Abstract
To explore the potential function of tricin in diabetic retinopathy (DR) and investigate whether Sestrin2 is closely involved in DR. A single intraperitoneal injection of streptozotocin-induced diabetes model in Sprague-Dawley rats and a high glucose-induced retinal epithelial cell model in ARPE-19 cells were established. The retinas were removed and examined by hematoxylin-eosin (HE) staining and dihydroethidium (DHE) staining. The proliferation ability and reactive oxygen species (ROS) level of ARPE-19 cells were detected by 5-ethynyl-2′-deoxyuridine (EdU) and flow cytometry. Then, the content of superoxide dismutase (SOD), malonaldehyde (MDA), and glutathione peroxidase (GSH-Px) in serum or cell supernatant was tested using enzyme linked immunosorbent assay (ELISA). In addition, the expression of Sestrin2, nuclear factor erythroid-2-related factor 2 (Nrf2), heme oxygenase-1 (HO-1), platelet endothelial cell adhesion molecule-1 (CD31), and vascular endothelial growth factor receptor 2 (VEGFR2) in retina tissue or ARPE-19 cells were validated through western blot and immunofluorescence assays. With the increase of MDA and ROS concentration, Sestrin2 expression was downregulated significantly, and Nrf2 and HO-1 expression was also reduced in retina tissue or ARPE-19 cells of model group, whereas CD31 and VEGFR2 expression was upregulated. However, tricin ameliorated the oxidative stress and angiogenesis and rectified the abnormal expression of Sestrin2/Nrf2 in diabetic retinopathy. Further mechanistic studies showed that silence Sestrin2 reduced the protective effect of tricin on ARPE-19 cells, as well as abolished its regulating effect on the Nrf2 pathway. These results suggested that tricin inhibits oxidative stress and angiogenesis in retinal epithelial cells of DR rats via reinforcing Sestrin2/Nrf2 signaling.
Introduction
Diabetic retinopathy (DR) has a very high incidence in the majority of patients who have had diabetes for 10 or more years. 1 DR causes irreversible damage to retinal function, and once vision is reduced or lost, it is unlikely to be restored to pre-onset vision levels with treatment. 2 To make matters worse, patients with advanced DR without timely treatment will inevitably lose their normal working and living abilities due to total blindness in both eyes. 2 Although strict control of glycaemia, hypertension and hyperlipidemia is emphasized in the clinic, DR still affects more than 145 million people globally. 3 Thus, it is important to find new targets and treatments for DR.
Sestrin2, a member of the Sestrin protein family, is a highly conserved stress-inducible metabolic protein. Sestrin2 can inhibit reactive oxygen species (ROS) and provide cellular protection against a variety of harmful stimuli, including genotoxic and oxidative stress, endoplasmic reticulum stress, and hypoxia. 4 Sestrin2 plays a key role in various cell signal transduction processes, and its dysregulation is associated with various diseases. 5 Pasha M et al. have found that the up-regulation of Sestrin2 under oxidative stress enhances the autophagy-directed degradation of Keap1, which targets and degrades nuclear factor erythroid-2-related factor 2 (Nrf2). 6 Nrf2 is a pleiotropic protein that regulates the cellular defense against toxic and oxidative insults. Activated Nrf2 can mediate the transcription of target genes such as heme oxygenase 1 (HO-1). 7 Collectively, as a cysteine sulfinyl reductase that plays an important role in regulating antioxidant effects, Sestrin2 is a possible cytoprotective target against oxidative stress-induced cell damage.
Tricin, a flavone isolated from rice bran, has been shown to have a variety of biological effects, including anti-inflammatory, anti-atherogenic, and anti-cancer.8,9 Although tricin is abundant in wheat hull and rice bran, it is rarely consumed in the daily diet. Study has found that tricin not only downregulates vascular endothelial growth factor receptor 2 (VEGFR2) signaling by reducing ROS production in endothelial cells, but also downregulates vascular endothelial growth factor (VEGF) expression by inhibiting hypoxia-inducible factor-1α (HIF-1α) accumulation in tumor cells. 10 Moreover, Tricin increased the protein expression levels of Nrf2 and HO-1, and promoted the nuclear translocation of Nrf2 in RAW264.7 cells induced by LPS. 11 However, the effect of tricin on DR has not been clearly studied. Therefore, this study investigated the role of tricin in DR through cell and animal experiments and verified whether its mechanism was related to sestrin2.
Materials and methods
Experimental animals
7-week-old male Sprague-Dawley (SD) rats (Vital River Laboratory Animal Technology Co., Ltd, China) were housed in a breeding room for 1 week to eliminate stress and remove unhealthy rats. 12 During this period, the rats increased in weight to 260–280 g. The experiments were performed in accordance with the recommendations of the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health (NIH Pub. No. 85-23, revised 1996). This study protocol was reviewed and approved by Animal Care and Use Committee of YanTaiShan Hospital, approval number 2022049.
Model and grouping
A diabetic rat model was established by a single intraperitoneal injection of STZ (Sigma-Aldrich; 60 mg/kg body weight) dissolved in citrate buffer solution (0.1 M, pH 4.5). An equal volume of citrate buffer was administered to control rats. Blood glucose levels were monitored with a glucometer once a week after STZ administration. Rats with random blood glucose levels higher than 16.7 mM were defined as having diabetes.
The tricin treatment was administered at 16 weeks after STZ injection. 13 40 rats were divided into control, diabetes, tricin-low (TRI-L, 75 mg/kg), tricin-medium (TRI-M, 100 mg/kg), and tricin-high (TRI-H, 150 mg/kg) groups, eight rats per group. Tricin was purchased from ChemFaces (Wuhan, China) and was >99% pure. Rats of the tricin groups were received different dosages of tricin by oral administration once daily for 4 weeks.14,15 Normal saline was administered to the control and diabetic groups.
Histological analysis
After 4 weeks, the eyeballs of diabetic or normal rats at 28 weeks of age were aseptically removed under anesthesia with zolazepam and hydroxyzine. After isolated, the retinas were fixed in 4% paraformaldehyde, embedded in paraffin and sectioned into 5 μm thick. Subsequently, sections were stained with hematoxylin-eosin (HE) and taken photos under light microscope (Olympus, Japan).
Dihydroethidium (DHE) staining
The retinas were incubated with DHE (1 μM in PBS) for 30 min at 37°C. Digital images were taken under fluorescence microscope (Olympus, Japan) and quantified by Image J 5.0 (NIH, USA) software.
Cell culture and treatments
ARPE-19 cells (ATCC, USA) were cultured in DMEM/F12 (1:1) growth medium (Life Technologies, UK) containing 2 mM L-glutamine (Life Technologies, USA), 10% fetal bovine serum (FBS, Solarbio, China), and penicillin (100 IU/ml)-streptomycin (100 μg/mL) (Solarbio, China) at 5% CO2 and 37°C incubator. HUVECs cells (ATCC, USA) were cultured in 1640 medium containing 10% serum with penicillin (100 IU/ml)-streptomycin (100 μg/mL) in incubator (5% CO2, 37°C).
Once the ARPE-19 cells reached to about 75% confluency, complete medium were replaced with FBS-free DMEM/F-12 for 24 h, and switched to high glucose (HG, 60 mM D-glucose) or normal glucose (NG, 5.5 mM d-glucose) conditions. The medium was changed every 2–3 days. To investigate the effects of tricin on cell proliferation and levels of Sestrin2 and Nrf2, high glucose-induced ARPE-19 cells were incubated with different concentrations of tricin (5 μM, 10 μM, and 15 μM). 16 For further mechanistic studies, high glucose-induced ARPE-19 cells were transfected with 50 nM Sestrin2 siRNA (RiboBio, China) or scrambled siRNA sequence through LipofectamineTM 3000 transfection reagent (Thermo Fisher Scientific, USA). 17 At 48 h after transfection, the cells were treated with 15 μM tricin for 1 h.
5-ethynyl-2′-deoxyuridine (EdU) assay
The proliferation capacity of ARPE-19 cells was assessed using the EdU kit (RiboBio, China) according to the instructions provided by the supplier. Images were captured by fluorescence microscope (Olympus, Japan) and quantified by Image J 5.0 (NIH, USA) software.
Intracellular ROS level determination
ARPE-19 cells were collected and resuspended in serum-free medium containing 10 μM DCFH-DA. After incubation at 37°C for 30 min, the medium was discarded, and the cells were washed with PBS. The luciferase intensity was measured with a flow cytometer (BD Biosciences, USA).
Enzyme-linked immunosorbent assay (ELISA)
The serum of rats or supernatant of ARPE-19 cells were homogenized in RIPA Lysis buffer (Solarbio Science& Technology Co.) and centrifugated at 4°C. The level of oxidative stress cytokines including superoxide dismutase (SOD), malonaldehyde (MDA), and glutathione peroxidase (GSH-Px) in serum or cell supernatant were detected by enzyme-linked immunosorbent assay (ELISA). All ELISA assays were conducted according to manufacturer’s instructions (Nanjing Jiancheng Co., China).
Tube formation assay
HUVECs (lower chamber) and ARPE-19 cells (upper chamber) were co-cultured in shuttle chamber and HUVECs were cured with 50 μL Matrigel (BD Bioscience, USA). After digested and centrifuged, HUVECs were inoculated on Matrigel and incubated at 37°C for 6 h. An inverted microscope (Olympus, NY) was used to observe the tube formation.
Immunofluorescence
The retinas or cells were fixed in 4% paraformaldehyde for 10 min. After washing, the retinas or cells were treated with 0.1%TritonX-100 for 30 min and blocked with bovine serum albumin for 30 min. Subsequently, the retinas were incubated with Sestrin2 and nuclear factor erythroid-2-related factor 2 (Nrf2) or platelet endothelial cell adhesion molecule-1 (CD31) at 4°C overnight. After incubated with Alexa Fluor 488-conjugated secondary antibody (Invitrogen, USA) for 1 h, fluorescence was observed under a fluorescence microscopy and the nucleuses were indicated by DAPI staining.
Western blot
The harvested retinal or cells homogenates were resolved with 12% SDS-PAGE electrophoresis and transferred to PVDF blocked with 5% skimmed milk. Then, the membranes were immunodetected with primary antibodies against Sestrin2 (1:1000, Abcam, USA), Nrf2 (1:500, Abcam, USA), heme oxygenase-1 (HO-1, 1:250, Dallas, USA), vascular endothelial growth factor receptor 2 (VEGFR2, 1:1000, Dallas, USA), and β-actin (1:2000, Abcam, USA). After incubated with secondary antibody Goat anti-Rabbit IgG (1:5000, Thermo Scientific, USA), protein bands were analyzed using Image Lab Software (Bio-Rad, USA).
Statistical analyses
All statistical analysis was conduct in GraphPad Prism version 6.0 software (San Diego, USA) and p-value <0.05 was considered statistically significant. Quantitative data were presented as means ± standard deviations (SD). Statistical differences were determined by one-way analysis of variance (ANOVA), followed by the least significant difference (LSD) test, Student’s t test was performed to analyze the data of the two groups.
Results
Tricin attenuates retinal morphological changes and inhibits oxidative stress induced by diabetes in rats
Figure 1(a) shows that in the normal rats at 28 weeks of age, the retinal structure is clear and complete, and the tissue cells in each layer of the retina are ordered and densely arranged. However, in the diabetic rats at 28 weeks of age, the number of structural layers was unclear, and the cells were arranged in disorder and loose. In addition, compared with the diabetes group, tricin had obvious improvement effect on the above pathological changes. Figures 1(b),(c) shows that the retinas from the diabetic rats at 28 weeks of age exhibited a significant increase in the intensity of DHE staining and MDA content in serum (p < 0.05). Meanwhile, the levels of SOD and GSH-Px of diabetic rats were significantly decreased compared with the normal control rats at 28 weeks of age (p < 0.05, respectively). However, with the increased levels of SOD and GSH-Px, the DHE intensity and MDA content of the tricin groups were significantly decreased compared with the diabetes group (p < 0.05), and the TRI-H group had the best effect. All the results indicated that tricin could attenuate retinal morphological changes and inhibits oxidative stress in diabetic rats. The effects of tricin on morphological changes and oxidative stress in diabetic rats. (a): Retinal pathological changes were detected by HE staining (400×). (b): ROS level were detected by DHE staining. (c): Contents of MDA, SOD, and GSH-Px in serum were detected by ELISA. *p < 0.05, **p < 0.01 vs. control group; ##p < 0.01 vs. diabetes group; ^^p < 0.01 vs. TRI-L group; $p < 0.05, $$p < 0.01 vs. TRI-M group.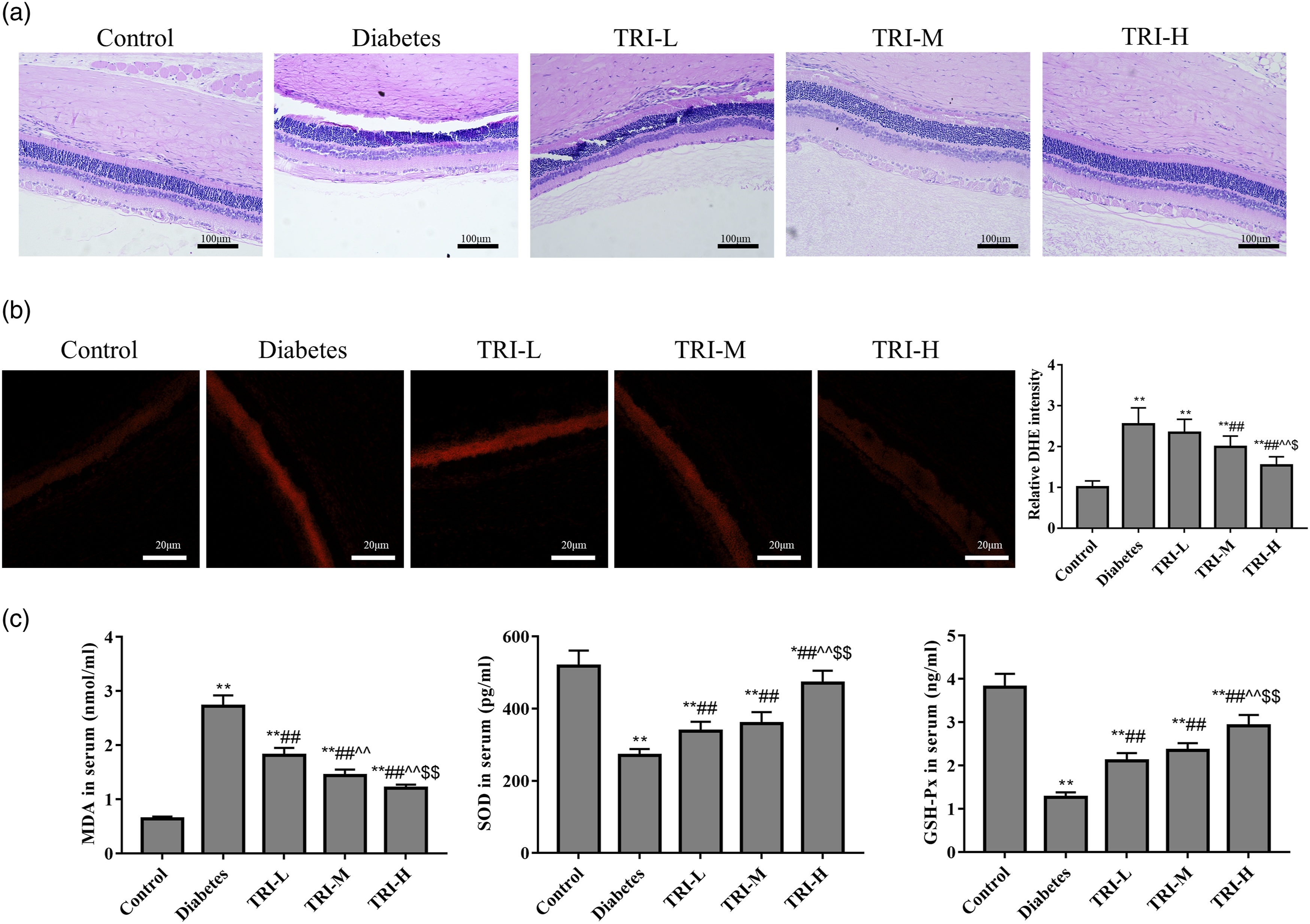
Tricin inhibits angiogenesis and reinforces Sestrin2/Nrf2 signaling in diabetic rats
Figure 2 shows that compared with control rats, the expression of CD31 and VEGFR2 in retina were significantly elevated in diabetes rats at 28 weeks of age, whereas the expression of Sestrin2, Nrf2, and HO-1 was down-regulated significantly (p < 0.05). However, when compared with the diabetes group, the expression of CD31 and VEGFR2 was significantly reduced and the expression of Sestrin2, Nrf2, and HO-1 was significantly increased in the tricin groups (p < 0.05). The results indicated that tricin could ameliorate angiogenesis and rectify the abnormal expression of Sestrin2/Nrf2 in diabetic retinopathy. The effects of tricin on angiogenesis and Sestrin2/Nrf2 signaling in diabetic rats. (a): CD31 was analyzed by immunofluorescence staining (red) and nucleus were observed by DAPI staining (blue). (b): Protein band diagrams were detected by western blot. (c): The expression of CD31, VEGFR2, Sestrin2, Nrf2, and HO-1. **p < 0.01 vs. control group; #p < 0.05, ##p < 0.01 vs. diabetes group; ^^p < 0.01 vs. TRI-L group; $p < 0.05, $$p < 0.01 vs. TRI-M group.
Tricin enhances the proliferation activity and expression levels of Sestrin2 and Nrf2 in ARPE-19 cells
We further investigated the effects of different concentrations of tricin on the proliferation of ARPE-19 cells and intracellular levels of Sestrin2 and Nrf2. The results show that compared with the NG group, the EdU staining ratio and expression levels of Sestrin2 and Nrf2 in the HG group decreased significantly (p < 0.05, Figure 3). However, compared with the HG group, tricin significantly enhanced the proliferation activity and expression levels of Sestrin2 and Nrf2 in ARPE-19 cells (p < 0.05, Figure 3). Those findings revealed that Sestrin2/Nrf2 signaling plays an important role in the protective effect of tricin on ARPE-19 cells. The effects of tricin on the proliferation ability and expression of Sestrin2 and Nrf2 in ARPE-19 cells. (a): EdU was used to detect the cell proliferation ability. (b): Western blot was used to detect the expression of Sestrin2 and Nrf2. (c): The s-phase fraction and the expression of Sestrin2 and Nrf2. **p < 0.01 vs. NG group; #p < 0.05, ##p < 0.01 vs. HG group; ^^p < 0.01 vs. TRI-L group; $$p < 0.01 vs. TRI-M group.
Tricin enhances the proliferation activity and inhibits oxidative stress by regulating Sestrin2 in ARPE-19 cells
For further mechanistic studies, ARPE-19 or HUVECs cells were divided into normal glucose (NG, 5.5 mM d-glucose), high glucose (HG, 60 mM D-glucose), tricin (TRI, 15 μM), tricin + scrambled siRNA (TRI + si-NC), and tricin + Sestrin2 siRNA (TRI + si-Sestrin2) groups. EdU (Figure 4(a)), flow cytometry (Figure 4(b)), and ELISA (Figure 4(c)) results shows that after transfection of Sestrin2 siRNA, the EdU staining ratio and the level of SOD and GSH-Px in TRI + si-Sestrin2 group decreased significantly compared with TRI and TRI + si-NC groups (p < 0.05). Moreover, the content of ROS and MDA were increased significantly compared with TRI and TRI + si-NC groups (p < 0.05). All these results suggested that tricin enhances the proliferation activity and inhibits oxidative stress of high glucose-induced retinal epithelial cells by regulating Sestrin2. Tricin enhances the proliferation activity and inhibits oxidative stress by regulating Sestrin2 in ARPE-19 cells. (a): EdU was used to detect the cell proliferation ability. (b): Flow cytometry was used to detect the intracellular ROS level. (c): ELISA was used to detect the contents of MDA, SOD, and GSH-Px. *p < 0.05, **p < 0.01 vs. NG group; #p < 0.05, ##p < 0.01 vs. HG group; %p < 0.05, %%p < 0.01 vs. TRI group; &p < 0.05, &&p < 0.01 vs. TRI + si-NC group.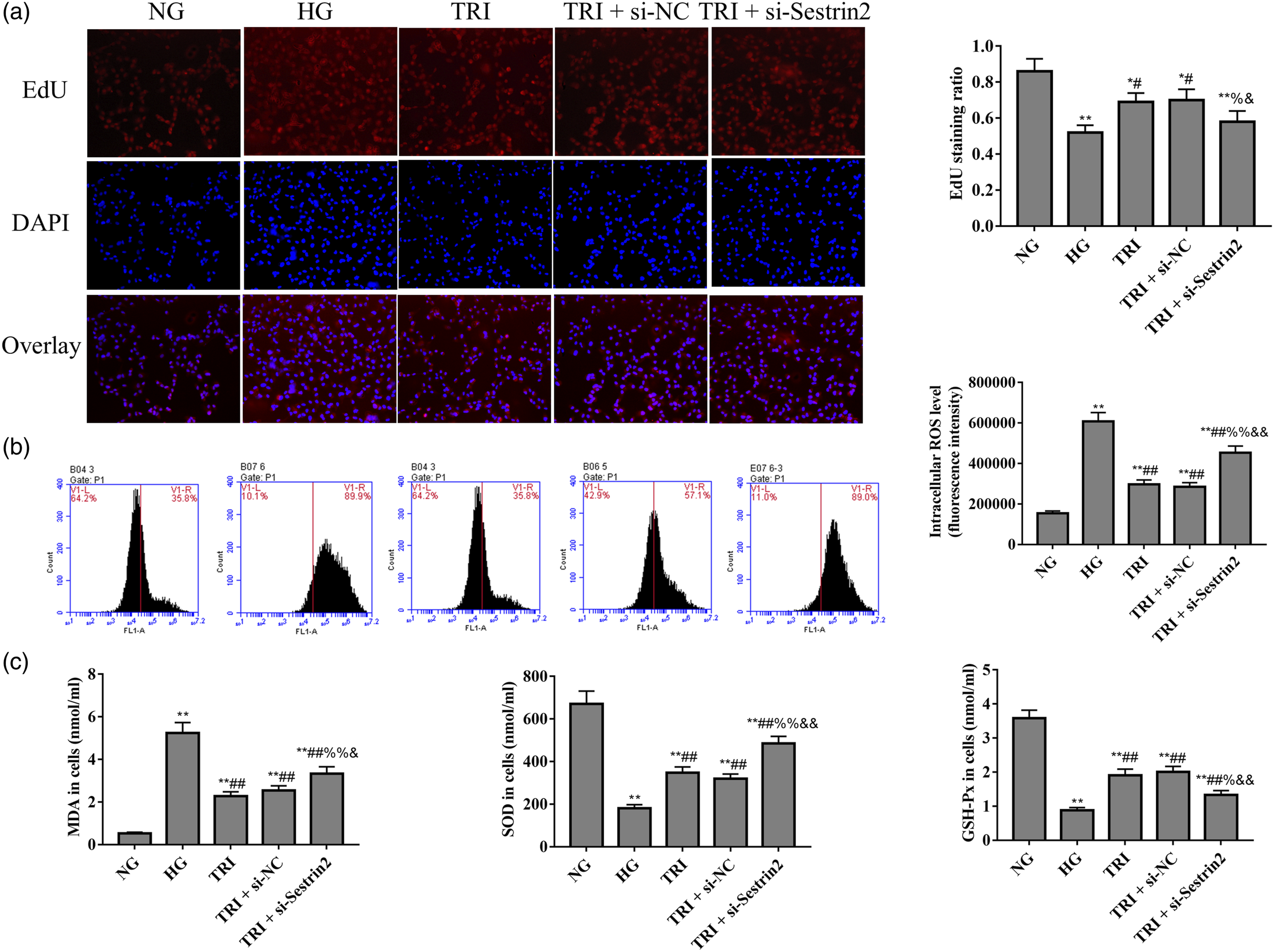
Tricin inhibits angiogenesis and reinforces Nrf2 signaling by regulating Sestrin2 in ARPE-19 cells
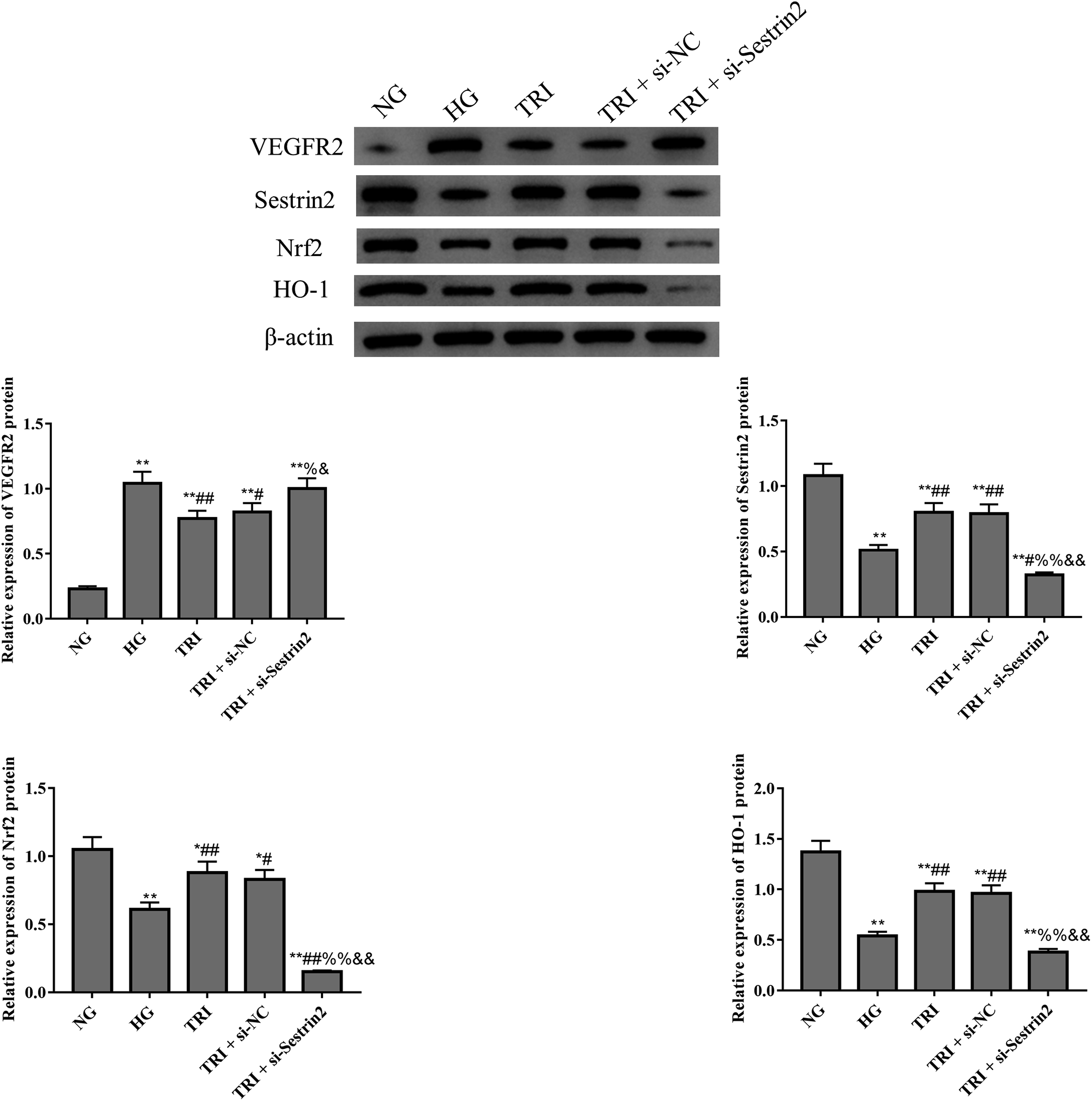
Figure 5(a) and Figure 6 revealed that the expression of CD31 and VEGFR were significantly increased in high glucose-induced retinal epithelial cells, and tricin significantly reduced the expression of CD31 and VEGFR2 in ARPE-19 cells (p < 0.05). Co-culture further revealed that ARPE-19 cells can influence the tube formation of HUVECs cells (Figure 5(b)). However, silence of Sestrin2 significantly reversed the anti-angiogenesis effect of tricin in high glucose-induced retinal epithelial cells. Figure 6 and Figure 7 show that after transfection of Sestrin2 siRNA, the Sestrin2 expression level of TRI + si-Sestrin2 group decreased significantly compared with another groups (p < 0.05), which indicated that the transfections were effectively. Moreover, with the silence of Sestrin2, the Nrf2 and HO-1 expression was also reduced in high glucose-induced ARPE-19 cells. Taken together, these results demonstrated that tricin inhibits angiogenesis of high glucose-induced ARPE-19 cells by reinforcing Sestrin2/Nrf2 signaling. Tricin inhibits angiogenesis by regulating Sestrin2 in ARPE-19 cells. (a): immunofluorescence was used to evaluate CD31 expression. (b): Tube formation assay was used to evaluate tube formation (×200). *p < 0.05, **p < 0.01 vs. NG group; ##p < 0.01 vs. HG group; %%p < 0.01 vs. TRI group; &&p < 0.01 vs. TRI + si-NC group. The expression of Sestrin2/Nrf2 signaling related proteins in ARPE-19 cells were analyzed by western blot. *p < 0.05, **p < 0.01 vs. NG group; #p < 0.05, ##p < 0.01 vs. HG group; %p < 0.05, %%p < 0.01 vs. TRI group; &p < 0.05, &&p < 0.01 vs. TRI + si-NC group. The expression of Sestrin2 and Nrf2 in ARPE-19 cells was analyzed by immunofluorescence staining. **p < 0.01 vs. NG group; #p < 0.05, ##p < 0.01 vs. HG group; %%p < 0.01 vs. TRI group; &&p < 0.01 vs. TRI + si-NC group.

Discussion
DR is one of the main complications of diabetes, and its incidence is increasing worldwide. 3 The occurrence and development of DR is related to the stimulation of high glucose environment, changes in hemodynamics, participation of inflammatory factors and many other factors. 1 Due to its complex metabolic disorders, it is often difficult to treat once it develops into advanced DR. Studying the pathogenesis and pathological characteristics of DR is of great significance for finding a more effective treatment.
Diabetes can cause oxidative stress in multiple tissues, and increased oxidative stress also activates other metabolic pathways that are detrimental to the development of diabetic complications. 18 Hanus J et al. found that Sestrin2 mediates the gossypol acetic acid function in antioxidative response and the retinal pigment epithelial survival upon oxidative stress. 19 Moreover, studies have shown that sestrin2 negatively regulates the proliferation of corneal epithelial cells, 2 and the Sestrin2/Keap1/Nrf2 axis may play an important role in glaucoma retinal degeneration. 21 As an important antioxidant sensor, the activation of Nrf2 is crucial for cellular defense mechanisms. 22 The activation of Nrf2 mediated transcription of multiple target genes, including HO-1, can enhance the resistance of cells to oxidative stress. 7 In this study, immunofluorescence and western blot experiments showed that Sestrin2 expression was downregulated significantly in retina tissue or ARPE-19 cells of model group, which indicated that Sestrin2 was a functional protein in the occurrence and development of DR. In addition, we report that, Sestrin2 small interfering RNA reduced the protective effect of tricin on ARPE-19 cells, as well as abolished its regulating effect on the Nrf2 pathway. These results suggest that Sestrin2 may be a potential and useful target for the treatment of DR by participating in the activation of the Nrf2 pathway.
Oxidative stress plays an important role in the pathophysiology of retinopathy since retina has the highest content of polyunsaturated fatty acids and the highest oxygen consumption of all tissues. 23 The retina is more vulnerable to oxidative stress in diabetes due to its ability to take up oxygen. Diabetic environment increases oxidative stress, and molecules or drugs associated with antioxidant functions may be ideal targets for attenuate diabetic retinopathy. Tricin has antioxidant properties, and Sestrin2/Nrf2 is the important component of endogenous antioxidant defense. While, the role of tricin in DR and its relationship with Sestrin2/Nrf2 have not been reported. Oxidative stress is a cell lesion caused by the inability of cells to resist excessive ROS. 24 Hyperglycemia leads to excessive production of ROS, causing oxidative stress in multiple tissues. Additional, ROS is one of the principal triggers of blood vessel growth. 24 Angiogenesis is a new blood vessel formed on the basis of existing blood vessels, which is essential for embryonic development and wound repair. However, dysregulated angiogenesis contributes to various pathologies such as diabetic retinopathy, atherosclerosis and cancer. 25 Clinically, non-proliferate diabetes retinopathy (NPDR) and proliferate diabetes retinopathy (PDR) are main stages of DR. 26 NPDR is characterized by microaneurysms, increased permeability, vascular occlusion, and degeneration. 27 Subsequently, PDR develops in some patients and is characterized by the formation of new blood vessels in the retinal vasculature. 28 In the present study, we revealed that tricin significantly ameliorated oxidative stress and angiogenesis and rectified the abnormal expression of Sestrin2/Nrf2 in diabetic retinopathy.
In sum, we found here that tricin attenuates DR in SD rats and ARPE-19 cells with a decrease in oxidative stress and angiogenesis. These events, coupled with increased oxidative stress and angiogenesis and reduced Nrf2 pathway activity caused by silenced Sestrin2, further confirmed tricin ameliorates DR injury by reinforcing Sestrin2. These results together suggest that tricin participates in the activation of Nrf2 pathway by regulating Sestrin2, thereby reducing DR in diabetic rats. However, in future studies, an in-depth study of age and age-related development of retina still needs perform to clarify the significance of age in the retina development during aging.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
