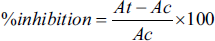Abstract
Background
Growing diabetes prevalence prompts investigation into plant-derived compounds’ potential to inhibit α-amylase, offering novel therapeutic avenues for treating diabetes mellitus (DM).
Objectives
In the current study, ethanolic and methanolic extracts of Fagonia cretica and Berberis lycium were evaluated against α-amylase.
Materials and Methods
The inhibitory activity of ethanolic and methanolic extracts was analyzed based on their IC50 values. An in vitro activity of α-amylase inhibition was performed, followed by molecular docking and molecular dynamics (MD) simulation of selected compounds.
Results
Results indicated that F. cretica and B. lycium extracts have strong inhibitory effects against α α-amylase. In ethanolic and methanolic extracts, the methanolic B. lycium extract was the most potent with an IC50 value of 2.10 µg/mL; the ethanolic B. lycium, ethanolic, and methanolic F. cretica extracts also showed significant anti-α-amylase effects with IC50 values of 3.88, 4.09, and 7.26 µg/mL, respectively. Further, a total of 36 phytochemicals were docked against the α-amylase enzyme to explore the binding mode of these phytochemicals. Docking results confirmed that most of the phytochemicals accommodate well in the active site of α-amylase and made strong interactions compared to the standard drug acarbose. Also, the MD simulation results confirmed that both phytochemicals revealed greater stability than the standard acarbose.
Conclusion
Based on these results, we concluded that the extract showed good effectiveness and further in vivo study is needed to manage DM.
Introduction
Diabetes mellitus (DM) is a metabolic disorder characterized by chronic hyperglycemia and impairment in the metabolism of proteins, lipids, and carbohydrates to varying degrees (Muhammad et al., 2021; Tamil et al., 2010). Hyperglycemia has been identified as a major risk factor in the development of DM and its complications. Therefore, control of glucose levels in the blood is crucial in the early treatment of DM and in reducing macro- and microvascular complications (Zinjarde et al., 2011).
At this time, diabetes is a health-related issue that affects people all over the world (Rahman, Muhammad, et al., 2021). Currently, insulin is used to treat type 1 diabetes mellitus (T1DM), and for type 2 diabetes mellitus (T2DM), other oral hypoglycemic medications such as sulfonylureas, thiazolidinediones, and peptide analogs are used (Bordoloi & Dutta, 2014). Synthetic medicines used to treat diabetes have several side effects, including nausea, vomiting, dysentery, alcohol flush, migraine anemia, and faintness, so research is focusing on medicinal antidiabetic drugs (Osadebe et al., 2014). The usage of medicinal plants in the management of DM is being investigated, and drugs from medicinal plants have shown superior activity compared to synthetic drugs due to fewer side effects and adverse effects. Medicinal antidiabetic drugs are used for curing and managing various lethal diseases (Rahman et al., 2019). Phytochemical-based medicines are often used when synthetic drugs are ineffectual in treating disease, and these are also cheap compared to all synthetic drugs (Kumar et al., 2014).
One therapeutic approach is preventing carbohydrate absorption after food intake, which is possible with the inhibition of the enteric enzyme α-amylase (Rahimzadeh et al., 2014; Sachan et al., 2019; Thengyai et al., 2020). The α-amylase is an essential secretory product of the pancreas and salivary gland that is known for the hydrolysis of complex carbohydrates in the intestinal mucosa into simple di- and oligosaccharides (Oyedemi et al., 2017; Sangeetha & Vedasree, 2012).
Medicinal plants are the main source of phytochemicals. The most common phytochemical groups are glycosides, alkaloids, polysaccharides, and phenolics such as flavonoids, terpenoids, and steroids (Firdous, 2014). These phytochemicals serve as a starting point for the research and creation of novel antidiabetic molecule types. There are numerous substances that have been identified from plant sources that exhibit antidiabetic action (Gaikwad et al., 2014).
Currently, the availability of many antidiabetic drugs, the fam of natural drugs is still gaining attention in treating diabetes by targeting α-amylase (Kashtoh & Baek, 2023). In a recent study, Li et al. (2023) investigated the α-amylase inhibitory activity of ethanolic, methanolic, and aqueous raspberry leaf tea extract. They found potent inhibitory activity with IC50 values of 1.26, 1.47, and 4.39 mg/mL for ethanolic, methanolic, and aqueous extracts, respectively. They also found an IC50 value of 5.12 mg/mL for acarbose (Li et al., 2023). Another recent study on the dark red shell of Sterculia nobilis Smith fruit (pericarp) has been investigated for α-amylase inhibition. Their result shows that the fruit extract effectively inhibits the enzyme with an IC50 value of 13.55 µg/mL when compared with acarbose (19.45 µg/mL) (Yang et al., 2023). Similarly, another recent study was conducted on an aqueous extract of Stachys lavandulifolia Vahl for its α-amylase inhibitory activity as well as for its hypoglycemic effect in vivo. The aqueous extract showed significant α-amylase inhibition activity with an IC50 of 0.99 mg/mL compared to an IC50 of 0.52 mg/mL for acarbose. It also showed a hypoglycemic effect on diabetic rats, with an AUCof 51.94 g/L/h (Remok et al., 2023). Likewise, various studies on different medicinal plants (Solanum oocarpum (Pereira et al., 2021), Solanum virginianum dried fruits (Saraswathi et al., 2021), and peanuts (Arachis hypogaea) (Sani et al., 2021)) showed α-amylase inhibitory activity.
Fagonia cretica belongs to the family Zygophyllaceae and has a lot of uses in traditional systems of medicine (Qureshi et al., 2016). Several studies reported their protective role in different diseases, like piles, digestive disorders, cancer (Marwat, 2008), skin diseases like eruptions, allergies (Qureshi et al., 2010), gastric troubles (Badshah & Hussain, 2011), gynecological problems (Iqbal & Sher, 2011), and in most regions of Pakistan, this plant is used traditionally for fever, typhoid, scabies, urinary discharges, asthma, bronchitis, liver troubles, and tumors (Ahmad, 2007). Berberis lycium belongs to the family Berberidaceae, and it is a common Pakistani medicinal plant famous for the name “Kashmal” (Qurat-ul-Ain et al., 2022). Traditionally, this plant is used in many diseases like antihyperlipidemic (Chand et al., 2007), antidiabetic (Aslam et al., 2015), antimicrobial, antifungal (Singh & Gupta, 2018), and antioxidant (Anwar et al., 2018).
Therefore, this study aimed to evaluate the antidiabetic activity of ethanolic and methanolic extracts of F. cretica and B. lycium to check their possible inhibitory activity against α-amylase.
Materials and Methods
Reagents and Chemicals Used
Methanol (CH3OH) and ethanol (C2H5OH) were used as extraction solvents. Phosphate-buffered saline (PBS) was made by dissolving 80 g NaCl, 244 g KH2PO4, 191 g Na2HPO4, and 2 g KCl in 1,000 mL of distilled H2O (at neutral pH), α-amylase enzyme, dimethyl sulfoxide (DMSO), starch which is a substrate for the α-amylase enzyme, acarbose, which is used as a regular inhibitor, dinitrosalicylic acid (DNS), and samples.
Sample Collection
The plant samples F. cretica and B. lycium were collected in healthy and disease-free conditions from District Dir Lower (34° 37 to 35° 07 North Latitudes and 71° 31 to 72° 14 East Longitudes), Khyber Pakhtunkhwa, Pakistan, in March 2020. This area is popularly known as the hub of medicinal plants because of abundant agriculture and rich hilly zones. This area is also famous for its traditional healers, who recommend different traditional plants for treating different diseases. So, we chose the plants with the help of a traditional healer and then further identified by Dr Mohib Shah (in-charge botanical garden) at the Department of Botany, Abdul Wali Khan University Mardan, Pakistan. After identification, the whole plant was washed in distilled water and was allowed to dry at room temperature for 2 weeks in the shade. An electric blender grinded the dried plant into a fine powder.
Extraction and Sample Preparation
Methanol (CH3OH) and ethanol (C2H5OH) were used to prepare extracts. To prepare the extracts, 180 g of powdered plant material was mixed with 600 mL of ethanol and methanol to immerse the plant material. The mouths of flasks were covered to avoid evaporation. The 0.45 µm filter paper was used to filter the dissolved plant extract solution. The methanol and ethanol were separated from each solution using a rotary evaporator method, leaving a semi-solid solution (i.e., crude extract) for more investigation. Lastly, 1.5 mg of F. cretica and B. lycium extracts were dissolved in 150 mL DMSO.
In Vitro α-Amylase Assay
The α-amylase assay was carried out by using the standard method with minor modifications (Chelladurai & Chinnachamy, 2018). Each well of the 96-well plate contained a total volume of 220 µL reaction mixtures (20 µL sample + 100 µL DNS + 20 µL DMSO + 50 µL PBS + 10 µL enzyme + 20 µL starch). To each well, 20 µL of DMSO was added, and the samples were serially diluted with 20 µL of DMSO. Afterward, 50 µL of PBS was introduced to the corresponding wells, and starch was used as a substrate. To each well, 20 µL of starch was introduced and incubated for 20 min at 37°C. Following the incubation, 10 µL of the α-amylase enzyme was introduced to each well. Finally, 100 µL of DNS was applied to all the wells separately and kept in an incubator for 30 min at 37°C. A spectrophotometer measured absorbance at 540 nm, and acarbose was used as standard.
The activity of α-amylase was calculated as follows:
where At and Ac represent the absorbance of test and control, respectively.
In silico Screening
Active Site Prediction
The active site was found using the site finder option of Molecular Operating Environment (MOE) software. The site finder option was used to predict possible active sites in α-amylase from the receptor’s three-dimensional (3D) atomic coordinates. Calculations were used to determine potential sites for ligand binding docking (Rahman, Shah, et al., 2021).
Preparation of Ligand for Docking Analysis
All the ligands included in the study were collected from available literature. The molecular structures were drawn using Chem-Draw ultra-version 12.0.2.1076 (2010), and then all the ligands were saved in mol format. The structures were protonated in 3D, and energy was minimized through MOE using default parameters.
Preparation of Protein and Molecular Docking
The α-amylase structure (downloaded from the protein databank, ID: 1B2Y) 3D protonation and energy minimization were performed by using MOE (Chemical Computing Group Inc., Canada, 2016) (Version 2014.09) software with default parameters. Protein-ligand docking score, ligand properties, and two-dimensional (2D) and 3D structures were analysed.
Molecular Dynamics (MD) Simulation
Amber 20 package was used for the MD simulations to study the atomic level stability of the enzyme α-amylase with the ligands. The initial system was set up for MD simulations using the tleap module. A transferable intermolecular potential with 3 points (TIP3P) hydrated cubic box of 10 Å was used to soak the protein–ligand complexes. The counter ions were added to the system so that the residual charges were neutralized. Energy minimization, heating, density equilibration, and equilibration under periodic boundary conditions were applied to the MD systems. As an NPT ensemble, the final production phase of 100 ns was completed at 310 K. To determine the strength of the protein–ligand interactions, the root mean square deviation (RMSD) and root mean square fluctuation (RMSF) analyses were performed after the 100 ns MD simulation (Rahman et al., 2023).
Statistical Analysis
The IC50 values were calculated from the mean inhibitory values using nonregression analysis and plots of % inhibition versus log inhibitor concentration. All the measurements were performed in triplicate, in the three independent experiments.
Results
In Vitro α-Amylase Assay Effects
F. cretica and B. lycium extracts were tested for α-amylase inhibitory activity at 7.81–1,000 µg/mL concentrations. The percent inhibition of in vitro α-amylase inhibitory assay of ethanolic and methanolic extracts of F. cretica and B. lycium is presented in Figure 1.
The Graph Represents the Inhibitory Potential of Ethanolic and Methanolic Extracts of F. cretica and B. lycium, Along With Standard Acarbose Against α-Amylase.
Methanolic B. lycium demonstrated high efficacy with an inhibitory activity of 70.34% and an IC50 value of 2.10 µg/mL in comparison to the standard acarbose, which showed 53.93% inhibition and IC50 value of 15.06 µg/mL. Ethanolic and methanolic F. cretica and ethanolic B. lycium also exhibited significant inhibitory activity with percent inhibition of 65.12%, 62.02%, and 68.00% with IC50 values of 4.09, 7.26, and 3.88 µg/mL, respectively.
Docking Analysis
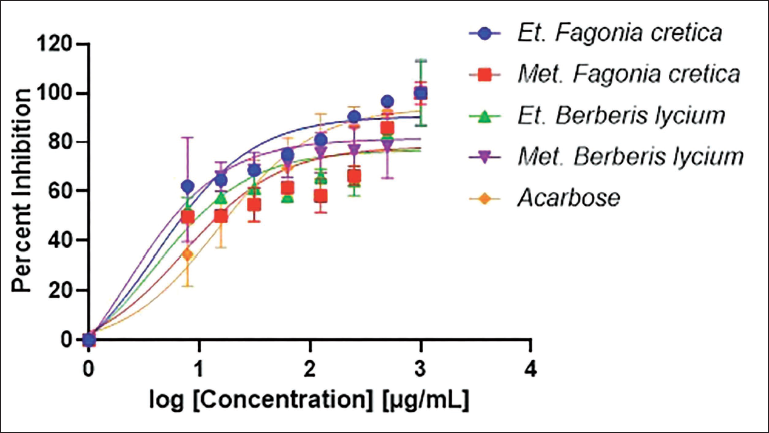
Docking analysis revealed that all the 36 phytochemicals docked well into the active site of α-amylase and made strong interactions. On the basis of the docking score, compound
(A) Active Site of Drug Target, (B) Three-dimensional (3D) Interaction of Compound 31 (Oxyberberin (B. lycium)), (C) Compound 35 (Chenabine (B. lycium)), and (D) Acarbose with the Active Site Residues of the Drug Target α-Amylase. Ligands are Shown as Cyan Sticks While the Green Dashed Line Represents the Bond.
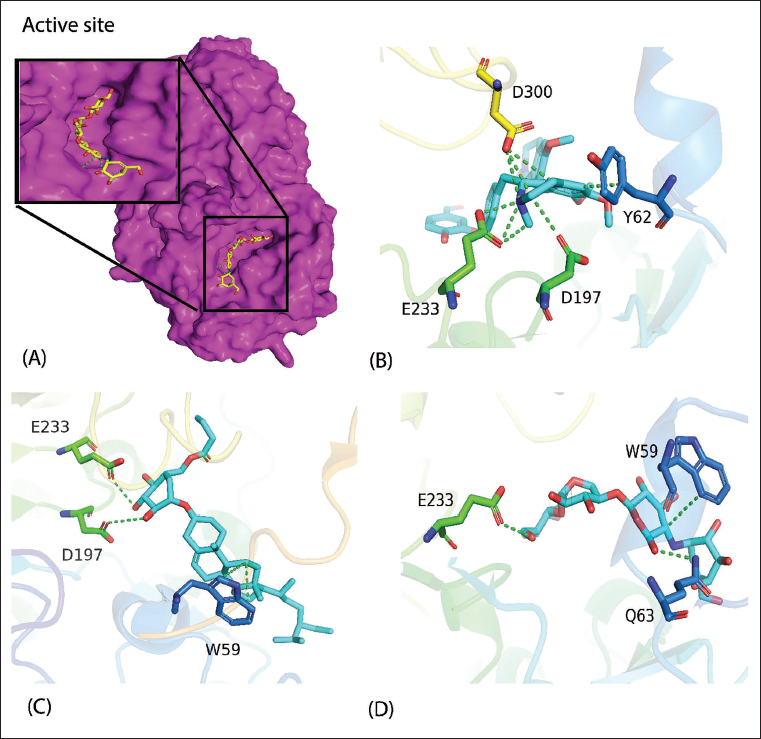
Docking Score and Interaction Pattern of Top 12 Compounds as well as the Standard Drug Acarbose Against α-Amylase.
MD Simulation Analysis
RMSD Analysis
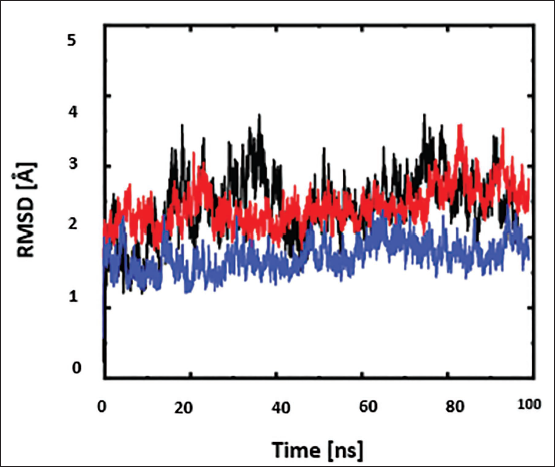
The RMSD values provide details regarding the structural conformation and the stability of the ligand bound to the protein during the simulation. The RMSD plot for the standard drug acarbose in complex with the target protein and top two best docking scored protein–ligand complexes is shown in Figure 3. With an average RMSD of 2.9, the standard drug bound with the protein demonstrated stability over the course of the 100 ns simulation (black). The standard complex highly fluctuated between 30 and 40 ns, but then it reached stability. The two complexes demonstrated protein backbone stability in the case of bound complexes throughout the simulation. The average RMSD was determined to be 1.5 Å for the protein complex Oxyberberin (blue), and 2.5 for the protein complex Chenabine (red). The predicted best docking scored and best-interacted hits-bound to protein has a somewhat lower RMSD value than the standard-bound protein, indicating the more stabilized complexes.
Root Mean Square Deviation (RMSD) Plot of Complex Oxyberberin (Blue), Complex Chenabine (Red), and Acrbose–Protein Complex (Black). The x-Axis Shows the Period of Simulation Time in Nanoseconds.
RMSF Analysis
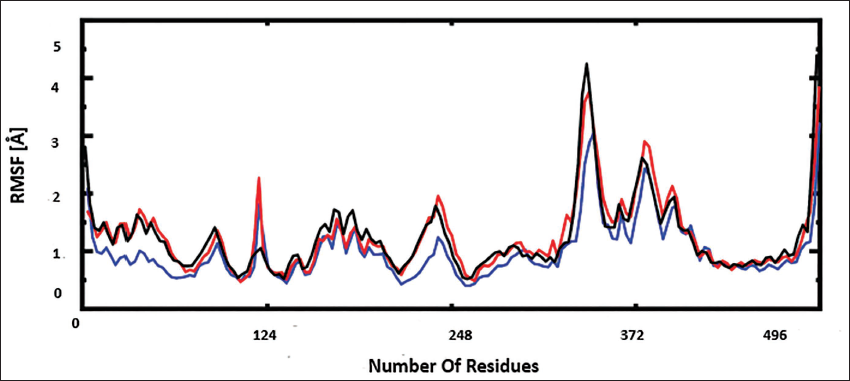
The fluctuations in amino acids were determined from the calculated RMSF. During the simulation run, the RMSF plot (Figure 4) showed stable secondary conformations. Due to the presence of a loop, the increase in RMSF in the range of 360–370 residues was estimated. The residues LYS200, HIS201, GLU233, ASP300, and HIS305, which make up the active site region, demonstrated strong stability, as seen by a lower RMSF value than the loop region. All three complexes were stable during the 100 ns MD simulation, according to the RMSF study.
Root Mean Square Fluctuation (RMSF) Plot of the Three Complexes Oxyberberin (Blue), Complex Chenabine (Red), and Acarbose–Protein Complex (Black). The Total Number of Residues is Shown on the x-Axis.
Discussion
There are different phytoconstituents having α-amylase inhibition potency (Chelladurai & Chinnachamy, 2018) reported an in vitro study on aqueous stem extract of Salacia oblonga; their findings showed that the extract inhibits the enzyme effectively with an IC50 value of 73.56 mg/mL and excellent percent inhibition of 59.46%. Maharjan et al. (2021) also screened leaves of Cinnamomum for the presence of phytochemicals and α-amylase inhibitory assay; their investigations revealed the presence of polyphenols, flavonoids, terpenoids, quinones, diterpenes, tannins, and good inhibitory potency of α-amylase with an IC50 value of 224.6 ± 2.76 µg/mL.
Gandhimathi et al. (2022) performed in vitro and in silico studies on phytoconstituents of Cyclea peltate roots to check its α-amylase inhibitory activity; according to their results, the extract inhibited the enzyme 69.42 ± 0.74% with an IC50 value of 484.08 µg/mL.
In our study, the α-amylase inhibitory activity of ethanolic and methanolic extracts of F. cretica and B. lycium was investigated. The result showed that these extracts efficiently inhibited α-amylase activity. Methanolic B. lycium showed the highest inhibitory activity with a percent inhibition of 70.34%, having an IC50 value of 2.10 µg/mL. Ethanolic and methanolic F. cretica and ethanolic B. lycium extracts inhibited α-amylase activity with a percent inhibition of 65.12%, 62.02%, and 68.00% with IC50 values of 4.09, 7.26, and 3.88 µg/mL, respectively. Similar results are also reported in the literature for methanolic extracts of Cinnamomum zeylanicum having an IC50 value of 130.55 µg/mL, Piper betle, having an IC50 value of 84.63 µg/mL, Artocarpus altilis having an IC50 value of 118.88 µg/mL, and Artocarpus heterophyllus having an IC50 value of 70.58 µg/mL for α-amylase inhibitory assay (Nair et al., 2013). Most medicinal plants tend to reduce blood glucose levels, and this is due to the availability of important phytoconstituents (Poongunran et al., 2015). With ample scientific literature documenting the genus Fagonia, it is widely reported that its biological activities are primarily associated with saponin glycosides. Notably, among these compounds, kaempferol triglycosides (Ibrahim et al., 2008) and triterpenoid saponins (Saeed & Sabir, 2003) are considered particularly significant. Astiti et al. (2021) isolated seven glycosides from Coccinia grandis leaves for α-amylase inhibitory activity. The most potent α-glucosidase inhibitor was kaempferol 3-O-robinobioside (3), exhibiting an IC50 value of 195.4 µM, 10.3 times more potent than the acarbose drug (Astiti et al., 2021). These intriguing results showed that F. cretica extracts’ activity may be due to the presence of kaempferol. However, kaempferol was not found in B. lycium extract (Landry et al., 2021).
Furthermore, the docking study was carried out to explore the binding mode of all the 36 phytochemicals. Several poses were generated for each compound with improved binding modes and interactions. The best docking score compounds that revealed strong interactions with the receptor were further short-listed for molecular dynamics study. Among all the 36 phytochemicals, compound
The present study revealed that the extracts of medicinally important plants play a crucial part in controlling DM by inhibiting α-amylase with less or no side effects compared to synthetic products. Based on these findings, we believe these phytochemicals could be useful in treating DM in the future.
Despite promising results, this study has several limitations. Firstly, the evaluation focused on the in vitro α-amylase inhibitory potential, and the transition to in vivo models is crucial for validating the therapeutic efficacy and safety of the identified extracts. Additionally, while suggestive of strong interactions, the molecular docking and MD simulation results may not fully capture the complexities of physiological conditions. The study did not explore the potential synergistic or antagonistic effects of compounds within the extracts, which could influence overall inhibitory activity. Also, the findings are specific to F. cretica and B. lycium, so generalizing to other plants or real-world applications requires further investigation.
Conclusion
The current study evaluated the α-amylase inhibitory potential of ethanolic and methanolic extracts of F. cretica and B. lycium. In the in vitro α-amylase inhibitory assay, the extracts showed 65.12%, 62.02%, 68.00%, and 70.34% inhibition having IC50 values of 4.09, 7.26, 3.88, and 2.10 µg/mL for ethanolic and methanolic F. cretica and B. lycium, respectively, which is higher than the standard acarbose, that showed 53.93% inhibition having an IC50 value of 15.06 µg/mL. Further, molecular docking and MD simulation were performed to find the lead compounds for α-amylase enzyme. The in silico study revealed that the two phytochemicals showed good interactions with the α-amylase and revealed better stability than the standard drug acarbose. These two phytochemicals have the potential to inhibit α-amylase and can be effective in combatting the diseases associated with α-amylase. We conclude that F. cretica and B. lycium extracts effectively inhibited the α-amylase enzyme. In the future, further studies are needed to use these extracts to manage DM.
Footnotes
Abbreviations
DM: Diabetes mellitus; DMSO: Dimethyl sulfoxide; DNS: Dinitrosalicylic acid; MD simulation: Molecular dynamics simulation; MOE: Molecular Operating Environment; RMSD: Root mean square deviation; RMSF: Root mean square fluctuation; TIP3P: Transferable intermolecular potential with 3 points; T1DM: Type 1 diabetes mellitus; T2DM: Type 2 diabetes mellitus; 3D: Three-dimensional.
Acknowledgments
The authors extend their appreciation to the Researchers Supporting Project Number RSP2025R457, King Saud University, Riyadh, Saudi Arabia.
Authors’ Contributions
Conceptualization, writing the original draft, reviewing, and editing: Imtiaz Ahmad, Yan-Kun Chen, Noor Rahman, Shafiullah, Haroon Khan. Formal analysis, investigations, funding acquisition, reviewing, and editing: Yan-Kun Chen; Luca Rastrelli, Said Alam, Yousef A. Bin Jardan. Resources, data validation, data curation, and supervision: Mohammed Bourhia and Changan Jiang.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Researcher Supporting Project (RSP2025R457), King Saud University, Riyadh, Saudi Arabia.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.