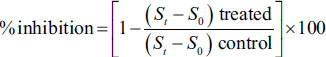Abstract
Background
Moroccan traditional medicine utilizes Juniperus thurifera and Juniperus phoenicea to treat numerous ailments.
Objectives
This research investigates the chemical composition, acute toxicity, and anti-inflammatory properties of J. thurifera (L.) and J. phoenicea (L.) hydroethanolic extracts.
Materials and Methods
High-performance liquid chromatography with a diode array detector (HPLC-DAD) identified the chemical makeup of J. phoenicea and J. thurifera hydroethanolic extracts. We tested the acute toxicity of hydroethanolic extracts of J. thurifera (T) and J. phoenicea (P) leaves in Wistar rats by gastric gavage at doses of 300, 1,000, and 2,000 mg/kg. Wistar rats were injected with 1% carrageenan in the plantar fascia to assess anti-inflammatory activity.
Results
Chemical investigation of the extracts revealed gallic acid, urocanic acid, linalool, ferulic acid, and mefenamic acid. Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels did not significantly vary based on biochemical markers, including urea, creatinine (CR), AST, and ALT. Both extracts increased CR and urea, especially J. phoenicea, at 1,000 mg/kg. The hydroethanolic extracts of J. phoenicea (P) and J. thurifera (T) demonstrated 100% and 40% edema-inhibiting action at 6 h, respectively.
Conclusion
The investigated extracts exhibited a strong anti-inflammatory effect. However, they may harm the kidneys, particularly J. phoenicea, which may explain why J. thurifera is overused.
Introduction
Coniferous plants of the Juniperus genus, belonging to the Cupressaceae family, play a significant role in arid and semiarid habitats throughout the northern hemisphere. In Morocco’s central High Atlas, Juniperus is one of the most genera frequently used by traditional healers (Gauquelin et al., 1999). More than 4,200 different species of vascular plants make up the Moroccan flora (Benabid, 2000). However, few of them have been examined for their pharmacological or chemical properties; the most predominant family among the conifers (gymnosperms) is the Cupressaceae (Hseini & Kahouadji, 2007). They leave a striking imprint on the plant landscape of the Mediterranean basin and constitute one of the world’s biodiversity hotspots (Gauquelin et al., 1999; Médail & Diadema, 2006).
Terpenoids, alkaloids, tannins, flavonoids, and other phytochemicals are present in medicinal plants and have a variety of pharmacological effects such as anti-microbial, anti-oxidant, anti-inflammatory, anti-bacterial, analgesic, anti-protozoal, cytotoxic, anti-viral, anti-pyretic, antitumor, anti-depressant, hypolipidemic, and anti-helmintic (Andersen & Jordheim 2005; Arasu et al., 2021; Oomah et al., 1996; Ouattar et al., 2022; Petrović et al., 2019). Ecological factors determine the role or significance of secondary metabolites in plants (Tiwari & Rana, 2015; Zouirech et al., 2022).
Juniperus species, crucial elements of Moroccan ecosystems, are facing varying degrees of degradation (Gauquelin et al., 1999). More specifically, J. thurifera is more degraded than J. phoenicea, despite belonging to the same Juniperus genus (Lafraxo et al. 2023). This disparity raises fascinating questions as to the factors underlying this divergence.
This study aims to assess the toxicity of J. thurifera extracts (T) compared with J. phoenicea extracts (P). It focuses on their chemical compositions. Extracts of both species were subjected to acute toxicity tests in animals in accordance with Organization for Economic Cooperation and Development (OECD) guidelines (425 and 423). In addition, the study evaluates the toxic effects of the two species on herbivores, examining the impact of these plants on animal health. Notably on markers such as aspartate aminotransferase (AST), alanine aminotransferase (ALT), and creatinine (CR), and subsequently analyzing the effects of these extracts on the organs of exposed animals by measuring the ROW results of biological tests (ROW kidney, ROW spleen, and ROW liver). A comparison of anti-inflammatory tests on the two juniper species, J. phoenicea and J. thurifera, also revealed significant differences in their ability to inhibit inflammation.
Materials and Methods
Plant Material
The leaves studied from J. thurifera and J. pheonicea were collected from plants in a mountainous region (lat: 33.68093368; long: 4.30823143) at the beginning of June 2023 where there is a population of trees of these two species (Figure 1 and Figure 2). The samples were deposited in a biology department herbarium under a reference number (FJT/02D20) after identification of the two plants concerned by a botanist from the Biology Department, Faculty of Sciences-FSDM-USMBA-Fez. The leaves were cleaned and dried at 35°C for 72 h in a ventilated oven. The dried samples were ground using an electric blender.
Preparation of Extracts
The studied parts were cleaned and dried under cover in an oven at 40°C for a few days. The dried parts were gutted and ground into a fine powder. 70 mL of ethanol and 30 mL of distilled water were added to 10 g in order to prepare an extraction of 10%. The mixture was left to macerate for 24 h and filtered through Whatman paper (No. 4). The liquid extract was dried in an oven at 40°C. The crude extracts were stored in tinted bottles at 4°C.
Chemical Analysis
Leaf extracts from J. phoenicea L. (P) and J. thurifera L. (T) were subjected to chemical analysis using reverse-phase high-performance liquid chromatography with a diode array detector (HPLC-DAD). We employed a Thermo Scientific HPLC system equipped with a MOS-1 HYPERSIL 250 × 4.6 mm SS Exsil ODS 5 µm analytical column. The analysis was performed in gradient mode with two solvents: A (water) and C (acetonitrile), following this elution gradient: 80% A, 20% C for 1 min; 60% A, 40% C for 2.5 min; and 80% A, 20% C for 4 min. The flow rate was maintained at 1 mL/min, and the injection volume was 5 µL. Compound identification relied on comparing their retention times and UV spectra with those of authentic standards (Bouslamti et al., 2022).
Acute Toxicity Test
Animal Model
Adult Swiss albino mice weighing approximately 25–30 g were obtained from the animal house of the Department of Biology, University Sidi Mohamed ben Abdellah, Faculty of Sciences, Fez. The animals were housed under controlled laboratory conditions of temperature (23 ± 2°C) and light (12 h light/dark cycles), with uninhibited access to food and water. The use of laboratory animals in all experimental procedures was conducted according to the ethical guidelines for the care and use of laboratory animals (National Research Council Committee).
Acute Toxicity
The acute toxicity assay was conducted following the OECD guidelines no. 423. Acute toxicity of J. phoenicea and J. thurifera extracts at different doses was performed using 12 h fasted mice. Seven groups, with five animals, each were used. Extracts were administered orally by gastric gavage as a single dose to animal groups as follows:
Group 1: Negative control (0.9% NaCl);
Group 2: Juniperus phoenicea hydroethanolic extract (300 mg/kg);
Group 3: J. phoenicea hydroethanolic extract (1,000 mg/kg);
Group 4: J. phoenicea hydroethanolic extract (2,000 mg/kg);
Group 5: Juniperus thurifera hydroethanolic extract (300 mg/kg);
Group 6: J. thurifera hydroethanolic extract (1,000 mg/kg);
Group 7: J. thurifera hydroethanolic extract (2,000 mg/kg).
Symptoms of toxicity such as the general appearance of the animal, including behavior, drowsiness, diarrhea, restlessness, and mortality (if any), were observed and recorded. The acute toxicity test was used to determine the median lethal dose (LD50) of J. phoenicea and J. thurifera extracts (Chinedu et al., 2013).
Biochemical Parameters
At the end of the experimental period of the test, the animals were anesthetized with sodium pentobarbital at a dose of 30 mg/kg before being sacrificed for blood collection. Blood samples were collected from the heart in heparinized tubes (0.2 mL with 10 U/mL heparin) and then centrifuged at 1,500 rpm for 10 min. Next, the recovered plasma was analyzed for AST, ALT, CR, and urea parameters.
Anti-inflammatory Activity
The anti-inflammatory effect of each studied extract was evaluated by carrageenan-induced paw edema. Animals were divided into four groups of five each and treated as follows:
Group 1: Negative control (0.9% NaCl)
Group 2: Positive control (10 mg/kg of indomethacin)
Group 3: J. phoenicea hydroethanolic extract (300 mg/kg)
Group 4: J. thurifera hydroethanolic extract (300 mg/kg).
After 1 h of gastric gavage, inflammation was induced by injecting 0.1 mL of the carrageenan agent in 0.9% saline into the rats’ right paws. Initial paw size was measured before carrageenan injection and after 3, 4, 5, and 6 h of treatment.
The average increase in the paw size of each group was determined and compared to both the positive and the negative control. The percentage of inhibition of edema was calculated as follows:
where St is the paw size after the carrageenan injection, and S0 is the initial paw size before the carrageenan injection.
Statistical Analysis
Experimental results were subjected to statistical analysis using GraphPad Prism 7.1 software (Microsoft, USA). Values are expressed as mean standard error. Data were evaluated using one-way analysis of variance (ANOVA), followed by Tukey’s multiple comparison test, with a significance level of 5%, to assess the significance of observed differences. A difference between values is considered significant if p < 0.05. Graphical representations were also generated using the ANOVA analysis method with the same software.
Results
Chemical Analysis
The chemical analysis of J. thurifera L. leaf extracts, conducted using reverse-phase HPLC-DAD, revealed several important findings (Figure 3). Peaks corresponding to specific compounds were identified and included gallic acid (retention time: 3.419 min), urocanic acid (retention time: 4.870 min), linalool (retention time: 15.234 min), ferulic acid (retention time: 16.549 min), and mefenamic acid (retention time: 23.807 min). These compounds were quantified based on the areas under their respective peaks, and their presence was confirmed by matching retention times and UV spectra with authentic standards.
Photo of a Juniperus thurifera tree on the BouIblane mountain.

Photo of a Juniperus phoenicea tree on the BouIblane mountain.

High-Performance Liquid Chromatography (HPLC) Chromatogram of Juniperus thurifera Extract at 280 nm.
Of these, urocanic acid was the most abundant, accounting for 16.24% of the total surface area, followed by gallic acid (0.89%), linalool (0.19%), ferulic acid (0.38%), and mefenamic acid (0.93%). The total area of identified compounds represented 29.83% of the analysis, suggesting that these compounds make up a significant part of the chemical composition of leaf extracts from J. thurifera L. However, it is important to recognize that some peaks presented purity problems or lacked sufficient valid data points, indicating potential difficulties in accurately characterizing certain compounds. Further research and validation studies may be required to resolve these purity and data quality issues. These results provide valuable insights into the chemical composition of J. thurifera leaf extracts, with urocanic acid being the most abundant compound among those identified, which could have implications for its potential pharmacological and therapeutic importance.
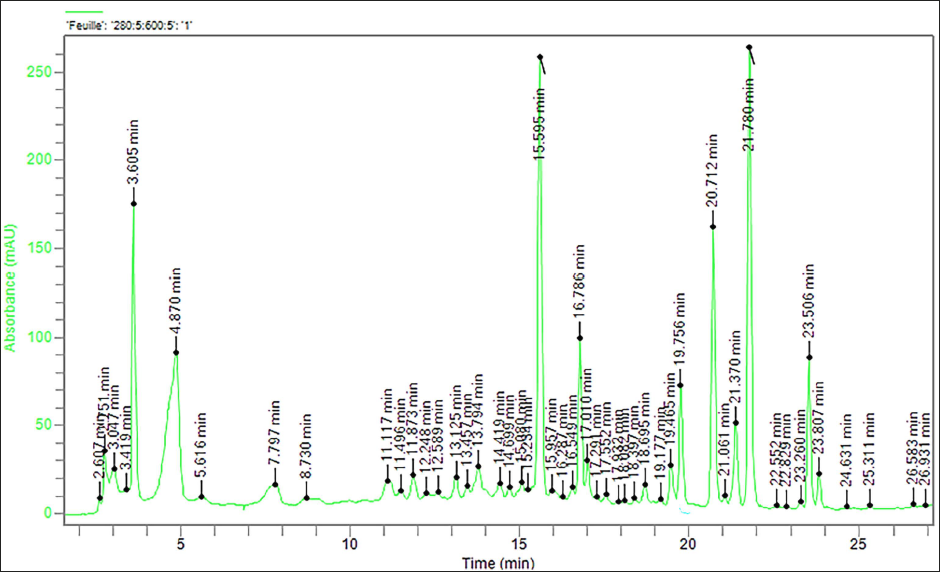
The HPLC-DAD analysis of J. phoenicea leaf extracts yielded insightful findings (Figure 4). Several compounds were identified, with gallic acid (retention time: 3.046 min) and urocanic acid (retention time: 4.621 min) being the most prominent. These compounds accounted for 17.21% and 8.84% of the total peak area, respectively. Gallic acid is known for its potent antioxidant properties and potential anti-inflammatory effects, making it a compound of interest for further research into its health benefits. Urocanic acid, derived from histidine, is associated with immune regulation and skin protection against harmful UV radiation. Its presence in the extracts suggests possible applications in skincare or UV protection products.
High-Performance Liquid Chromatography (HPLC) Chromatogram of Juniperus phoenicea Extract at 280 nm.
Additional compounds identified included linalool (retention time: 15.089 min) and mefenamic acid (retention time: 23.890 min), constituting 0.51% and 0.07% of the total area, respectively. Linalool, apart from its pleasant floral scent, has demonstrated antimicrobial, anti-inflammatory, and anxiolytic properties, making it suitable for aromatherapy and potential therapeutic applications. Mefenamic acid, commonly used as a non-steroidal anti-inflammatory drug (NSAID), is known for its pain-relieving and anti-inflammatory effects. Its presence opens possibilities for traditional medicine or new therapeutic applications.
The total area of identified compounds represented 36.63% of the overall analysis. However, it is noteworthy that certain peaks displayed challenges related to peak purity or insufficient data points, which may affect the accuracy of quantification. These results provide valuable insights into the chemical composition of J. phoenicea leaf extracts, with gallic acid and urocanic acid being the major constituents, warranting further investigation into their potential biological activities and pharmacological relevance. Addressing issues with peak purity and data quality will be essential for comprehensive characterization and future research endeavors related to these extracts.
Acute Toxicity Test
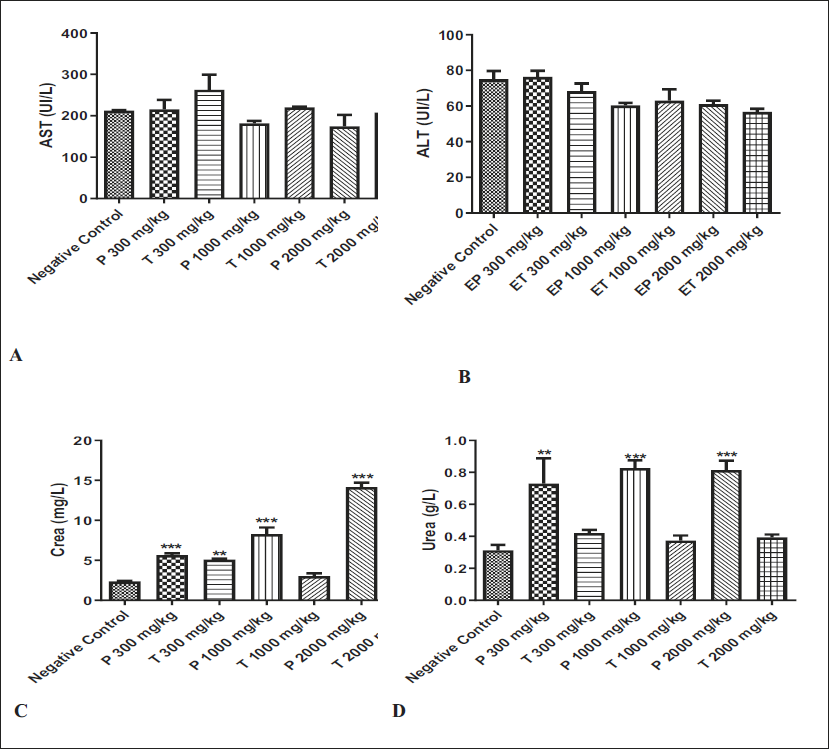
The results of AST, ALT, CR, and urea levels in the serum recovered from the mice treated with (P) and (T) extracts are presented in Figure 5. Compared to the negative control group, the enzymatic activities of AST (A), and ALT (B) measured in the plasma from mice treated with (P) and (T) at all tested doses showed no significant difference. However, both (P) and (T) extracts at all tested doses demonstrated a significant increase in CR and urea levels. Additionally, CR levels were significantly higher in the serum of mice treated with the (T) extract at a dose of 300 mg/kg.
Effects of Juniperus phoenicea and Juniperus thurifera Hydroethanolic Extract on Aspartate Aminotransferase (AST) (A), Alanine Aminotransferase (ALT) (B), and the Kidney Crea (C) and Urea (D) Quantification During the Acute Toxicity Test. * Indicates a Significant Difference at p < 0.05 from the Negative Control Group. Values are Expressed as Mean ± Standard Error of the Mean (SEM) (n = 5).
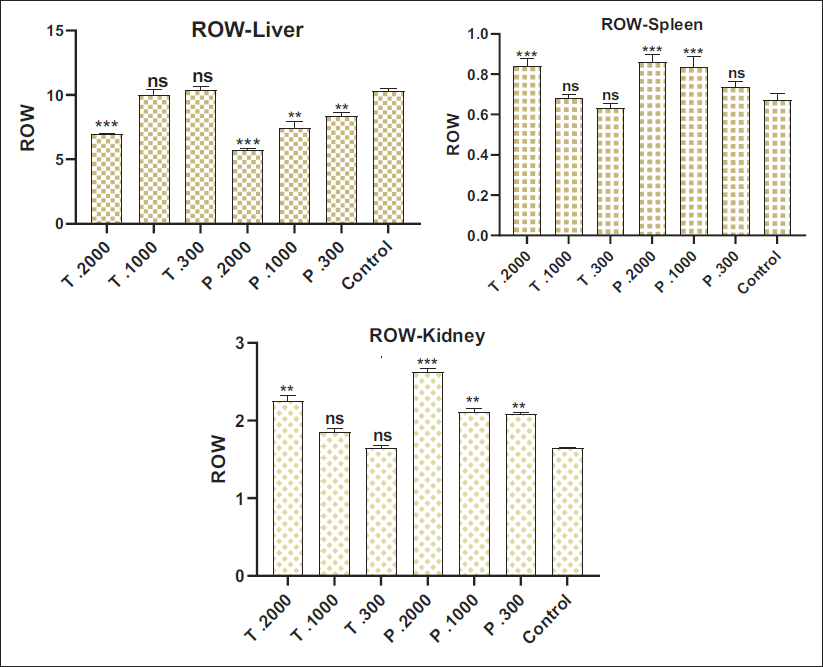
When exploring the different concentrations within each plant, a dose-dependent trend emerges. Organ measurements increase consistently with concentration, indicating a relationship between the amount of extract and its effect on the relative weights of the liver, kidney, and spleen (Figure 6). The findings suggest a notable disparity in toxic effects between the two plants. Analysis of the different concentrations within each plant reveals significant variations, highlighting a dose-dependent relationship. This observation is particularly evident in organ measurements, where increasing concentrations are often associated with an intensification of toxic effects.
Effect of Extracts Juniperus phoenicea and Juniperus thurifera on Animal Organ Weights.
Evaluation of the Anti-inflammatory Effects of Extracts
The results of the anti-inflammatory effects of EP and ET extracts at a dose of 300 mg/kg are summarized in Figure 7.
Evaluation of the Anti-inflammatory Effects of EP and ET Extracts on Carrageenan-induced Edema.
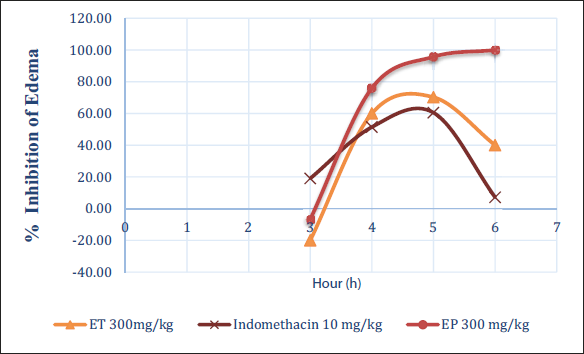
Edema was intentionally induced in the left hind leg of rats. Changes in edema were assessed 1 h after oral administration of the different extracts. The results were compared with those obtained using indomethacin. The outcomes for the treated rats were also compared with those from the edematous control. Observations showed that the extract of EP (J. phoenicea) and ET (J. thurifera) at a dose of 300 mg/kg had a significant anti-inflammatory effect compared with the positive control “indomethacin.”
EP extract showed a marked anti-inflammatory activity compared with ET extract. The results indicate that the anti-edematous effect of EP extract is comparable, if not superior, to that of indomethacin. These results suggest that the EP and ET extracts may have promising anti-inflammatory potential. The superior efficacy of the PE extract compared with ET underlines possible differences in the chemical composition and pharmacological properties of the two extracts. The obtained results showed that hydroethanolic extract of EP and ET at 300 mg/kg showed an important anti-inflammatory effect compared to positive control (Indomethacin). On the other side, the EP extract presented a significant anti-inflammatory activity compared to the ET extract.
Discussion
Plants contain various potentially toxic compounds that, once absorbed, tend to accumulate in the liver, a key detoxification organ (Saad et al., 2006; Zouirech et al., 2023). Indeed, the liver plays an essential role in metabolizing foreign substances, sometimes converting them into potentially hepatotoxic compounds (Moselhy & Ali, 2009; Perveen et al., 2017). This process makes the liver the first target of toxicity, being the first organ exposed to anything absorbed through the small intestine (Bischoff et al., 2018; Maxcy & Wallace, 2012).
The liver works closely with the kidneys to remove toxic substances from the blood, underlining the importance of assessing renal and hepatic function when using herbal medicines (Abou Seif, 2016). In-depth studies of these functions can provide crucial clues regarding the possible toxic effects of medicinal plants. Tests commonly used for this purpose include the determination of AST, ALT, CR, and other relevant parameters (Huang et al., 2006).
A significant increase in serum AST, ALT, and enzyme levels is observed in cases of liver cell necrosis, which serves as a reliable indicator of adverse effects on the liver. These markers are crucial for assessing liver health and can be used as indicators of the liver’s response to potentially toxic compounds present in medicinal plants. In short, careful monitoring of liver and kidney function through these specific tests is essential to ensure safety when using medicinal plants (Ksheerasagar et al., 2011; Ozer et al., 2008).
Results of biochemical parameter analysis in rats treated with hydroethanolic extracts of the two plants studied, J. phoenicea and J. thurifera, revealed no significant variations in levels of AST and ALT, two important enzymes associated with liver function. ALT is a cytosolic enzyme released during hepatic necrosis, and AST, although present in the liver, is found in other organs such as the heart, skeletal muscles, lungs, and kidneys (Nadia et al., 2023; Scheig, 1996). In particular, the results of the study revealed a marked reduction in AST activity in rats, accompanied by a significant decrease in ALT (Rasool et al., 2020).
AST and ALT are liver enzymes that play a crucial role in amino acid metabolism (Jegatheesan et al., 2016). Elevated AST levels are often associated with liver damage, while a decrease in ALT may indicate reduced liver activity (Gowda et al., 2009). These results suggest that aqueous extracts of the plants studied have no specific effects on liver function in rats. However, an increase in AST may indicate liver damage, but it may also be influenced by other factors, such as muscle damage. Similarly, the decrease observed in AST and ALT levels in animals treated with different doses of hydroethanolic extracts, although not statistically significant, suggests a possible hepatoprotective effect of J. phoenicea and J. thurifera extracts. However, a decrease in ALT can be interpreted in different ways and may not necessarily reflect an improvement in liver health.
It is important to note that the observed decrease in the AST and ALT levels, although not statistically significant in this study, tended to intensify with increasing doses administered. This could indicate a possible dose-dependent relationship between these hepatoprotective effects (Nathwani et al., 2005; Oulkheir et al., 2017). Elevated levels of these enzymes are generally associated with organ damage, including hepatic necrosis, cirrhosis, and drug-induced liver toxicity (Jegatheesan et al., 2016; Licata, 2016). In this context, an observed reduction in these markers suggests a potential protective effect of the hydroethanolic extracts of the plants studied on the livers of treated rats.
With regard to renal parameters such as CR and urea, the presence of significant changes suggests that, in this particular study, treatment with the hydroethanolic extracts of the plants has marked effects on the renal function of the rats (Arsad et al., 2013; Meneses et al., 2023). Comparative analysis of the hydroethanolic extracts of J. phoenicea and J. thurifera reveals more pronounced toxicity for J. phoenicea, according to the three doses tested (300 mg, 1000 mg, and 2000 mg). These results suggest that J. phoenicea may be more toxic than J. thurifera, particularly concerning CR and urea parameters. These findings may be reassuring regarding the potential impact on the kidneys. However, it is important to emphasize that further studies are needed for a more complete assessment of renal safety.
The flavonoid-rich chemical composition of J. phoenicea and J. thurifera appears to be a key element in the search for the compounds responsible for their marked effects on renal function. Flavonoids are plant compounds known for their antioxidant and hepatoprotective properties, as demonstrated by several studies (Farghali et al., 2015; Madrigal et al., 2014). The abundance of flavonoids in J. phoenicea and J. thurifera (Lafraxo et al., 2022) suggests that these compounds could play a significant role in the beneficial effects observed on liver function in rats.
The observed inflammatory effect can be due to gallic acid, linalool, and mefenamic acid, identified in the J. phoenicea extract by HPLC (Figure 7) (Nayebi et al., 2017; Uritu et al., 2018). Gallic acid has been demonstrated to have a powerful anti-inflammatory activity by exerting an effect on MAPK and NF-κB signaling pathways (Bai et al. 2021; Locatelli et al., 2013). NSAIDs, such as mefenamic acid, are widely used to treat inflammatory diseases, and their effect is related to the inhibition of the conversion of arachidonic acid to prostaglandins, which are mediators of the inflammatory process (Bindu et al., 2020).
Conclusion
The results suggest a significant difference in renal toxicity between J. thurifera and J. phoenicea, although both species show no liver toxicity as measured by the enzymes AST and ALT. It appears that the higher renal toxicity observed in J. phoenicea may be attributed to specific compounds present in its hydroethanolic extracts, potentially having adverse effects on renal function. Although both species have demonstrated anti-inflammatory properties, the prevalence of greater renal toxicity in J. phoenicea raises questions about its use despite these anti-inflammatory benefits. The absence of kidney toxicity in J. thurifera, in contrast to J. phoenicea, suggests that J. thurifera could be considered a safer source for grazing and other uses, particularly in high mountain environments. However, to ensure responsible use of this resource, a holistic approach is essential.
Abbreviations
ALT: Alanine aminotransferase; ANOVA: Analysis of variance; AST: Aspartate aminotransferase; CR: Creatinine; EP: The hydroethanolic extracts of J. phoenicea (EP); ET: The hydroethanolic extracts of J. thurifera (ET); HPLC-DAD: High-performance liquid chromatography equipped with diode array detector.
Footnotes
Acknowledgments
The authors extend their appreciation to the Researchers Supporting Project number (RSPD2024R686), King Saud University, Riyadh, Saudi Arabia.
Author’s Contribution
All authors contributed significantly to the article.
Data Availability Statement
Data will be available upon request from the corresponding author.
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The procedures used to perform this study are in agreement with the international guidelines used for the use of laboratory animals. The ethical committee of the Faculty of Sciences of Fez, Morocco, revised and approved this work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is financially supported by the Researchers Supporting Project number RSPD2024R686, King Saud University, Riyadh, Saudi Arabia.