Abstract
Background
1,8-Cineole is used in traditional folk medicine to treat various diseases. This study evaluates the protective effects of 1,8-cineole on the respiratory system by investigating its impact on inflammatory factors in animal studies.
Materials and Methods
We conducted searches in PubMed, Medline, Embase, Cochrane, and the Web of Science databases until December 2023. This study utilized Get Data Graph Digitizer 2.26, EndNote, and RevMan 5.4. The random effects model and standard mean difference (SMD) were applied for data analysis.
Results
The results showed tumor necrosis factor-alpha (TNF-α) (SMD: −8.19, 95% CI: −11.30 to −5.08, p < 0.00001), interleukin (IL)-6 (SMD: −7.38, 95% CI: −10.61 to −4.15, p < 0.00001), IL-1β (SMD: −19.18, 95% CI: −32.57 to −5.79, p = 0.005), NF-κB (SMD: −6.26, 95% CI: −7.88 to −4.65, p < 0.00001), neutrophils (SMD: −8.16, 95% CI: −11.65 to −4.67, p < 0.00001), and macrophages (SMD: −5.06, 95% CI: −8.06 to −2.05, p = 0.001).
Conclusion
The study suggests that 1,8-cineole significantly inhibits the expression of inflammatory factors, indicating its potential for further study and development.
Introduction
The Global Burden of Disease has reported that 545 million people worldwide suffer from chronic respiratory diseases (CRDs), including chronic obstructive pulmonary disease (COPD) (50%) and asthma (50%), representing an increase of 39.8% since 1990. There were 3.2 million deaths from COPD and 495,000 from asthma (GBD Chronic Respiratory Disease Collaborators, 2020; Viegi et al., 2020). COPD has been the leading cause of death from CRDs, while asthma has the highest prevalence among CRDs (GBD Chronic Respiratory Diseases Collaborators, 2023). COPD and asthma are both inflammatory lung diseases (Liu et al., 2024; Polverino & Sin, 2024). Smoking (Gan et al., 2022), viruses (Globenko et al., 2023), and bacteria (Marsh et al., 2019) can all cause and exacerbate inflammatory responses in the respiratory tract. Inflammation is an important cause of physiological, biochemical, psychosocial, microbiological, and comorbidity traits that cause and exacerbate respiratory diseases. Many factors induce the release of inflammatory factors [such as tumor necrosis factor-alpha (TNF-α), interleukin (IL)-6, IL-1β, NF-κB] and mediators (such as neutrophils and macrophages), triggering a series of pathological processes. The injury to endothelial cells and myocytes causes the recruitment of neutrophils and macrophages, leading to the release of oxygen radicals and inflammatory factors, exacerbating airway inflammation and oxidative stress, and worsening lung injury (Arora & Chopra, 2018; Brightling & Greening, 2019; Domagała-Kulawik et al., 2006; Eapen et al., 2017; Gioia et al., 2017).
Natural compounds remain the primary sources of drugs, as scholars purposefully search for compounds with specific medicinal properties, thereby contributing to the discovery of drugs from natural products (Zhang et al., 2020). Based on previous reports, 1,8-cineole (Petry et al., 2024), Mucuna pruriens (Yadav et al., 2017), ursolic acid (Rai et al., 2016), and Withania somnifera (Prakash et al., 2014) have demonstrated significant anti-inflammatory activity. Therefore, the study and development of natural compounds with potential anti-inflammatory effects and the exploration of the relationship between natural compounds and respiratory diseases are also worthy of further study.
1,8-Cineole, also known as eucalyptol, is the main component of eucalyptus oil. It is mainly extracted from Eucalyptus globulus of the Myrtaceae family [E. globulus (Labill.)], Camphor of the Lauraceae family [Cinnamomum camphora (L.) Presl], and other plants of the same genus (Cai et al., 2021; Elaissi et al., 2012; Külheim et al., 2011). CAS: 470-82-6, molecular formula is C10H18O. It is a kind of camphor-like special fragrance, insoluble in water, and a colorless liquid cyclic monoterpene compound. 1,8-Cineole widely exists in many Chinese herbal medicines. It has been found that 1,8-cineole possesses anti-inflammatory and antioxidant properties, particularly significant anti-inflammatory effects for treating respiratory diseases (Juergens et al., 2020), cardiovascular diseases (Wang et al., 2021), and Alzheimer’s disease (Paul et al., 2020). Some clinical randomized controlled trials have reported that 1,8-cineole effectively improves lung function and has a significant effect on respiratory diseases (Juergens et al., 2003; Worth & Dethlefsen, 2012; Worth et al., 2009).
This meta-analysis evaluates the protective effects of the natural compound 1,8-cineole on the respiratory system by probing into its inhibitory effect on inflammatory factors in animal models of respiratory disease.
Materials and Methods
This protocol was registered in the PROSPERO (CRD42022294114).
Literature Search
In this study, a comprehensive search for relevant literature was conducted in the PubMed, Cochrane, Medline, Embase, and Web of Science databases. The search terms used in this study were [(1,8-cineole or eucalyptol) and (respiratory diseases or asthma or chronic obstructive pulmonary disease or pulmonary or COPD or airway or inflammation)]. There were no limitations set regarding race, place, sex, age, ethnicity, or publication dates. The search was conducted from the inception of the databases up until December 2023, and there was no restriction on the language of publication.
Inclusion and Exclusion Criteria
Inclusion Criteria
(a) The study included rats and mice as subjects, without specific requirements for their sex and age. (b) The animal models examined were related to respiratory diseases such as asthma, COPD, and lung injury, with no specific requirements regarding the mode of damage. (c) The intervention of interest involved any form (whole, powder, extract, standardized mixture) of 1,8-cineole (eucalyptol). The dosage, timing, and frequency of the intervention were not restricted. (d) The observed outcomes focused on inflammatory markers associated with respiratory diseases, including TNF-α, IL-6, IL-1β, NF-κB, neutrophils, and macrophages. (e) The study designs included controlled studies with a separate control group. The comparison groups consisted of a model control group and an experimental group (with the reference group being the dose group that showed the best effect, excluding other dose groups). (f) The publications included in the study were not limited by language or publication date but excluded comments and meeting summaries.
Exclusion Criteria
(a) 1,8-Cineole (eucalyptol) was not the main intervention factor. (b) No significant correlation was observed with respiratory diseases. (c) This study was not rats or mice. (d) There was no correlation between outcome indicators and inflammatory factors. (e) Comments and summaries of the Meeting. (f) The literature was of low quality.
Data Collection and Extraction
Data Collection
The screening process entailed two separate reviewers conducting initial screening based on the title and abstract of the articles. Subsequently, the eligible articles underwent full-text screening to determine final inclusion. At each stage, two independent reviewers evaluated the literature independently. Any discrepancies were resolved through discussion or consultation with a senior investigator (Balkhi et al., 2023).
Data Extraction
Two independent reviewers conducted the completion of the review. In case of any discrepancies, discussions were held between the reviewers and senior reviewers to reach a consensus. To address any missing or unclear data, we reached out to the corresponding author via their provided email address. Our process involved first extracting numerical data from figures, tables, and text. If the data was not explicitly provided, we employed Get Data Graph Digitizer 2.26 software to obtain the data from graphics. If the data was continuous, we extracted the data with the highest efficacy.
Assessment of Risk of Bias
Independently assessed risk of bias based on the SYRCLE’s risk of bias tool (Hooijmans, Rovers, et al., 2014). The studies were examined separately by two independent reviewers, including (a) sequence generation; (b) baseline characteristics; (c) allocation concealment; (d) random housing; (e) blinding of performance bias; (f) random outcome assessment; (g) blinding of detection bias; (h) incomplete outcome data; (i) selective outcome reporting; (j) other sources of bias. Evaluation result: “Yes” or “no” or “unclear,” mean “low deviation risk,” “high deviation risk” or “unclear risk of bias,” respectively.
Statistical Analysis
The RevMan 5.4 software (Puvvada et al., 2022) was used for data analysis. The standard mean difference (SMD) was used for the analysis of continuous data. The data reported in a few pieces of literature was standard deviation (SD) rather than standard error of the mean (SEM), so we recorded the data using mean ± SEM. A conversion was performed between SD and SEM (SEM = SD/the square root of sample size). If appropriate, the random effects model was used for all analyses to pool data across the studies for a meta-analysis. Between-study heterogeneity was assessed by using the I2-statistic. This statistic showed that I2 > 30% equated to moderate heterogeneity, I2 > 50% equated to substantial heterogeneity, and I2 > 75% equated to considerable heterogeneity (Higgins & Green, 2011). We used subgroup analysis and sensitivity analysis to investigate potential sources of heterogeneity.
Results
Literature Search Results
A systematic literature search yielded a total of 183 studies related to our research. After eliminating 110 duplicate and overlapping studies, we reviewed the titles and abstracts of the remaining 73 studies. Based on this screening, 33 studies were excluded from further consideration. The full texts of the remaining 40 studies were then assessed for their adherence to the inclusion criteria. As a result, an additional 32 studies were excluded, leaving us with a final selection of 8 studies. The detailed process of literature screening is presented in Figure 1.
The Flow Chart of Literature System Screening.
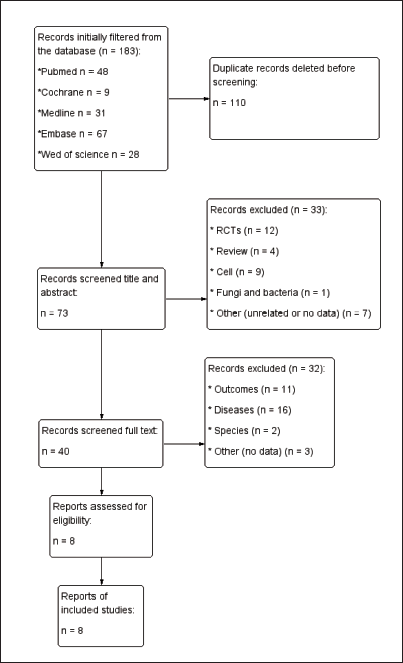
Literature Features
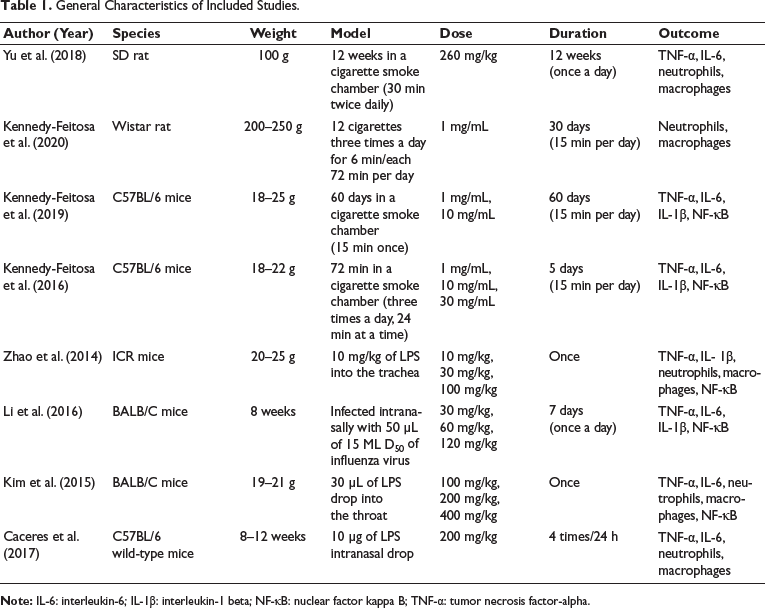
This study incorporated eight studies that met the inclusion and exclusion criteria. A total of 500 animals were included, with 252 animals comprising the experimental group and 248 animals in the control group. The included literature encompassed publications from 2014 to 2023. The study included two rat models and six mouse models, including one study (Kim et al., 2015) from Korea, one study (Caceres et al., 2017) from the USA, three studies (Li et al., 2016; Yu et al., 2018; Zhao et al., 2014) from China, and three studies (Kennedy-Feitosa et al., 2016, 2019, 2020) from Brazil. Four studies (Kennedy-Feitosa et al., 2016, 2019, 2020; Yu et al., 2018) of smoke-induced airway inflammation or lung injury. Three studies (Caceres et al., 2017; Kim et al., 2015; Zhao et al., 2014) of lipopolysaccharide (LPS)-induced acute pneumonia. One study (Li et al., 2016) of virus-induced lung injury. The general characteristics of these eight studies are shown in Table 1.
General Characteristics of Included Studies.
Results of Bias Risk Assessment
After reading the full text of the eight studies, we found that some information reports were not detailed enough, resulting in unclear reports of “Sequence generation,” “Baseline characteristics,” “Allocation concealment,” “Blinding of performance bias,” “Blinding of detection bias,” and “Incomplete outcome data” of all the studies. Only “Selective outcome reporting” and “Other sources of bias” were assessed as low risk of bias in all studies. “Random housing” was assessed as a low risk of bias in seven studies. There was no high risk of bias in eight studies. The bias risk assessment results of the included studies are shown in Supplementary Table S1.
Meta-Analysis Results
Primary Outcome
TNF-α was evaluated in seven studies (Caceres et al., 2017; Kennedy-Feitosa et al., 2016, 2019; Kim et al., 2015; Li et al., 2016; Yu et al., 2018; Zhao et al., 2014). The results showed significant heterogeneity (p = 0.0005, I2 = 75%). Therefore, we used a random-effects model for the meta-analysis. 1,8-Cineole significantly reduced TNF-α levels in the experimental group compared with the control group (SMD: −8.19, 95% CI: −11.30 to −5.08, p < 0.00001) (Figure 2).
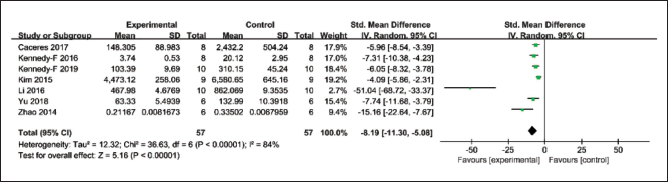
Secondary Outcomes
IL-6 was evaluated in six studies (Caceres et al., 2017; Kennedy-Feitosa et al., 2016, 2019; Kim et al., 2015; Li et al., 2016; Yu et al., 2018). The results showed that 1,8-cineole significantly reduced IL-6 levels in the experimental group (SMD: −7.38, 95% CI: −10.61 to −4.15, p < 0.00001) (Supplementary Figure S1). IL-1β was evaluated in four studies (Kennedy-Feitosa et al., 2016, 2019; Yu et al., 2018; Zhao et al., 2014). The results showed that 1,8-cineole significantly reduced IL-1β levels in the experimental group (SMD: −19.18, 95% CI: −32.57 to −5.79, p = 0.005) (Supplementary Figure S2). NF-κB was evaluated in five studies (Kennedy-Feitosa et al., 2016, 2019; Kim et al., 2015; Li et al., 2016; Zhao et al., 2014). The results showed that 1,8-cineole significantly reduced NF-κB levels in the experimental group compared with the control group (SMD: −6.26, 95% CI: −7.88 to −4.65, p < 0.00001) (Supplementary Figure S3). Neutrophils were evaluated in five studies (Caceres et al., 2017; Kennedy-Feitosa et al., 2020; Kim et al., 2015; Yu et al., 2018; Zhao et al., 2014). The results showed that 1,8-cineole significantly reduced the number of neutrophils in the experimental group (SMD: −8.16, 95% CI: −11.65 to −4.67, p < 0.00001) (Supplementary Figure S4). Macrophages were evaluated in five studies (Caceres et al., 2017; Kennedy-Feitosa et al., 2020; Kim et al., 2015; Yu et al., 2018; Zhao et al., 2014). The results showed that 1,8-cineole significantly reduced the number of macrophages in the experimental group (SMD: −5.06, 95% CI: −8.06 to −2.05, p = 0.001) (Supplementary Figure S5).
Sensitivity Analysis
The results of sensitivity analysis showed that after deleting one study (Li et al., 2016), meta-analysis of TNF-α and IL-6 showed decreased heterogeneity. When one study (Zhao et al., 2014) was also deleted, the heterogeneity of TNF-α was significantly reduced (I2 = from 84% to 25%). We found that only one of the included studies had a different intervention style compared to the others. In this study (Li et al., 2016), 1,8-cineole alone was administered for 2 days prior to virus challenge, followed by combined treatment with oseltamivir for 5 days after infection. Mice were sacrificed 6 days after infection and relevant samples were collected. The remaining studies used 1,8-cineole alone as the primary intervention throughout. Thus, we believed that this factor introduced heterogeneity in TNF-α and IL-6 results. For TNF-α, one study (Zhao et al., 2014) considered the vehicle group as the data for the model group, which may have contributed to increased heterogeneity in TNF-α and neutrophils. Furthermore, we identified that two studies (Kennedy-Feitosa et al., 2016, 2019) shared the same first and corresponding authors, with similar subjects, experimental protocols, animal species, and disease models. However, significant heterogeneity remained, and after excluding one of the studies, heterogeneity in IL-1β and NF-κB decreased. Thus, we attributed this study as the cause for the significant increase in IL-1β and NF-κB heterogeneity. Our findings indicated that variations in models and species contributed to the heterogeneity in meta-analyses, as well as heterogeneity within each group. This was primarily due to the difficulty in finding two or more animal studies with identical species, models, diseases, drugs, doses, dosage forms, and treatment cycles. The diversity in animal studies offers reliable evidence and abundant resources for preclinical studies but also leads to increased heterogeneity in meta-analysis (Hooijmans, IntHout, et al., 2014).
Discussion
Inflammatory substances elicit response in tracheal endothelial cells and monocytes, resulting in the secretion of various proinflammatory factors (TNF-α, IL-6, IL-1β) (Mukhopadhyay et al., 2006). Moreover, these substances stimulate the release of arachidonic acid from lipid membranes, which in turn induces the production of eicosenoic acid by goblet cells, resulting in mucus secretion and tissue inflammation (Arora & Chopra, 2018). The presence of proinflammatory factors has detrimental effects on lung structure, and overexpression of TNF-α and IL-1β is linked to the promotion of pulmonary fibrosis (Lundblad et al., 2005). Additionally, proinflammatory cytokines play a crucial role in recruiting inflammatory cells to the site of injury (Hackel et al., 2021; Ishii et al., 1997). Among the first cells to be recruited are macrophages and neutrophils (Kariyawasam & Robinson, 2007), which migrate to the injured area through chemotactic-directed movement to perform phagocytosis (Murugan & Peck, 2009). The stimulated macrophages and neutrophils contribute to the inflammatory response by releasing a multitude of proinflammatory factors and lipid mediators, including TNF-α, IL-6, IL-1β, and arachidonic acid metabolites (Barnes et al., 2003; Botelho et al., 2011; Hilda & Das, 2016; Navrátilová et al., 2021). TNF-α and IL-6 induce the production of endothelin, leading to the contraction of blood vessels and trachea (Endo et al., 1992), while also promoting the production of ICAM-1, which enhances the adhesion of leukocytes and eosinophils to the affected areas (Adler et al., 1994; Wegner et al., 1990). The secretion of IL-1β by macrophages, in combination with its receptor IL-1RI, results in the production of substantial amounts of GM-CSF (Lukens et al., 2012), a growth factor that promotes the proliferation and proper functioning of neutrophil cells and macrophages (Ataya et al., 2021). TNF-α, IL-1β, and toll-like receptors (TLRs) have the ability to activate IKKs, which subsequently phosphorylate IκB, resulting in the release of NF-κB (Amaro-Leal et al., 2020; Schuliga, 2015). In chronic airway inflammatory diseases, NF-κB plays a pivotal role in regulating the release of inflammatory cytokines and related mediators such as TNF-α, IL-6, IL-1β, ICAM-1, and GM-CSF, which contribute to the recruitment of inflammatory cells and the modulation of inflammation (Schuliga, 2015; Zhang et al., 2022).
Meta-analyses examined the effects of 1,8-cineole on various inflammatory markers, including TNF-α, IL-6, IL-1β, NF-κB, neutrophils, and macrophages, and consistently demonstrated a significant reduction in their expression levels in the experimental groups. However, even after sensitive sources had been removed from the analysis, the observed effects remained significant and stable, underscoring the robustness of these findings. These results strongly indicate that 1,8-cineole effectively inhibits the expression of inflammatory factors, thereby exhibiting remarkable anti-inflammatory properties. It is worth noting that these findings are supported by other studies as well. This intervention effectively prevents antigen-presenting-induced decrease in mucociliary clearance and alleviates airway inflammation in vivo studies (Bastos et al., 2011). Additionally, in diabetic-atherosclerotic rats, 1,8-cineole has improved the activity of glyoxalase-1, lipid profile, and renal function, thereby potentially averting atherosclerotic lesions and improving renal function, with these effects being attributed to the inhibition of TNF-α expression (Mahdavifard & Nakhjavani, 2021). 1,8-Cineole has demonstrated its ability to inhibit IL-1β, IL-6, NLRP3, and NF-κB levels in an animal model of inflammatory skin disease induced by Propionibacterium acnes (Lee et al., 2019). In a study involving rats, the administration of 1,8-cineole has resulted in the upregulation of Ho-1, NRF2, and Bcl-2 levels in brain tissue. Additionally, it has been found to regulate the phosphorylation of NF-κB and exhibit inhibitory effects on TNF-α, IL-6, and IL-1β (Xu et al., 2021). In poultry, the use of 1,8-cineole microcapsules has shown significant inhibition of TNF-α, IL-6, IL-1β, and NF-κB at higher temperatures, leading to a reduction in inflammatory response and improved body weight gain (Jiang et al., 2020). In vitro studies have revealed that 1,8-cineole exerts significant inhibitory effects on TNF-α, IL-1β, leukotriene B4, and thromboxane B2 in monocytes isolated from the blood of normal subjects (Juergens, Stöber, Vetter, 1998). Treatment with 1,8-cineole has also shown significant inhibition of arachidonic acid metabolism in vitro, as demonstrated by isolated monocytes stimulated with the calcium ionophore A23187 (Juergens, Stöber, Schmidt-Schilling, et al., 1998). Furthermore, pretreatment of Aβ(25-35)-induced inflammation in PC12 cells with 1,8-cineole has effectively restored cell viability and has inhibited the production of TNF-α, IL-1β, and IL-6 (Khan et al., 2014). In human lymphocytes, 1,8-cineole has demonstrated significant inhibition of inflammatory cytokines, particularly TNF-α (92%) and IL-1β (84%). The inhibitory effects of TNF-α and IL-1β have been even more pronounced in human monocytes (Juergens et al., 2004). Finally, in a murine lung alveolar macrophage model, pretreatment with 1,8-cineole has significantly inhibited the production of TNF-α, IL-1 (α and β) and regulated the phosphorylation of NF-κB and P38 in response to LPS-induced inflammation (Yadav & Chandra, 2017). Additionally, 1,8-cineole has inhibited TLR4-induced inflammation in human bronchial epithelial cells, leading to decreased expression of IL-8, IL-6, and GM-CSF through the regulation of P38 MAPK and Akt phosphorylation (Lee et al., 2016). Finally, the combination of 1,8-cineole and curcumin has effectively inhibited IL-6 and cigarette extract (CSE)-induced inflammation and cell death (Reis et al., 2021). The effect of 1,8-cineole on the inflammatory mechanisms of respiratory diseases is shown in Figure 3.
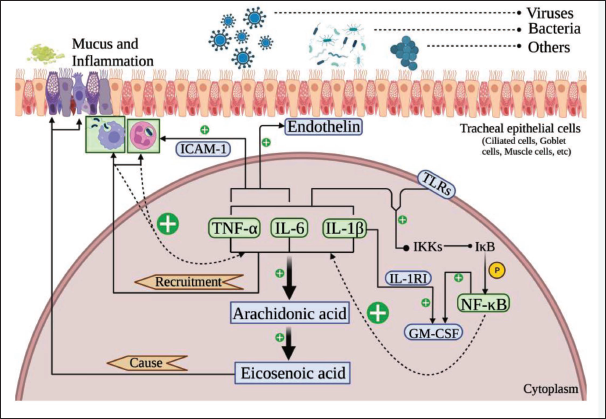
In related studies, the administration of 1,8-cineole has been investigated through oral or inhalation routes, employing different administration and dosage forms for various diseases. Ultrasonic atomization, nasal drops, and smears are employed for the management of conditions such as COPD, asthma, anxiety, sinusitis, and inflammatory skin disorders (Cai et al., 2021). In recent years, a new drug delivery system has been developed to enhance the oral availability of 1,8-cineole (Abada et al., 2019). This system has involved the utilization of self-microemulsion and cyclodextrin inclusion compounds, which have improved solubility, increased absorption, reduced volatility, exudation, and limited the occurrence of side effects associated with 1,8-cineole administration (Jiang et al., 2019). Moreover, the inhalation and local administration of 1,8-cineole has proven effective in avoiding first-pass effects, reducing irritation, and providing rapid therapeutic effects with minimal side effects. These routes of administration are deemed as the most suitable for the treatment of respiratory diseases (Bastos et al., 2011; Lee et al., 2016). The absorption characteristics of 1,8-cineole after oral administration are similar to intravenous administration (Sa et al., 2021). Notably, the primary metabolite detected in human plasma and urine has been α2-hydroxy-1,8-cineole, which accounts for 70% of the metabolite and can also be secreted through breast milk (Kirsch & Buettner, 2013). Additionally, 1,8-cineole can easily cross the blood–brain barrier (Dao et al., 2023; Moss & Oliver, 2012). Moreover, acute and long-term toxicity studies in male and female mice have demonstrated that 1,8-cineole exhibits low toxicity and relative safety in vivo (Caldas et al., 2016).
Therefore, based on the significant anti-inflammatory activity of 1,8-cineole, future studies could utilize nanotechnology to encapsulate it in suitable carriers, enhancing its targeted delivery to the lungs and improving therapeutic efficacy. Additionally, optimizing inhalation therapy, including the development of improved inhalation devices and formulations, as well as creating regular inhalers based on 1,8-cineole, could increase local drug concentration in the respiratory tract, reduce systemic adverse effects, and achieve synergistic benefits. This approach would facilitate daily medication management for patients, enabling easier home-based disease management and significantly improving their quality of life.
Limitations
(a) This meta-analysis only searches English databases, which may ignore studies published in other languages. (b) The small sample size of included studies may cause potential bias. (c) The data in this study are from animal studies rather than clinical studies, and there are genetic differences between the two, but the outcomes are closely related to clinical medicine.
Conclusion
This meta-analysis examines the anti-inflammatory and respiratory protective effects of 1,8-cineole in animal studies for the first time. We find that 1,8-cineole significantly inhibited inflammatory factors (TNF-α, IL-6, IL-1β, NF-κB) and inflammatory cells (neutrophils and macrophages), demonstrating a beneficial effect on the respiratory system.
Footnotes
Abbreviations
Bcl-2: B-cell lymphoma 2; COPD: Chronic obstructive pulmonary disease; CRDs: Chronic respiratory diseases; CSE: Cigarette extract; GM-CSF: Granulocyte-macrophage colony-stimulating factor; Ho-1: Heme oxygenase-1; ICAM-1: Intercellular adhesion molecule-1; IκB: Inhibitor of kappa B; IKKs: IκB kinases; IL-1β: Interleukin-1 beta; IL-6: Interleukin-6; IL-1RI: Interleukin-1 receptor type I; LPS: Lipopolysaccharide; NF-κB: Nuclear factor kappa B; NLRP3: NOD-like receptor family, pyrin domain containing 3; NRF2: Nuclear factor erythroid 2-related factor 2; SD: Standard deviation; SEM: Standard error of mean; SMD: Standard mean difference; TLRs: Toll-like receptors; TNF-α: Tumor necrosis factor-alpha.
Acknowledgments
The authors extend their gratitude to the developers of the software (Get Data Graph Digitizer, EndNote, RevMan, and BioRender) used in this study, as well as to the contributors of past research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Fund of Guizhou Provincial Department of Education [2022XMSB00035072].
