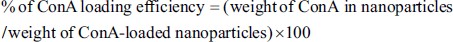Abstract
Background
Non-small-cell lung cancer is the most ubiquitous malignancy, and the present treatments do not offer the most effective outcomes for therapy. Natural drug therapy, which directly targets tumor cells, is now recognized as a safe technique for cancer treatment. The use of tailored nanoparticles also improves the efficacy of cancer therapy.
Objectives
To synthesize, characterize, and evaluate the natural drug concanavalin A (ConA) coated with chitosan nanoparticles (ConA-CS NPs) for cytotoxicity against lung cancer cell line (NCI-H358).
Materials and Methods
The current study demonstrates the development of ConA-CS NPs as well as their unique characteristics, anticancer processes, and possible applications in treatment. Using Uv-vis, Fourier transform infrared, X-ray diffraction, field emission scanning electron microscopy, and dynamic light scattering studies, ConA-CS NPs were generated and evaluated. The mitochondrial toxicity test assay was used to examine ConA-CS NP-induced cytotoxicity. Dual (acridine orange/ethidium bromide) staining was performed to examine apoptotic modifications, and mitochondrial membrane potential levels in NCI-H358 cells were examined using the appropriate fluorescence staining assays.
Results and Conclusion
The use of ConA-CS NPs for enhanced tumor treatment, which combines natural drugs to eradicate the tumor and prevent relapses, is supported by in vitro investigations. The ability of ConA-encapsulated CS NPs to impede human lung carcinoma cell growth has not yet been investigated. Therefore, this investigation may provide a new paradigm for cancer detection and therapy, and the results may surely help to enhance the quality of life for cancer patients.
Introduction
Cancer has emerged as the globe’s deadliest ailment in recent decades (Sung et al., 2021). It is among the most prevalent global causes of death and morbidity. According to the futuristic projections issued by Sung et al. (2021), there will be 1.8 million fatalities caused by lung cancer and 2.2 million new cases of most forms of cancer worldwide in 2020. It has been determined to be the primary cause of cancer-related morbidity and mortality in men. The illness ranks as the third most common cause of morbidity and the second most common cause of death for women. Tung et al. (2013) reported that of all cases of lung cancer, 85–88% are non-small-cell lung cancer (NSCLC) and 12–15% are small-cell lung cancer (SCLC) (Tung et al., 2013; Zhang et al., 2018). The development of novel and potent cancer preventive therapies is essential given the global malignant illness rate’s steady rise. According to estimates by Sung et al. (2021), there will be 28.4 million more cases of cancer globally by 2040, a 50% increase over the next 20 years. Traditional cancer treatments have several unfavorable side effects, including intolerance, drug resistance, and detrimental effects on healthy tissue. Therefore, the development of a novel cancer prevention technique is highly necessary. Therefore, plant-derived natural products are considered safe therapeutic possibilities, given their significant therapeutic benefits, which include fewer side effects, improved health, and the potential to treat tumors. With the addition of nanotechnology, it has become a new avenue for cancer treatment (Kemp & Kwon, 2021).
Biodegradable nanoparticles have garnered a lot of attention lately because of their efficient bodily elimination and safe treatment administration. To produce biodegradable nanoparticles, a variety of substances were employed, such as proteins, polysaccharides, and synthetic polymers (Cheung et al., 2015). Because of their superior biocompatibility, biodegradable polymers are now used extensively in the production of nanoparticles as carriers of drugs (Venkatesan et al., 2016).
Chitosan is a polymer that occurs naturally and is formed from chitin that possesses a broad variety of bioactivities. It has previously been widely used as an excellent biocompatibility carrier of drugs (Sultan et al., 2022). Chitosan-based nanoparticles (CS NPs) are showing a great deal of promise for the successful and secure delivery of biomolecules and drugs against cancer (Narayan et al., 2021). When juxtaposed with alternative nanoscale drug carriers, CS NPs show various benefits, such as heightened stability, accommodating production circumstances, and utilizing aqueous solutions rather than organic solvents. A variety of methods can be used to establish CS NPs (Ahmad et al., 2023).
Plant lectin is an instance of a noxious protein that is found in naturally occurring plants. It works by binding selectively to glycans to cause physiological consequences (Estrada-Martínez et al., 2017). Plant lectin is a toxic protein that exhibits antiproliferative properties against tumor cells. Furthermore, plant lectin’s unique capacity to bind sugars has been used to distinguish between tumors that are not malignant and those with varying levels of glycosylation (Bloise et al., 2021; Lawanprasert et al., 2020; Oliveira et al., 2019).
Concanavalin A (ConA) is a typical legume lectin that has been investigated extensively. It is believed to target programmed cell death (PCD) and diversify human tumor cell death. Apoptosis and autophagy, two evolutionary-conversed mechanisms for preserving homeostasis and eradicating pathogenic cells, are referred to as PCD (Ouyang et al., 2012). ConA was found to trigger apoptosis in mouse macrophage cells and human melanoma cells in earlier investigations. Additionally, ConA caused HeLa cells to undergo autophagic cell death (Faheina-Martins et al., 2012; Suen et al., 2000). Consequently, ConA possesses noteworthy abilities to induce apoptosis and autophagy, rendering it a promising antineoplastic drug for future cancer treatments. However, no research has been done on the fabrication of ConA drug-loaded nanoparticles with anticancer efficacy employing natural polymers, namely chitosan. In this study, we synthesized durable, biocompatible, and ConA-loaded chitosan nanoparticles (ConA-CS NPs) for the therapy of cancer. The chitosan’s sensitivity to pH might enable tumor-specific drug release. After ConA-CS NPs were produced and described, in vitro anticancer activity was examined. ConA-CS NPs that were produced have the potential to be a promising therapeutic agent for the treatment of lung carcinoma.
Materials and Methods
Materials
Dimethyl sulfoxide (DMSO), hydrochloric acid, sodium hydroxide pellets, low molecular weight chitosan, sodium tripolyphosphate (NaTPP), glacial acetic acid, acridine orange/ethidium bromide (AO/EtBr), rhodamine-123 (Rh-123) staining, dichloro-dihydro-fluorescein diacetate (DCFH-DA), and ConA were acquired from Sigma-Aldrich (St. Louis, MO, USA). Despite any additional extraction, most of the compounds constituted of excellent analytical quality.
Synthesis of ConA-coated CS NPs (ConA-CS NPs)
ConA-CS NPs were developed via an ionic gelation technique employing NaTPP as a gelating substance, somewhat altering the earlier described procedure (Othman et al., 2018; Venkatesan et al., 2016). To make a solution comprising chitosan (with purity > 90%, viscosity 60–300, and molecular weight of 60–160 kDa) with a value of 2.5 mg mL–1, 1.25 g of chitosan, and 0.25% (1.25 mL) of Tween 80 were dispersed in 500 mL of 1% (v/v) acetic acid. Overnight at the ambient temperature, the chitosan solution was constantly agitated and 1 M NaOH aqueous solution was employed to bring the pH of the solution down to 5.5. Due to the formation of nanoparticles, the subsequent foggy solution parted ways, and the nanoparticles were separated in the solution by centrifuging (18,000 rpm) at 10°C for 30 minutes (Moosazadeh Moghaddam et al., 2023). The nanoparticles were centrifuged 3 times at 18,000 rpm for 10 minutes at 10°C to eliminate unprocessed polymers. They were then stored at 4°C for further investigations.
ConA was successfully encapsulated within the CS NPs by adding a ConA suspension in anhydrous ethanol (3 mg mL–1) to the chitosan solution (2.5 mg mL–1, 10 mL) and agitating at 1,000 rpm for 1 hour. Next, with gentle stirring, a drop-by-drop addition of NaTPP mixture in water (0.25 mg mL–1, 25 mL) was made to the chitosan/ConA combination. To generate the ConA-CS NPs, the mixture that resulted was stirred for 1 hour at room temperature. ConA-CS NPs were separated by centrifugation for 30 minutes at 12,000 rpm to exclude unbound ConA and unprocessed chitosan. After being separated, the nanoparticles were lyophilized after being washed with purified water. The identical production method was used for producing ConA-free, conventional CS NPs; the only difference was the inclusion of ConA.
Evaluating the Loading Capacity of ConA
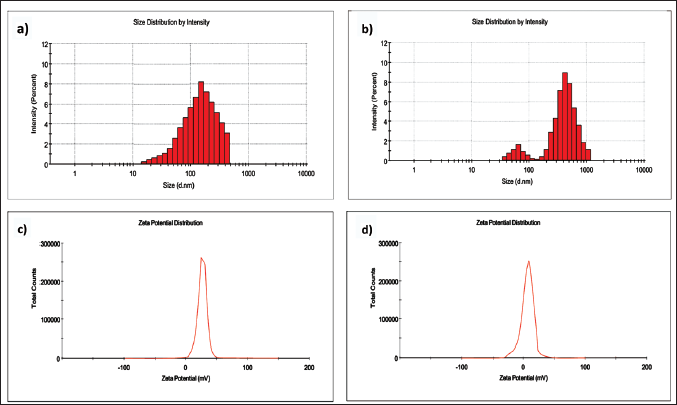
The granule containing ConA-CS NPs was separated from the supernatant [containing free pellet (PL)] by centrifuging the mixture at 12,000 rpm to measure the ConA loading efficiency of the ConA-CS NPs. UV–Vis spectroscopy at 328 nm was used to calculate how much ConA was still in the supernatant. The quantity of ConA in the supernatant was subtracted from the total amount of ConA utilized in the mixture to determine the volume of ConA loaded in the ConA-CS NPs. For later use, a particle of ConA-CS NPs was obtained and kept at 4ºC. The following equation was used to compute the ConA loading efficiency:
Characterization of ConA-CS NPs
The shape, size, and structure of the generated ConA-CS NPs were characterized using a variety of characterization techniques, such as UV–Vis, X-ray diffraction (XRD), Fourier transform infrared (FT-IR), scanning electron microscopy (SEM), dynamic light scattering (DLS), and zeta potential.
UV–Vis Spectroscopy
Utilizing a UV–vis spectrophotometer with a frequency range of 200–800 nm distance deviation, the powdered synthesized ConA-CS NPs were dispersed in deionized (DI) water to determine the maximum peak absorption.
FT-IR Spectroscopy
Functionality entities, molecules in the body, and metal expansion were found to be responsible for reducing the amount and stability of ConA-CS NPs through the use of FT-IR. Using an FT-IR spectrophotometer set to 4,000–400 cm–1, the spectra for the dried-out powder of the ConA-CS NPs that were generated were recorded.
Morphological Analysis of ConA-CS NPs
Employing the findings of the SEM and energy dispersive X-ray analysis (EDAX) assessment, the structure and average dimensions of the produced ConA-CS NPs were investigated using a scanning electron microscope. To verify the elemental makeup of ConA-CS NPs made using synthetic methods, the device was connected to an EDAX.
DLS and Zeta Potential Analysis
The fabricated ConA-CS NPs stability and size distribution scattering were evaluated using a zeta potential device and DLS, accordingly. The ConA-CS NPs material was placed in a zeta and DLS cuvette for analysis after ultrasonication, scattering, and dispersal in Milli-Q H2O (1 mg in 1 mL).
Drug Release Capacity
Dialysis was used to examine drug release with various pH buffer conditions. A dialysis membrane bag (MW = 10 kDa) was immediately filled with a solution of ConA-CS NPs (10 mg mL–1). Twenty milliliters of buffers (pH 5.0 and pH 7.4) were added to the dialysis bag and stirred magnetically at 120 revolutions per minute at 37°C. An equivalent volume of brand-new buffer was added to the sample solution (1 mL) at the prearranged time. Using a UV measurement at 328 nm, the quantity of ConA released into the solution was calculated.
In Vitro Cytotoxicity Assay
ConA-CS NPs cytotoxicity was assessed using the mitochondrial toxicity test (MTT) (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) test. On 96-well plates, lung cancer cell line (NCI-H358) (5 × 103) cells were cultivated in each well, and the cells were allowed to multiply for a whole day. Each well received 100 µL of the MTT agent following an additional 48 hours of incubation in cell culture with ConA-CS NPs at varying levels. After incubating for 4 hours, the absorbance of every well was measured at 490 nm using a microplate reader. Equation cell viability = (OD treatment/OD control) × 100% was used to estimate the cell viability.
Intracellular Generation of Reactive Oxygen Species (ROS)
The ROS generation experiment was conducted by previous guidelines (Xu et al., 2019). The NCI-H358 cells were treated for 24 hours with ConA, CS NPs, and ConA-CS NPs. Afterward, they were washed with phosphate-buffered saline (PBS) and reconstituted in culture media (separate from serum) with 10 µM DCFH-DA. Following that, ROS production was determined by flow cytometry.
AO/EB Staining
In NCI-H358 cells treated with ConA, CS NPs, and ConA-CS NPs, the proportion of cell apoptosis was ascertained using AO/EtBr staining. NCI-H358 cells were plated (1 × 103/well) and they were incubated at 37°C for 24 hours. Following ConA, CS NPs, and ConA-CS NPs therapy, the cells were incubated for 24 hours at 37°C. The cells that were treated were stained for 10 minutes with 100 µg/mL of AO/EtBr dye in a 1:1 ratio after the treatment process stage. The amount of apoptotic cell death in the treated cells was quantified using a fluorescent microscope.
Mitochondrial Membrane Potential (MMP) Assay (∇ΨM)
Employing Rh-123 staining techniques, the amount of MMP (∇ΨM) in NCI-H358 cells exposed to ConA, CS NPs, and ConA-CS NPs was assessed. In order to accomplish this, 1 × 103 NCI-H358 cells were inserted into each well of a 24-well plate, and the cells were then incubated at 37°C for 24 hours. After that, the cells were treated with ConA, CS NPs, and ConA-CS NPs for 24 hours at 37°C. Following 30 minutes of Rh-123 staining the cells at a dose of 10 µg/mL, the fluorescence brightness was measured with a fluorescence microscope.
Apoptosis Detection via Flow Cytometry
The NCI-H358 cells subjected to ConA-CS NPs were examined for the progression of apoptosis using annexin V-FITC labeling. By the integrity of the membrane, necrotic and apoptotic cells were distinguished using propidium iodide (PI) staining. In summary, ConA, CS NPs, and ConA-CS NPs were applied to NCI-H358 cells for 24 hours at an inhibitory concentration (IC50) and a final concentration of 200 mg/mL (based on the weight of chitosan). The cells were subsequently washed twice with cold PBS. Following rinsing, 1 × 104 NCI-H358 cells were stained with PI staining and annexin V-FITC (BD Sciences) for 15 minutes in the dark. A flow cytometer was used to determine the staining cells’ mean fluorescence intensity. The percentages of viable (annexin V–, PI–), necrotic (annexin V–, PI+), early (annexin V+, PI–), and late (annexin V+, PI+) apoptotic/dead cells were used to express the results. ConA-CS NPs exposure resulted in a greater proportion of apoptotic cells than in untreated cells.
Evaluation of Cell Cycle Arrest
NCI-H358 cells (1 × 104) in culture have been treated with ConA, CS NPs, and ConA-CS NPs for a duration of 24 hours. After being removed, the cells were kept in ice-cold 70% ethanol for 12 hours at 4°C. Once washed with PBS, the sample was once more submerged in a solution containing 25 µL PI. After adding 10 µL of RNase A to 0.5 mL of buffer, the mixture was allowed to settle in the dark at 37°C for 30 minutes. Next, the phase organization of the cell cycle was determined with a flow cytometer.
Statistical Examination
In triplicate statistics, mean ± standard deviation was provided after a one-way assessment of variance (ANOVA) was performed at a significance threshold of p < 0.01.
Results
Preparation of PL–CS NPs
In this study, an ionic gelation method was used to prepare CS NPs encapsulating ConA. Chitosan was mixed with NaTPP at a 4:1 weight ratio. The physicochemical properties of the formulated CS NPs have been known to be strongly influenced by the degree of deacetylation of chitosan, NaTPP concentration, chitosan concentration, chitosan/TPP molar ratio, and pH condition. The formation of CS NPs was carried out under pH 5.5 conditions because chitosan can be actively self-assembled into nanoparticles in an acidic aqueous solution through ionic cross-linking with multivalent anions. ConA-CS NPs were analyzed with FT-IR, SEM, zeta potential, and DLS.
Characterization of ConA-CS NPs
FT-IR spectroscopy was used to analyze the ionic gelation process that produced the ConA-CS NPs. Figure 1 shows the FT-IR spectra of ConA, CS NPs, and ConA-CS NPs. The significant range in the chitosan spectrum was found to be approximately 3,412 cm–1, which is the OH vibration. The C–N stretching vibration of type I amine has a peak at 1,384 cm–1, while the asymmetric stretch of C–O–C has a peak at about 1,159 cm–1. The peak of 3,412 cm–1 in the chitosan spectrum was displaced to 3,405 cm–1 and widened in the CS NP spectra, suggesting enhanced hydrogen bonding. Furthermore, the peak values for the amide II carbonyl stretch at 1,646 cm–1 and the amine I N–H bending vibration at 1,380 cm–1 were moved to slight modification, respectively. ConA-CS NPs (Figure 1c) show that ConA has coated CS NPs by showing all of the peaks and connections that are associated to chitosan and ConA (546, 690, 732, 1,010, 1,380, 1,621, 1,728, 2,110, 3,100, and 3,405 cm–1). The incorporation of chitosan to the construction, which functions as a chelator and deposits the amine and hydroxyl groups out of chitosan on the structure of ConA, is what causes a reduction in intensity at the peak in the ConA-CS NPs when compared to ConA. Additionally, there is a little peak displacement in the ConA-CS NPs when compared to ConA. This is because of the regional interactions among O, Co, and N, as well as the electric charge exchange between the constituent parts of the ConA-CS NPs. The crystalline structure of ConA was not destroyed by the addition of chitosan in the ConA-CS NPs, indicating that ConA’s structure was intact.
Concanavalin A-loaded Chitosan Nanoparticles (ConA-CS NPs), Concanavalin A (ConA), and Chitosan Nanoparticles (CS NPs) (a, b) were Analyzed Using Fourier Transform Infrared (FT-IR). The Amine Groups, Hydroxyl Rings, and Stretching Vibration of the Water Bond are Responsible for the Large Peaks in the 3,446 cm−1 Range of the Chitosan Sample’s Wavelength of Absorption (a). Within the Spectrum of 3,437, 3,285, 2,780, 2,150, 1,830, 1,650, and 697 cm−1 and 45, 1,335, 1,450, 1,590, 1,760, 2,120, 2,878, 3,332, and 3,620 cm−1, Absorption Bonds are Seen for the ConA Sample (b), Indicating the Encapsulation of CS NPs by ConA. Between 400 and 4,000 cm−1 was the Range Obtained.
SEM and EDAX Analysis
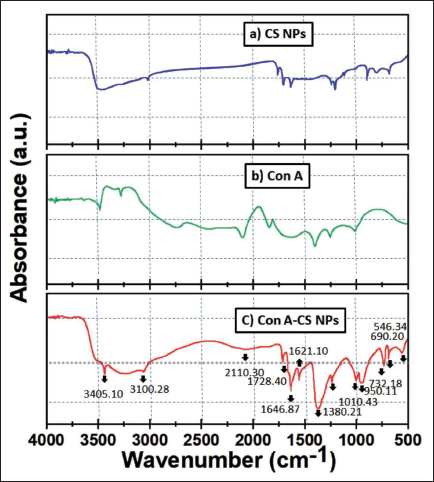
The dimensions, structure, and dispersal of created nanoparticles were all described using SEM. The representative micrographs of the produced ConA-CS NPs are shown in Figure 2a. It was found that the dispersed, spherical, smooth-surfaced nanoparticles had a mean particle dimension of 198–220 nm. They produced NPs’ high surface energy, the tiny spaces between them, and the particles themselves all contributed to this type of aggregation.
Scanning Electron Microscopy (SEM) Images of Concanavalin A-loaded Chitosan Nanoparticles (ConA-CS NPs). Energy Dispersive X-ray Analysis (EDAX) Spectra of Synthesized ConA-CS NPs.
EDAX was utilized to count the volume of oxygen as well as metals contained on the outside of the fabricated ConA-CS NPs. The EDAX and dispersal of the particle dimensions of the produced ConA-CS NPs are shown in Figure 2b. Oxygen from C metal NPs was seen in the EDAX spectra, peaking at 1.5 keV. A few faint peaks of C and O were also present; they were connected because the extracts contained molecules.
Analysis Using DLS and Zeta Potential
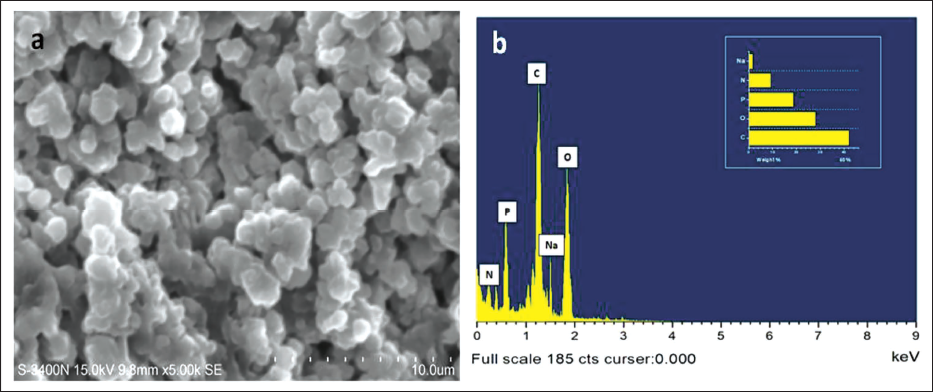
The obtained ConA-CS NPs’ particle size distribution and stability were assessed using DLS and zeta potential techniques. CS NPs and ConA-CS NPs were believed to be necessary components for a variety of biological and abiotic systems based on zeta potential and DLS studies. The polydispersity index (PDI), charge, and particle size distribution of CS NPs and ConA-CS NPs are observable in the DLS spectra, as Figure 3 illustrates. With a PDI of 0.462 and 0.418, the expected particle size for CS NPs as well as ConA-CS NPs was 192.5 ± 6 and 286 ± 2 (Figure 3a and b). Conversely, studies of the zeta potential for CS NPs and ConA-CS NPs made via ionic gelation creation revealed that it was 26.3 and 23.4 mV (Figure 3c and d). Due to the decreased aggregation risk of negative zeta potential particles, the ConA-CS NPs typically exhibit greater stability.
Particle Size Distribution of (a) Chitosan Nanoparticles (CS NPs) (c, d) Zeta Potential Analysis of Synthesized CS NPs and Concanavalin A-loaded Chitosan Nanoparticles (ConA-CS NPs).
ConA Loading and Release Property
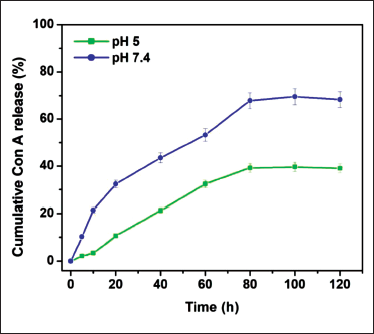
ConA-CS NPs’ ability to load drugs increased in a concentration-influenced manner, up to 78% of their weight. Conversely, Table 1 shows that the encapsulation efficiency (EE) had a maximum value of 74.6% and showed an inverse association with the concentration of ConA. Last but not least, in vitro drug release from the ConA-CS NPs was equivalent in a variety of physiological pH conditions, including pH 7.4 of healthy tissues and pH 5.0 corresponding to the acidification of endosomes (Figure 4). Consequently, the ConA-CS NPs may release the drug cargo in a range of physiological conditions, regardless of pH. ConA-CS NPs showed impressive stability in a typical physiological microenvironment and blood circulation, as shown by Figure 4, where less than 5% of ConA was released at pH 7.4 in PBS after 24 hours of incubation and approximately 19.5% of ConA was released by ConA-CS NPs over a period of 24 hours at pH 5.0. ConA-CS NPs’ pH sensitivity suggests that, given that tumor microenvironments are somewhat more acidic than physiological tissues, they have great potential for releasing drugs to treat cancer.
Drug Loading (DL) and Encapsulation Efficiency (EE) of Concanavalin A-loaded Chitosan Nanoparticles (ConA-CS NPs).

Drug Release Profile from Concanavalin A-loaded Chitosan Nanoparticles (ConA-CS NPs) at Different pH Values. Data were Expressed as the Mean ± SD (n = 3) Using an Assessment of Variance (ANOVA).
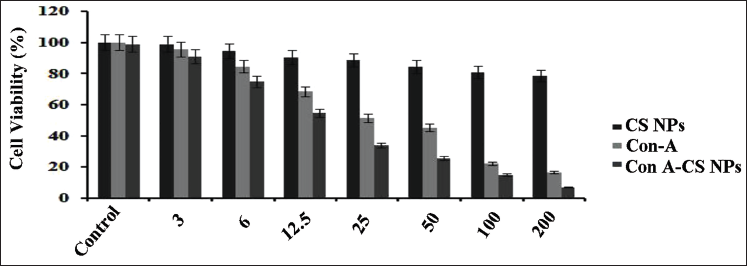
Cytotoxicity of ConA-loaded CS NPs
In this investigation, we used an MTT-based experiment to investigate the prospective anticancer effects of a few formulations, such as ConA, CS NPs, and ConA-CS NPs, on NCI-H358 cells. Figure 5 shows that the cells were treated separately for 24 hours with different amounts of these compositions, ranging from 3 to 200 µM/mL. It is noteworthy that the control group showed no symptoms of cytotoxicity. On the other hand, cytotoxicity increased progressively as the cells received exposure to higher doses of ConA and ConA-CS NPs. This may be related to CS NPs’ effective drug release. At a concentration of 12.5 and 50 mM ConA, 50% of NCI-H358 cells were killed by both free and ConA-CS NPs. Meanwhile, research was done on the cytotoxicity of simple ConA-CS NPs. Incredibly, substantial doses of CS NPs (50 µM/mL) showed that the NCI-H358 cells treated with simple CS NPs demonstrated cell viability greater than 80%. These findings show that CS NPs are extremely biocompatible and suitable for use as a secure delivery system for anticancer drugs. Nevertheless, the use of ConA-CS NPs increased the cytotoxic impact. From these findings, it was clear that the ConA-loaded CS NPs had the greatest amount of cytotoxic activity when compared to the unloaded type.
Viability of Lung Cancer Cell Line (NCI-H358) Cells Cultured for 24 Hours at Different Rates of Concanavalin A (ConA), Chitosan Nanoparticles (CS NPs), and Concanavalin A-loaded Chitosan Nanoparticles (ConA-CS NPs). Using the Traditional Mitochondrial Toxicity Test (MTT) Assay, the Anticancer Effect of ConA-CS NPs towards NCI-H358 Lung Cancer Cells was Determined. In a Dosage-dependent Way, ConA and ConA-CS NPs Demonstrated Strong Cytotoxicity against NCI-H358 Cells. Remarkably High Concentrations of CS NPs, the NCI-H358 Cells Treated with Pure CS NPs Demonstrated Cell Viability of Over 80%. These Findings Show that CS NPs are Extremely Biocompatible and Suitable for Use as a Secure Delivery System for Anticancer Drugs. An Assessment of Variance (ANOVA) was Used to Express the Data as the Mean ± SD (n = 4).
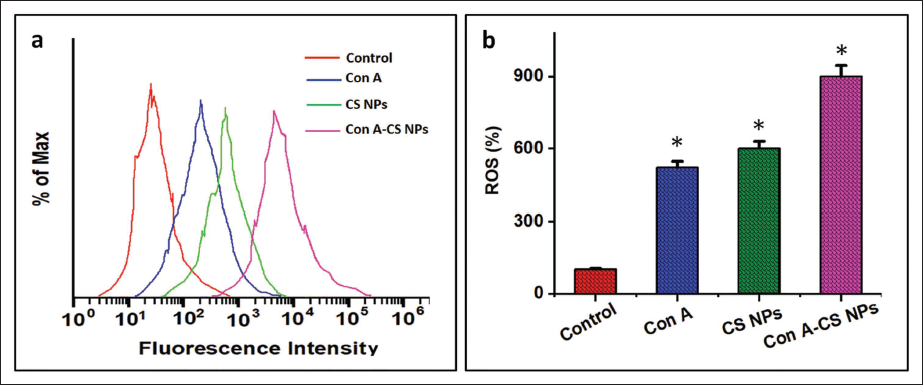
ROS Production of ConA-CS NPs
The current work measured the cellular quantity of ROS generation in lung carcinoma cells employing a DCFH-DA probe. Flow cytometry and confocal visualization were used to confirm the results. The flow cytometry findings showed that the ConA-CS NPs significantly enhanced ROS generation after treatment with ConA, CS NPs, and conjunction usage of ConA-CS NPs for 24 hours (Figure 6a). The green fluorescence value significantly increased after being exposed to ConA-CS NPs for 24 hours, as shown in Figure 6a. Using flow cytometry, the intensity of DCF fluorescence was assessed (Figure 6b), and intracellular ROS levels were quantitatively assessed. NCI-H358 cells produced more ROS following treatment with ConA-CS NPs, and this increase was dose-related for ConA. Thus, it is hypothesized that ConA-induced apoptosis in NCI-H358 carcinoma cells is partly due to elevated intracellular ROS caused by ConA-CS NPs.
Using 2,7-Dichlorofluorescein Diacetate Labeling, It was Possible to Examine in A549 Lung Cancer Cell Line (NCI-H358) Cells the Effect of Concanavalin A (ConA), Chitosan Nanoparticles (CS NPs) and Concanavalin A-loaded Chitosan Nanoparticles (ConA-CS NPs) on the Generation of Intracellular Reactive Oxygen Species (ROS). Flow Cytometry (a): A Flow Cytometer was Used to Quantify the Amount of Intracellular ROS Accumulation and Color It with Dichloro-dihydro-fluorescein Diacetate (DCFH-DA). (b) Measuring Intracellular ROS Levels Quantitatively. When Compared to the Control Group, the Results Show the Mean and Standard Deviation of Three Replicates at the Level of Significance of *p < 0.05.
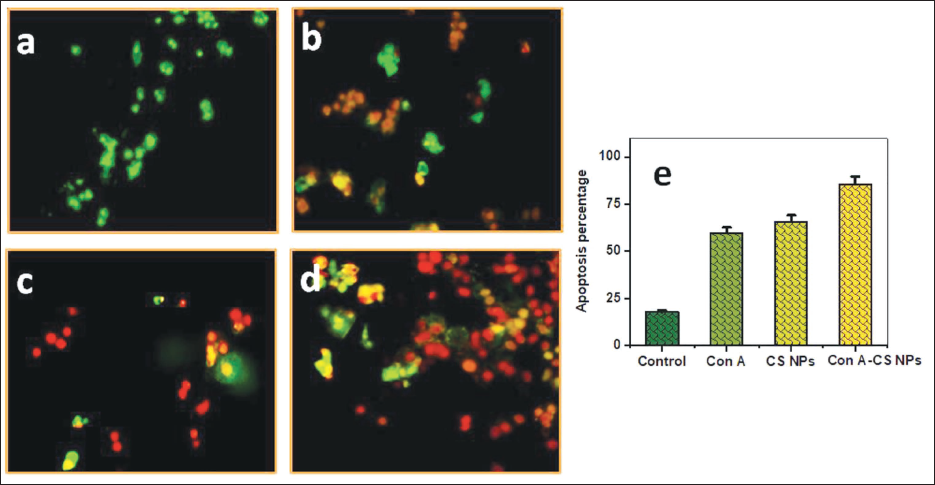
Activation of Apoptosis by ConA-CS NPs
The potential of ConA, CS NPs, and ConA-CS NPs to affect lung cancer cells’ capacity to complete apoptosis was demonstrated in this instance using AO/EtBr staining. Under a fluorescence microscope, chromatin, which abridged throughout apoptosis, may be seen using the widely used AO/EtBr staining. Fluorescence microscopy is used to show how NCI-H358 cells have been subjected to different concentrations of ConA for a whole day (Figure 7a). Green fluorescence was continually seen in the nucleus of the control cell. Rather, fluorescence imaging demonstrated that an impairment in the membrane’s stability resulted in an unbalanced nucleus shape in the treated NCI-H358. Formulations including cells treated with ConA-CS NPs exhibited reddish-orange structures called apoptotic bodies, which involved nuclear regression and puffing. The enhanced antitumor capacity of the formed ConA-CS NPs has been attributed to the lung cancer cells. In the groups that were treated with ConA-CS NPs, the proportion of apoptotic cells was 83.12% (Figure 7b). The little cell death seen by the simple CS NP-treated cells indicated their great biocompatibility.
To Assess the Apoptosis Phase Using a Concanavalin A (ConA), Chitosan Nanoparticles (CS NPs), and a Combination of Concanavalin A-loaded Chitosan Nanoparticles (ConA-CS NPs), the Cells were Further Stained with Acridine Orange/Ethidium Bromide (AO/EtBr).
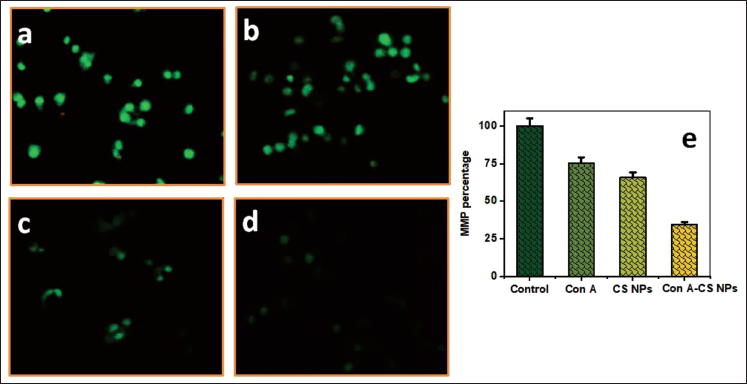
MMP Level in NCI-H358 Cells Induced by ConA-CS NPs
NCI-H358 cells undergo apoptosis and produce more MMP in response to ConA, CS NPs, and ConA-CS NPs. Using a fluorescence microscope, the MMP changes were found (Figure 8a). Most people agree that MMP levels decrease during apoptosis. The study examined the role of ConA-CS NPs treatment using Rh-123 staining, which is reliant on MMP changes, an early indicator of apoptotic characteristics. Figure 8b shows the different dosages of ConA, CS NPs, and ConA-CS NPs that were applied to NCI-H358 cells over the course of a day. Initial signs of apoptosis were brought on by the treatment’s impact on MMP.
The effect of Concanavalin A (ConA), Chitosan Nanoparticles (CS NPs), and Concanavalin A-loaded Chitosan Nanoparticles (ConA-CS NPs) on Lung Cancer Cell Line (NCI-H358) Cell Mitochondrial Membrane Potential (MMP) Levels.
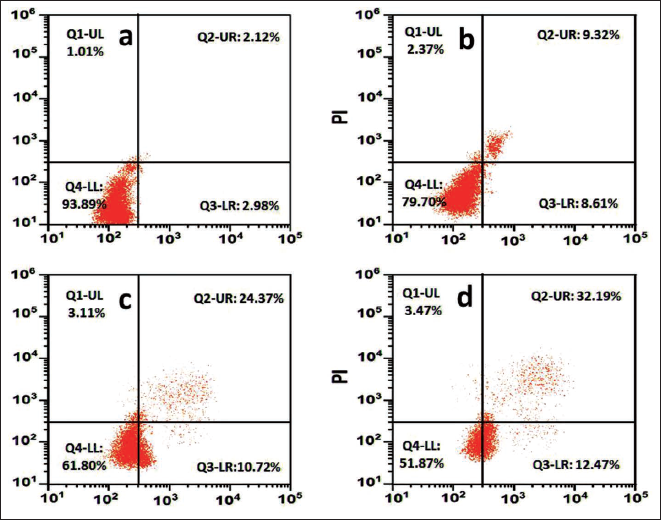
Apoptosis by Flow Cytometry
Using an apoptosis kit that included fluorescein isothiocyanate (FITC)-labeled annexin V and PI staining, it was possible to determine the proportion of necrotic and apoptotic cells presented in Figure 9. ConA, CS NPs, and ConA-CS NPs treating NCI-H358 cells differed significantly from the control group, according to the findings. Consequently, 32.19% of ConA-CS NPs were apoptotic cells. In reaction to treatment with ConA-CS NPs, we observed an increase in the number of cells in the Q2 and Q3 states of apoptosis in NCI-H358 cells. The fraction of apoptotic cells was shown to increase in a dose-related manner when ConA-CS NPs were added to NCI-H358 cells.
Apoptotic Analysis by Flow Cytometry; (a) Control, (b) Chitosan Nanoparticles (CS NPs), (c) Concanavalin A (ConA), and (d) Concanavalin A-loaded Chitosan Nanoparticles (ConA-CS NPs).
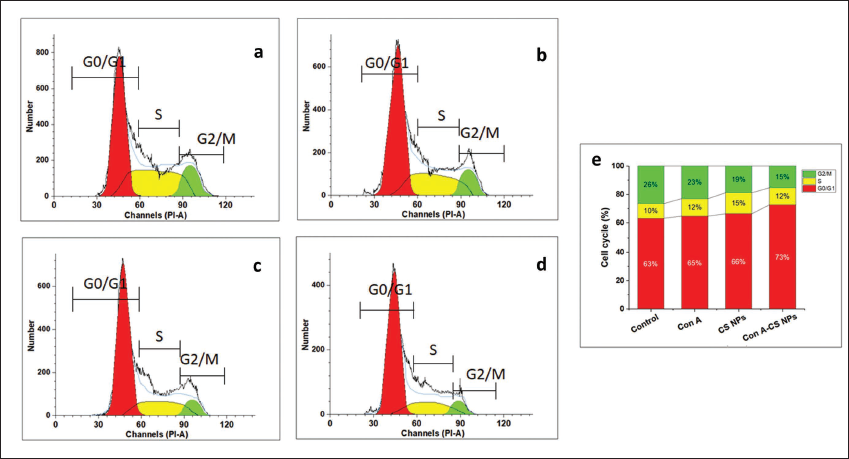
Cell Cycle Arrest
Preventing the growth of cancer cells requires the cell cycle to be advanced. To further comprehend the potential process leading to the synergistic application of ConA and CS NPs’ antiproliferative characteristics, the effects of the ConA-CS NPs on cell cycle dispersion were examined. The results of the cell cycle investigations indicated that applying ConA-CS NPs at selected doses to NCI-H358 cells caused the cells to arrest in the G2/M phase. This suggests that the simultaneous administration of ConA-CS NPs inhibited cell growth by stopping the cell cycle (Figure 10).
Cell Cycle Analysis; (a) Control, (b) Chitosan Nanoparticles (CS NPs), (c) Concanavalin A (ConA), and (d) Concanavalin A-loaded Chitosan Nanoparticles (ConA-CS NPs). According to This Result, the Lung Cancer Cell Line (NCI-H358) Cells were Markedly Inhibited by the ConA-CS NPs via the G2/M Phase Cell Cycle Arrest.
Discussion
The most often detected variety of lung cancer worldwide is NSCLC, which is one of the least common invasive cancers with a high mortality rate (Bade & Dela Cruz, 2020). Although several medications are often used to treat NSCLC, their resistance to small cell lung carcinoma relapse limits their clinical utility (Lin et al., 2019). Because lung cancer has a high rate of recurrence, resistance to treatment develops quickly, and current medicines are ineffective after a recurrence. Byers and Rudin (2015) argue that the development of new medications is essential to improving and reducing fatal lung tumors. A variety of combinations are being tested for the treatment of cancer to treat the illness more efficiently (Bayat Mokhtari et al., 2017; Choi et al., 2022).
Many plant lectins that have been scientifically shown to possess antiproliferative impacts on cancer cells both in vivo and in vitro are well-known in recent decades. Legume lectins, one of the 12 lectin families, have drawn the most interest from researchers due to their exceptional antiproliferative properties and their uses in cancer treatments. The earliest known legume lectin was ConA. Due to its ability to trigger autophagy and mitochondrial apoptosis, it may be used as an antineoplastic drug in upcoming cancer treatments (Shi et al., 2014). Comparing natural material-based drug delivery systems (DDSs) to traditional treatments, encouraging outcomes have been seen. Their numerous advantages include being biodegradable, recyclable, environmentally friendly, and non-immunogenic, as well as being easily modified for integration (Wong et al., 2020). Effective stability, uniform size dispersion, excellent drug encasing, adjustable drug release, and tunable physicochemical characteristics are among the benefits of CS NPs (Muller et al., 2019). Promoting the use of plant-based substances as anticancer treatments requires conducting clinical studies to evaluate their anticancer efficacy. Thus, in the current study, CS NPs were employed as a vehicle to deliver natural drugs. The resulting ConA-CS NPs provoked cell death and impeded the proliferation of lung cancer cells, helping to establish a feasible, effective therapy for the disease.
The FT-IR spectroscopic examination demonstrated the exterior chemistry of groups of functional importance in addition to innate molecules, as demonstrated by distinctive Fourier transform bands with varying wave numbers that corresponded to distinct substantial modifications in the reflection bands’ impact and location (Manimaran et al., 2022). The encapsulation of CS NPs by ConA is shown by most of the peaks and interactions in the ConA-CS NPs that are associated to chitosan and ConA (745, 1,335, 1,450, 1,590, 1,760, 2,120, 2,878, 3,332, and 3,620 cm−1). The effect of the chitosan, furthermore, which functions as a chelator and absorbs the amine and d-hydroxyl group of chitosan and deposits them in the ConA matrix, is that the extent of the peaks in the ConA-CS NPs data is lower than in the ConA data. The electric charge exchanges among the ConA-CS NPs parts that constitute it as well as the local connections among O, Co, and N are the reasons for the sample’s peak shift in comparison to ConA.
An essential physicochemical characteristic that influences the physical stability and adhesive qualities of nanoparticles is their zeta potential (Bayat et al., 2008). Zeta potential levels between <–30 mV and >+30 mV are, in theory, regarded as stable ranges. Regarding zeta potentials, the ICN-K04 formulation in this investigation had one of +39 mV, indicating significant interaction stability. The range of particle sizes is measured by the PDI, which has values between 0 and 1. Within this investigation, PDI values near 0 demonstrated a uniform dispersion, whereas values over 0.5 indicated significant heterogeneity (Manimaran et al., 2022). The PDI values of the produced ConA-CS NPs ranged from 0.1 to 0.4, indicating that monodispersed particles are present.
The external form of the generated ConA-CS NPs could be observed by SEM examination at a nanoscale level, and EDAX disclosed their elemental composition. SEM analysis confirmed bunches of spherical ConA-CS NPs formations that ranged in size from 50 to 60 nm. On the surface of ConA-CS NPs, the creation of intermolecular hydrogen bonds did not exhibit any appreciable shearing impact. Previous reports of comparable results may be found here (Moosazadeh Moghaddam et al., 2023).
CS NPs and concanavalin distinguish themselves out between important natural drug-depending nanomaterials because of their multiple therapeutic applications and their cancer prevention, antiviral, antimicrobial, and antifungal properties (Manimaran et al., 2022; Moosazadeh Moghaddam et al., 2023; Muller et al., 2019; Shi et al., 2014). To learn more about the potential for therapy of ConA-CS NPs, we looked at their anticancer properties.
In a prior work, it was examined how isolongifolene-loaded CS NPs affected the killing capacity of the particles against the apoptotic A549 cells (Manimaran et al., 2022). In this work, we examined the 24-hour cytotoxicity of ConA-CS NPs at NCI-H358 cell lines. The outcome demonstrated a 50% dose-dependent inhibition of cancer cell survival, and we also discovered that the 50% IC50 of ConA-CS NPs was 12.5 µM at all other doses.
Tumor formation requires the inhibition of apoptosis. The processes behind apoptosis-mediated death of cells, which have been connected to several human disorders, have drawn more attention from researchers lately (Chenyang et al., 2021). The apoptotic response of ConA, CS NPs, and a mixture of ConA-CS NPs against NCI-H358 lung tumor cells was examined using the AO/EtBr dye and a dual staining technique. Despite EtBr, which produces cell detritus stained with a reddish-orange hue, the AO stain may be permeable to living cells (green). The red intense fluorescence of the ConA-CS NPs treated cells was higher than that of the other kinds of treated cells, indicating that the natural drug carried ConA-CS NPs accelerated apoptosis in the cancerous lung cell line. According to Venkatesan et al. (2016), piperlongumine-loaded CS NPs significantly suppress gastric cancer cells by increasing intracellular ROS levels, which causes elevated cell apoptosis. Similarly, studies have demonstrated the effectiveness of isolongifolene-loaded CS NPs in preventing lung cancer cells.
To better understand the effects of ConA, CS NPs, and ConA-CS NPs combination on mitochondria, we assessed MMP using an indicator termed Rh-123 dye. We discovered that ConA-CS NPs treatment significantly reduced MMP in NCI-H358 cells. Consequently, it is being shown that CS NPs loaded with piperlongumine induce ROS-mediated mitochondrial damage, which results in apoptosis (Venkatesan et al., 2016).
Together with other indicators associated with apoptosis, mitochondria are the most significant organelles for regulating cellular death. Free radicals (ROS) are highly reactive molecules with detached electrons in their valence shells that greatly increase oxidative damage to cells (Chenyang et al., 2021). It is believed that raised ROS concentrations are the main factor causing cell death. Furthermore, one of the ways that ConA-CS NPs together have an anticancer effect is by the generation of ROS in cancer cells. The fluorescence indices ConA-CS NPs may be combined to determine the amounts of intracellular ROS. The ROS experiment’s findings indicate that NCI-H358 cells treated with ConA-CS NPs produce higher ROS in comparison to the control group. Lastly, as compared to ConA and CS NPs alone in the current investigation, the impacts of the ConA-CS NPs mixtures may explain the created ConA-CS NPs increased therapeutic efficacy.
Numerous treatments with chemotherapy and phytochemical-conjugated nanoparticles have been found to target the trigger of apoptosis and cell cycle arrest as viable targets for therapy (Chang et al., 2021; Singh Tuli et al., 2022). Using flow cytometry, the impact of ConA-CS NPs on the cell cycle phase was assessed. When cancer cells underwent treatment with ConA-CS NPs, the cells gathered in the G2/M phase of the cell cycle; however, when cells were treated with ConA alone, the cells stabilized in the S phase of the cell cycle. This result suggested that the G2/M phase cell cycle arrest of lung cancer cells was strongly triggered by the ConA-CS NPs. Our results were found to be in line with the earlier study that demonstrated how ConA acts by stopping the cell cycle in different cancers. Furthermore, ConA induces a temporary cell-cycle arrest by post-translational activation of p53, thereby inducing cellular DNA repair (Chang et al., 2021; Huldani et al., 2022). The results of this study demonstrated that ConA-CS NPs were highly effective at treating lung cancer cells.
Conclusion
Using ionic gelation, biodegradable CS NPs encasing ConA were created and then used in anticancer treatment. The effective preparation and characterization of ConA-CS NPs were accomplished for characteristics such as loading efficiency, zeta potential, and PDI. It was discovered that the ConA-CS NPs formulation was the best of the nanoformulations that were created. The formulation of ConA-CS NPs XRD spectrum attests to the nanoparticles’ weak crystallized character. SEM verified the spherical shape of ConA-CS NPs. ConA-CS NPs demonstrated dose-dependent, potent anticancer action towards NCI-H358 lung cancer cells. ConA-CS NPs, on the other hand, show extremely minimal cytotoxicity, indicating their exceptional biocompatibility. Additionally, it was shown that ConA-CS NPs dramatically raised the amount of intracellular ROS in NCI-H358 cancer cells, which raised the cell apoptotic threshold. These findings show that CS NPs are biocompatible DDSs with strong anticancer properties in lung cancer cells and effective ConA loading. Consequently, ConA-CS NPs show potential as a successful chemopreventive method for halting the development of lung cancer by causing cell death. In fact, with further research to establish controlled dosages, ConA-CS NPs therapy may be utilized in clinical care.
Abbreviations
NSCLC: Non-small-cell lung cancer; ConA: Concanavalin A; CS NPs: Chitosan nanoparticles; FT-IR: Fourier transform infrared; SEM: Scanning electron microscopy; XRD: X-ray diffraction; DLS: Dynamic light scattering; EDAX: Energy dispersive X-ray analysis; MTT: Mitochondrial toxicity test; DMSO: Dimethyl sulfoxide; NaTPP: Sodium tripolyphosphate; MMP: Mitochondrial membrane potential; PBS: Phosphate-buffered saline; ROS: Reactive oxygen species; PDI: Polydispersity index.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.