Abstract
Background
p53 is a phosphorylated glycoprotein whose expression is abnormally elevated in various human tumors. p53 also regulates a variety of tumor cells, but research on cervical cancer has not yet been reported.
Objectives
This study intends to investigate the role of targeting p53 in cervical cancer.
Materials and Methods
Electron microscopy was used to assess the size of protein particles, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) was used to detect the OD value of nanocomposites at different concentrations using a UV spectrophotometer, MTT and cell cloning were used to detect cell proliferation expression ability, and reverse transcriptase polymerase chain reaction (RT-PCR) to detect p53 expression. Immunofluorescence measured nuclear factor kappa B (NF-κB) expression in cells, and western blotting assessed the relative expression of p53 and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) proteins. The detection mechanism was further explored through in vivo studies.
Results
Glutaraldehyde (GLA)-loaded liposomal albumin particles showed a positive zeta value, indicating that the CS coating process of GLA was successful. Our data show that the experimental group showed a concentration-dose-dependent growth relationship, indicating that GLA-loaded liposome albumin particles have better antitumor activity in cervical cancer cell lines. Compared with most GLA-loaded liposomal albumin particle groups, p53 expression was inhibited, and the expression of p53 was downregulated in GLA-loaded liposomal albumin particle cervical cancer. In vivo results showed that the measured tumor volume decreased after treatment with GLA-loaded liposomal albumin particles, and mice treated with GLA-loaded liposomal albumin particles had longer survival times. Induction of NF-κB in tumors may be a powerful strategy allowing GLA-loaded liposomal albumin particles to avoid apoptosis and possibly after cytotoxic chemotherapy.
Conclusion
This study demonstrates that GLA-loaded liposomal albumin particles may participate in the development and progression of human cervical cancer cells by targeted regulation of NF-κB activity.
Introduction
Cervical cancer is a common gynecological tumor, which is particularly serious in Asia. Although progress has been made in the past decade, its development mechanism has not been studied (Shrestha et al., 2018). Abnormal signal expression induces tumorigenesis and affects downstream signal transduction. These studies aim to find new treatments for intestinal cancer, thereby reducing its mortality (Marth et al., 2017).
Previous studies using emodin to treat cervical cancer patients found that caspase-3 and caspase-9 expression was abnormally increased, inhibiting cancer cell proliferation and promoting cell apoptosis (Saei Ghare Naz et al., 2018). Glutaraldehyde (GLA) is a diterpene compound with rich biological functions and antitumor effects. GLA has anti-inflammatory and stomachic properties. As early as the 19th century, GLA was proven to have an inhibitory effect on tumor cells. Although these drugs have a specific antitumor effect in recent years, due to their poor water solubility and instability, the therapeutic effect is somewhat unsatisfactory. Previous studies have shown that the main factor affecting the effect of tumor treatment is cancer stem cells, but most chemotherapy drugs have certain limitations, such as short half-life, low stability, poor water solubility, and so on. The main features in the past were passive targeting and long half-life. In recent years, active targeted drug delivery has become a research hotspot (Mboumba Bouassa et al., 2017; Nahand et al., 2019). Human serum albumin nanoparticles can be used as carriers for a variety of tumor cells. Loaded with GLA nano-albumin particles, the nano-albumin particles can significantly improve the stability and water solubility of GLA drugs, thereby improving the therapeutic effect. Studies have confirmed the suitability of lipid nanocarriers for nose-to-brain delivery in monolayer cells in vitro (Acharya Pandey & Karmacharya, 2017).
p53 is inactivated by viral proteins in HPV-infected cervical tissue (Chen, Li, et al., 2018; Shang et al., 2018; Voci et al., 2021; Yazghich & Berraho, 2018). Therefore, some people believe that activating p53 may be a target for treating cervical cancer (Hu & Ma, 2018; Lin et al., 2019). We explored the mechanism of GLA-loaded liposomal albumin particles by establishing a cervical cancer cell xenograft tumor model.
Abnormal nuclear factor kappa B (NF-κB) activation was associated with human cervical cancer (Chen, Liu, et al., 2018). Cervical epithelial hyperplasia and duct branching that lack κBα (iκBα) protein inhibitors increase, and their development is interfered with by the NF-κB signaling pathway (Zhang et al., 2015). APC-driven c-Rel mice develop various pathological types of late-onset intestinal cancer. Inhibition of NF-κB activation decreased cervical cancer xenograft growth (Wang et al., 2019). Previous in vitro experiments have shown that blocking the NF-κB pathway with pyrrolidine dithiocarbamate (PDTC) successfully inhibits the expression of aging-related proteins, revealing the tumor development and progression mediated by the NF-κB/p53 pathway. Active NF-κB signaling exists in epithelial cells, including multiple gene-positive subtypes, and its activation can lead to cell apoptosis and shrink tumors (Kang et al., 2017; Zhu et al., 2018). Here, we propose for the first time the relationship between the level of GLA-loaded liposomal albumin particles and antitumor effects in cervical cancer, as well as its underlying regulatory mechanisms.
Materials and Methods
Tissue Sources and Cell Culture
After cells were seeded, they were transfected for 6 h in serum-free using Lipofectamine 2000 reagent (Shanghai Thermo Fisher Scientific Co., Ltd.), and cells were switched to fresh medium containing 10% FBS and cultured for 48 h.
Synthesis and Preparation of GLA-loaded Liposomal Albumin Particles
Human serum albumin nanocarrier (HSANC) was added to a methanol solution and dispersed by ultrasound. After 1 h, centrifuge, wash, add mannitol, sonicate, and freeze–drying. Nanoparticles were characterized using transmission electron microscopy. Set up two groups, the control group (n = 5) was given 0.9% 0.12 mol/mg normal saline, and the GLA group (n = 15) was given the same dose of GLA-HSANC drug. Then, according to the drug concentration, they were divided into low-dose group (0.06 mol/mg), medium-dose group (0.12 mol/mg), and high-dose group (0.18 mol/mg). Transplanted tumor volume V = L/(V control group at the beginning of treatment – V control group at the end of treatment) × 100%. Mice were sacrificed using the neck fracture method, and the transplanted tumor tissue was harvested. Tumor inhibition rate (%) = (mass of transplanted tumors in NS control group – mass of transplanted tumors in NSGLA group)/mass of transplanted tumors in NS control group × 100%. The tests were conducted on 1d, 4d, 7d, 10d, 13d and 16d after administration. After the last administration, nude mice were fasted for 6 h under anhydrous conditions, and tumor markers (CA125, CEA) were detected.
The particles’ mean size and electrokinetic potential were measured using dynamic laser scattering (DLS) and a Zetasizer 3000 HS particle analyzer (Malvern Instruments Ltd, Worcester, UK). Size and zeta potential values are expressed as the average of three measurements.
Transmission Electron Microscope (TEM)
Observe exosomes by TEM. Exosomes were fixed and then washed. Approximately 10 µL was placed on a 300-mesh copper electron microscope grid for 5 min. Exosomes were then counterstained with 2% uranyl oxalate, washed, and air-dried followed by observation under a TEM (JEM-2100, Jeol, Japan).
Reverse Transcriptase Polymerase Chain Reaction (RT-PCR)
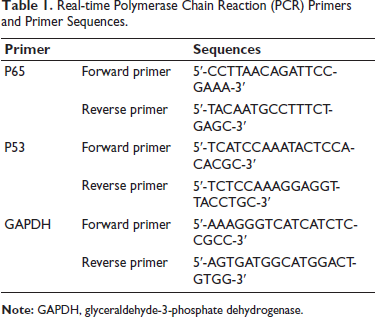
Trizol was used to extract ribonucleic acid (RNA), which was used for qRT-PCR with conditions: 95°C 10 min, followed by 40 cycles of 95°C 15 s and 60°C 1 min. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and 18S RNA were quantified before calculation using the ∇∇Ct method. Primer sequences are shown in Table 1.
Cell Cycle Determination
Take the above successfully transfected MG63 cells, wash with PBS, centrifuge at 1,000 r/min for 5 min, make a single cell suspension, add Annenin V-PE, 7-AAD, incubate for 15 min, add 70% ethanol solution, dose 1 mL, add detection of cell cycle.
Antitumor Effects and Systemic Toxicity in vivo
A total of 80 BALB/c mice, half male and half female, were used in the experiment, all from the Experimental Animal Center of our hospital. Subcutaneous tumor transplantation technology was used to inject tumor cell lines under the skin of mice. After four weeks, the main organs of the mice were collected. The tissues were fixed in formalin and stained. Immunohistochemistry and ImmunoSilver staining were subsequently performed.
Immunofluorescence
After transfection of six-well plates with transfection plasmids in cell lines, cells were washed, fixed with formalin, and permeabilized with Triton-X100 solution. After serum blocking, the primary antibody was incubated overnight with a secondary antibody in the dark. Next, a blocking solution containing 4,6-diamino-2-phenylindole was added for fixation for nuclear counterstaining, and proteins were observed with fluorescence microscopy. Negative control photos were processed similarly, using PBS as a control.
Western Blot
After protein extraction, they were separated by SDS-PAGE for western blot using primary antibodies (Proteintech, Chicago, USA, 1:1,000, Cat). After overnight incubation with a secondary antibody for 1 h. Western blot bands were detected and analyzed by an Odyssey imaging system (LI-COR, Inc., Lincoln, NE, USA).
Cell Proliferation Ability Detection
Mocks and inhibitors were transfected into cells, and a blank well with no cells and only culture medium was set as a blank control group. Twenty-four hours after transfection, cells were seeded in a 60-well plate at 1 × 103 (200 µL). In addition, the MD microplate reader (USA) was used to detect OD450 nm, and SoftMax pro 7.0 was used to calculate the final results. Proliferation levels were further determined. Generally, 10 mm of DCFH-DA is added to cultured cells for half an hour.
Statistical Method
Data were displayed as mean ± standard deviation (SD) and assessed by Student’s t-test. p < 0.05 means a difference.
Results
Synthesis and Characterization of GLA-loaded Liposomal Albumin Particles
As shown in Figure 1, the average particle sizes of each formulation were similar, and all show positive zeta values, indicating the successful CS coating GLA process. Observed by electron microscope, the nanoparticles were uniform in size and showed no abnormalities (Table 1).
Real-time Polymerase Chain Reaction (PCR) Primers and Primer Sequences.
Transmission Electron Microscope Photo of Albumin Particles Loaded with Glutaraldehyde (GLA) Liposomes (Scale Bar 1:200 nm) (Magnification ×30,000).

Effects of GLA-loaded Liposomal Albumin Particles Inducing p53 on Cervical Cancer Cells
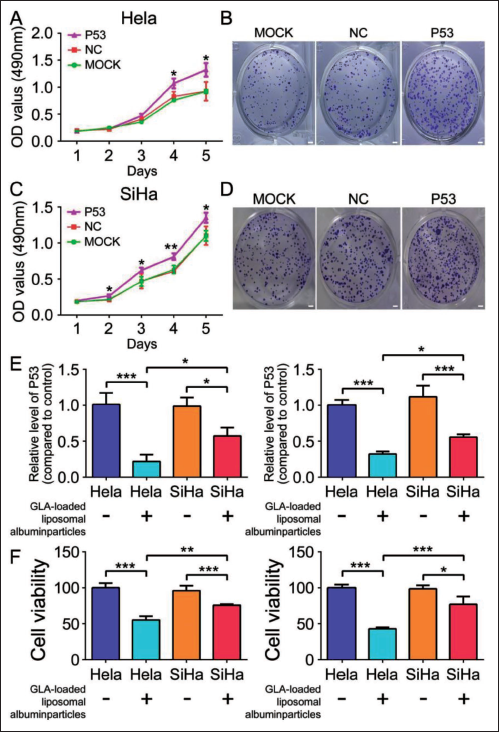
Lung cancer cells were transfected with P53 overexpression plasmid to observe whether overexpression of P53 could reverse cervical cancer cell proliferation (Figure 2A–D). Upregulation of P53 aggravates cell proliferation expression. The RT-PCR results showed that stimulation with GLA-loaded liposomal albumin particles decreased P53 expression, as shown in Figure 2E. Importantly, GLA-loaded liposomal albumin particles could reverse P53-induced cervical cancer cell death proliferation (Figure 2F). Overall, GLA-loaded liposomal albumin particles can inhibit the development and progression of cervical cancer cells induced by P53.
GLA-loaded Liposomal Albumin Particles Mediate NF-κB Transduction by Regulating P53
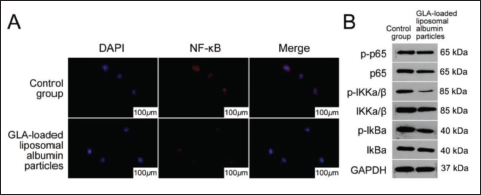
Given that NF-κB is an important signaling pathway that regulates expression in cervical cancer, we speculate that NF-κB is negatively correlated with GLA-loaded liposome albumin particles in cervical cancer tissues. As expected, compared with the control group, the NF-κB immunofluorescence detection signal expression was inhibited in most GLA-loaded liposomal albumin particle groups, as shown in Figure 3A. p-IκBα and p-P65 expression in cervical cancer tissues was down-regulated after GLA-loaded liposome albumin particles were screened by fluorescence detection, as shown in Figure 3b. Therefore, GLA-loaded liposome albumin particles can inhibit NF-κB signaling activation.
GLA-loaded Liposomal Albumin Particles have Obvious Effects on Inhibiting Tumor Proliferation in vivo
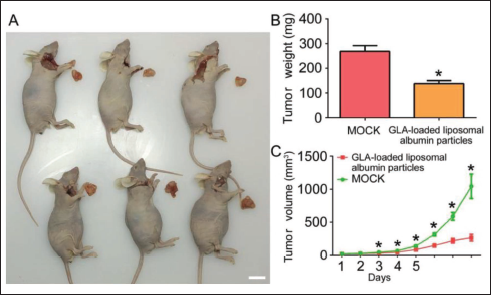
Based on the above findings, we speculate that GLA-loaded liposomal albumin particles can promote tumor growth in vivo. To further support our hypothesis, we established a xenograft tumor model by injecting infected SiHa cells into nude mice to investigate whether GLA-loaded liposomal albumin particles have similar inhibitory long-term effects in vivo. GLA-loaded liposomal albumin particles inhibited the growth of xenograft tumors in nude mice (Figure 4A). Compared with the control group, both tumor weight (p < 0.05) and volume (p < 0.05), GLA-loaded liposome albumin particles inhibited tumor cell proliferation (Figure 4b and C). In general, GLA-loaded liposome albumin particles inhibited tumor cell proliferation. Body albumin particles inhibit cells in cervical cancer and significantly promote tumor growth, suggesting that GLA-loaded liposomal albumin particles play an inhibitory role in the growth of cervical cancer.
Discussion
Cervical cancer is highly malignant, and its mechanisms of occurrence and metastasis have not yet been clarified. GLA has been reported to regulate cell death, cycle arrest, metastasis, stress response, and proinflammatory response in cancer cells, especially in solid tumors (Gao et al., 2018; Kori & Yalcin Arga, 2018). Its cell permeability and tendency to precipitate in aqueous solutions result in its diminished antitumor effect. With the development of nanotechnology, it has become possible to use it as a carrier drug because it can significantly enhance the efficacy and reduce toxic and side effects (Wu et al., 2018). Therefore, carbon nanotubes, polymers, inorganic metal nanolayers, and graphene oxide have been used in the past as nanocarriers for different drugs (Hardy et al., 2023). It is attractive due to its ease of synthesis, controllable particle size, and large drug-loading surface area. Although without additional surface coating, in order to exhibit dose-dependent toxicity, good functionalization with a biocompatible coating, such as PEG, was found to reduce apparent toxicity (Vaughn et al., 2022). Other studies have explored GLA functionalized with PEI for deoxyribonucleic acid (DNA) plasmid and siRNA delivery (Kim et al., 2020). Previous studies have found that PEG and PEI-functionalized GLA can enhance gene delivery (O’Young et al., 2011). Some studies have used GLA to carry liposomal albumin and determined that the maximum drug loading capacity of 100 µg/mL GLA to carry liposomal albumin is 56 µM. Next, to verify the enhanced intracellular transport of GLA-loaded liposomal albumin particles, we evaluated the cellular uptake of labeled GLA-loaded liposomal albumin particles. Our results show that GLA-loaded liposomal albumin particles significantly enhanced cellular uptake and resulted in a significant increase in GLA volume. Consistently, we showed that GLA-loaded liposomal albumin particles are also used as a potential alternative to interventional therapy in cervical cancer (Ghasemi et al., 2019).
p53 can promote cervical cancer cell behaviors and GLA-loaded liposome albumin particles can reverse p53’s effect. Overall, GLA-loaded liposomal albumin particles can inhibit the progression of cervical cancer induced by P53. NF-κB is required for K-ras-induced cervical cancer. Some studies using low-passage immortalized human cervical cancer cell lines and control cells found that NF-κB activity was upregulated in K-ras-transformed cells. Furthermore, deletion of NF-κB p65/RelA subunit inhibited cervical cancers, and p65/RelA-deficient tumors had a greater number of apoptotic cells and lower tumor grade (Tilborghs et al., 2017; Zhang et al., 2020). In most studies, genetic alterations enhance signaling mediated by HER family receptors. There are studies exploring the association between GDNF and NF-κB in cervical cancer cells. In cell lines, GLA signaling stimulation leads to NF-κB activation, consistent with NF-κB’s role in the enhanced tumorigenic capacity of cell lines due to elevated GLA levels in these cells (Ou et al., 2020). In addition, GLA blocks NF-κB activation, reflecting the effect of a specific inhibitor of IKK (Zhang et al., 2019). Our study data indicate that GLA-loaded liposomal albumin particles inhibit NF-κB activity when expressed. It has also been reported before that both GLA-loaded liposomal albumin particles and NF-κB have antiapoptotic effects. Rapidly growing tumors often contain large numbers of apoptotic cells.
Conclusion
In conclusion, this study partially clarified the antitumor relationship between GLA-loaded liposomal albumin particles and cervical cancer, inhibiting NF-κB activity when GLA-loaded liposomal albumin particles were expressed. Induction of NF-κB in tumors may be a powerful strategy allowing GLA-loaded liposomal albumin particles to avoid apoptosis and possibly after cytotoxic chemotherapy. This study demonstrates that GLA-loaded liposomal albumin particles may participate in the development and progression of human cervical cancer cells.
Footnotes
Abbreviations
GAPDH: Glyceraldehyde-3-phosphate dehydrogenase; GLA: α-Galactosidase A; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide; NF-κB: Nuclear factor kappa B; RT-PCR: Real-time reverse transcriptase polymerase chain reaction.
Acknowledgments
The authors gratefully acknowledge the Yulin Traditional Chinese Medicine Hospital Laboratory for providing the necessary equipment for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the ethnic committee of Yulin Traditional Chinese Medicine Hospital.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
