Abstract
Background
Diabetic retinopathy (DR) is a major cause of visual problems and ultimately results in vision loss. Nearly 25% of diabetic patients have suffered from DR.
Objectives
The present research has assess the salutary properties of monotropein against streptozotocin (STZ)-induced DR in rats.
Materials and Methods
The experimental rats were challenged with STZ, and after the initiation of diabetes, the animals were treated with monotropein for 60 days. An investigation was conducted to assess the glucose, food intake, and body weight levels of the experimental rats. The antioxidants, angiogenic protein levels, and inflammatory cytokines were assessed using assay kits. The apoptotic protein levels and lipid profiles were examined by kits. A morphometric analysis was performed on the retinal tissues.
Results
The treatment of monotropein successfully reduced the glucose level, food consumption, and glycosylated hemoglobin (HbA1c) while increasing the body weight of DR rats. The monotropein also enhanced antioxidant levels and significantly decreased inflammatory cytokines and angiogenic protein levels in the DR rats. The lipid profiles and proapoptotic protein levels were also decreased by the monotropein.
Conclusion
The present research shows that monotropein successfully reduced dyslipidemia, inflammatory responses, and oxidative stress in DR rats. These data demonstrate that monotropein has the potential to mitigate STZ-induced DR in rats.
Introduction
Diabetes mellitus refers to a collection of metabolic complications that typically include aberrant regulation of glucose levels in the body, resulting from either impaired insulin production, decreased insulin action, or both (Cole & Florez, 2020). Patients with diabetes often experience a range of problems as the condition progresses, including diabetic retinopathy (DR). DR is a condition affecting the small blood vessels in the retina, which can ultimately result in blindness (Wang & Lo, 2018). DR is the prevailing microvascular problem and prevalent ocular condition in the majority of individuals with diabetes and affects over 60% of patients with diabetes. Researchers have identified various causes of DR, including chronic diabetes, sustained high blood sugar levels, hypertension, and dyslipidemia (Altman & Schmidt, 2018). At the clinical level, DR contributes significantly to visual problems and vision loss in diabetes patients. Hyperglycemia primarily affects the retina, a highly metabolically active tissue in the body. Chronically elevated blood sugar levels cause changes to small blood vessels, blood flow, and nerve function, ultimately leading to DR. The causes of DR are complex and involve various factors, such as oxidative stress, inflammation, angiogenesis, ischemia, and neuronal apoptosis (Al-Kharashi, 2018).
Although there have been tremendous improvements in controlling blood sugar levels through diet and medication, effectively dealing with the consequences of diabetes, such as vision problems, continues to be a major concern in medical practice. Due to the limitations of conventional drug treatments for diabetic problems, there is an increasing interest in alternative and complementary therapies (Xu et al., 2018). DR is a disease that causes vascular oxidative stress and inflammation. In this condition, reactive oxygen species (ROS) and inflammatory markers are generated locally and play a key role in its progression as well as other harmful effects (Mendonca et al., 2020). Both hyperglycemia and inflammation play important roles in this mechanism, but their impact varies depending on the disease’s severity. Both hyperglycemia and inflammation have significant roles in triggering DR in patients with diabetes (Tomić et al., 2020). From this perspective, an elevated influx of systemic inflammatory cytokines can damage the diabetic retina. This damage occurs through various mechanisms, including the impairment of the blood–retinal barrier (BRB), higher vascular permeability, the amplification of ROS production, the activation of microglial cells and infiltration of macrophages, and the promotion of pericyte and neural cell apoptosis (Ucgun et al., 2020).
From a clinical perspective, DR is categorized into two stages: Proliferative and nonproliferative DR. In nonproliferative types, there are intraretinal microvascular aberrations. These abnormalities include high permeability of retinal blood vessels, swelling of the central part of the retina (macular edema), thickening of the membrane surrounding the small blood vessels, and capillary occlusion (Romero-Aroca et al., 2016). The detection of irregular angiogenic mechanisms in the retina and the development of retinal fibrosis can indicate the proliferative stage. The modifications resulted in significant visual impairments when the newly formed anomalous blood vessels infiltrated the vitreous cavity, resulting in vitreous hemorrhage. Early intervention can help prevent visual deficits in the initial stages of the disease, but managing them becomes increasingly challenging as the disease progresses (Vujosevic et al., 2020).
Inhibiting the advancement of DR typically involves strict regulation of glucose levels. Oral hypoglycemic medications and insulin continue to be the primary pharmaceuticals used in diabetes treatment. Conversely, a number of natural remedies have shown encouraging outcomes in managing diabetes while causing few negative effects (Tahrani et al., 2010). Monotropein, a natural bioactive compound, is an iridoid glycoside found extensively in the Morinda officinalis plant. Several previous studies have highlighted that monotropein has renoprotective (Zhang et al., 2020), hepatoprotective (Chen et al., 2020), anti-inflammatory and antioxidant (Jiang et al., 2020), antinociceptive (Choi et al., 2005), antiarthritic (Wang et al., 2014), and alleviating muscle atrophy (Wang et al., 2022) properties. However, no scientific study has yet explored its beneficial effects on DR. Therefore, the current study aims to investigate the therapeutic effects of monotropein on streptozotocin (STZ)-induced DR in rats.
Materials and Methods
Experimental Rats
The present work was conducted on male Wistar rats weighing approximately 230 ± 10 g. The rats were housed in a controlled laboratory environment, and careful attention was paid to their well-being. The circumstances were maintained at 22–26°C temperature, air moisture levels between 40 and 70%, and a 12-h dark–light sequence. During the trial, animals were given purified water and regular rat food. Before commencing the studies, a one-week acclimation time was provided for all rats to adapt to the laboratory environment.
Treatment Methods
The experimental rats were divided into four groups, with six animals in each. Group I is a control group and was not subjected to any treatments. The control rats were administered a 0.1 M saline concentration without STZ or monotropein. Group II rats were given STZ (60 mg/kg) to induce DR. After administering STZ for 48 h, the blood glucose of the rats was analyzed. Rats with 250 mg/dL or higher glucose levels were classified as diabetic. Group III consisted of diabetic animals that were treated with 30 mg/kg of monotropein orally for 9 weeks. Group IV consisted of diabetic animals treated with metformin (350 mg/kg), serving as the standard treatment for 60 days.
Analysis of Body Weight, Glycosylated Hemoglobin (HbA1c), Blood Glucose, and Food Consumption Levels
The average food consumption of the rats was meticulously assessed and recorded. The rats’ body weight was measured using an electronic balance, and the recorded results were scrutinized. The level of blood was assessed using a glucometer. The level of HbA1c was analyzed using a kit following the instructions provided by the manufacturer (Abcam, USA).
Quantification of Lipid Profiles
The high-density lipoprotein (HDL), low-density lipoprotein (LDL), triglycerides, total cholesterol, and atherogenic index were examined in the rats using kits. The specifications provided by the manufacturer (Elabscience, USA) were followed during the assays. Every assay was done three times, and the outcomes are presented in mg/dL.
Analysis of Antioxidant Levels
The catalase (CAT) and superoxide dismutase (SOD) activities in the retinal tissues were examined with the help of kits following the directions provided by the manufacturer (Elabscience, USA). The concentrations of glutathione (GSH) and the ratio of GSSG/GSH in the retina of experimental rats were examined using kits, following the directions provided by the manufacturer (Abcam, USA).
Analysis of Proinflammatory Cytokines
The levels of nuclear factor (NF)-κB, tumor necrosis factor (TNF)-α, interleukin (IL)-6, and IL-1β in the serum were measured using specialized kits provided by the manufacturer (Elabscience, USA), following the recommended protocols. Each assay was done in triplicate, and the findings are given in pg/mL.
Analysis of MMP-9, MCP-1, and VEGF Levels
The MMP-9, MCP-1, and VEGF were examined in experimental rats using the specific kits. The instructions provided by the manufacturer (Abcam, USA) were followed during the analyses. The tests were done in triplicate, and the findings are given in ng/L.
Morphometrical Analysis
The retinas obtained from the rats were evaluated morphometrically using a computer-based image analysis method. Using the morphometric analysis feature of ImageJ software, the thickness of the retina and the total cell numbers inside the ganglion cell layer (GCL) were investigated.
Assessment of Apoptotic Protein Levels
The Bcl-2, Bax, and the Bax/Bcl-2 ratio in the retina of the rats were analyzed using commercially available assay kits. The analysis was conducted following the manufacturer’s protocols (Elabscience, USA).
Statistical Analysis
The values are studied using SPSS software. The results are given as the mean ± standard deviation (SD) of three repeated measurements and analyzed using one-way analysis of variance (ANOVA) and Duncan’s post hoc method. The significance was fixed based on a “p” value of less than 0.05.
Results
Effect of Monotropein on the Diabetes-induced Changes in Experimental Rats
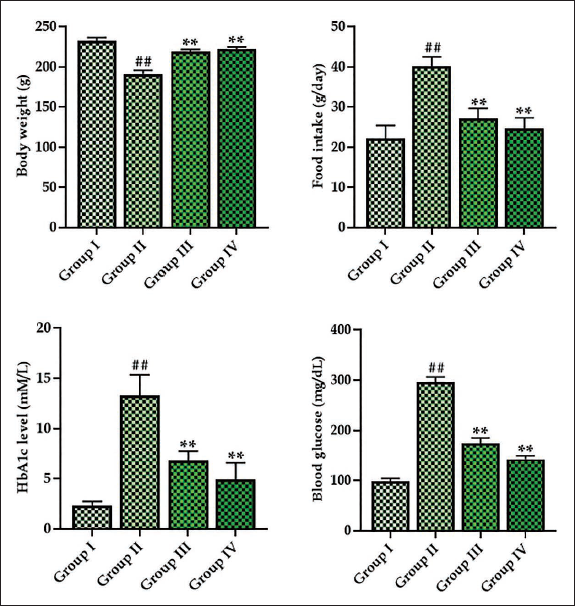
Figure 1 illustrates the regulatory effects of monotropein on diabetes-induced physiological abnormalities, including food intake, body weight, glucose, and HbA1c levels in diabetic rats. Food consumption, glucose, and HbA1c levels were found to be increased in the DR rats. Rats with DR also exhibited a decrease in body weight compared to the control. Captivatingly, the 30 mg/kg of monotropein significantly reduced the food uptake, glucose, and HbA1c levels. The monotropein treatment resulted in an improvement in the body weight of the rats with DR. These findings are consistent with the outcomes of the metformin treatment.
Effect of Monotropein on the Lipoprotein Levels in Experimental Rats
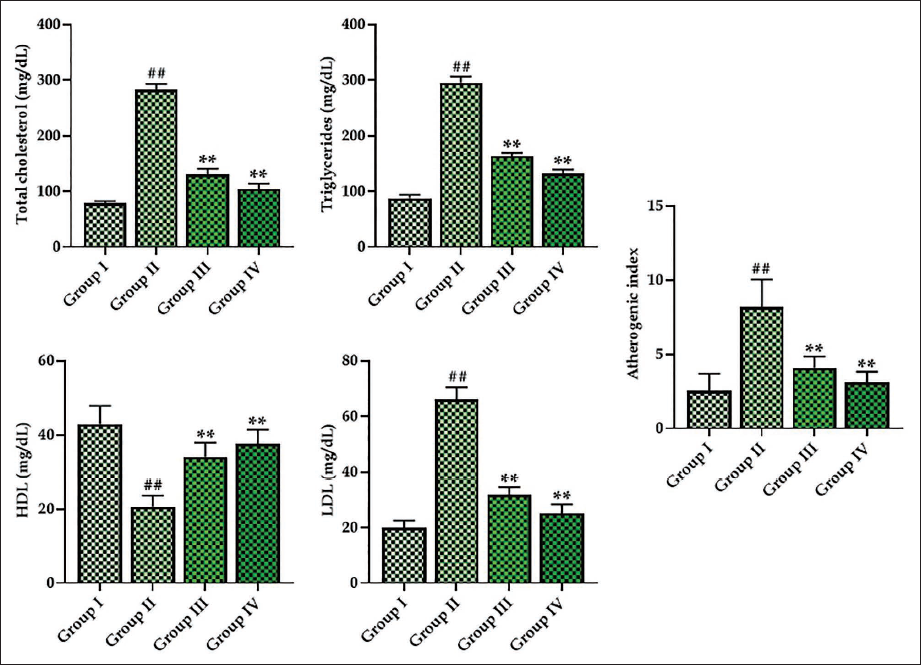
Figure 2 displays the lipoproteins, including triglycerides, LDL, HDL, cholesterol, and atherogenic index levels, in the DR rats. The serum of the STZ-induced rats exhibited reduced levels of HDL, whereas it increased LDL, triglycerides, cholesterol, and atherogenic index compared to the control. Remarkably, the monotropein treatment considerably enhanced the level of HDL. The 30 mg/kg of monotropein-treated rats also exhibited reduced levels of cholesterol, LDL, triglycerides, and atherogenic index in their serum. Metformin treatment also regulated these lipoprotein levels.
Effect of Monotropein on the Antioxidant Levels in Experimental Rats
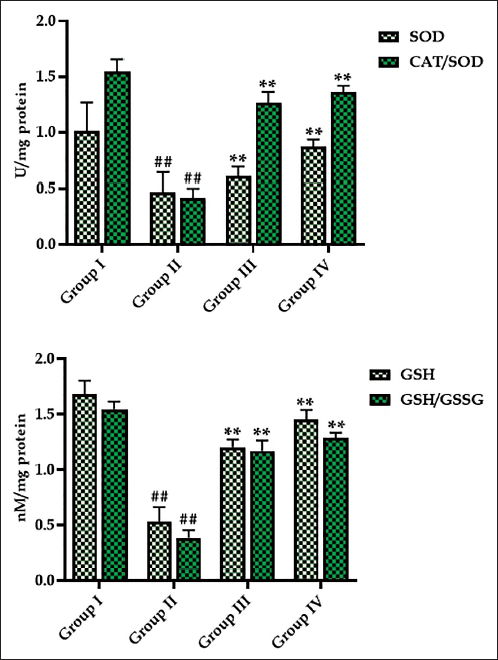
Figure 3 illustrates the monotropein treatment’s effect on the antioxidants in the DR rats. The STZ-induced DR rats exhibited decreased SOD and GSH levels, as well as reduced relative ratios of SOD/CAT and GSH/GSSG, compared to the control. Fascinatingly, the 30 mg/kg of monotropein successfully elevated the SOD and GSH levels, as well as the relative ratios of SOD/CAT and GSSG/GSH in their retina. This provides evidence for the antioxidant effects of monotropein. The antioxidant effects of monotropein were comparable to the metformin treatment.
Effect of Monotropein on the Inflammatory Markers in Experimental Rats
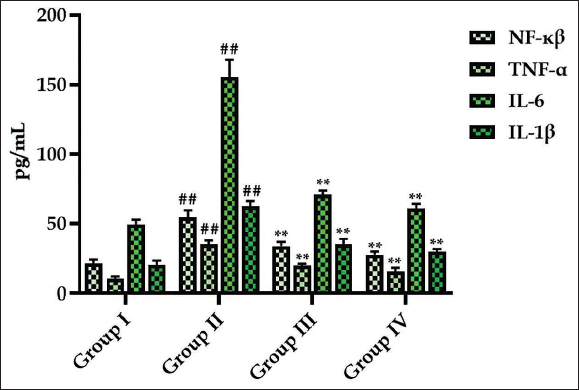
The NF-κB, IL-6, IL-1β, and TNF-α levels in the serum of experimental rats are presented in Figure 4. Elevated inflammatory cytokine levels were noted in the serum of DR rats when compared to control. Contrastingly, treatment with 30 mg/kg of monotropein significantly decreased these cytokine levels in the serum of DR rats (Figure 4). Treatment of DR rats with metformin also resulted in a substantial reduction in the inflammatory cytokine levels in their serum.
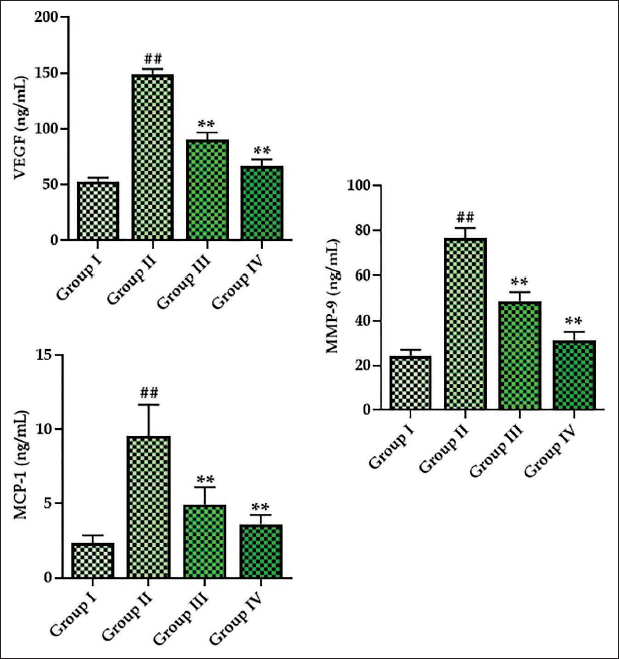
Effect of Monotropein on the Angiogenic Proteins in Experimental Rats
The levels of angiogenic markers, including MCP-1, MMP-9, and VEGF, in the experimental rats are given in Figure 5. The present results demonstrate that MCP-1, VEGF, and MMP-9 levels were elevated in the serum of DR rats. Nevertheless, the treatment with monotropein revealed a drastic reduction in the VEGF, MCP1, and MMP-9 levels, as depicted in Figure 5. The treatment with metformin also significantly reduced these angiogenic marker levels in the DR rats.
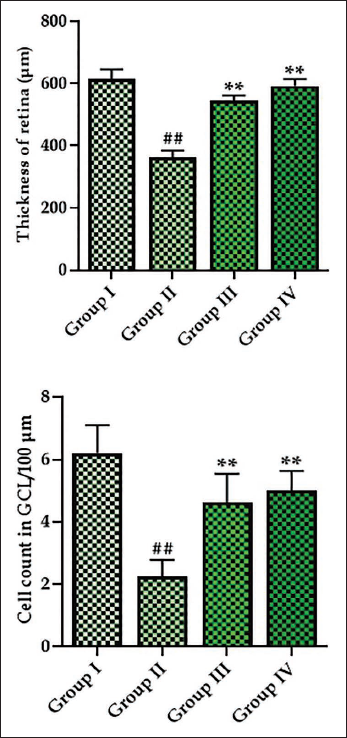
Effect of Monotropein on the Thickness of Retina and Number of Cells in Ciliary Ganglion Layer (CGL) of Experimental Rats
Figure 6 exhibits the results of a morphometric examination performed on the retina of the experimental rats. The results revealed that the thickness of the retina and the cell counts in the CGL were reduced in the DR rats. Remarkably, the treatment with monotropein successfully regulated these changes in the DR rats. The thickness of the retina of monotropein-treated rats showed a considerable increase in both cell counts and thickness in the CGL. Metformin treatment also mitigated these changes in the DR rats.
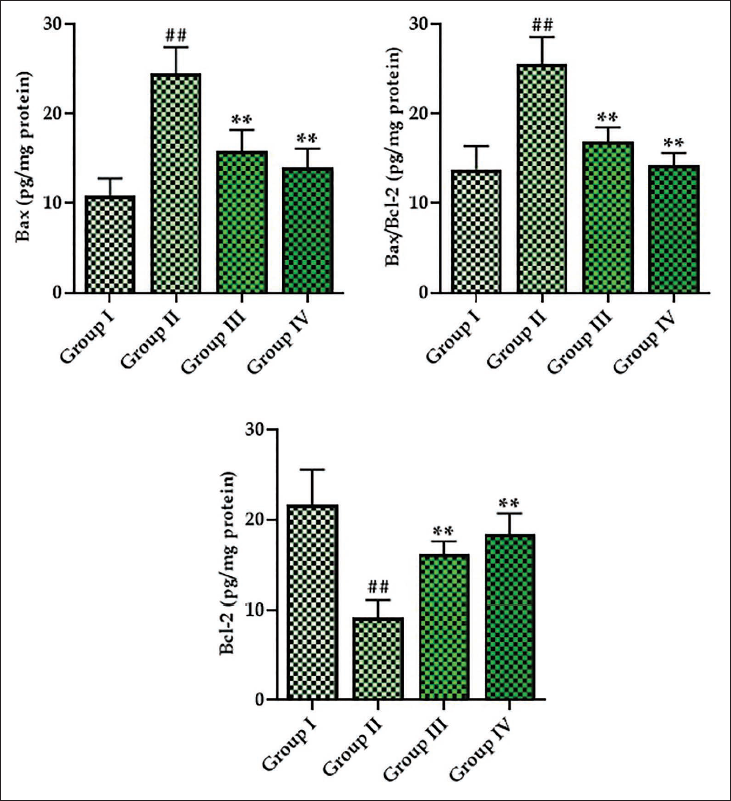
Effect of Monotropein on the Apoptotic Protein Levels in the Experimental Rats
Figure 7 illustrates the apoptotic protein levels, including Bcl-2, Bax, and Bcl-2/Bax ratio, in the retina of experimental rats. The retina of DR rats showed elevated Bax protein and the Bcl-2/Bax ratio, whereas the Bcl-2 levels decreased in their retina. Surprisingly, the monotropein treatment significantly reduced these protein levels. The monotropein treatment at 30 mg/kg concentration in the DR rats resulted in a significant decrease in the Bax and Bcl-2/Bax levels and increased the Bcl-2 protein levels in their retina.
Discussion
DR is a major cause of visual impairment and vision loss, and the number of incidents is rapidly increasing worldwide. Nearly 25% of diabetic patients have been affected by DR. A previous study reported that the world prevalence of DR is nearly 22% (Saeedi et al., 2019). Clinically, DR is characterized by changes in the pathophysiology of the retinal small blood vessels, resulting in significant visual impairment and permanent blindness. The modifications mentioned encompass capillary blockage, hemorrhages, microaneurysms, and neovascularization (Duh et al., 2017). Currently, the specific pathophysiological mechanisms that cause DR are still not well understood. Inflammation and oxidative stress play a significant role in DR progression. Research has shown that prolonged increases in blood glucose levels stimulate ROS accumulation and proinflammatory mediators in the retina (Dubey et al., 2023). DR is worsened by the presence of macular edema, retinal detachment, and neovascular glaucoma, leading to significant visual problems (Yao et al., 2018).
STZ is the predominant drug used to generate diabetes in rodents. STZ selectively targets the pancreatic cells responsible for insulin secretion, leading to a decline in the number of functioning pancreatic cells and the development of diabetes (Zhu, 2022). It is known to cause identical clinical symptoms as those observed in diabetic patients. STZ induces oxidative damage to the pancreas, resulting in hyperinsulinemia and persistent hyperglycemia. In addition, the administration of STZ is linked to adverse effects on the liver and kidneys. Furthermore, STZ is linked to characteristic manifestations of diabetes, such as a reduction in body weight, excessive hunger, increased urine production, and excessive thirst. The presence of hypoinsulinemia and hyperleptinemia primarily causes the rise in food consumption in individuals with diabetes. Nevertheless, the primary cause of weight loss in diabetes patients is the heightened depletion of fat reserves and the dependence on fatty acids as a source of energy (Nørgaard et al., 2020). This study shows that STZ causes diabetes by elevating blood glucose levels. Elevated HbA1c levels in diabetic rats result from persistent hyperglycemia, leading to hemoglobin glycation. The concentration of HbA1c is positively correlated with DR. It is considered a valuable diagnostic and predictive tool for assessing diabetes-related complications (Wium-Andersen et al., 2021). Our study showed that the treatment with monotropein significantly reduced food consumption, HbA1c, and glucose levels in DR rats. The monotropein treatment also elevated the body weight of the STZ-induced rats. These findings highlight its potential for mitigating diabetes-related complications.
Oxidative stress refers to a disproportion between antioxidant mechanisms and ROS accumulation. It is widely recognized as the primary mechanism responsible for the harmful effects and diseases associated with DR (Alfonso-Muñoz et al., 2021). Oxidative stress in the retina leads to an elevation in the thickness of the basement membrane and triggers damage to the BRB, hypoxia, inflammation, apoptosis, angiogenesis, and neovascularization. Nevertheless, the use of antioxidant therapy effectively prevents retinal damage by reducing the intensity of these mechanisms (Cecilia et al., 2019). The antioxidant system in most cells consists of nonenzymatic components, such as GSH, as well as antioxidant enzymes, including SOD, CAT, GPx, GRx, and others (Hennig et al., 2018). Hyperglycemia is commonly recognized to cause changes in multiple metabolic systems that produce ROS. DR is extensively known as one of the most common consequences of diabetes. Retinal cells are susceptible to oxidative assault due to various polyunsaturated fatty acids. Elevated levels of glucose oxidation in the retina also play a significant role in DR development (Darenskaya et al., 2021). In this work, the results exhibited a drastic reduction in the SOD, GSH, CAT/SOD proportion, and GSH/GSSG ratios in the retina of the DR rats. Captivatingly, the treatment with monotropein remarkably boosted the antioxidants in the DR rats. These outcomes highlight the antioxidant potential of monotropein.
Inflammation has a critical role in DR development. The inflammatory cytokines, including IL-1β and TNF-α, and inflammatory transcription factors, including NF-κB, have been observed to be increased in the retinal tissues of diabetic patients (Gasparini & Feldmann, 2012; Singh & Rai, 2019). NF-κB, a heterodimer consisting of a p65 subunit, regulates the transcription of various genes that have a role in immunity and inflammation. Inhibiting NF-κB activation decreases the expression of inflammatory markers, including IL-6. Elevated levels of IL-6 are linked to the disruption of the BRB and alterations in retinal blood vessels in a hyperglycemic condition (Robinson et al., 2020). IL-1β, a very influential inflammatory cytokine in DR, stimulates the production of chemokines and the recruitment of macrophages, therefore promoting the degeneration observed in DR. TNF-α is crucial in both the initial and final phases of BRB deterioration in DR because it promotes cell death through mitophagy (Liu et al., 2021). DR patients have elevated inflammatory cytokine levels in their ocular fluids compared to diabetic patients without DR. Furthermore, there is a direct link between the escalation of inflammatory markers and the advancement of DR (Semeraro et al., 2015). The present findings showed that STZ-induced rats revealed drastic elevations in their serum of the IL-6, NF-κB, IL-1β, and TNF-α levels. Fascinatingly, the treatment of monotropein successfully reduced these cytokines in the STZ-induced rats, which highlights its anti-inflammatory properties.
In progressive DR, the mechanisms of inflammation and angiogenesis are closely linked. In physiological conditions, the process of angiogenesis maintains normal parameters. However, in pathological conditions like DR, the expression of angiogenic markers like VEGF increases (Saleh et al., 2019). Elevated inflammatory cytokine levels stimulate the generation of VEGF, and subsequently, VEGF triggers the production of inflammatory cytokines. VEGF is widely acknowledged as the primary marker for angiogenesis in DR, and its expression escalates as the disease progresses in severity. VEGF regulates proangiogenic factors that disrupt the BRB through several methods, resulting in heightened vascular permeability and retinal edema (Rossino et al., 2020). Given the importance of VEGF and inflammation in the development of DR, there is a focus on using drugs that have anti-VEGF and anti-inflammatory actions to alleviate problems in diabetic patients (Zhao & Singh, 2018). The current medical treatment for DR involves the use of monoclonal antibodies to inhibit VEGF binding to its receptors, thereby reducing VEGF signaling. The efficiency of anti-VEGF medication in delaying the progression of DR has been demonstrated (Bonnin et al., 2019). In this study, the results exhibited that STZ-induced rats demonstrated increased VEGF levels. However, the monotropein treatment considerably decreased the VEGF levels in the STZ-induced rats.
Research has indicated that MMP-9 is also significantly involved in neovascularization. Furthermore, MCP-1 is crucial in advancing many diabetes-related problems (Forsyth et al., 1999; Robison et al., 1997). MCP-1, belonging to the family of inflammatory chemokines, primarily functions through paracrine and autocrine mechanisms. MCP-1 has the ability to stimulate the movement and penetration of inflammatory cells. It also plays a pivotal role in phagocytic cell activation and inflammation (Wakabayashi et al., 2011). Research has proved that the MCP-1 expression is elevated in the retinal tissues of DR patients. Furthermore, the TNF-α expressions are directly associated with MCP-1 activation (Dong et al., 2012). The present study also highlighted that the monotropein treatment successfully reduced the MMP-9 and MCP-1 levels in the DR rats. These findings prove that monotropein effectively mitigated the angiogenic processes in the DR rats.
Cell apoptosis, which can result in organ damage, can be triggered by oxidative stress and inflammatory cytokines. The caspase family of enzymes is well-recognized as the primary initiator of cell death (McIlwain et al., 2015). Cell apoptosis often encompasses both extrinsic and intrinsic mechanisms of cell death. The inflammatory cytokine TNF-α activates death receptors, triggering extrinsic and intrinsic cell death pathways (Elmore, 2007). In contrast, intracellular signaling can trigger the cell apoptosis pathway by increasing p53/Bax expression, decreasing Bcl-2 expression, and activating Bax. This ultimately results in mitochondrial injury and cytochrome-C release (Khalfaoui et al., 2010). The present work, which found increased Bax and Bcl-2/Bax ratios and diminished Bcl-2 protein levels in the retina of the DR rats, proves the occurrence of apoptotic incidences. Fascinatingly, the monotropein treatment successfully reduced the Bax and Bcl-2/Bax ratios as well as elevated the Bcl-2 protein levels in the retina of the DR rats. These findings highlighted that the monotropein treatment has antiapoptotic effects on the retina of DR rats.
Conclusion
In summary, the present research shows that monotropein successfully reduced dyslipidemia, inflammatory response, and oxidative stress in rats with DR. The treatment of monotropein significantly regulated the levels of angiogenic markers, reduced the inflammatory regulators, and increased the antioxidants in rats with DR. These data demonstrate that monotropein has the potential to mitigate DR induced by STZ in rats. Further investigations within this context could enhance our understanding of the underlying mechanisms responsible for the salutary properties of monotropein in combating DR. This, in turn, may facilitate the development of new therapy options in the future.
Footnotes
Abbreviations
BRB: Blood–retinal barrier; DR: Diabetic retinopathy; HbA1c: Glycosylated hemoglobin; HDL: High-density lipoprotein; LDL: Low-density lipoprotein; ROS: Reactive oxygen species; STZ: Streptozotocin.
Acknowledgments
This work was supported by the Department of Ophthalmology, Hangzhou Huaxia Eye Hospital, Hangzhou 310030, China.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This work was approved by the institutional ethical committee Hangzhou Huaxia Eye Hospital, Hangzhou 310030, China (Ethical number: 2023/53).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
