Abstract
Inflammation is one of the mechanisms involved in the acute kidney injury (AKI) caused by cisplatin (CP)-induced nephrotoxicity. Tempol (4-hydroxy-2,2,6,6-tetramethylpiperidin-1-oxyl) has powerful antioxidant activity. We investigated its potential nephroprotective effects and the underlying mechanisms that may add further benefits to its clinical usefulness in a CP-induced AKI model. Male Swiss albino mice were divided randomly into four groups: control, CP (20 mg/kg intraperitoneally), tempol (100 mg/kg/day, per os) + CP, and tempol only treatments. Blood samples were collected to analyze renal function parameters. Immunoblotting and immunohistochemical analysis were used to assess the level and localization of inflammatory markers. Tempol afforded protection to animals from CP-induced elevation of inflammatory markers as indicated by reduced expression of nuclear factor-kappa B, cyclooxygenase-2, and tumor necrosis factor-α in kidney tissue. Histological findings and analysis of kidney function markers corroborated with these findings confirming a nephroprotective role for tempol. In conclusion, this study provides important evidence for the promising anti-inflammatory effects of tempol which appears to contribute significantly to its nephroprotective action.
Introduction
The growing interest in the role of oxidative stress, apoptosis, and inflammation in a range of illness processes has lead to the search of drugs that prevent the generation of reactive oxygen species and reduce the inflammatory response. 1,2 Of a hoard of antioxidants developed and investigated tempol (4-hydroxy-2,2,6,6-tetramethylpiperidin-1-oxyl) has some unique properties. 2 It is a membrane-permeable radical scavenger, which has superoxide dismutase (SOD) and catalase-like (mimetic) activities with potential to prevent organ toxicity of brain, heart, liver, and kidney. 3,4 It has been reported that tempol reduced cerebral edema in hypoxic brain and also protected the neurodegenerative disorder in mice. 5,6 Tempol exerts the protective effect by restoring oxidative balance in mitochondria against oxidative damage inside cells. 3 Its therapeutic role against acute liver injury via restoration of nuclear factor-kappa B (NF-κB) expression has been reported by Abouzied et al. 7 Tempol was effective in alcoholic and nonalcoholic models of liver disease. 8 The antioxidant activities of tempol have been already demonstrated experimentally in gentamicin and vancomycin-induced nephrotoxicity. 9 Chatterjee et al. reported that tempol protect rat proximal tubular cells against H2O2-induced cellular injury or death. 10 It is also reported to protect kidney from acute injury through PI3K/Akt/Nrf2 signaling pathway. 11 Gonzalez et al. demonstrated experimentally that tempol ameliorated cyclophosphamide-induced oxidative stress in rat micturition reflex. 12
Nephrotoxicity is the main concern of anticancer drugs. Especially, in case of cisplatin (cis-diamminedichloroplatinum [II]) (CP)-treated patients, nephrotoxicity is the dominant side effect. 13,14 Studies reveal severe toxicity of CP to renal cells in patients in more than 30–35% of cases. 13 –15 There is also a strong evidence showing involvement of inflammatory mediators in the pathogenesis of CP-induced acute kidney injury (AKI). 16,17 Tempol has been shown to ameliorate CP-induced mitochondrial dysfunction in kidney, lipid peroxidation, and DNA damage. 4,18 Tempol has also shown protective effects against toxicity caused by cyclophosphamide, alcohol, and antibiotics. 8,9,12 Since there is involvement of inflammatory cascade in CP-induced nephrotoxicity, it would be interesting to study whether tempol afforded protection also involves the anti-inflammatory mechanism. Hence, the present study takes into account this aspect of nephroprotective property of tempol.
Materials and methods
Chemicals
CP and tempol were purchased from Dr Reddy’s Laboratories (Hyderabad, India) and Sigma-Aldrich Co. (St Louis, Missouri USA), respectively. Hematoxylin, Ponceau S, and Triton X-100 were purchased from Sigma-Aldrich. Acrylamide, bisacrylamide, diacetyl monoxime, ethylene-diaminetetraacetic acid, eosin, β-mercaptoethanol, and tris-hydroxymethyl aminomethane were procured from Sisco Research Laboratories Pvt. Ltd. (Mumbai, India). Peroxidase from horseradish was from E. Merck Ltd. (Mumbai, India). Nonidet P-40 was obtained from Amresco Llc. (Celveland, Ohio, USA). All other chemicals and reagents were of the high purity grade obtained locally from accredited vendors.
Animals and treatment
Male Swiss albino mice (28 ± 5 g) obtained from the Central Animal House Facility were divided into four groups (n = 6). Animals were maintained on a standard laboratory diet and water ad libitum, a 12-h light/dark cycle, and temperature of 22 ± 2°C. The study protocols were approved by the Institutional Animal Ethics Committee of the University (project # 1198). Animals were acclimatized for minimum 7 days before their use. To generate the CP-induced AKI, mice were injected with a single dose of CP (20 mg/kg in normal saline), intraperitoneally (i.p.). Tempol (100 mg/kg) was dissolved in water. CP and tempol dose schedules were based on the preliminary investigation involving a range of doses and also from previously published reports. 4,16 Tempol was administered through oral gavage once a day for 4 days followed by i.p. CP injection. Mice were anesthetized with pentobarbital (30 mg/kg), subsequently killed at 72 h after CP injection. Renal tissue and blood were collected for further analysis. A pictorial representation of animal treatment regimen is shown in Figure 1.

Schematic representation of animal treatment groups.
Assessment of renal function biochemical markers
Serum was prepared from blood and blood urea nitrogen (BUN) and creatinine were analyzed by the standard methods. 19,20
Western blot analysis
The protein level of inflammatory markers, namely NF-κB, cyclooxygenase-2 (COX-2), and tumor necrosis factor-α (TNF-α), was analyzed by western blotting. β-Actin was used as house-keeping protein. For western blot analysis, an equal amount of protein (100 μg) was loaded and subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis. The separated proteins were then blotted onto polyvinylidene difluoride (PVDF) membrane (IPVH00010; Merck-Millipore, Burlington, Massachusetts, USA). The PVDF membranes were incubated with a blocking buffer containing 5% BSA in phosphate buffer saline Tween-20 (PBST) and incubated with diluted rabbit polyclonal antibodies, namely anti-NF-κB (p65) (dilution 1:1000; Bio Legend, San Diego, California, USA), anti-TNF-α (dilution 1:1000; eBioscience, San Diego, California, USA), and anti-COX-2 (dilution 1:1000; Santa Cruz Biotechnology Inc., Dallas, Texas, USA). The blots were washed with PBST and incubated with horseradish peroxidase-conjugated secondary antibody. Signals were visualized by chemiluminescent detection kit according to the manufacturers’ protocol (WBKLS0500; Merck-Millipore, Burlington, Massachusetts, USA). The relative optical density of bands was determined using Image J software Java Version 6. 21 Comparison between groups of different treatments was carried out by densitometry as the percentage of β-actin.
Immunohistochemical analysis
The protective effect of tempol on CP-induced inflammation in kidney tissue was evaluated by immunohistochemical analysis of inflammatory markers. Tissues were fixed in neutral formalin and embedded in paraffin; 5 mm thick sections of tissue were cut onto poly-lysine-coated glass slides. Sections were deparaffinized three times (5 min each) in xylene followed by dehydration in graded ethanol and finally rehydrated in running tap water. For antigen retrieval, sections were boiled in 10 mM citrate buffer (pH 6.0) for 5–7 min. Sections were incubated with hydrogen peroxide for 15 min to minimize nonspecific staining and then rinsed three times (5 min each) with 1× PBST (0.05% Tween-20). Blocking solution was applied for 10 min and sections were incubated with diluted rabbit polyclonal antibodies namely anti-COX-2 (dilution 1:200) and anti-NF-κB (dilution 1:300) overnight at 4°C in a humid chamber. Further processing was done according to the instructions of Ultra Vision plus Detection System Anti-Polyvalent, HRP/DAB (Ready-To-Use) staining kit (Thermo Fischer Scientific, Waltham, Massachusetts, USA). The peroxidase complex was visualized with 3,3′-diaminobenzidine (DAB). Finally, the slides were counterstained with hematoxylin, dried and mounted with DPX Mountant using cover slips. Slides were observed under the light microscope (Leadz, Uxbridge, Greater London, UK). Analysis was carried out by Image J Software at 40× magnification. 22
Histopathological analysis
Kidney tissues were excised out and fixed in 10% neutral formalin and embedded in paraffin. Sections of 5 µm thickness were cut from the tissues. The paraffin-embedded tissues were deparaffinized using xylene and ethanol. Slides were washed using PBS and permeabilized with 0.1 M citrate + 0.1% Triton X-100 permeabilization solution. The deparaffinized sections were stained with hematoxylin and eosin and observed at 40× magnification using the light microscope. To avoid any type of bias slides were coded and examined by the histopathologist in a blinded manner.
Estimation of protein concentration
The protein concentration in samples was measured using bovine serum albumin (BSA) as standard following the method of Bradford. 23
Statistical analysis
Data were expressed as the means ± standard error and analyzed using analysis of variance followed by Newman–Keuls multiple comparison test. Values of p < 0.05 were considered as significant. All the statistical analyses were performed using Graph Pad Prism 5 software (Graph Pad Software Inc., San Diego, California, USA).
Results
Effect of tempol on CP-induced changes in kidney function markers
CP treatment (group II) caused a significant (p < 0.001) increase in both BUN and creatinine levels in serum of animals when compared with control (group I; Figure 2). Pretreatment of tempol (group III) in CP treated animals caused a significant (p < 0.001) reduction in both the parameters, BUN (Figure 2(a)) and creatinine (Figure 2(b)) when compared with that of CP treatment group. Tempol alone (group IV) treatment caused no significant change (Figure 2).
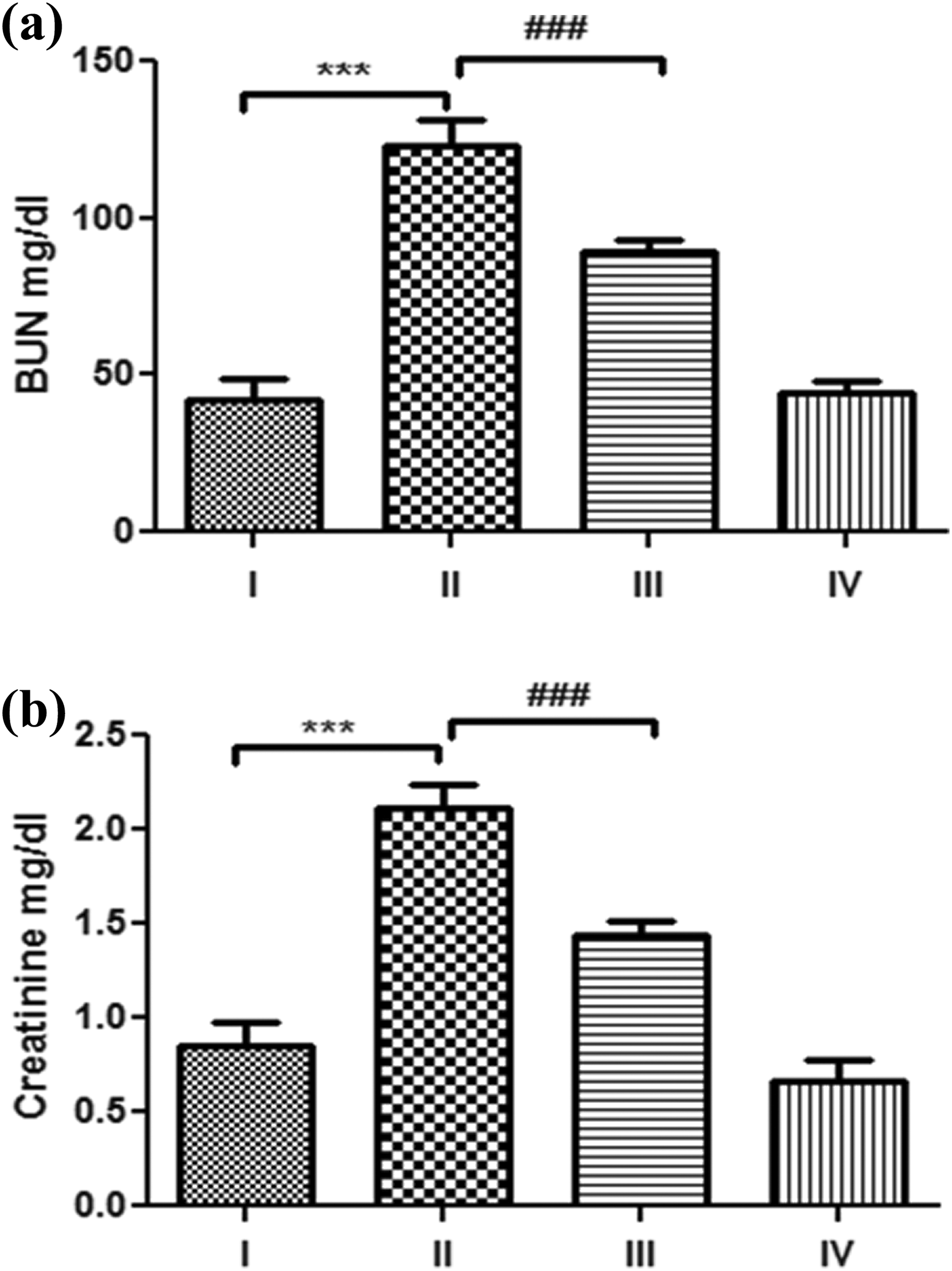
The effect of tempol and CP on the levels of BUN (a) and creatinine (Cr) (b). A single dose of CP caused a significant (***p < 0.001) increase in both BUN and creatinine when compared with controls (I). Tempol pretreatment in CP-treated animals (III) caused a significant (###p < 0.001) restoration in both BUN creatinine when compared with the CP-treated animals (II). Tempol alone caused no significant change. Data are expressed as means ± standard error of means (SEM) (n = 6). CP: cisplatin (cis-diamminedichloroplatinum [II]).
Effect of tempol on CP-induced histological changes
Histopathological findings of kidney (Figure 3) showed normal glomerular and tubular histology both in the cortical and medullary regions in control animals (a). CP caused glomerular and peritubular damage and vacuolation, swelling, and necrosis, which resulted in the invasion of inflammatory cells into the kidney cortical section in CP-treated animals (b). Pretreated with tempol (c) demonstrated a marked reduction in the histological abnormalities in renal tissues. Treatment of tempol alone (d) (100 mg/kg) showed no remarkable histological changes.

Effect of tempol on CP-induced renal tubular damage. Histological sections of the kidney at 72 h after treatment. (a) Control showing normal histoarchitecture. (b) Animals treated with CP (20 mg/kg) showed necrosis, inflammatory cell invasion vacuolation, and desquamation of epithelial cells in the renal tubules. (c) These changes were attenuated by the pre-treatment of tempol to a great extent with lesser necrosis in proximal tubules compared to CP alone treated animals. (d) Tempol alone treated animals showed no adverse tissue changes. Original magnification—40× (HE). CP: cisplatin (cis-diamminedichloroplatinum [II]).
Immunohistochemical response of NF-κB protein
Immunohistochemical analysis of NF-κB (Figure 4(a)) in the kidneys of control mice (group I) showed no remarkable expression. In contrast, strong NF-κB immunopositive expression was found in renal tubular cells of CP-treated animals, with numerous positive nuclei. Treatment of CP-intoxicated mice with tempol reduced NF-κB immunopositivity (group III) and mice treated with tempol (group IV) showed low NF-κB immunoreactivity. The histogram (Figure 4(b)) represents a significant increase in group II (CP) (p < 0.05) when compared to that of group I. Tempol pretreatment with CP-treated animals in group III caused a significant (p < 0.05) restoration in NF-κB expression. There were no significant changes in group IV tempol only treated animals.

Photomicrographs depicting immunohistochemical staining of NF-κB. Effect of tempol pretreatment on CP-induced NF-κB expression. Photomicrographs of kidney sections (a) depicting control group (I), CP-treated group (20 mg/kg b. wt.) (II), tempol (100 mg/kg b. wt.) + CP (III), and tempol alone (100 mg/kg b. wt). For immunohistochemical analyses, dark brown color indicates specific immunostaining of NF-κB and blue color indicates nuclear hematoxylin staining. The kidney section of CP treated group (II) has more NFκB immunopositive staining (arrows) as indicated by brown color as compared to control group (I) while pretreatment of tempol in groups III reduced NF-κB immunostaining as compared to group II. No significant difference was observed between groups IV and I. Original magnification: 40×. The histogram (B) represents a significant increase in group II (CP) (**p < 0.01) when compared to that of group I. Tempol pretreatment with CP-treated animals (III) caused a significant (#p < 0.05) restoration in NF-κB expression. There is no significant change in tempol only treated animals (IV). CP: cisplatin [cis-diamminedichloroplatinum [II]); NF-κB: nuclear factor-kappa B.
Immunohistochemical response of COX-2 protein
Immunohistochemical analysis of COX-2 (Figure 5(a)) in the kidneys of control mice (group I) showed no notable expression. In contrast, strong COX-2 immunopositive expression was found in renal tubular cells of CP-treated animals, with numerous positive nuclei. Treatment of CP-intoxicated mice with tempol reduced COX-2 immunopositivity (group III) and mice treated with tempol (group IV) showed low COX-2 immunoreactivity. The histogram (Figure 5(b)) represents a significant increase in group II (CP; p < 0.01) when compared to that of group I. Tempol pretreatment with CP-treated animals in group III caused a significant (p < 0.05) restoration in COX-2 expression. There was no significant change in group IV tempol only treated animals.

Photomicrographs depicting immunohistochemical staining of COX-2. Effect of tempol pretreatment on CP-induced COX-2 expression. Photomicrographs of kidney sections (a) depicting control group (I), CP-treated group (20 mg/kg b. wt.) (II), tempol (100 mg/kg b.wt.) + CP (20 mg/kg b.wt) (III), and tempol only (100 mg/kg b. wt.). For immunohistochemical analyses, dark brown color indicates specific immunostaining of COX-2 and blue color indicates nuclear hematoxylin staining. The kidney section of CP-treated group (II) has more COX-2 immunopositive staining (arrows) as indicated by brown color as compared to control group (I) while pretreatment of tempol in groups III reduced COX-2 immunostaining as compared to that of group II. No significant difference was observed between groups IV and I. Original magnification: 40×. The histogram (b) represents a significant increase in group II (CP) (**p < 0.01) when compare to group I. Tempol pretreatment in CP-treated animals (III) caused a significant (#p < 0.05) restoration in COX-2 expression. There is no significant change in tempol only treated animals (IV). CP: cisplatin (cis-diamminedichloroplatinum [II]); COX-2: cycloxygenase-2.
Inflammatory response of inflammatory markers via western blot
The protein concentration of inflammatory markers NF-κB, TNF-α, and COX-2 in western blot analysis of renal tissue is shown in Figure 6(a). The treatment of CP significantly elevated the expression of NF-κB (p < 0.01; Figure 6(b)), TNF-α (p < 0.01; Figure 6(c)), and COX-2 (p < 0.001; Figure 6(d)) in group II as compared to group I and treatment of tempol with CP restored the level of NF-κB, TNF-α, and COX-2 (p < 0.05) in group III. There were no significant changes in NF-κB, COX-2, and TNF-α expression in tempol only group (IV).

Effects of tempol on the expression of NF-κB, TNF-α, and COX-2 proteins in kidney tissues as studied in western blot analysis. In this figure, (a) showing the expressions of inflammatory proteins. The histograms (b), (c), and (d) represent integrity density of NF-κB, for TNF-α, and COX-2 protein, respectively. In the group (II), CP (20 mg/kg) treatment caused a significant increase in NF-κB (**p < 0.01), TNF-α (**p <0.01), and COX-2 (***p < 0.001) versus control animals. Pretreatment of tempol (100 mg/kg) in group III attenuated these changes in case of all the markers significantly (#p < 0.05) was compared with CP-treated animals. No significant effect was elicited on these markers in the animals treated with tempol only (IV) at the same dose level. Data are expressed as means ± SEM. CP: cisplatin (cis-diamminedichloroplatinum [II]); NF-κB: nuclear factor-kappa B; COX-2: cycloxygenase-2; TNF-α: tumor necrosis factor-α.
Discussion
We observed that tempol has distinct anti-inflammatory action against CP-induced inflammatory response in mouse kidney. It has been reported that tempol is a well-tolerated protective agent against oxidative stress and has no negative effect on cell growth. 24 Tempol is a low-molecular-weight piperidine nitroxide, which cumulates in the cytosol through membranes. 25 It has shown beneficial effects in cerulein-induced pancreatic injury, endotoxin-induced organ injury, and hemorrhagic shock in animal studies. 26 –28 Beneficial effects of tempol depend on scavenging hydroxyl radicals within the cell and also reduction in tissue injury due to inflammation. 10,24 It was revealed in western blotting and immunohistochemical studies that tempol protected animals from inflammatory onslaught of CP which significantly contributed to nephroprotection. Such protection was evident at tissue level also. Earlier, tempol has been reported for the protective effect against drug-induced nephrotoxicity. 4,14,29 Tempol is a membrane permeable SOD-mimetic agent, which makes it a suitable candidate of choice for anti-apoptotic and anti-inflammatory actions. 4,7,29,30
In our study, we also found that 20 mg/kg CP caused a significant change in renal markers. Tempol (100 mg/kg p.o.) significantly reduced the level of renal markers such as BUN and creatinine. Soni et al. also reported a similar action for tempol. 18 Furthermore, tempol also protected the kidney from oxidative damage by its antioxidant action. 4,30,31 Histological examination of kidney under the light microscope revealed the protective effect of tempol in kidney tissues. Besides protection against CP-induced nephrotoxicity reported here by us, tempol has demonstrated protective effect against methotrexate-induced liver injury in rats. 30
NF-κB plays a vital role by participating in the regulation of cell signaling under various physiological conditions and COX-2 has a key role in CP-induced acute kidney toxicity in mice. 17 Activation of NF-κB promotes inflammatory mediators via transcription of specific genes producing anti-apoptotic and inflammatory responses. 14 This response leads to increased expression of TNF-α in kidney cells, an important cytokine involved in systemic inflammation and CP-induced acute-phase response. We observed that animals pretreated with tempol and then exposed to CP showed a marked decrease in the expression of inflammatory markers such as NF-κB and COX-2. This observation supports the anti-inflammatory role of tempol. It has been reported that oral administration of tempol reduced the expression of NFκB and TNFα in the soleus muscle of ren-2 rats that are transgenic for the renin gene. 32 Moreover, a study by Li et al. showed that CP increases the expression of NF-κB and COX-2. 17 Tempol has been reported to inhibit the mRNA expression of COX-2 and to reduce the contusion spinal cord injury. 32 Abouzied et al. also observed that tempol has a potential therapeutic role in acute liver injury via restoration of NF-κB expression. 7 Recently, using male Wistar rats having the left kidney clipped (2K1C) Nunes et al. reported protective effect of tempol against chronic ischemic renal injury from stenosis. 33 They also reported that tempol prevented renal dysfunction via its antihypertensive and antioxidant properties. 33 Recently, we have reported on the protective effects of a natural product, S-allyl cysteine (SAC) against urotoxicity of another anticancer drug, cyclophosphamide. 34,35 Urotoxicity and nephrotoxicity are two most common side effects of chemotherapy and therefore, natural as well synthetic anticancer compounds hold a significant value in the effective management of cancer related maladies. Taken together the antioxidant action and anti-inflammatory role against CP-induced nephrotoxicity and high membrane permeability tempol may prove to be a promising protective agent (Figure 7).

CP-induced nephrotoxicity showed oxidative stress, increase inflammatory mediators, NF-κB, TNF-α, and COX-2. Tempol treatment restores inflammatory mediators and downregulate the expression of NF-κB, TNF-α, and COX-2. NF-κB: nuclear factor-kappa B; COX-2: cycloxygenase-2; TNF-α: tumor necrosis factor-α; CP: cisplatin (cis-diamminedichloroplatinum [II]).
In conclusion, findings of the present study reinforce the significant anti-inflammatory role of tempol in nephroprotection against CP-induced AKI and associated toxicopathological consequences. So far, antioxidant role of tempol has been highlighted. Our study offers a new insight into the mechanism of tempol afforded nephroprotection. Being a highly permeable compound from bioavailability viewpoint, its clinical use is further underscored.
Footnotes
Acknowledgement
Dr A.K. Tiwari, Veterinary Officer, Central Animal House Facility, Jamia Hamdard, New Delhi, India, facilitated interpretation of histopathological findings.
Author contributions
MAA and SR were responsible for the design and execution of the studies. SAHA contributed to western blotting analysis and its interpretation. SS, MF, SD, and JA contributed to the analysis of biochemical data. All authors contributed to the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Mohd Amir Afjal, Mahwash Fatima and Juheb Akhter received Research Fellowship from the University Grants Commission (UGC), Government of India under Basic Scientific Research (BSR) Scheme (Grant No. F. 25-1/2013-14(BSR)/7-91/2007(BSR).
