathological Assessment of Liver
NC and RM-alone administered rats presented typical liver histopathology. DEN-stimulated HCC model rats unveiled ballooning degeneration, irregular nuclei, enlarged hepatocytes with focal nodular hyperplasia, fat deposits, and inflammatory infiltration. Administration of RM (40 mg/kg bw) could mitigate nodules, hyperplasia, and enlargement. DEN-induced hepatocyte changes were expressively inverted by RM in contrast to DEN-stimulated HCC rats by restructuring the hepatocellular architecture (Figure 5).
Influence of Rosmanol (RM) on Liver Histopathology of Diethylnitrosamine (DEN)-prompted Hepatocellular Carcinoma (HCC) Rats. The Histopathological Changes of Hepatic Tissue of Control and Experimental Rats (Hematoxylin & Eosin (H&E) Staining, 200×), Scale Bar = 50 µm. (Group I) Normal Control (NC) Rats Showing a Typical Hepatic Histology; (Group II) DEN-stimulated Rats Showing Ballooning Degeneration, Fat Deposits, Enlarged Hepatocytes with Focal Nodular Hyperplasia, and Inflammatory Infiltration; (Group III) RM (40 mg/kg bw) Administered Rats Mitigate Nodules, Hyperplasia, and Enlargement; (Group IV) RM (40 mg/kg bw) Alone Showing Normal Hepatic Histology.
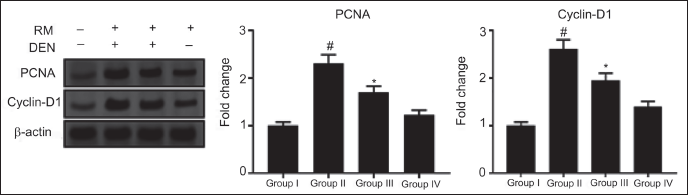
Effect of RM on Hepatocyte Proliferation
To explore the antiproliferative activity of RM on DEN-stimulated HCC, the proteins cyclin-D1 and PCNA expression were assessed. Protein levels of cyclin-D1 and PCNA were expressively increased (p < 0.05) by DEN-stimulated rats versus NC. Remarkably, RM could reduce (p < 0.05) cyclin-D1 and PCNA protein expression counter to the DEN-model group. Comparable protein expressions were observed in NC and RM-alone treated rats (Figure 6).
Influence of Rosmanol (RM) on the Hepatic PCNA and Cyclin-D1 Protein Expression in Diethylnitrosamine (DEN)-induced Hepatocellular Carcinoma (HCC) Rats. PCNA and Cyclin-D1 Protein Expression Levels were Examined by Western Blot. Results were Displayed as Mean ± SD of Six Observations and the Significance is Considered as *p < 0.05 versus Normal Control (NC), #p < 0.05 versus the DEN-model.
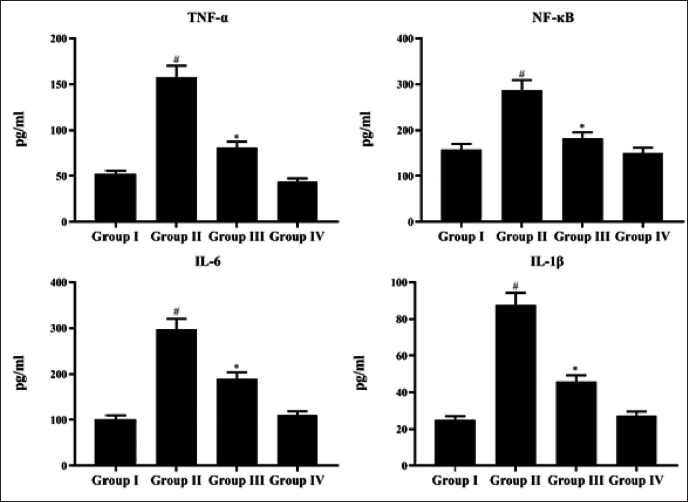
Anti-inflammatory Activity of RM on the DEN-stimulated HCC in Rats
In the DEN-prompted group, inflammatory mediators containing TNF-α, NF-κB, and IL-6 were considerably enhanced (p < 0.05). Supplementation of RM (40 mg/kg bw) to the DEN-induced group could reduce significantly as compared to the model group. Parallel results were detected in both NC and RM-alone administered rats. These findings established the anti-inflammatory potential of RM against DEN-prompted HCC in rats (Figure 7).
Anti-inflammatory Activity of Rosmanol (RM) Against Diethylnitrosamine (DEN)-induced Hepatocellular Carcinoma (HCC) Rats. The Inflammatory Mediators such as TNF-α, NF-κB, and IL-6 were Measured by Enzyme-linked Immunosorbent Assay (ELISA) Kit. Results were Displayed as Mean ± SD of Six Observations and the Significance is Considered as *p < 0.05 versus Normal Control (NC), #p < 0.05 versus the DEN-model.
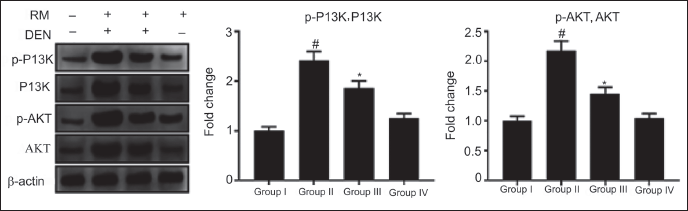
RM Regulated PI3K/Akt Pathway
The protein expression level of p-PI3K and p-AKT is intentionally elevated (p < 0.05) in the DEN-stimulated model mice versus NC. Administration of RM (40 mg/kg bw) expressively alleviated the expression levels of p-PI3K and p-AKT. The ratio of p-PI3K/PI3K and p-AKT/AKT were elevated in the DEN-prompted HCC group, whereas RM could mitigate the ratio of phosphorylation of these proteins. These emphasized the anticancer and apoptotic activity of RM against DEN-prompted HCC in rats (Figure 8).
Influence of Rosmanol (RM) on PI3K/Akt Signaling. PI3K, p-PI3K, AKT, and p-AKT Protein Expression were Evaluated by Western Blot Assay. Results were Displayed as Mean ± SD of Six Observations, and the Significance is Considered as *p<0.05 versus Normal Control (NC), #p < 0.05 versus the Diethylnitrosamine (DEN)-stimulated.
Discussion
HCC is the most fatal carcinoma in the universe (Mak et al., 2020; Sung et al., 2021). Establishing HCC animal models is an exceptionally vital implement to recognize cellular and molecular variations throughout liver tumorigenesis and emerging effective treatments (Ding et al., 2017; Jo et al., 2011; Kurma et al., 2021). DEN has become an excellent chemical carcinogen for producing HCC in rats as an investigational human model of LC (Kurma et al., 2021). DEN is a renowned hepatocarcinogenesis, which is generally used to make LC in rodent HCC models owing to its capability to interrupt nuclear enzymes contained in DNA replication and repair (Arboatti et al., 2018). Previous reports revealed that DEN-induced HCC development was caused by oxidative damage and ROS generation described by antioxidant reduction, moderated antioxidant enzyme activities, and inflammation (Abdu et al., 2022). Hence, a natural antioxidant with antitumor and anti-inflammatory activity is a suitable alternative to treat HCC. In this report, we demonstrated the chemopreventive efficacy of RM against DEN-stimulated HCC through anti-inflammatory and antitumor activity via targeting the PI3K/Akt pathway as a potential mechanism.
Similarly, RM inhibited COX-2 and iNOS gene expression along with their downstream products, PGE2 and NO, correspondingly (Lai et al., 2009), which primes to the declining translocation of NF-κB subunits (Cheng et al., 2011). As designated formerly, RM suppresses human cancer cell proliferation of colon carcinoma (Cheng et al., 2011), neuroblastoma (Tabata et al., 2012), and breast cancer (Jiang et al., 2021). Previously, it has been investigated that the RM-triggered caspase cascades and includes both mitochondria-facilitated and death receptor-enabled apoptotic pathways in colon cancer cells COLO 205 (Doolaege et al., 2007). RM involves the mitochondrial-aided apoptosis on the repression of MDA-MB 231 and MCF-7 mammary carcinoma cell proliferation by ROS-facilitated JAK2/STAT3 and PI3K/Akt inhibition (Jiang et al., 2021). In contrast, carnosic acid isolated from Rosemary showed diminished apoptosis in neuroblastoma cells by the protective action related to the modulation of PI3K/Akt pathway, cytochrome-c release depleted, and declining caspases (De Oliveira et al., 2015).
The anticancer effectiveness of RM on DEN-prompted HCC was exposed in male rats. DEN-stimulated rats revealed body weight loss and elevated liver weight, while RM administration attenuates the body weight deficit and liver weight, which designates the remedial action of the RM counter to DEN-prompted HCC. This substantial increase in hepatic weight observed by the DEN-model group could be ascribed to enhanced metabolic activity (Assar et al., 2021), cellular swelling, liver cell destruction, along hepatic fat variations (Miller & Zachary, 2017). An earlier investigation demonstrated that gambogenic acid inverted the impact of DEN as it arose in body weight, and diminished liver weight was noticed, which specifies the amelioration of hepatocytes (Zhang et al., 2023). In hepatic histopathology, images show irregular nuclei, fat deposits, ballooning degeneration, enlarged hepatocytes with focal nodular hyperplasia, and inflammatory infiltration in DEN-stimulated HCC model rats. Administration of RM (40 mg/kg bw) could mitigate nodules, hyperplasia, and hepatic cell enlargement. DEN-induced hepatocyte changes were expressively inverted by RM against DEN-stimulated HCC rats by restructuring the hepatocellular architecture. RM could moderate the loss of body weight and liver weight, as well as liver damage due to its chemopreventive, anticancer, and hepatoprotective effects. RM is a phenolic diterpene chiefly extracted from R. officinalis L. (Rosemary). Simultaneously, we found survival rates and tumor incidence in the control and experimental rats. From this, the survival rate was higher in RM-induced and controlled tumor growth development compared to the control rats. Finally, it was concluded that RM was not more toxic. It naturally found vitamin compounds. Accumulating evidence has stated that rosemary and its phenolic components employ antiproliferative, anticancer, antioxidant, and anti-inflammatory activities (Altinier et al., 2007; Cheung & Tai, 2007). Numerous studies have documented the hepatoprotective activity of R. officinalis extracts due to the presence of phenolic compounds in the experimental model of alcoholic liver disease (Martínez-Rodríguez et al., 2019), azathioprine-induced toxicity in rats (Amin & Hamza, 2005), and paracetamol-induced liver damage (Fadlalla & Galal, 2020). These documents emphasize the hepatoprotective activity of RM.
The pathogenesis of HCC is related to the existence of OS biomarkers in serum owing to the incidence of tissue damage. Herein, DEN-stimulated rats elevated the hepatic injury markers such as ALT, AST, ALP, LDH, and GGT. Related data were provided by Zhang et al. (2023) and Ahmed et al. (2022). The augmented biomarkers in serum were pointedly diminished by oral gavage of RM as presented in the data. Hepatic enzymes ALT and AST are powerful signs of hepatocellular impairment (Limdi & Hyde, 2003). GGT shows a vital role in transpeptidation, biotransformation, nucleic acid metabolism, hydrolysis of glutathione glutamyl group, and carcinogenesis (Ma et al., 2014). During hepatic damage, an excess quantity of these cytoplasmic enzymes was discharged into the bloodstream owing to the membrane permeability alterations. DEN stimulation considerably augmented ALT, AST, ALP, GGT, and LDH levels. The abnormal enzyme intensities in serum were normalized with RM (40 mg/kg bw) treatment. In an experimentally induced hepatic toxicity model, Rosemary and its phenolic constituents could moderate the hepatic toxicity enzyme marker, including ALT, AST, ALP, and GGT (Amin & Hamza, 2005; Fadlalla & Galal 2020; Martínez-Rodríguez et al., 2019). These findings established that RM could alleviate DEN-induced hepatic damage in rats.
OS is the main cause of HCC and has developed as a critical element in the initiation, development, and progression (Fu & Chung, 2018; Heindryckx et al., 2009; Jo et al., 2011). DEN is processed in the body to generate extremely reactive free radicals by cytochrome-P450, commencing the LPO sequence. Extreme free radicals react with lipids to liberate LPO. It is a reactive molecule able to interrelate with DNA to stimulate mutation and, eventually, tumorigenesis. DEN has been exposed to prompt HCC by enhancing OS and liver injury (Arboatti et al., 2018; Kurma, 2021). These results very well relate with the current report that displays a substantial rise in the LPO level when induced with DEN, when compared with NC rats. RM unveils its chemopreventive efficiency on DEN-stimulated HCC by reducing the LPO. Antioxidant generation is primarily recognized as an applied method for defending cells against exogenous toxic carcinogens and immune suppression. Collecting evidence described that RM exerts a strong antioxidant; it averts superoxide anion generation, LPO, and free radicals rummaging activity (Altinier et al., 2007; Del Baño et al., 2003; Doolaege et al., 2007). In the present study, RM treatment enhanced the hepatic antioxidant enzyme status. Enzymatic antioxidants are controllers of antioxidants and are accountable for the ejecting of ROS and other cellular free radicals (Gnanaraj et al., 2016). DEN alone treated rats showed elevated TBARS as well as LOOH developments and lessened enzymatic antioxidant actions. Enzymatic antioxidants comprising GPx, GST, SOD, GR, and CAT were increased in RM-administered rats owing to their antioxidant nature. These data highlight that the administration of RM forages unnecessary free radicals, establishing its antioxidant potential against HCC.
Pro-inflammatory cytokines are discharged in reaction to inflammation, infection, and carcinogen-prompted harm. In this study, NF-κB, TNF-α, and IL-6 were found to be raised in DEN-stimulated rats, while RM-administered rats presented these higher levels recurring to the near normal level. NF-κB is a principal controller of inflammation and apoptosis in the expansion of liver damage, liver fibrosis, and HCC. It has been established that suppression of NF-κB activity is directly relative to the stimulation of apoptosis (Chen et al., 2020; Crusz & Balkwill, 2015). RM could reduce NF-κB activity and may activate apoptosis in malignant cells. The prior investigation documented that RM inhibited COX-2 and iNOS gene expression along with their downstream products, PGE2 and NO, correspondingly (Lai et al., 2009), which primes to the declining translocation of NF-κB subunits (Cheng et al., 2011). This current study unveils the anti-inflammatory effect of RM against DEN-prompted HCC in rats.
An imbalance between cell proliferation and apoptosis plays a significant part in tumor enlargement. Cell proliferation is crucial for the growth of instigated cells, whereas apoptosis involves a central involvement in growing regulation that is stimulated by DNA impairment and unrestrained propagation (Hanahan & Weinberg, 2011; Pfeffer et al., 2018). Thus, the generation of malignant cell fatality by apoptosis is a key objective of tumor management. Herein, RM exhibited a momentous attenuation of the cyclin-D1 and PCNA protein expression against the DEN-stimulated model. It is recognized that cyclin-D1 protein plays a significant role in the advancement of the G1/S stage cell cycle. The data presented here intensely supports that RM subdues cell multiplication by the attenuation of cyclin-D1, subsequent in cell cycle block in the G1 stage, hence averting cell propagation. Apoptosis is generally controlled by signaling networks and one of the noticeable signaling networks is the PI3K/Akt pathway, a crucial oncogenic kinase in tumor formation (Ding et al., 2017; Jo et al., 2011). In this work, RM attenuated the PI3K/Akt protein expression in DEN-stimulated rats. p-PI3K and p-AKT protein expression levels are substantially increased (p < 0.05) in DEN-stimulated model animals relative to NC. The expression levels of p-PI3K and p-AKT were eased expressively upon administration of RM (40 mg/kg bw). In the DEN-induced HCC group, the ratio of p-PI3K/PI3K and p-AKT/AKT was increased; however, RM was able to reduce the phosphorylation of both proteins. These highlighted the apoptotic and anticancer properties of RM against DEN-induced HCC in rats. An earlier research reported that RM involves the mitochondrial-facilitated apoptosis on the repression of MDA-MB 231 and MCF-7 mammary carcinoma cell proliferation by ROS-facilitated JAK2/STAT3 and PI3K/Akt inhibition (Jiang et al., 2021). Niu et al. (2024) examined that the RM (20 mg/kg bw) administered rats showed TBARS level was substantially alleviated and restored the antioxidant status in RM L administered rats. Histopathologic assessment of gastric tissue unveiled that an MNNG-induced group presented squamous cell carcinoma with keratin pearls and also reduced GC incidence, and only mild dysplasia was observed. Further, RM alleviated Bcl-2, PI3K, AKT, and HMGB1, as evidenced through RT-PCR and Western blot analysis. From these, we selected the 40 mg/kg bw, non-toxic dose for our current study. Therefore, PI3K/Akt downregulation inhibits HCC cell proliferation via triggering apoptosis, proposing that HCC chemopreventive mechanisms induced by RM.
Conclusion
In conclusion, the current study established RM as a beneficial antihepatocarcinogenesis phenolic compound against DEN-induced HCC, signifying that the antitumor actions are perhaps facilitated by increased survivability of rats by regulating the PI3K/Akt signaling through its antioxidative, antiproliferative, and apoptotic effects. RM markedly suppressed OS, LPO, proliferation, inflammation, and hepatic damages, while enhancing antioxidant enzyme status. Hence, RM can be acclaimed for further study as a possible drug aspirant for ameliorating chemoresistance and metastasis, advancing the HCC patient’s prognosis.
Summary
RM could reduce the loss of body weight and liver weight against DEN-stimulated HCC rats. RM markedly inhibited proliferation, inflammation, and hepatic damages, while enhancing antioxidant enzyme status on DEN induced in LC rats.
Footnotes
Abbreviations
HCC: Hepatocellular carcinoma; RM: Rosmanol; DEN: Diethylnitrosamine; LC: Liver cancer; OD: Oxidative stress; RNS: Reactive nitrogen species; NC: Normal control; LDH: Lactate dehydrogenase; GGT: Gamma-glutamyl transpeptidase; AST: Aspartate aminotransferase; ALP: Alkaline phosphatase; ALT: Alanine aminotransferase; GPx: Glutathione peroxidase; CAT: Catalase; GR: Glutathione reductase; SOD: Superoxide dismutase; GST: Glutathione-S-transferase; LPO: Lipid peroxidation; H&E: Hematoxylin & eosin.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The Study was approved by the ethical committee Qian County People’s Hospital Animal Care (Ethical Committee approval number: 2024-811).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
