Abstract
Background
Although it does not represent a threat to healthy humans, Pseudomonas aeruginosa is a type of aerobic, gram-negative bacteria that can cause catastrophic infections in patients with immune system abnormalities and cystic fibrosis.
Objectives
The current study investigated the effect of Pseudomonas aeruginosa-mannose sensitive hemagglutinin (PA-MSHA) on liver cancer cell viability, colony formation, and invasion potential, and explored the underlying mechanism.
Materials and Methods
Proliferation potential and clonogenic survival of the cells were studied using MTT and colony formation assays, respectively. Transwell and Western blotting assays were used for the assessment of invasive potential and protein expression, respectively.
Results
The results revealed that exposure to PA-MSHA led to a time-dependent suppression in HepG2 and HEP3B cell proliferation. Incubation with PA-MSHA decreased HepG2 cell proliferation to 92%, 75%, 50%, 35%, and 20%, respectively, after 12, 24, 36, 48, and 72 hours. Similarly, incubation for 12, 24, 36, 48, and 72 hours reduced the proliferation of HEP3B cells to 94%, 79%, 55%, 41%, and 26%, respectively. The rate of clonogenic survival was significantly lower in HepG2 and HEP3B cells on incubation with PA-MSHA. The cell invasion potential was also significantly reduced on incubation with PA-MSHA. In HepG2 cells, incubation with PA-MSHA significantly reduced the expression of Bcl-2 (26 kDa) protein and promoted Bax (21 kDa) level. Following incubation with PA-MSHA, HepG2 cells showed higher cleaved caspase-3 level compared to the control cells. Compared to control cells, the PA-MSHA incubation of HepG2 cells resulted in a prominent reduction in Notch3 protein expression.
Conclusion
In summary, PA-MSHA treatment prevents liver cancer cell proliferation and targets the survival of clonogenic cells. It reduces invasiveness, targets Notch3 protein expression in liver cancer cells, and increases the level of proapoptotic and suppresses antiapoptotic protein expression. Therefore, PA-MSHA shows encouraging anticancer effects on HepG2 and HEP3B cancer cells, suggesting that it could be developed as a viable treatment option for liver cancer.
Introduction
Although it does not pose a threat to healthy individuals, Pseudomonas aeruginosa is a gram-negative, aerobic bacteria that causes devastating infections in patients with immune system abnormalities and cystic fibrosis (Faure et al., 2018). Although P. aeruginosa is primarily an aerobic bacterium, it has been shown to function as a facultative anaerobe in the presence of low oxygen levels, using other electron acceptors to generate energy (Arai, 2011). Gene-modified, attenuated, or inactivated P. aeruginosa has been shown to significantly inhibit tumor growth in vivo in mice models by inducing an antitumor response in host cells (Wang et al., 2016; Zhang et al., 2014), inducing cancer cell death in a programmed manner (Cao et al., 2009; Chang et al., 2014; Qi et al., 2020; Zhao et al., 2016), and inhibiting proliferative signaling pathways (Liu et al., 2010; Wei et al., 2016; Xiu et al., 2020). Numerous virulence factors have been found to be linked with P. aeruginosa-induced cytotoxicity against cancer cell lines (Bernardes et al., 2014; Goldufsky et al., 2015; Kwan et al., 2009; Rahimi et al., 2019; Vázquez-Rivera et al., 2015; Wolf & Elsässer-Beile, 2009). Exotoxin A (ExoA) is the most toxic of these virulent factors, and it is essential for the production of immunotoxins and targeted cancer therapy (Weldon & Pastan, 2011).
Bacteria can be used in cancer therapy either after genetic modification or by attenuating them to reduce their toxicity and potential for replication. The MSHA fimbriae are expressed more frequently on the surface of Pseudomonas aeruginosa-mannose sensitive hemagglutinin (PA-MSHA), a new strain of the bacterium created through genetic engineering. Shielding of P. aeruginosa from additional detrimental virulence factors on its surface leads to a decrease in the pathogenic potential of the bacteria (Mu, 1986). Additionally, it has been discovered that MSHA fimbriae binds to toll-like receptor 4 (TLR4) (Mossman et al., 2008).
The discovery that the presence of bacteria inhibited tumor growth in patients infected with Streptococcus pyogenes was made by W. Busch and F. Fehleisen (Trivanović et al., 2021). The first time bacteria were used to treat cancer was when William Coley gave S. pyogenes vaccinations to cancer patients (Coley, 1991). It has been discovered that hypoxia brought on by the fast growth and multiplication of cancer cells characterizes solid tumors (Emami Nejad et al., 2021). Additionally, the oxygen-deficient environment promotes tumor cell proliferation and angiogenesis (Wang et al., 2017). Numerous facultative and obligatory anaerobes are known to form colonies and multiply quickly in the hypoxic areas of tumors (Dróżdż et al., 2020). Clinicians worldwide are becoming interested in using bacteria-mediated therapeutic strategies to treat cancer because they can circumvent many of the drawbacks of traditional chemotherapeutic and radiation therapy approaches (Sedighi et al, 2019). The current study examined the effect of PA-MSHA on the growth of liver cancer cells in vitro. Moreover, the study also explored the underlying mechanism of PA-MSHA-induced suppression in liver cancer cell growth.
Materials and Methods
Cell culture
Shanghai, China’s Stem Cell Bank Academy of Sciences supplied the HepG2 and HEP3B liver cancer cell lines. In order to cultivate the cells, 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin were combined with DME-medium. For the whole night, the cells were cultured in an incubator with a temperature of 37°C and 5% CO2.
Cell Proliferation Assay
The cells were propagated in DME-medium containing 10% FBS in 96-well plates, with a seeding density of 2.5 × 104 cells per well. For the entire night, the cells were incubated at 37°C with 5% CO2 in the medium supplemented with 1% penicillin/streptomycin. The cells were then incubated for 72 hours at 37°C with a fresh medium containing PA-MSHA in place of the old one. Following the TFT incubation period, each well was filled with 20 µl of MTT (5 mg/ml), and an additional 4 hours of incubation were completed. The formazan crystals that had formed in the wells were then dissolved by adding dimethyl sulfoxide (150 µl) to each of the wells. Absorbance measurements were made for the calculation of cell viability at 570 nm using the microplate reader (Bio-Rad Laboratories Inc., Hercules, CA, USA).
Colony Assay
HepG2 and HEP3B cells were plated in six-well plates and incubated in DME-medium at 37°C for the entire night. After incubation, the cells were treated with a 2 ml solution of Base Agar Matrix Layer, trypsinized, counted, and cleaned with PBS. To allow for solid formation, the plates were kept at a temperature of 25°C. Each well of the plates received a 2 ml agar layer containing the cells, which were subsequently kept at room temperature until the cells formed a monolayer. The plates were treated with PA-MSHA in DME-medium, and the cells were incubated in it at 37°C under 5% CO2 until visible growing colonies formed. Following incubation for 10–14 days, the cell colonies were mostly visible, and those with 50 cells or more were regarded as the surviving colonies.
Cell Invasion Assay
Matrigel was applied to the upper side of the Transwell filters of the polycarbonic membrane having a diameter of 6.5 mm and pore sizes of 8 µm. The matrigel acted as an extracellular matrix for determining the invasive potential of the tumor cells after solidification for 45 minutes. The cells, after incubation with PA-MSHA for 72 hours, were placed in 200 µl of serum-free medium on the upper side of the Transwell chamber. The 500 µl of DME-medium combined with 10% FBS was placed in the lower chamber, serving as a chemoattractant. Cotton swabs were used to mechanically remove the non-invaded cells following 12 hours of invasion of the cells. Methyl alcohol was used to fix the cells that had invaded the bottom chamber, and crystal violet dye was used to stain them for half an hour. The membranes after removal were placed onto coverslips and analyzed at ×100 magnification for cell invasion under an inverted microscope.
Western Blot Analysis
HepG2 and HEP3B cells were treated with RIPA buffer containing phenylmethylsulfonyl fluoride on ice for 30 minutes after being incubated for 72 hours at 37°C with PA-MSHA or left untreated (control). After the lysate was extracted from the supernatant by centrifuging at 13,000 × g for 20 minutes at 4°C, the protein content was measured using the Bradford Protein Assay kit. Protein samples were electrophoretically loaded onto a 10% SDS-PAGE and subsequently transferred onto PVDF membranes, following the manufacturer’s recommendations. Previously, the nonspecific sites in the membranes were blocked by an overnight incubation (at 4°C) with 5% skim milk powder. Subsequently, primary antibodies were incubated on the membranes for the whole night at 4°C. Membranes were subjected to three times TBST washing before being incubated for 1 hour at room temperature with secondary goat antimouse IgG conjugated horseradish peroxidase antibodies. The enhanced chemiluminescent (ECL) kit was used to develop the membranes.
Statistical Analysis
The information displayed consists of the mean ± standard deviations from the triplicate experiments that were conducted independently. Group differences were evaluated using the Student’s t-test, one-way analysis of variance (ANOVA), and Tukey’s post hoc test. A statistical program called SPSS version 10.0 (SPSS, Inc., Chicago, IL, USA) was utilized to analyze the data. p < 0.05 was employed to assess the statistical significance of the variations.
Results
PA-MSHA Suppresses HepG2 and HEP3B Cell Proliferation
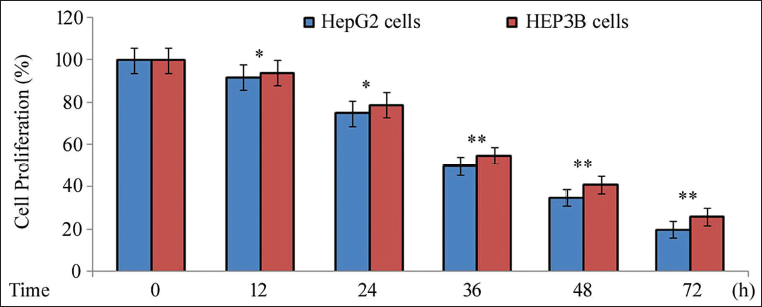
Incubation with PA-MSHA was followed by MTT assay to assess the impact on proliferation in HepG2 and HEP3B cells (Figure 1). It was found that the cells treated with PA-MSHA had a time-dependent suppression of cell proliferation. Following treatment with PA-MSHA, HepG2 cell proliferation decreased to 92%, 75%, 50%, 35%, and 20%, respectively, after 12, 24, 36, 48, and 72 hours. Similarly, incubation for 12, 24, 36, 48, and 72 hours reduced proliferation of HEP3B cells to 94%, 79%, 55%, 41%, and 26%, respectively.
Effect of PA-MSHA on Cell Proliferation. PA-MSHA was Incubated with the Cells for 12, 24, 36, 48, and 72 hours. The MTT Assay was Used to Assess the Changes in the Cells’ Proliferative Potential. The Results are Presented as the Mean ± Standard Error of Three Measurements. *p < 0.05 and **p < 0.01 Versus Control Cells.
PA-MSHA Decreases Clonogenic Survival
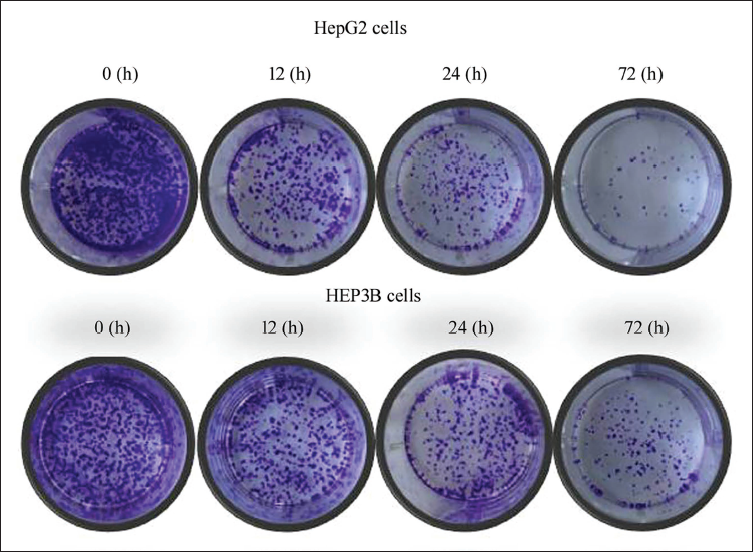
To investigate the changes in clonogenic survival rate, the cells were incubated with PA-MSHA (Figure 2). The results showed that the cells when incubated with PA-MSHA, the rate of clonogenic survival was down to a remarkable extent than when the cells were left as controls. The reduction in the clonogenic survival rate became more pronounced when the incubation period was extended from 12 to 72 hours.
Effect of PA-MSHA on Clonogenic Survival of HepG2 and HEP3B Cells. The Clonogenic Survival of Cells Treated with PA-MSHA or Left Untreated (Control) was Assessed Using the Colony Formation Assay.
Effect of PA-MSHA on Cell Invasiveness
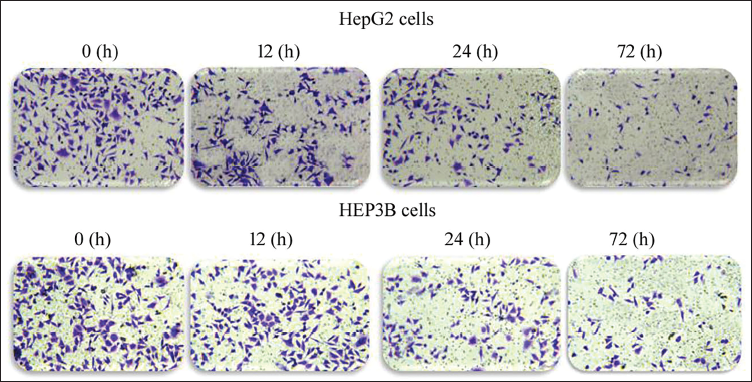
Transwell assay was conducted to monitor the impact of PA-MSHA on the invasion potential of HepG2 and HEP3B cells following 12, 24, and 72 hours of incubation (Figure 3). HepG2 and HEP3B cell invasion potential was significantly reduced by PA-MSHA incubation as compared to control cells. Furthermore, the invasion potential of HepG2 and HEP3B cells was found to gradually decline with longer incubation times, ranging from 12 to 72 hours.
The PA-MSHA Treatment Decreases Invasion Potential of the Cells. The Transwell Assay was Utilized to Evaluate How the PA-MSHA Incubation Affected the Invasion Potential of the Cells. The Number of Infiltrated Cells was Counted, and ×100 Magnification Photos were Taken.
PA-MSHA Influences the Expression of Apoptotic Proteins in HepG2 Cells
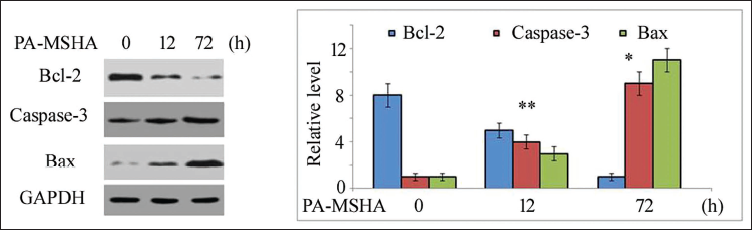
Following the incubation of HepG2 cells with PA-MSHA, the level of apoptotic proteins was measured at 12 and 72 h (Figure 4). In HepG2 cells, it was found that incubation with PA-MSHA led to a remarkable reduction in the expression of the Bcl-2 (26 kDa) protein. In HepG2 cells, incubation with PA-MSHA led to a notable rise in the expression of Bax protein (21 kDa) when compared to the control cells. After incubation with PA-MSHA, HepG2 cells showed higher expression of cleaved caspase-3 compared to the control cells.
Effect of PA-MSHA on the Expression of Apoptotic Proteins in HepG2 Cells. Incubation with PA-MSHA for 12 and 72 hours was Followed by Determination of the Expression of Proteins in HepG2 Cells. *p < 0.05 and **p < 0.01 Versus Control Cells.
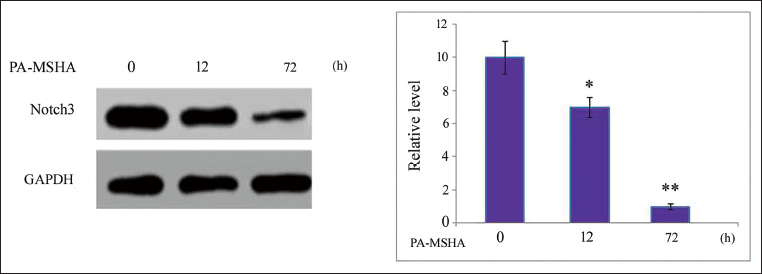
PA-MSHA Targets Notch3 Expression in HepG2 Cells
Incubation of HepG2 cells with PA-MSHA for 12 and 72 hours was followed by assessment of expression of Notch3 protein by Western blotting (Figure 5). The results showed that, in comparison to control cells, the PA-MSHA incubation of HepG2 cells led to a remarkable reduction in Notch3 protein expression.
Effect of PA-MSHA on Level of Notch3 Protein in HepG2 Cells. PA-MSHA was Poured into the Cell Cultures and Incubation was Carried out for 12 and 72 hours at 37°C. In HepG2 cells’ Notch3 Protein Expression was Evaluated. *p < 0.05 and **p < 0.01 Versus Control Cells.
Discussion
Chemotherapeutic agents manipulate oncogenic signaling transduction, which plays an essential role in preventing tumor cell division and limiting the growth and development of tumors (Bhullar et al., 2015; Czabotar et al., 2014; Gali-Muhtasib et al., 2015; Zhang et al., 2016). Proapoptotic protein expression upregulation has been demonstrated to trigger the apoptotic pathway and subsequently eliminate the undesirable cells through an innate multi-step process (Czabotar et al., 2014). In the current study, PA-MSHA treatment has been found to inhibit the proliferation of the tested cells in a time-dependent manner. Moreover, clonogenic survival of HepG2 and HEP3B cells was effectively suppressed in a time-dependent manner on incubation with PA-MSHA. Exposure of HepG2 and HEP3B cells with PA-MSHA led to a pronounced suppression in invasive potential compared to the control cells.
It has been reported that overexpression of the Bcl-2 family of proteins increases the permeability of the mitochondrial membranes, causing the enhancement in cytochrome c and, ultimately, the induction of cell death (Czabotar et al., 2014). The p53 typically controls the cellular expression of proapoptotic proteins during the induction of apoptosis, including Bcl-2 and Bax (Haldar et al., 1994; Harn et al., 1996). Activation of apoptotic pathways has been explored as one of the leading and most effective methods for inhibition of tumor growth. Treatment of the cells with PA-MSHA in the current study decreased cell viability in a time-dependent manner. Furthermore, when the cells were incubated with PA-MSHA, the rate of clonogenic survival was significantly lower than in the control cells. These preliminary results suggest that PA-MSHA has anticancer activity against liver cancer cells and, as such, warrants additional research to determine whether it has antitumor properties. The Transwell assay showed that PA-MSHA incubation significantly and time-dependently reduced the invasion potential of the tested cells. Furthermore, it was discovered that extending the PA-MSHA incubation period from 12 to 48 hours resulted in a gradually diminished capacity for invasion of cells. The Bcl-2 protein expression in HepG2 cells was significantly reduced because of PA-MSHA treatment, according to the results of the Western blotting experiment. On the other hand, HepG2 cells treated with PA-MSHA showed a significant, time-dependent increase in the expression of Bax and caspase-3.
Notch3 expression is noticeably higher in many types of tumor cells, including lymphoblastic leukemia T-cells, prostate carcinoma, and triple-negative breast carcinoma cells (Bellavia et al., 2000; Pedrosa et al., 2016; Turner et al., 2010; Zhang et al., 2010). Overexpression of the Notch3 pathway is crucial in the induction of carcinoma. It has been reported that overexpression of Notch3 significantly increases the rate of proliferation and metastatic potential in cancer cells. Furthermore, the ability of cancer cells to invade apoptosis and exhibit stem cell-like properties is also caused by Notch3 overexpression (Zhang et al., 2010). Notch3 expression is controlled by the transcription factor p53, which is crucial in preventing tumor growth by arresting the cell cycle and triggering apoptotic signals (Bieging et al., 2014; Giovannini et al., 2009). In the current study, HepG2 cells on treatment with PA-MSHA showed a prominent decrease in the expression of Notch3 protein in comparison to the control cells. Thus, it appears that PA-MSHA incubation inhibits liver cancer cell growth partly by targeting the expression of the Notch3 protein.
Conclusion
PA-MSHA treatment prevents liver cancer cell proliferation and targets the survival of clonogenic cells. It reduces invasiveness, targets Notch3 proteins in liver carcinoma cells, and increases the level of proapoptotic and suppresses antiapoptotic proteins. Therefore, PA-MSHA shows encouraging anticancer effects on HepG2 and HEP3B cancer cells, suggesting that it could be developed as a viable treatment option for liver cancer.
Footnotes
Abbreviations
PA-MSHA: Pseudomonas aeruginosa-Mannose Sensitive hemagglutinin; ExoA: Exotoxin A.
Acknowledgments
None.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the research committee, General Hospital of Western Theater Command, Chengdu, China.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
