Abstract
Background and Objectives
Stroke threatens neurological function. Salidroside is widely used to treat neurological diseases such as stroke. Previous studies have shown that miR-155 plays an important regulatory role in the pathogenesis of stroke and that the Notch pathway plays an important role in nervous system development and regeneration.
Materials and Methods
PDA NPs-SAL complex was prepared, a stroke rat model was constructed, and the cerebral infarction area of rats was measured. Analyze the changes of PDA NPs-SAL on rat brain tissue and evaluate the neuroprotective effect. Detect the changes in miR-155 and Notch pathway-related proteins in stroke rat models to further understand the role of miR-155 and Notch pathway in the pathogenesis of stroke and their relationship.
Results
Salidroside had certain protective effects on the nerves of stroke rats, and the effect of PDA NPs-SAL was more prominent. At the same time, PDA NPs-SAL promotes the Notch pathway by inhibiting miR-155, thereby exerting a neuroprotective effect on stroke rats.
Conclusion
Under the intervention of PDA NPs-SAL, the neurological function scores of rats were reduced, and the effect of PDA NPs-SAL was more significant. In addition, PDA NPs-SAL inhibited miR-155 and activated the Notch pathway, thereby reducing the water content of rat brain tissue and cerebral infarction area, thereby exerting a neuroprotective effect on stroke rats.
Introduction
Some traditional Chinese medicine ingredients, such as baicalin and tanshinone, can reduce cerebral edema and inflammatory reactions caused by cerebral hemorrhage by regulating the expression and activity of AQP4, thereby improving cerebral hemorrhage damage (Wei et al., 2020). Ginsenosides, curcumin, and so on can inhibit neuron apoptosis by regulating the p53/Bax/Caspase-3 molecular pathway, thereby reducing nerve damage caused by cerebral hemorrhage (Huang et al., 2019). Berberine, puerarin, and so on can reduce the inflammatory response by regulating key molecules of the inflammatory response, such as NF-κB and TLR4, thereby improving stroke’s pathological process and prognosis (Bai et al., 2020). These all highlight the application potential of Traditional Chinese Medicine in neurological diseases. Although many Traditional Chinese Medicine ingredients are effective in neuroprotection, there are few reports on salidroside in this regard, and the mechanism is unclear and needs further exploration.
Research shows that salidroside has a certain effect on the treatment of Alzheimer’s disease, Parkinson’s disease, and depression to a certain extent (Zhang et al., 2017). It can inhibit inflammation and oxidative stress reactions, further improving the cognitive function and pathological changes of Alzheimer’s disease mice to a certain extent. However, the mechanism of stroke has not been studied yet. Some studies have reported that salidroside reduced the protein and mRNA expression levels of MAP3K6 and MMP24 genes while upregulating the miRNA expression level of the miR-1343-3p gene (Zhao et al., 2019). The presence of miR-155 can inhibit the expression of phosphatase and tensin homolog (PTEN), thereby weakening its inhibitory effect on Akt, making the PI3K/Akt signaling pathway more active, thereby inhibiting the expression of Bcl-2, and improving the survival ability of nerve cells (Wang et al., 2016). miR-155 can inhibit the expression of negative regulatory factors such as SOCS1, A20, and SHIP1, thereby promoting the activation of the NF-κB signaling pathway, inducing the expression of antioxidant enzymes such as SOD and GPx, which is beneficial to scavenging free radicals and thereby alleviating the effects of oxidative stress on cranial nerve damage. Although miR-155 has a positive role in brain neuroprotection, whether it is regulated by salidroside to exert neuroprotection in stroke is unclear, and further research is needed to determine.
However, salidroside has poor stability and is susceptible to degradation due to factors such as light and oxygen. Its solubility is low, which limits its absorption and utilization in the body (Sun, Zhou, et al., 2018). Modification with polydopamine nanoparticles cannot only improve the stability of salidroside and reduce its degradation and inactivation (Liu, Lei, et al., 2022), but also improve the solubility of salidroside, making it easier to dissolve and release. In addition, nanoparticles have smaller particle sizes and good surface activity, which can accurately deliver drugs to specific tissues or cells through passive or active targeting mechanisms, improving the accumulation and effect of drugs in the target area (Liu, Li, et al., 2022). In order to solve this problem, researchers combined salidroside with polydopamine nanoparticles to prepare polydopamine nanoparticle-modified salidroside (PDA-SAL) in order to improve its solubility and bioavailability in water thereby enhancing its neuroprotective effect.
The Notch pathway focuses on nervous system development and diseases (Lin et al., 2019). Studies have shown that activating the Notch pathway can promote the proliferation and differentiation of neural stem cells, promote neuron regeneration and repair, and thus improve the prognosis of stroke (Tan et al., 2022). MiR-155 is an important microRNA that can regulate the activity of the Notch signaling pathway. Studies have shown that inhibiting the expression of miR-155 can activate the Notch signaling pathway, thereby promoting neuron regeneration and repair (Ni et al., 2017). Therefore, this study aimed to explore whether salidroside modified with polydopamine nanoparticles can regulate the Notch signaling pathway by inhibiting miR-155 and exerting neuroprotective effects on stroke rats.
Materials and Methods
Instruments, Reagents, and Animals
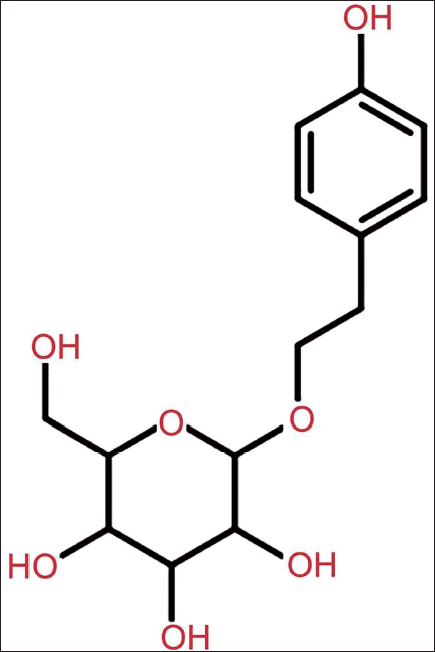
Salidroside (specification: 5 mg, molecular formula: C21H18O5, molecular structure shown in Figure 1) was purchased from Shanghai Yuanye; the secondary antibody (Shanghai Youningwei Biotechnology); scanning electron microscope (SEM) was purchased from Shanghai Carle Zeiss, inverted microscope (Leica Microsystems, Shanghai). C57BL/6 mice, male, were purchased from Shanghai Guandao Biotechnology Co., Ltd.
Molecular Structural Formula of Salidroside.
Methods
Preparation of PDA NPs-SAL
Add dopamine to a solution containing sodium hydroxide and 10% hydrogen peroxide and react at 25°C to synthesize polydopamine. After that, clean it with an ultrasonic wave for half an hour. Dissolve salidroside and polydopamine in methanol, respectively, to obtain a salidroside solution and a polydopamine solution. The salidroside solution and the polydopamine solution were mixed at a ratio of 1:2 and sonicated at 25°C for 1 h. The solution of salidroside and polydopamine conjugate was slowly dropped into the solution of polydopamine nanoparticles, ultrasonicated at 25°C for 1 h, and then the polydopamine nanoparticles in the mixed solution were precipitated by centrifugation. Then, the polydopamine nanoparticles were washed with pure water and dried at low temperatures in a desiccator. That is, PDA NPs-SAL is obtained.
Construction of Stroke Rat Model
A stroke rat model was constructed using the suture method, and 40 Sprague–Dawley (SD) rats were selected, 5 of which were used as the sham group (sham group). Only the skin was incised without any intervention. The remaining rats were anesthetized using 5% isoflurane. Sterilize the skin of the rat’s neck, make an incision in the rat’s neck, and carefully expose the common carotid artery and its branches. Under the guidance of a microscope, insert the suture plug into the middle cerebral artery, stabilize it for 20 min, and block the blood supply for 30 min. Pull out the suture after 2 h. After ensuring arterial return, the incision is closed and sutured. After the rat wakes up, a tail-lifting test is performed to observe the flexion state of the right forelimb. The mold formation rate is 85%. According to the single-blind method (the experimenters did not know which group received which treatment), they were further randomly divided into eight groups, with five mice in each group, including NC group, positive control group (nimodipine), SAL group, PDA NPs-SAL group, PDA NPs-TSL + miR-155 mimic group, PDA NPs-TSL + miR-155 inhibitor group, PDA NPs-TSL + Jagged1 group, and PDA NPs-TSL + LY411575 group. At the same time, special identifiers or codes were used to identify drugs, mice, and so on before the study was carried out, and it was necessary to ensure that the experimenters could not identify the specific treatment methods of each group.
Cognitive Function
The cognitive function of all rats was tested using a water maze experiment. A circular pool (1.5 m in diameter) was divided into four quadrants. An escape platform with a diameter of 12 cm was set in the center of the pool. Rats were placed in the four quadrants and guided to find the escape platform. After 7 consecutive days of training, the time it took for each group of rats to find the escape platform (escape latency) was recorded, and the number of times the rats crossed the platform within 1 min after the platform was removed was also recorded.
Neurobehavioral Score
The degree of nerve damage in rats was evaluated by the nervous system deficit score, which used a scoring standard of 0–4. 0 points: Normal performance, no obvious neurological defects. 1 point: Slight defect, slightly abnormal walking posture, but able to maintain balance. 2 points: Moderate defect, unstable gait, mild limb weakness. 3 points: Obvious defects, severe abnormality in gait, weakened grip, and difficulty in balancing. 4 points: Serious defect, complete loss of walking ability, inability to stand, and move.
2,3,5-Triphenyltetrazolium Chloride (TTC) Cerebral Infarction Volume Determination
After the rats were euthanized, the brain was cut into seven 3 mm thick brain slices and incubated in TTC solution at 37°C for 30 min. After staining, the infarcted area was white due to ischemia and was unable to metabolize TTC, and normal brain tissue was red. After shooting, Image-Pro software was used to measure the cerebral infarction volume.
Apoptosis of Neuronal Cells in Brain Tissue
In the DAPI experiment, the sample to be tested was fixed to remove excess impurities, fixed with 4% paraformaldehyde for 15–30 min, and washed again with PBS. Add drops of DAPI staining solution onto the sample to cover the entire sample area. Stain for 5–10 min to allow the DAPI staining solution to fully penetrate into the cell nucleus. Observe the sample under a fluorescence microscope. The excitation wavelength is generally 350–400 nm. You can see that the cell nucleus exhibits blue or blue-violet fluorescence to determine whether the cell has undergone apoptosis. The nuclei of apoptotic cells usually appear to be condensed, fragmented, and marginalized, and the fluorescence intensity will be correspondingly enhanced.
For TUNEL experiment, the following procedures were performed: dewaxing, in short, glass drying, add 50 µL TUNEL reaction mixture (negative control group 50 µL dUTP solution) and seal, react in a dark humid box, 37°C × 60 min. Wash and take photos before counter-staining. Count and take pictures using a light microscope.
Western Blot Detection
100 mg of brain tissue from each group was taken, the cells were extracted and digested in a dissolving buffer, and the total protein content was measured. Separate by 10% SDS-PAGE and then incubate with primary antibody at 4°C overnight. Wash and incubate the membrane with HRP-labeled secondary antibody for 1 h. Detection was enhanced by the internal reference glyceraldehyde 3-phosphate dehydrogenase (GAPDH).
Enzyme-Linked Immunosorbent Assay (ELISA) to Detect the Expression of Inflammatory Factors in Hippocampal Tissue
Two rats were taken from each group. After anesthesia and sacrifice, the hippocampal tissue on the injured side was removed, and the tissue pieces were cut into pieces at low temperatures and homogenized according to the operating procedures of the ELISA kit.
Quantitative Reverse Transcription Polymerase Chain Reaction (qRT-PCR) Detection
Total ribonucleic acid (RNA) was extracted by the TRIzol method and reverse transcribed into complementary deoxyribonucleic acid (cDNA). Gene expression was measured by using SYBR fluorescent PCR technology. GAPDH is an intrinsic control between ROCK/MLCK and miRNA, and the qRT-PCR results were analyzed. Relative expression levels were estimated using the 2−∇∇Ct method. Table 1 lists the primers and primer sequences.
Primer Sequences.

Statistical Methods
The data obtained in each of the above experiments were analyzed using SPSS 21.0 and GraphPad Prism software. Comparisons were analyzed by one-way analysis of variance (ANOVA) with Newman–Keuls multiple comparison post hoc analysis. If there are no special requirements, p < 0.05 is used as the test standard.
Results
Successfully Constructed PDA NPs-SAL
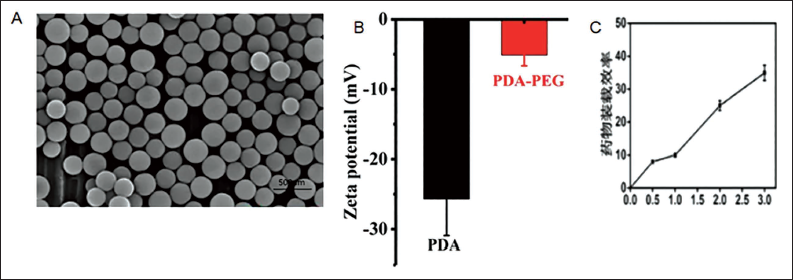
The nanoparticles were round when observed under a transmission electron microscope, and the shell appeared lighter gray or white (Figure 2A). The zeta potential values of PDA and PDA NPs-SAL were −(25.7 ± 5.2) and −(5.1 ± 1.53) mV, respectively (Figure 2B). The SAL drug loading ratio (SAL:PDA) increased with the increase in the amount of SAL added (Figure 2C). At the same time, the encapsulation efficiency of PDA NPs-SAL was (43.06 ± 5.29)%, indicating that PDA NPs-SAL was successfully constructed.
Electron Microscope Image of PDA NPs-SAL. A, Electron Microscopy Image of PDA NPs-SAL; B, Zeta Potential Image of PDA NPs-SAL; C, Drug Loading Rate of PDA NPs-SAL.
Stroke Modeling
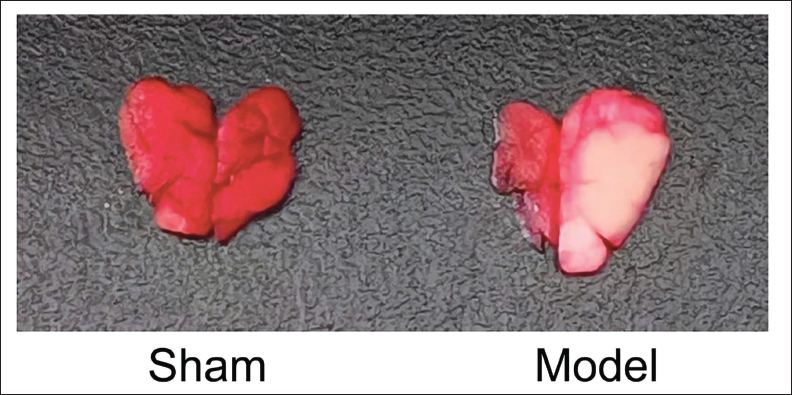
After the rats woke up, there were no abnormal neurobiological responses in the sham group, while the model group had varying degrees of contralateral limb weakness and reduced touch. A tail-raising experiment found that the rats in the model group showed a flexion state of the contralateral forelimb, and when walking on level ground, Horner’s sign is present on the affected side, unable to walk straight, or only turning in circles to the opposite side. The neurobiological score is 1–3 points. Among them, the average scores of sham, modeling 4 h, modeling 8 h, modeling 12 h, and modeling 1 day are 0, 1, 3, 3, 3.55, and then the rats were decapitated and taken. The back of the brain was used for the TTC test. Compared with the sham group, the model group showed obvious stroke lesions, ischemia, and large lesions (Figure 3), indicating successful modeling.
Results of 2,3,5-Triphenyltetrazolium Chloride (TTC) Test on Stroke Lesions.
PDA NPs-SAL has a Neuroprotective Effect on Stroke Rats and is Closely Related to the Phosphorylation Inhibition of the NOTCH Pathway
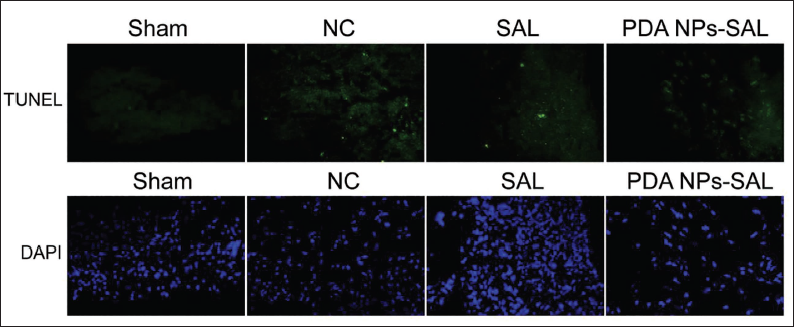
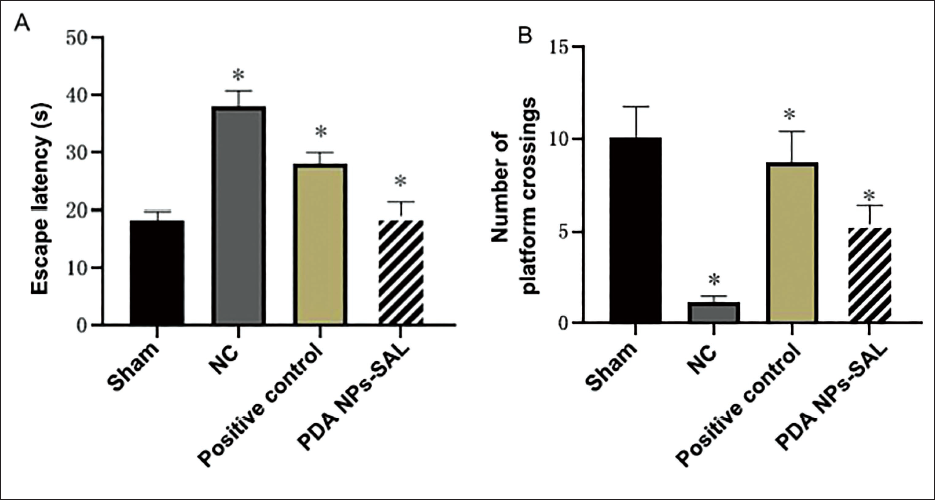
We used PDA NPs-SAL in a stroke rat model and found that the neuronal cell apoptosis rate was significantly reduced under SAL intervention. In particular, this apoptosis phenomenon was further amplified under PDA NPs-SAL intervention (Figure 4). To confirm that PDA NPs-SAL has a protective effect on the neurological function of stroke rats, we used the common neuroprotectant nimodipine to intervene in stroke model rats. The results showed that compared with the positive control group, the escape latency of the rats in the PDA NPs-SAL group was significantly shorter (Figure 5A), while the number of times they crossed the platform was significantly more (Figure 5B).
Apoptosis of Neuronal Cells in Each Group.
Effects of PDA NPs-SAL on Cognitive Function of Stroke Rats. A, Escape Latency; B, Number of Platform Crossings.
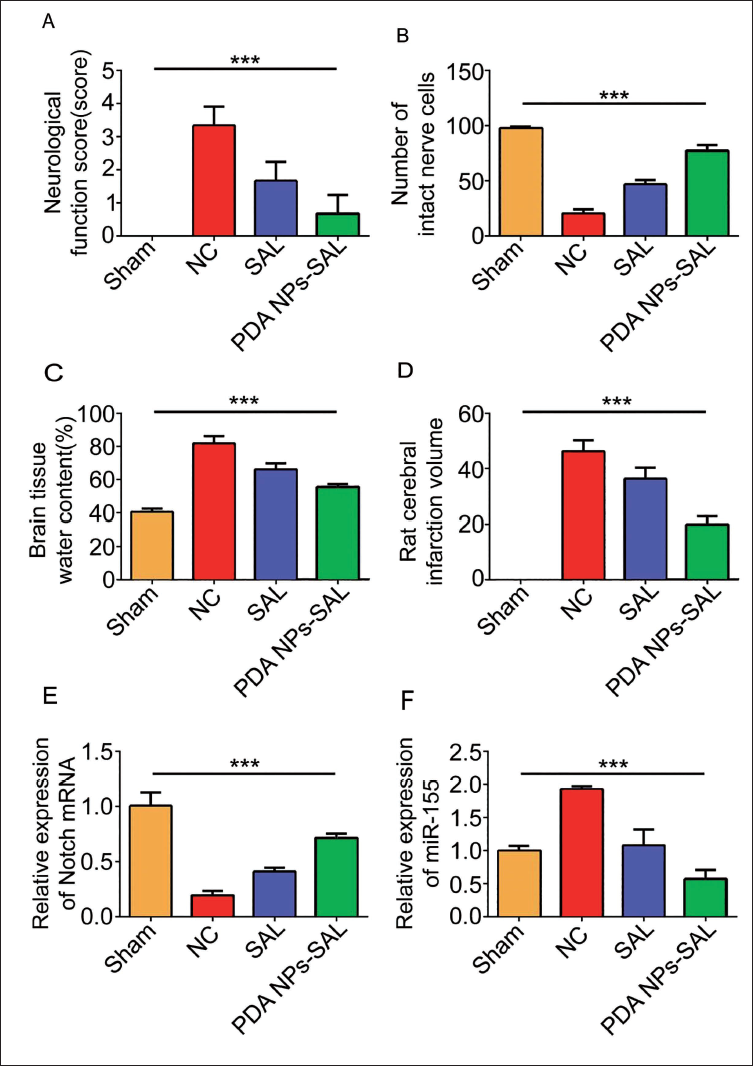
When compared with the sham group and NC group, it was found that the score of the sham group was 0 points, the neurological function score of the rats in the NC group was the highest, the scores of the SAL group and the PDA NPs-SAL group showed a gradual decreasing trend, and the PDA NPs-SAL group decreased most obvious (Figure 6A). At the same time, in the results of Figure 6B, in terms of the number of intact nerve cells in the brains of rats in each group under the electron microscope, SAL and PDA NPs-SAL can slow down the death of nerve cells caused by stroke. This phenomenon deserves further in-depth exploration of the neuroprotective effect of PDA NPs-SAL. After measuring the brain tissue water content and cerebral infarction area of stroke rats, it was found that under the intervention of SAL or PDA NPs-SAL, the brain tissue water content was significantly reduced, and cerebral infarction area was significantly reduced (Figure 6C and D), and the PDA NPs-SAL. The intervention effect is the most significant. In addition, the results of measuring the relative expression of NOTCH mRNA and the expression of miR-155 in rats in each group found that the effect of PDA NPs-SAL on increasing the relative expression of NOTCH mRNA was stronger than that of SAL (Figure 6E), inhibiting miR-155. The effect of expression is more prominent (Figure 6F), which not only shows that PDA NPs-SAL has a neuroprotective effect on stroke rats, but is also found to be closely related to the phosphorylation of the NOTCH pathway.
Effect of PDA NPs-SAL on Neurological Function in Stroke Rats. A, Neurological Function Score; B, Number of Intact Nerve Cells; C, Brain Tissue Water Content; D, Rat Cerebral Infarction Volume; E, Relative Expression of Notch mRNA; F, miR-155 Relative Expression.
PDA NPs-SAL Exerts Neuroprotective Effects on Stroke Rats by Inhibiting the Expression of miR-155 and Activating the Notch Pathway
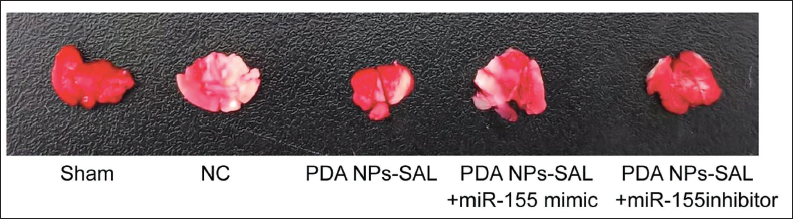
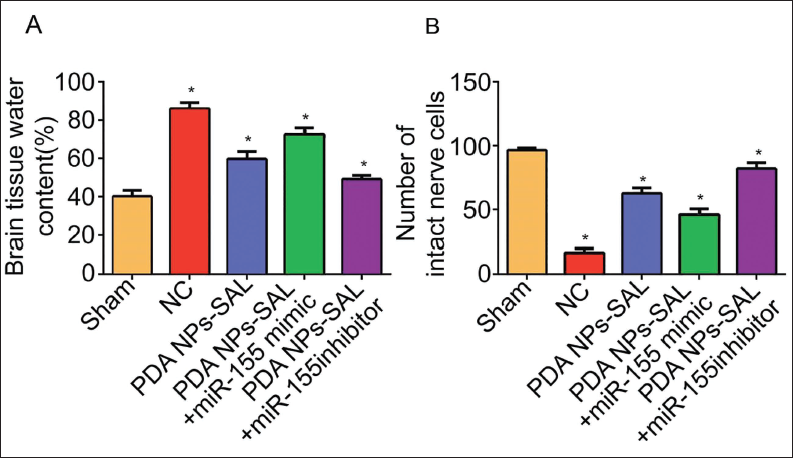
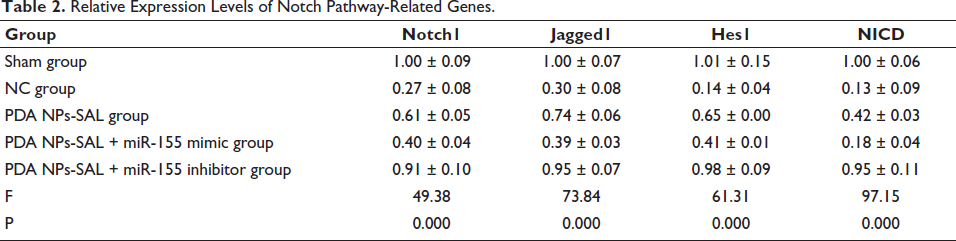
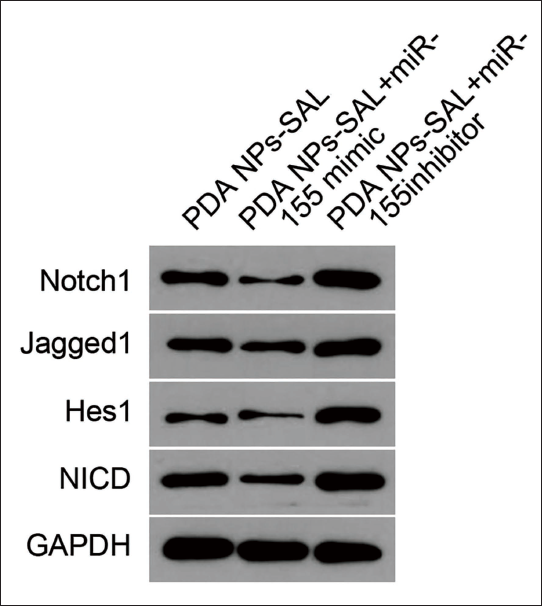
By comparison, it was found that with the further intervention of miR-155 inhibitor, the cerebral infarction area (Figure 7) and brain tissue water content (Figure 8A) of rats were significantly reduced, while the miR-155 mimic group showed opposite results, and the number of intact nerve cells after PDA NPs-SAL intervention was reduced (Figure 8B). The results of the combined use of PDA NPs-SAL and miR-155 inhibitor showed that it had the strongest neuroprotective effect on stroke rats. What is extremely interesting is that in terms of the relative expression of Notch pathway-related genes, the pathway-related genes were activated under the intervention of PDA NPs-SAL, and the change was more significant under PDA NPs-SAL + miR-155 inhibitor (Table 2). It shows that PDA NPs-SAL has the same neuroprotective effect in stroke rats when inhibiting the expression of miR-155. The expression levels of Notch1, Jagged1, and other proteins contradict the above results (Figure 9). In addition, during this experiment, we found that both the PDA NPs-SAL and miR-155 inhibitors could increase the expression of Notch1, Jagged1, and Hes1, and the effect was stronger under the intervention of PDA NPs-SAL + miR-155 (Table 2 vs. NC group, p < 0.05). It is suggested that PDA NPs-SAL inhibits the expression of miR-155 and promotes the phosphorylation of the Notch pathway, exerting a neuroprotective effect on stroke rats.
2,3,5-Triphenyltetrazolium Chloride (TTC) Test Results of Stroke Lesions.
Comparison of Water Content and Number of Intact Nerve Cells in Infarcted Brain Tissue.
Relative Expression Levels of Notch Pathway-Related Genes.
WB Detection of Protein Expression.
Discussion
Research has found that Bupleurin can promote the survival and repair process of nerve cells, increase the production of nerve growth factors NGF and BDNF (Li et al., 2020), and regulate signaling pathways in nerve cells such as PKC and MAPK, thereby promoting the survival and repair of nerve cells. Emodin can interfere with the combination of Keap1 and Nrf2, and prevent the degradation of Nrf2 (Sun, Ma, et al., 2018), allowing it to accumulate in the nucleus, thereby promoting the activation of the Nrf2-ARE pathway, increasing the expression of GPX, SOD, and so on and then protect nerve cells from oxidative stress to a certain extent. Salidroside has certain instability under light, high temperature, acid–base environment, or oxidative conditions. At the same time, its bioavailability in the body is low (Liao et al., 2019). Orally ingested salidroside is easily degraded by gastric acid and intestinal enzymes, resulting in reduced bioavailability. In addition, salidroside also has certain solubility problems, which limits its absorption and utilization in the body. In this study, the PDA NPs-SAL composite was successfully prepared. Studies have reported that salidroside can inhibit the activity of PDK-1, thereby indirectly reducing the activation level of Akt, thereby reducing platelet activation and aggregation. In ischemic stroke, salidroside can activate the TGF-β1/Smad3 signaling pathway (Zhang et al., 2021), thereby reducing the expression of Bax (Feng et al., 2018) and increasing the expression of Bcl-2, thereby reducing neurological damage and protecting nerve cells not damaged. In further animal experiments, this study found that under SAL intervention, the neurological function scores of rats decreased, the number of intact nerve cells in the brain increased, the water content in the brain decreased, and the area of cerebral infarction decreased. The regulation of neuroprotection tended to go in a better direction. The intervention effect of PDA NPs-SAL is more obvious than that of SAL. This shows that SAL has a neuroprotective effect on stroke rats, and the protective effect of PDA NPs-SAL is stronger. This is due to the fact that polydopamine nanoparticles serve as an excellent carrier material to carry salidroside, which can increase salidroside to be water-soluble and bioabsorbable, thereby improving its absorption and utilization in the body. By controlling the interaction between salidroside and polydopamine nanoparticles, its efficacy, stability, and solubility can be regulated, thereby enhancing its biological activity. Salidroside-modified nanoparticles utilize the effect of controlling the release rate (Chen et al., 2020) to achieve the purpose of delayed release and targeted delivery of drugs.
This study found that the apoptosis rate of neuronal cells was significantly reduced under the intervention of PDA NPs-SAL. This apoptosis phenomenon was further amplified under the intervention of PDA NPs-SAL, indicating that PDA NPs-SAL has a neuroprotective effect and can reduce the number of neuron deaths. During stroke, excessive accumulation of intracellular calcium ions (Ca²+) is one of the key factors leading to neuronal damage and death, and nimodipine can protect neurons from calcium overload by blocking voltage-dependent calcium channels and reducing calcium ion influx. In this study, it was found that compared with the positive control group intervened by nimodipine, the escape latency of rats in the PDA NPs-SAL group was significantly shorter, while the number of times they crossed the platform was significantly higher, which further illustrates that PDA NPs-SAL has a certain improvement effect on cognitive function in stroke. Further observations showed that compared with the sham group and the NC group, the neurological function scores of rats in the SAL group and the PDA NPs-SAL group gradually decreased, and the decrease was most obvious in the PDA NPs-SAL group. At the same time, electron microscope observation found that SAL and PDA NPs-SAL can slow down the death of nerve cells caused by stroke. These results further support the neuroprotective effect of PDA NPs-SAL. PDA NPs-SAL has a stronger effect on increasing the relative expression of NOTCH mRNA than SAL, and its effect on inhibiting the expression of miR-155 is more prominent. This suggests that the neuroprotective effect of PDA NPs-SAL on stroke rats may be related to the phosphorylation of the NOTCH pathway and inhibition of the expression of miR-155. Analysis suggests that, on the one hand, the downregulation of miR-155 not only helps promote neuron regeneration and synapse reconstruction (Wu et al., 2016) and improves the damage of brain diseases, but may also participate in the tissue repair process by promoting the proliferation and differentiation of stem cells. Inhibiting the expression of miR-155 and reducing the levels of angiogenesis factors HIF-1α (Wu et al., 2019) and VEGF are helpful in the treatment of lacunar cerebral infarction. In response to cerebral ischemia-reperfusion injury, downregulation of miR-155 leads to a decrease in brain tissue water content. In the process, the expression of Notch1 and Hes1 is increased, thereby improving cerebral ischemia-reperfusion injury. Some studies have found that salidroside can inhibit the expression of miR-155 by directly targeting regulatory factors such as NF-κB (Lu et al., 2017) and AP-1 (Huang et al., 2017), which are upstream of miR-155, and inhibiting their activity. AMPK is an important energy sensor (Luo et al., 2016). Salidroside can inhibit the expression of miR-155 by activating the AMPK pathway (Gui et al., 2017), and PDA NPs-SAL further amplifies this effect and shows an extremely excellent inhibitory effect.
DAPT can block the Notch pathway, improve blood lipids in AS ischemic stroke rats, inhibit the release of TNF-α, IL-6, and so on and the activation of the NF-κB/TLR4 pathway, thereby maximizing improvement. The symptoms of AS ischemic stroke (Yan et al., 2021). In the study, the cerebral infarct area and brain tissue water content of rats were significantly reduced under the intervention of miR-155 inhibitors, while the opposite results were seen when using miR-155 mimics, indicating that miR-155 is involved in the brain pathological process of stroke, and inhibiting its expression may help reduce brain damage. The combined use of PDA NPs-SAL and miR-155 inhibitors has the strongest neuroprotective effect on stroke rats, further supporting the synergistic effect of PDA NPs-SAL and inhibiting the expression of miR-155 in neuroprotection. Genes related to this pathway were activated under the intervention of PDA NPs-SAL, and this change was more significant under the intervention of PDA NPs-SAL + miR-155 inhibitor, which suggested that the PDA NPs-SAL and miR-155 inhibitors to a certain extent can increase the expression of Notch1, Jagged1, and Hes1, and the effect is stronger under the intervention of PDA NPs-SAL + miR-155. This further supports that PDA NPs-SAL inhibits the expression of miR-155 and promotes phosphorylation of the Notch pathway, thereby exerting a neuroprotective effect on stroke rats.
Conclusion
In summary, PDA NPs-SAL promotes the Notch pathway by inhibiting miR-155, thereby exerting a neuroprotective effect on stroke rats, and shows that PDA NPs-SAL and inhibiting the expression of miR-155 play a role in stroke rats. It synergizes in neuroprotection and may act through the activation of the Notch pathway. These findings provide the experimental basis for the development of new stroke treatments.
Footnotes
Abbreviations
ELISA: Enzyme-linked immunosorbent assay; GAPDH: Glyceraldehyde 3-phosphate dehydrogenase; PDA-SAL: Polydopamine nanoparticle-modified salidroside; PTEN: Phosphatase and tensin homolog; SD: Sprague–Dawley; TTC: 2,3,5-Triphenyltetrazolium chloride.
Acknowledgments
The authors gratefully acknowledge the Dongping People’s Hospital Laboratory for providing the necessary equipment for this study.
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest regarding the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the ethics committee of Dongping People’s Hospital.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
