Abstract
Background
One of the leading causes of acute brain injury is cerebral ischemia/reperfusion (I/R) injury. It is an ailment that occurs when the brain does not receive enough oxygen-rich blood supply, which damages brain cells. Oxidative stress is recognized as a significant mechanism that causes I/R injury and is a critical focal point for therapy. Apoptosis and inflammation are all pathogenic processes underpinning I/R damage. Eucalyptol (EU), a plant-derived substance, protects neuronal damage. We postulate that EU inhibits apoptosis and oxidative stress after focal cerebral I/R damage in mice.
Objectives
Thus, the present work sought to examine the neurological protective benefits of EU in mice models of cerebral ischemia/reperfusion (CI/R) injury.
Materials and Methods
The measurement of neurological function, infarct volume, and the amount of water in the brain were evaluated subsequently. The brain tissue was examined to assess the activities of oxidative stress, inflammatory cytokines, and matrix metalloproteinases (MMP) levels, and histopathology was determined. Using enzyme-linked immunosorbent assay (ELISA), apoptotic marker expression was used to measure the levels of B-cell lymphoma 2 (Bcl-2), caspase-3, and Bcl-2 associated X-protein (Bax) proteins that exist as indicators of apoptosis.
Results
EU treatment malondialdehyde (MDA) levels decreased while glutathione (GSH), superoxide dismutase (SOD), A-TOC, and catalase (CAT) activity increased. Following EU therapy dramatically reduced cerebral dysfunction, which was supported by reduced histological damage in I/R injury. Furthermore, with lower levels of MMP-2 and 9, EU treatment could be a novel approach for preventing CI/R injury. EU-treated mice had a notable reduction in cleaved Bax, caspase-3, and increased Bcl-2 protein activities. EU therapy shields the brain against CI/R injury by inhibiting cell death.
Conclusion
According to the present study’s findings, EU is a promising neuroprotective agent that can be used to treat CI/R injury in mice. By focusing on these aspects, nurses can provide comprehensive care to patients involved in studies on the effects of EU on CI/R injury, promoting better outcomes through careful monitoring, supportive interventions, and education.
Introduction
Stroke is a major global health hazard that threatens both the economy and people’s well-being worldwide. It is the most significant cause of mortality and lasting impairment (Liu et al., 2009). The defining feature of this condition is the obstruction of blood vessels caused by the occurrence of an occluded thrombus in the brain, which results in insufficient blood and oxygen flow (Jiang et al., 2014). The most effective treatment for patients suffering from a stroke caused by ischemia is to restore blood circulation to the brain as quickly as possible. Reperfusion carries a hidden risk of harming brain tissue, including neuronal death, blood–brain barrier (BBB) leakage, hemorrhagic transformation, and cerebral edema (Ma et al., 2014; Yao et al., 2014). This condition, known as cerebral ischemia/reperfusion (CI/R) injury, is facilitated through a succession of processes such as glutamate excites toxicity, inflammation, oxidative stress, and apoptosis ultimately resulting in cell death (Tabassum et al., 2015; Zhang et al., 2013).
The resumption of blood circulation after a period of CI and hypoxia causes CI/R injury (Wang et al., 2021). Characterized by a high death and disability rate, CI/R injury adversely affects patients’ health while also significantly reducing their standards of life (Tian et al., 2020). Nevertheless, because of the intricate mechanisms underlying the ischemia/reperfusion (I/R) process, recombinant tissue plasminogen activator (rtPA) exists as the exclusive drug approved by the Food and Drug Administration (FDA) for treating stroke caused by ischemia to date. The usefulness of this medicine in clinical therapy is severely limited because of its bounded pharmacological scope and heightened danger of subarachnoid hemorrhage (Micieli et al., 2009). The exploration of the pathogenic causes and the establishment of safe and efficient neuroprotective medicines to combat the effects of ischemic stroke are therefore of paramount therapeutic importance.
Natural products have been important parts of traditional medicine since ancient times, with various promising health effects (Cerqua et al., 2022; Ferrarini et al., 2022; Rai et al., 2019; Şahin et al., 2023). Eucalyptol (EU), a natural monoterpene in eucalyptus oil, has potential therapeutic benefits for treating cerebral I/R injury. Its neuroprotective properties reduce oxidative stress, inflammation, and apoptosis, preserving brain tissue and improving neurological function. EU’s mechanisms include scavenging free radicals, enhancing antioxidant enzyme activity, inhibiting proinflammatory cytokines, and improving cerebral blood flow. There has recently been an increase in interest in the use of natural products therapeutically for I/R, particularly those that are plant-based (Açar et al., 2023; D’Avino et al., 2023; Mostofa et al., 2024; Singh et al., 2018). Eucalyptus species are one such plant material that has gained the interest of academics and environmentalists all over the world for their enormous source of wood and oil. The terpenoid oxide, EU existing with a chemical name 1,8-cineole, extracted from eucalyptus species, is extensively known for its anti-inflammatory, antibacterial, and antioxidant characteristics. A prior study revealed that EU alleviated cerulein-induced acute pancreatitis by reducing stress caused by oxidation in mice (Lima et al., 2013). A clinical trial found that inhaling eucalyptus oil, which contains EU, can reduce inflammation in individuals who have undergone complete knee replacements (Jun et al., 2013). Because of its secretolytic characteristics, eucalyptus has been utilized to treat respiratory disorders (Bastos et al., 2011). EU, when inhaled, reduces inflammation in the airways of guinea pigs challenged with ovalbumin. According to reports, EU helps reduce asthmatic inflammation (Zhou et al., 2007). EU, having the potential to cross BBB, may directly regulate brain receptors and enzymes (Moss & Oliver, 2012). EU may therefore have significant uses in acute lung inflammation.
As part of our investigation, we developed a CI/R injury-triggered mice model to determine if EU can reduce neurological impairment by acting as an antioxidant and preventing apoptosis. Further research was executed to validate the efficacy of EU in treating CI/R injury and to determine whether it can be used to decrease neuronal degeneration, cellular oxidative stress, and inflammation in the brain. EU, a medication used to treat CI/R injuries, has various mechanisms that help reduce oxidative stress and prevent cell death. It is essential to monitor the patient’s neurological status, manage oxygenation and perfusion, administer EU, provide nutritional support, maintain fluid balance, prevent secondary complications, and initiate rehabilitation activities.
Materials and Methods
Materials
Eucalyptol (Eucal) and nimodipine were purchased from Sigma–Aldrich (USA). We bought tumor necrosis factor (TNF)-α, interleukin-1 beta (IL-1β), interleukin (IL)-6, and IL-10 enzyme-linked immunosorbent assay (ELISA) kits from R&D Systems (Minneapolis, MN). The investigations employed only analytical-grade chemicals; all other chemicals were purchased from standard suppliers.
Animals
The applicable national laws and institutional guidelines for animal use and care were followed, and mice were procured from the Approved Animal Center with the appropriate ethical approval for animal experimentation study. Featuring a 22–24℃ temperature, a 12-h light–dark cycle, and a relative humidity of 60 ± 5%, the animals were acclimated to their surroundings and were given unlimited accessibility to water and nourishment. The mice’s cage was lined with autoclaved rice husk bedding, which was replaced daily.
An Illustration of Localized Cerebral Ischemia and Reperfusion
Before middle cerebral artery occlusion (MCAO) surgery, the mice were fasted overnight to create models of localized cerebral ischemia. To anesthetize the mice before surgery, intraperitoneal injections of (w/v) chloral hydrate were administered at 37°C. Intraluminal filaments were used to cause focal cerebral ischemia. The mice’s neck was incised midline while it was fixed on a table. The right internal, external, and common carotid arteries were exposed by a skin incision. To establish an occlusion, the Vagus nerve was separated from the common carotid artery on the right. Next, a 6-0 nylon monofilament was administered into the middle cerebral artery from the external carotid artery incision. The occlusion lasted for 1 h. Brain blood circulation was determined by utilizing a laser to confirm successful occlusion (Wang et al., 2021; Zhang et al., 2023). Reperfusion began after 2 h of MCAO by withdrawing the monofilament. After anesthesia, mice were given free access to nourishment and water. The mice were observed for heart rate, temperature, and blood pressure throughout the entire surgical operation. The control group lacked clamping CCA.
Grouping and Treatment
Following MCAO surgery, the mice were separated into four different groups, with the sham-operated animals serving as group I controls. Group II includes MCAO-induced animals (I/R group) who receive no treatment. Group III was induced with MCAO and given 50 mg/kg of EU by oral gavage 2 h after the MCAO procedure. Group IV received oral gavage treatment of 15 mg/kg nimodipine (positive control) 2 h post-MCAO surgery. The I/R group used Zea Longa’s approach to block the internal carotid artery for 2 h, following 24 h of reperfusion. Normal saline was administered to the I/R group intragastrically once a day for 7 days before the surgery. The I/R and sham groups both had identical preoperative care and surgery; however, there was no nylon suture occlusion in the sham group. The treatment phase ended with the mice’s euthanasia, wherein the brain tissue and blood samples were obtained for the ensuing examinations (Zhang et al., 2023).
Assessment of Neuro Infarct Volume, Neurological Deficiency Score, and Brain Water Content
Neuro Infarct Volume
2,3,5-Tetrazolium chloride (TTC) staining was used to assess cerebral infarct volumes. Five equally spaced coronal slices were cut from the mice’s brain tissue. The samples were stained with 1% TTC and sustained at 37°C for 20 min, subsequently with 4% formaldehyde fixation overnight (Zhao et al., 2015). Following incubation, portions of tissue were examined beneath a microscope. The area of infarction was calculated after examining the images with the Image J software. Upon examination, infarcted tissue stayed white and unstained, but the normal tissue turned red (Wu et al., 2019). To determine the overall infarction volume of brain slices, the infarction area was multiplied by the thickness of each slice.
Neurological Deficit Score
An examiner blinded to the experimental groups evaluated neurological impairment scores 24 h after MCAO using a five-point scale based on the Zea longa standard.
Grade 0: The mice exhibited normal behavior and showed no signs of neurological impairments. Grade 1: Limited ability to fully stretch the forelimb on the other side. Grade 2: Rotating to the right (contralateral side). Grade 3: Tumbling across the other side whilst trying to move forward. Grade 4: Unable to step independently and absence of consciousness (Li et al., 2024). The severity of motor motion injury is proportional to the neurological deficiency score.
Brain Water Content
After reperfusion, the water content of the brain was assessed. The brain tissues extracted from mice in each group were measured using an electronic scale to determine the moist weight of the entire brain. To eliminate the water content or dry weight, the tissues of the brain were desiccated to a consistent weight in an oven set at 105°C. The percentage of brain water content was obtained by multiplying ([wet weight − dry weight]/wet weight) by 100 (Wu et al., 2019).
Determination of Superoxide Dismutase (SOD), Catalase (CAT), Glutathione (GSH), Total Antioxidant Capacity (T-AOC), and Malondialdehyde (MDA) Activities
The activities of SOD, GSH, CAT, T-AOC, and MDA were measured using ELISA kits. The brain tissues were promptly taken out of each group and cleaned in cooled saline. A 10% (w/v) homogenate was then created by homogenizing the brain tissues for 20 min in an ice-cold saline solution. The supernatant was then collected and tested for MDA, SOD, GSH, CAT, and T-AOC using a microplate reader, following the directions included with the test kits. The assay findings are represented in nmol/mg protein or U/mg protein (Zhao et al., 2015).
Estimation of Pro and Anti-inflammatory Cytokines
The study used a commercial ELISA kit to assess cytokines that promote inflammation, such as TNF-α, IL-1β, and IL-6, and those that prevent inflammation, like IL-10 from the brain samples of several experimental groups. The assay was carried out as directed by the instructions in the kit. The absorbance of both the samples and standards was determined at 450 nm (Zhang et al., 2023).
The Estimation of Matrix Metalloproteinase Levels
Using the purchased ELISA kits, matrix metalloproteinases (MMP)-2 and MMP-9 levels in the brain tissues of various experimental groups were determined. The experiment followed the manufacturer’s directions. The experiment was conducted in three replicates. MMP levels were determined using a standard curve based on standard concentration absorbance values (Wang et al., 2021).
Estimation of Apoptotic Proteins
The mice were chosen at random and beheaded after 24 h of reperfusion. Brain tissues were rapidly taken from mice and kept at ‒20°C on ice. The acquired brain cells were quantified and processed in a 1:10 (w/v) ice-cold whole-cell lysis buffer. The concentrations of B-cell lymphoma 2 (Bcl-2), Bcl-2 associated X-protein (Bax), and Caspase 3 were determined in the samples using an ELISA kit (Zhao et al., 2015).
Histological Examination
The brain tissues were sliced with 5-µm thickness, and stained with hematoxylin and eosin (H&E). Stained tissues were measured under a Light microscope (Olympus, Tokyo, Japan) using 10× magnification (scale bar indicates 10× magnification) (Zhao et al., 2015).
Statistical Analysis
Results were shown as mean ± standard deviation (SD). The statistical assessment was accomplished with GraphPad Prism. A one-way analysis of variance (ANOVA) was performed to compare differences across groups. The differences between the control and sample means were considered statistically significant if p < 0.05.
Results
Eucalyptol’s Effect on Infarct Volume, Neurological Deficiency Score, and Brain Water Content
Figure 1a shows a significantly higher infarct in the I/R group (35–40%) when compared to the sham operation group, which showed negligible volume. When the EU-treated group was examined, it was discovered that the infarct was significantly reduced in size, accounting for 20% of the total volume. The treatment of EU and nimodipine (NIM) dramatically reduced the percentage of infarct area. The mice in the sham group received a score of 0 out of 4, signifying that they had no neurological damage. The I/R group had a much higher score (four) than the sham group, which indicates the severity of brain injury. The EU and nimodipine-treated groups’ scores significantly fell to 2 and 1, respectively, when compared to the I/R group, indicating a considerable improvement in neurological function (Figure 1b).
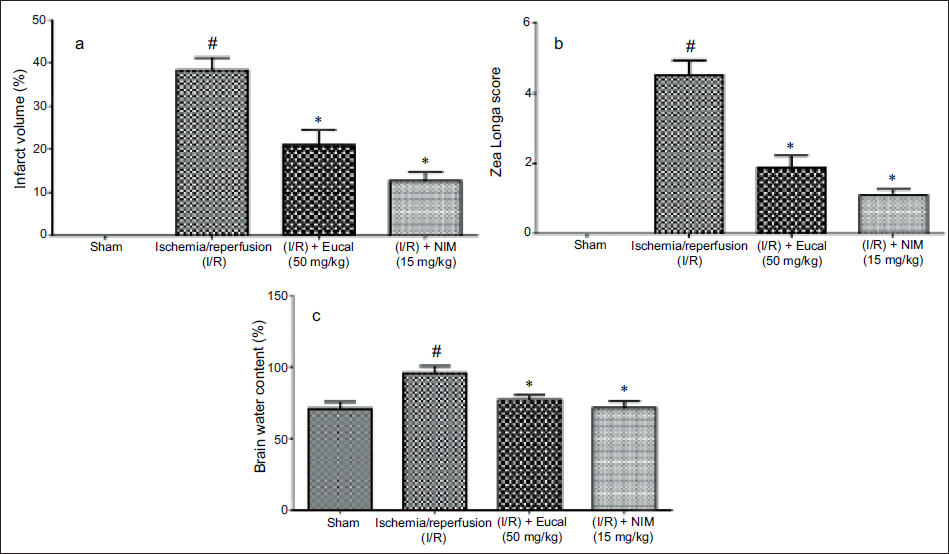
While evaluating the brain water content, it was obvious, as shown in Figure 1c, that 65–70% water was seen in the sham-operated mice brain; however, it was significantly elevated to 90–100% in group II mice. EU therapy dramatically reduced water content in the brain of MCAO-operated mice, with an effect similar to that of the positive control nimodipine. As a result, it was clear that the EU was beneficial in treating I/R brain injury.
Eucalyptol Alleviates Oxidative Stress and Antioxidants in Brain Tissues
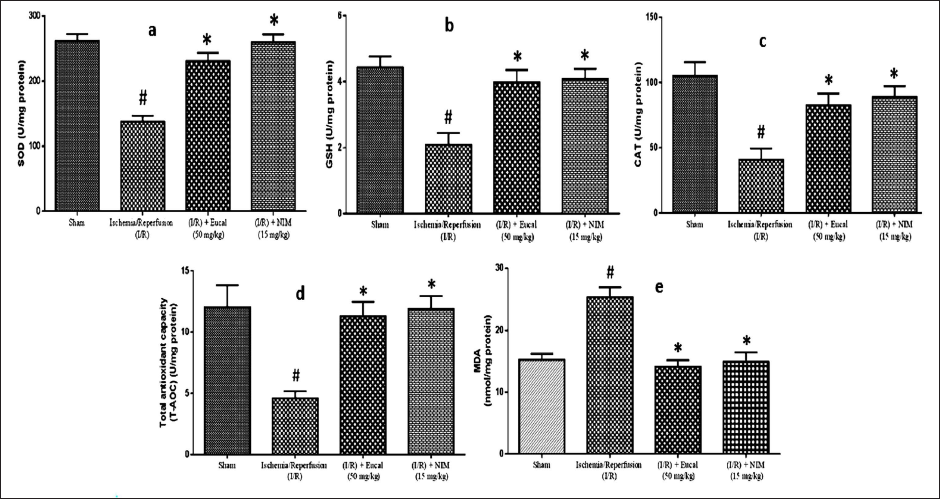
Our study evaluated the antioxidant state of EU-treated MCAO-performed mice’s brain tissue to that of other groups. In mice, MCAO significantly reduced the level of antioxidants compared to sham-operated control mice. EU therapy elevated the levels of oxidative stress indicators (SOD, CAT, GSH, and T-AOC) in MCAO-performed animals, as shown in (Figure 2a–d). EU treatment in mice had a similar effect on antioxidant levels as nimodipine treatment in the positive control group. Untreated MCAO animals had higher levels of MDA compared to control mice. Both EU and positive control-treated mice demonstrated a considerable decrease in MDA levels, as seen in Figure 2e. EU was found to increase SOD, GSH, CAT, and T-AOC activity while decreasing MDA activity.
Impact of Eucalyptol on Inflammatory Cytokines in Mice with Cerebral Ischemia
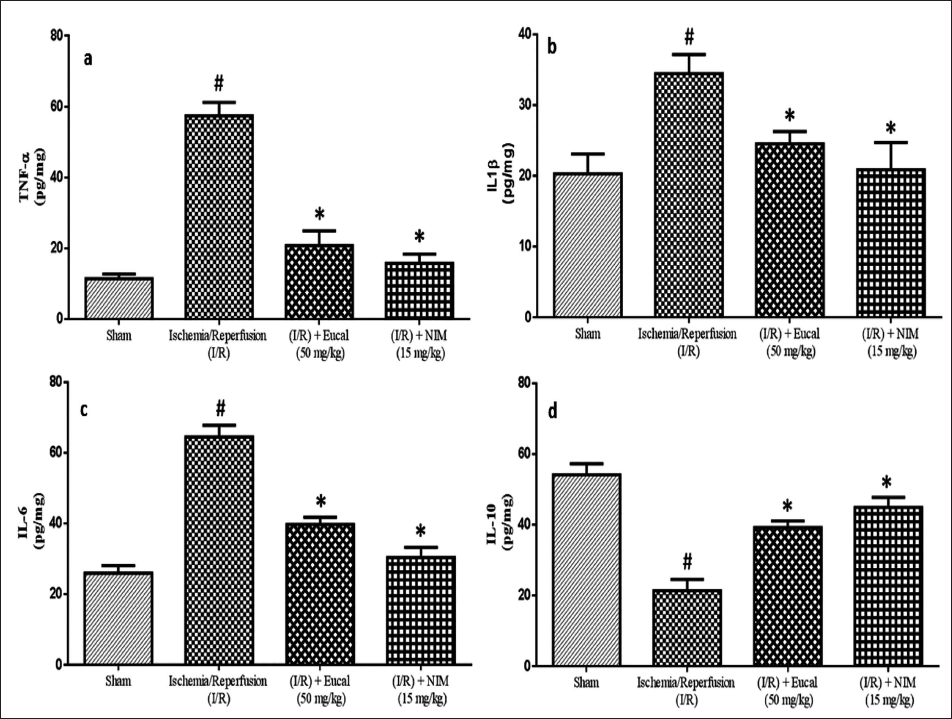
Figure 3 displays the levels of proinflammatory and anti-inflammatory cytokines in mice treated with EU, nimodipine, and MCAO. ELISA results showed that the I/R group had higher levels of cytokines TNF-α, IL-1β, and IL-6 compared to control group I, whereas IL-10, the anti-inflammatory cytokine levels decreased. The EU and nimodipine groups had significantly lower levels of TNF-α, IL-1β, and IL-6 compared to the I/R group, but IL-10 levels increased significantly (Figure 3a–d). EU was found to effectively reduce inflammation caused by I/R damage, comparable to nimodipine at a specific concentration.
Eucalyptol’s Effect on the Levels of Matrix Metalloproteinase in Mice Induced with Cerebral Ischemia
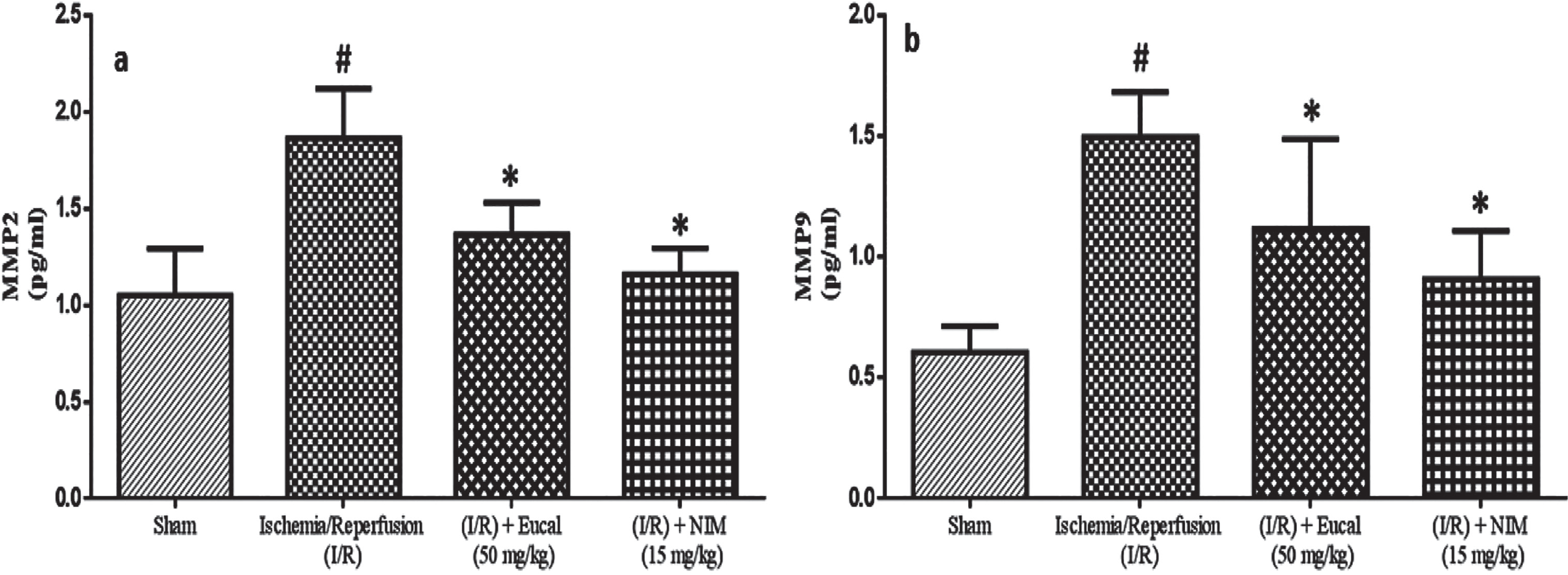
To assess the efficacy of matrix metalloproteinase inhibitors in treating stroke, we measured MMP levels in EU-treated mice and compared them to other experimental groups (Figure 4a and b). The levels of MMP-9 and MMP-2 were significantly elevated in the untreated mice group; however, the EU-treated group lowered the MMP levels in the tissues.
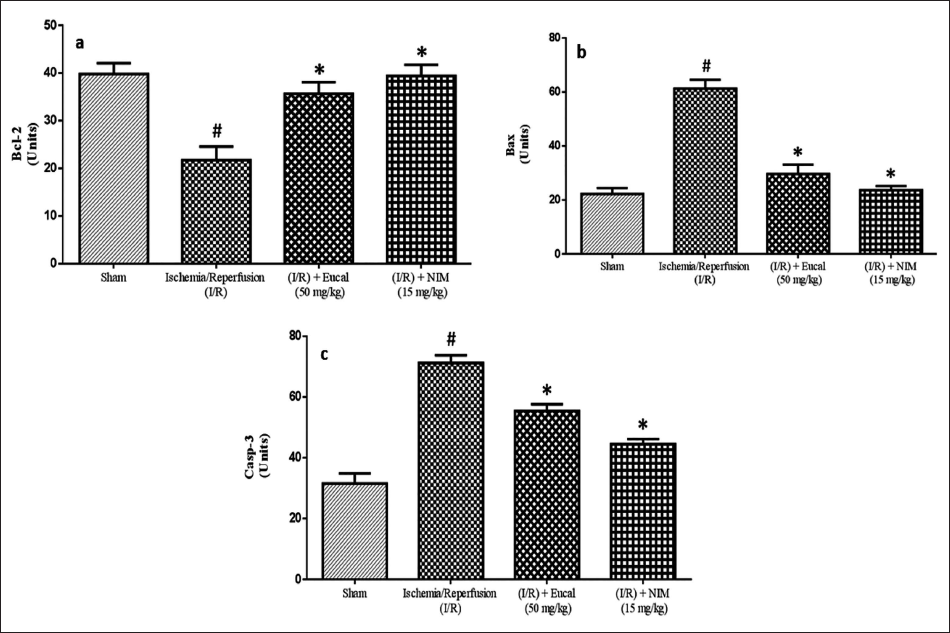
Effects of Eucalyptol on Brain Tissue Apoptosis-related Factors
The proapoptotic protein (Bax and Caspase-3) and antiapoptotic protein (Bcl-2) levels in all four groups were assessed by using an ELISA kit. Group II had elevated levels of caspase-3 and Bax, but lower Bcl-2 levels than the sham group. EU therapy dramatically boosted Bcl-2 levels while inhibiting caspase-3 and Bax activity (Figure 5a–c).
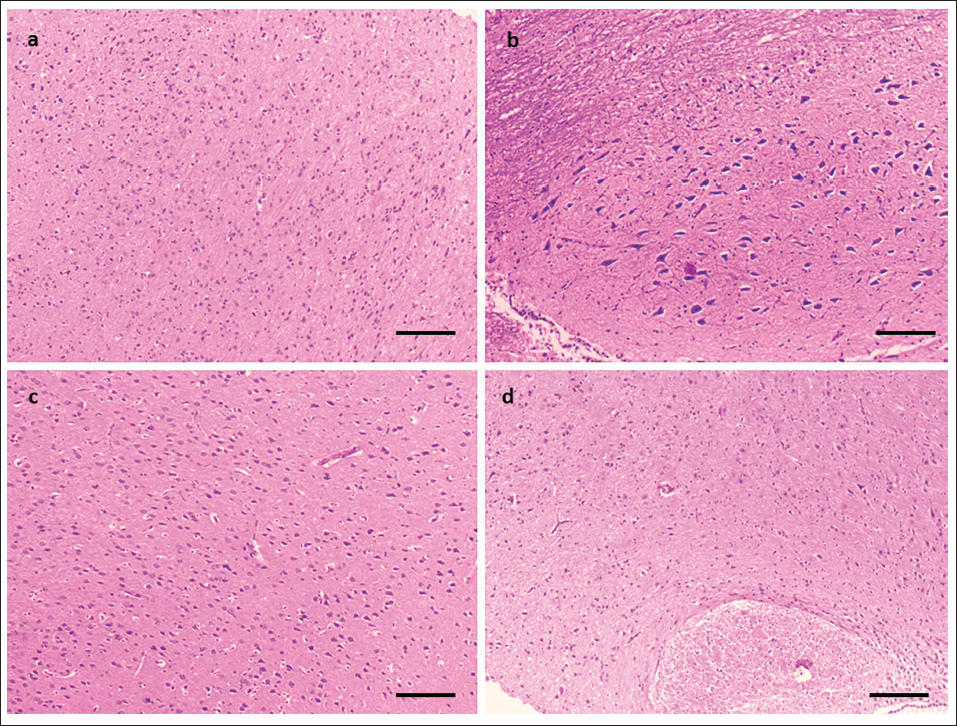
Effect of Eucalyptol on Histological Changes in Brain Tissues
H&E staining was applied to examine histological alterations in the neurons of mice brains among different groups. As demonstrated in Figure 6a, group I was closely aligned and well-structured, with obvious light blue staining concentrated on the nucleus. In group II, Figure 6b, neurons displayed shrunken nuclei and a disorganized array with deep blue staining. Figure 6c shows that EU therapy reduced apoptotic cells in the area, similar to positive control. Furthermore, the damage was greatly reduced, while the number of normal neurons rose. Overall, the EU appears to efficiently prevent I/R-induced neuronal death.
Eucalyptol (Eucal) affects Histological Alterations in the Hippocampus. A Photograph of Hematoxylin and Eosin (H&E) Staining in the Area of the Hippocampus. (a) Depicts the Sham Group, (b) Ischemia/Reperfusion (I/R), (c) EU with 50 mg/kg Group, and (d) Nimodipine with 15 mg/kg Group (Scale Bar indicates 10× Magnification).
Discussion
Blood flow disruption brought on by thrombus or emboli clogs blood arteries, resulting in cerebral ischemic stroke (Zhang et al., 2023). Following cerebral ischemia, blood flow recovery causes major functional and anatomical changes leading to CI/R injury. This can cause lasting effects such as brain cell death and edema (Wang et al., 2021). The neurological, histological, and biochemical alterations, as well as the processes of cerebral ischemia injury in mice, are all studied in great detail using this technique (Zhao et al., 2015). A less intrusive and dependable model of transitory regional ischemia used in classical stroke research is called MCAO. Research on MCAO-related localized cerebral I/R injury in animal models is crucial. Studies on EU have shown that it possesses antibacterial, anti-inflammatory, antioxidant, analgesic, and spasmolytic properties in treating several illnesses, such as sinusitis, rhinitis, influenza, colds, and other respiratory infections (Seol & Kim, 2016).
Assessing the efficacy of cerebrovascular medications in the management of ischemic brain injury requires consideration of both the infarct volume and neurological deficit score (Zhao et al., 2015). Based on our findings, the I/R group’s infarct volume was significantly higher than that of the sham group. EU dramatically reduced I/R-induced infarct volumes while also improving neurological scores. Additionally, brain water content was drastically lowered, indicating the efficacy of EU against brain injury. Research indicates that administering natural plant extracts at an early stage can improve CI/R injuries (Wang et al., 2021). Eucalyptus globulus extracts have also been demonstrated to shield SH-SY5Y cells from oxidative stress caused by H2O2 by increasing GSH levels, antioxidant enzyme activity, and cell viability while reducing lipid peroxidation and reactive oxygen species (ROS) production. Hence, these extracts have the potential to serve as a source of antioxidants with potential health advantages against oxidative stress, as well as a raw material for dietary, pharmacological, and nutritional supplements (González-Burgos et al., 2018). In this work, we found that treatment with EU dramatically boosted GSH, SOD, T-AOC, and CAT activities while decreasing MDA levels. These findings imply that EU shields the brain from CI/R injury through antioxidant activity. Supplementary products from gut microbiota are crucial for brain health and disease processes, with dysbiosis leading to inflammation and brain damage. Probiotics can reduce injury severity and enhance neuroprotective SCFAs. Understanding these interactions could lead to therapeutic strategies targeting the gut–brain axis (Ağagündüz et al., 2022, 2023).
It has been demonstrated that EU has impacts on several inflammatory diseases, such as pancreatitis, respiratory disorders, and cardiovascular and neurological diseases, in addition to lessening colon damage. Studies have demonstrated that EU has analgesic properties in an inflammatory model and is a powerful inhibitor of proinflammatory cytokines, including TNF-alpha and IL-1 beta. The findings of previous investigations demonstrate that EU has a potent inhibitory effect on both TNF-α and IL-1β (Bhowal & Gopal, 2015). Also, the EU anti-inflammatory properties may contribute to its preventive effects against neurodegenerative disorders (Seol & Kim, 2016). The current study result shows decreased levels of IL-6, TNF-α and IL-1β and increased levels of IL-10 which was supported by earlier research, which found that 1,8-cineole lowers proinflammatory factors significantly. Matrix metalloproteinases are proteolytic enzymes that destroy the extracellular matrix. Brain edema, breakdown of BBB, inflammation, and bleeding are all brought on by disruption in MMPs. When an acute stroke situation arises, MMP-2 is first activated, disrupting the extracellular matrix proteins and then triggering MMP-9, which in turn causes an influx of inflammatory cells (Zhang et al., 2023). EU therapy reduced MMP-9 and 2 levels in rats that were performed in the MCAO, perhaps avoiding ischemic-induced BBB breakdown and decreasing cerebral edema.
Apoptosis following I/R injury significantly contributes to stroke-related brain injury (Li et al., 2024). Studies show that the caspase family promotes and executes apoptosis in mammalian cells. One of these, caspase-3, is essential to the caspase cascade “waterfall” as a downstream apoptotic protease (Li et al., 2024). Caspase-3 serves a vital part in apoptosis signaling and regulates neuronal apoptosis (Wu et al., 2019). According to our analysis, the EU-treated group’s levels of active caspase-3 and Bax are considerably decreased in comparison to the vehicle group. Bcl-2 levels were significantly higher in the EU-treated group compared to the vehicle group. This demonstrates that the EU provides neuronal protection through antiapoptotic pathways. H&E staining results revealed that histologic features, such as nucleolus karyopyknosis, were present in the neurons of group II. The administration of EU reduced the number of deteriorated neurons while significantly increasing the number of undamaged neurons.
EU has been found to have potential clinical implications in treating cerebral I/R injury. It reduces infarct volume, improves neurological function, reduces brain edema, and has antioxidant properties. EU protects brain cells from oxidative damage, enhancing neuroprotection and recovery. It also modulates the inflammatory response, reducing inflammation-induced damage and promoting healing in stroke patients. EU also inhibits MMPs, associated with BBB disruption and neuronal damage. These findings suggest EU potential as a therapeutic agent for stroke patients.
The study highlighted EU’s neuroprotective benefits by analyzing the activities of Bcl-2, Bax, and caspase-3 proteins, as well as oxidative indicators such as MDA, SOD, GSH, T-AOC, and CAT activities in mice subjected to MCAO. However, due to the diversity in the efficacy of EU, its specific molecular mechanism in improving CI/R injury still needs to be verified by subsequent studies, thus ensuring its safety and efficacy in clinical prevention and treatment of CI/R injury. It is crucial to educate the patient and family about the condition, emphasize medication adherence, and suggest lifestyle modifications to improve overall health. Accurate record-keeping and effective communication are essential for providing comprehensive care. By focusing on these aspects, nurses can provide comprehensive care for patients involved in studies on EU’s effects on CI/R injury, promoting better outcomes through careful monitoring, supportive interventions, and education.
Conclusion
Cerebral ischemic stroke is a fatal condition that leads to poststroke complications and long-term disability for sufferers. Currently, treating stroke victims is costly and sometimes leads to difficulties over time. This study examined the impact of EU, a bioactive chemical, on MCAO-performed mice. EU dramatically reduced infarct volume and brain water content by increasing antioxidant levels and lowering MMPs. By suppressing the synthesis of proinflammatory cytokines via TNF-α, IL-1β, and IL-6, EU also lowers oxidative stress by controlling signaling pathways and scavenging free radicals. It also suppressed histological changes. Our findings demonstrate that EU ameliorates cerebral ischemia in mice considerably. Additional study on its efficacy and safety is required before subjecting the chemical to additional trials and formulation as an anti-ischemic medication. In the future, nurses can enhance patient outcomes in EU-induced CI/R injury studies by providing comprehensive care, monitoring, supportive interventions, and education.
Footnotes
Abbreviations
ANOVA: Analysis of variance; Bax: Bcl-2 associated X-protein; BBB: Blood–brain barrier; Bcl-2: B-cell lymphoma 2; CAT: Catalase; CI/R: Cerebral ischemia–reperfusion; EBI: Early brain injury; ECL: Enhanced chemiluminescence; ELISA: Enzyme-linked immunosorbent assay; EU: Eucalyptol; FDA: Food and Drug Administration; GSH: Glutathione; H&E: Hematoxylin and eosin; IL: Interleukin; IL-1β: Interleukin-1 beta; I/R: Ischemia/reperfusion; LPS: Lipopolysaccharide; MCAO: Middle cerebral artery occlusion; MDA: malondialdehyde; MMP: Mitochondrial membrane potential; ROS: Reactive oxygen species; rtPA: Recombinant tissue plasminogen activator; SAH: After subarachnoid hemorrhage; SD: Standard deviation; SOD: Superoxide dismutase; T-AOC: Total antioxidant capacity; TNF: Tumor necrosis factor; TTC: 2,3,5-Tetrazolium chloride.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the Ethics Committee of Shanxi Bethune Hospital (Approval Number: SBQDL-2023-232).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
