Abstract
Background
The manifestation of severe sepsis is sepsis-induced myocardial dysfunction (SIMD), which is the chief cause of elevated death in sepsis patients. Sepsis mainly attacks the heart, and the effective switch of the inflammatory cascades is of prodigious implication in relieving sepsis-allied myocardial impairments. Natural antioxidants with anti-inflammatory and antiapoptotic properties are promptly required as typical anti-septic agents.
Objectives
Morus alba root bark contains a flavonoid sanggenol L (SL), which exhibits antioxidant, apoptotic, and anti-inflammatory activities. Nevertheless, the anti-septic action of SL has not yet been described. Hence, we explored the antioxidative, antiapoptotic, and anti-inflammatory actions of SL on lipopolysaccharide (LPS)-triggered sepsis in rats.
Materials and Methods
Rats were divided into four sets: group 1: control, group 2: LPS (20 mg/kg bw), group 3: LPS (20 mg/kg bw) + dexamethasone (2 mg/kg bw), and group 4: LPS (20 mg/kg bw) + SL (10 mg/kg bw). We assessed cardiotoxicity, antioxidants, cytokines, histopathology, protein expressions of apoptosis, and inflammatory enzymes.
Results
Data unveiled that SL inhibited toxicity markers, oxidative stress, inflammation, apoptosis, and histological changes. SL effectively attenuated LPS-induced acute myocardial injury by stimulating the PI3K/AKT/mTOR signaling and suppressing the nuclear factor kappa B (NF-κB) pathway.
Conclusion
Thus, SL might be a possible anti-septic agent for the treatment of SIMD.
Introduction
Sepsis, a systemic inflammatory response syndrome (SIRS), is generally produced by a dysregulated host immune response to the infection (Liang et al., 2021). Sepsis is a complex disorder with multiorgan dysfunction, particularly cardiovascular disease (CVD). The rate of sepsis mortality with myocardial dysfunction is 70–90%, related to 20% in sepsis patients deprived of myocardial injury (Song et al., 2021). Sepsis-induced myocardial dysfunction (SIMD) commonly occurs in patients with severe forms of sepsis. Lipopolysaccharide (LPS) is the foremost external membrane constituent of gram-negative bacteria and has been experimentally used to create cardiomyocyte injury (Jiang et al., 2016; L’Heureux et al., 2020). SIMD has been allied with the extreme generation of cytokines comprising interleukin-6 (IL-6), interleukin-1β, and tumor necrosis factor-α (TNF-α), which affect myocyte death and impairment (Sun et al., 2018; Zhao et al., 2020). Furthermore, preceding reports established that SIMD has latent mechanisms consisting of inflammatory intermediaries, structural modifications, cardiomyocyte contractility defect, mitochondrial dysfunction, declined energy metabolism, and cell demise (Bayeva et al., 2014; Wang & Zhou, 2020; Zhu et al., 2021). An extreme reactive oxygen species (ROS) formation at inflammatory locations contributes to oxidative stress (OS), which is in sequence to prompt destruction in the mitochondria (Chang et al., 2021). The bacterial toxins and inflammatory intermediaries produced in response exert a tranquilizing influence on the myocardium, producing reversible or irreversible harm to cardiomyocytes by apoptosis (Antonucci et al., 2014); a key mechanism that involves septic cardiac damage (Avlas et al., 2011). Thus, regulating extreme inflammatory response, OS, and subduing apoptosis in the myocardium may protect against SIMD. Therefore, the development of an innovative antioxidative agent with anti-inflammatory and antiapoptotic activity is required for the treatment of SIMD.
Sanggenol L (SL) is a well-known flavonoid component that is isolated from the root bark of the Morus alba (mulberry). It has been shown to have therapeutic benefits, including anti-inflammatory and anticancer properties (Nam et al., 2016; Qin et al., 2015). By activating caspases and suppressing nuclear factor kappa B (NF-κB), SL triggers antiproliferative and apoptotic activities in ovarian cancer cells (Nam et al., 2016). It has been documented that SL triggers apoptosis in both caspase-dependent and caspase-independent melanoma cells (Won & Seo, 2020a). Formerly, it has been explored that the protective effect of SL prompts apoptosis that constrains inflammatory and cell proliferative signaling pathways in 7,12-dimethylbenz[a]anthracene (DMBA)-induced hamster buccal pouch carcinogenesis (Fu et al., 2023). An earlier work recognized that SL has lessened inflammation and ankle joint mutilation in type II collagen-prompted arthritis in investigational rats by the suppression of the PI3K/AKT signaling (Sun et al., 2023). A moment ago, SL caused cell cycle block and apoptosis by the initiation of p53 and suppression of PI3K/AKT/mTOR pathways in prostate cancer (Won & Seo, 2020). However, the antioxidative, antiapoptotic, and anti-inflammatory action of SL in LPS-prompted sepsis has not yet been examined. This current research is the first to scrutinize how SL can reduce inflammation, cardiotoxicity markers, OS, and apoptosis in the LPS-stimulated septic model.
A typical cell signaling pathway PI3K/AKT/mTOR is correlated to apoptosis (Jiang et al., 2013). In recent times, several reports have established that PI3K/AKT/mTOR signaling not only averts apoptosis but also involves suppressing inflammatory reactions and alleviating sepsis-related mortality (Jiang et al., 2013; Liu et al., 2015; Zhang et al., 2007). The PI3K triggers the AKT, as a downstream signaling molecule that has a significant deed in an array of biological processes comprising cellular proliferation, inflammation, and apoptosis, which are comprised of cardiac illnesses (Franceschelli et al., 2017). NF-κB is a vital nuclear transcription element and switches numerous inflammatory gene expressions. Prior documents described that NF-κB exerts a crucial deed in the SIMD pathogenesis (Alvarez et al., 2016). Thus, the possible cross-regulation between the PI3K/AKT/mTOR and NF-κB p65 signaling pathways offers an innovative direction to study the pathogenesis of sepsis. Hence, in this study, the anti-septic role of SL on PI3K/AKT/mTOR and NF-κB p65 signaling pathway in the LPS-prompted rat model was investigated through cardiac markers, antioxidants, inflammation, and apoptosis.
Materials and Methods
Reagents
LPS, dimethyl sulfoxide (DMSO), dexamethasone (Dex), and SL were acquired from Ruicong Ltd. (Shanghai, China). Biochemical kits were purchased from Elabscience (Wuhan, China). Enzyme-linked immunosorbent assay (ELISA) kits were obtained from Nanjing KeyGENBioTECH. Co., Ltd. (Nanjing, China). Western blot antibodies were attained from Ese-Bio (Shanghai, China).
Test Animals
Sprague-Dawley male rats (220–250 g) were achieved in the university animal store and sustained at regular 12-hour light/dark cycle in germ-free polypropylene cages in secure laboratory conditions. Animals were fed with a consistent pellet diet with drinking water. They were adapted for 1 week before investigations.
Trialplan
All rats were arbitrarily set apart into four arrays consisting of ten each. Group 1: control, group 2: LPS (20 mg/kg bw), group 3: LPS (20 mg/kg bw) + Dex (2 mg/kg bw in DMSO), group 4: LPS (20 mg/kg bw) + SL (10 mg/kg bw in DMSO). LPS induction and treatments follow according to the previous method (Shang et al., 2019). Subsequently, in the trial, entire rats were sedated and sacrificed by cervical dislocation.
Preparation of the Sample
Blood was collected from rats through cardiac rupture and underwent 4,000 rpm centrifugation. The serum was alienated and conserved at –80°C for cytokines estimation. The residual serums were recollected at 4°C to determine cardiac toxicity enzymes. The heart was dissected and segregated into dual portions. The single part was sustained at –80°C for anti-inflammatory, antioxidant, and cardiotoxicity assessment. The residual fragment of the tissues was shifted to formalin (10%) for the valuation of histopathology.
Assay of Cardiotoxicity Markers and Antioxidant Enzymes
The levels of cardiotoxicity marker proteins such as creatinine kinase (CK), lactate dehydrogenase (LDH), and the antioxidant enzymes activities of catalase (CAT), glutathione (GSH), superoxide dismutase (SOD), and glutathione peroxidase (GSH-px) were estimated as per the kit purchased from Nanjing Jiancheng Bioengineering Institute (Jiangsu, China).
Histopathological Examination
Heart tissues were immediately excised and conserved with formaldehyde (10%), secured in paraffin blocks, and cut into pieces, then hematoxylin and eosin staining. Sequentially, stained tissues were recognized for histopathological damage and swelling under the light microscope (Nikon, Tokyo, Japan).
Determination of Cytokines
IL-6 and TNF-α cytokines levels in serum and tissue supernatant levels were measured by ELISA affording to the company’s directions. All estimations were achieved thrice.
Western Blotting Analysis
Concisely, the cardiac tissue was dissected and homogenized. These were washed with phosphate-buffered saline (PBS) and lysed by the cold lysis buffer, then sustained on ice for 30 minutes. Total protein was assessed by the procedure of the BCA Test Protein Kit (Elabscience, Wuhan, China). The lysate was cold (4°C) and centrifuged for 10 minutes at 13,000 rpm and the resulting solution was distributed through SDS-PAGE (10%) and shifted electrophoretically to the PVDF film. At that moment, 5% of fat-less milk was working to block the membrane, then preserved with their exact primary antibodies, later adding horseradish peroxidase-conjugated secondary antibodies. Protein bands were quantified using densitometry and homogeneous to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) expression using the software ImageJ.
Statistical Study
The statistics analysis was accomplished by GraphPad Prism software version 9.0 and expressed as mean ± SEM. It was accomplished by an analysis of variance (ANOVA) and subsequently Duncan’s assessment. p < 0.05 was reflected as significant statistically.
Results
SL Alleviates Cardiotoxicity Enzyme Markers in Serum and Cardiocyte Isolates
To identify myocardial damage, the toxicity marker enzymes such as CK-MB in serum and LDH in serum and cellular supernatant were determined. The levels of serum CK and LDH in serum and cardiocyte supernatant were intensely elevated (p < 0.05) after exposure to LPS. Administration of SL efficiently reduced these cardiotoxicity markers in serum and cardiac tissue extracts. These results revealed that SL could attenuate LPS-induced cardiac injury (Figure 1).
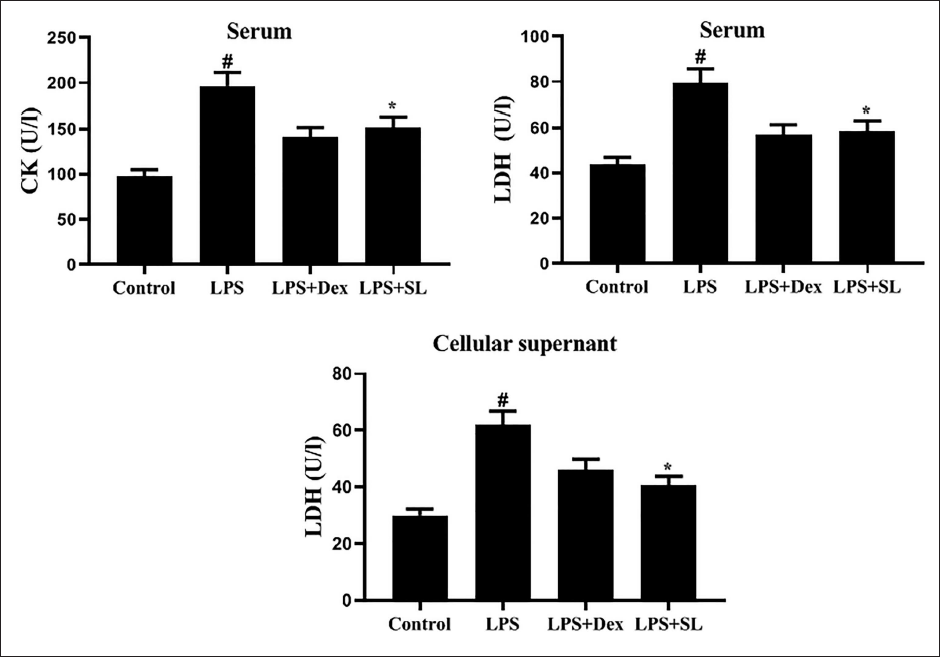
SL Enhances the Antioxidant Enzyme Status in Serum and Cardiocyte Isolates
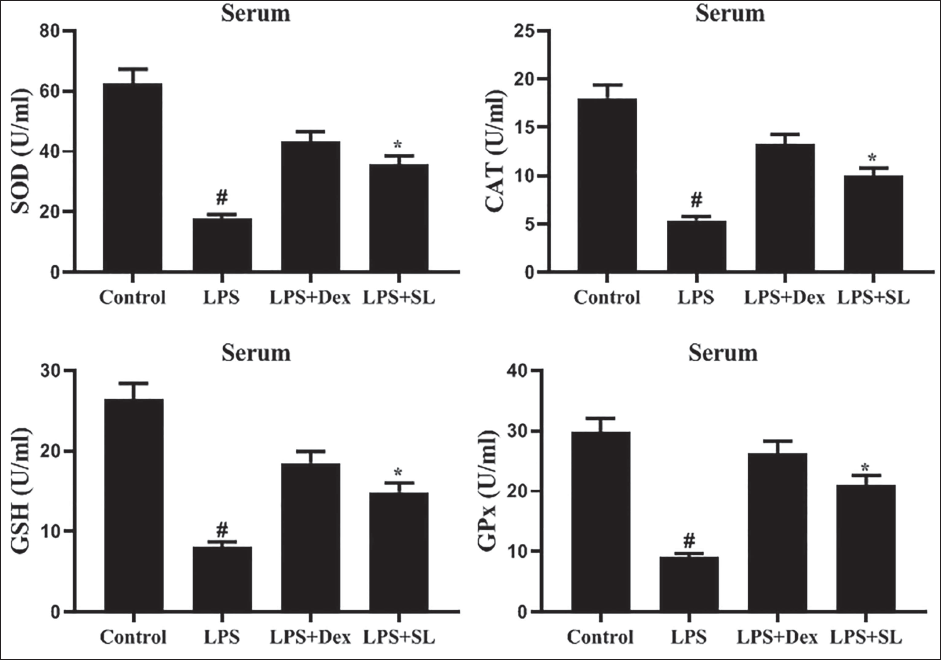
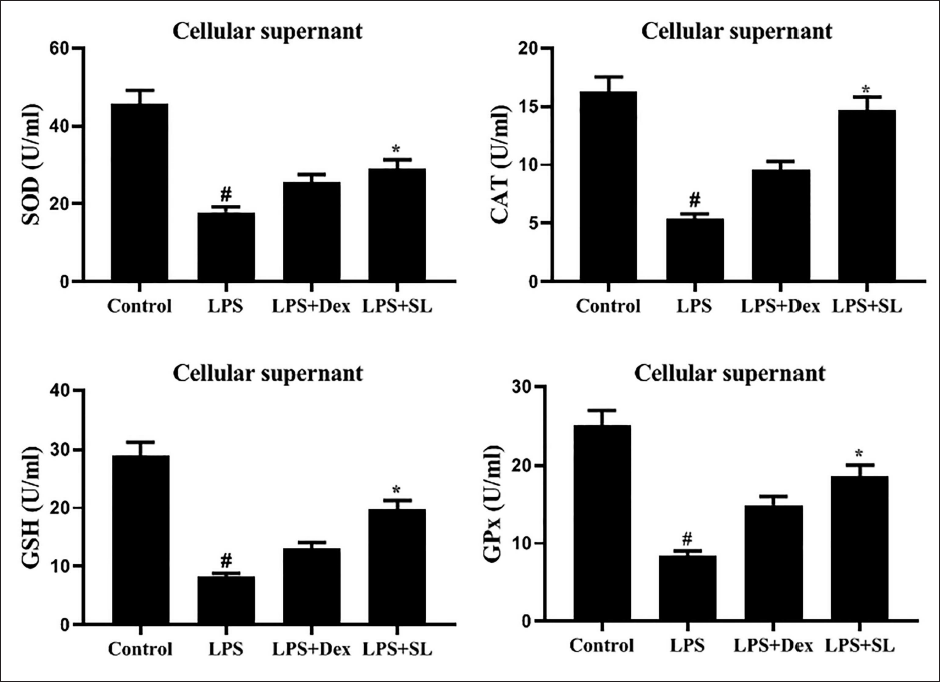
LPS-triggered lipid peroxidation in serum and cardiocyte extracts was estimated by the antioxidant activity of CAT, SOD, GSH, and GSH-px (Figures 2 and 3). Administration of LPS considerably dropped (p < 0.05) these levels of antioxidants status, whereas SL could restore CAT, SOD, GSH, and GSH-px levels in serum and cardiocytes isolates. These investigative findings recommended that SL was proficient in reducing LPS-triggered OS.
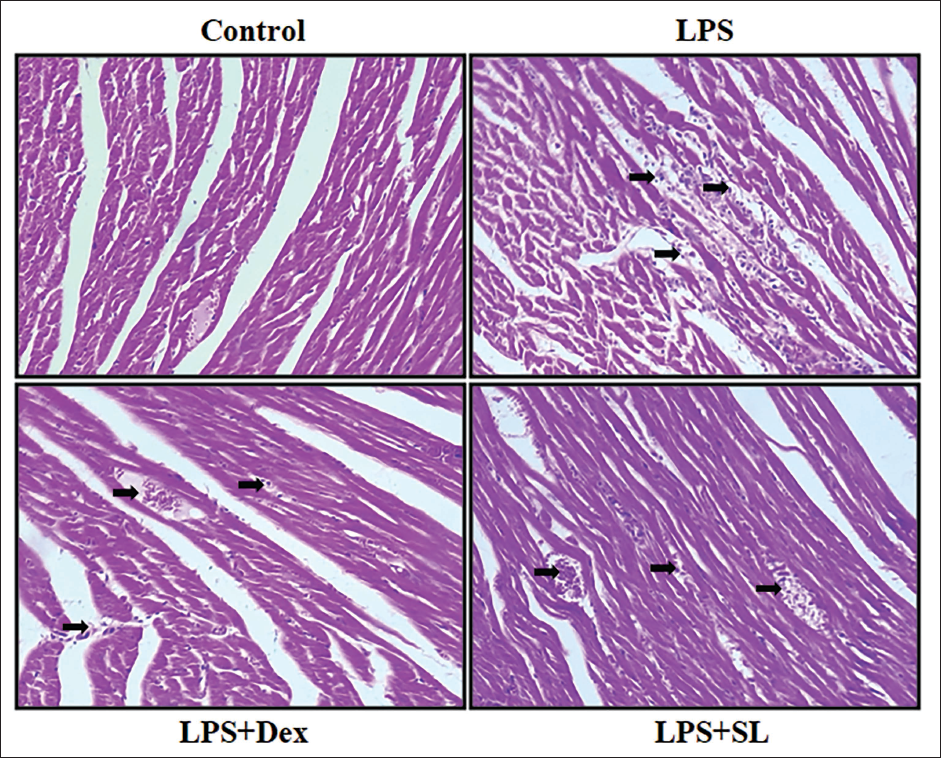
SL Reduces Histopathological Changes in Heart Tissues
Figure 4 depicts the histopathological examination of the cardiac tissue in control and experimental animals. LPS-stimulated rats showed cardiomyocyte necrosis and neutrophil infiltration. SL treatment decreased the myocardium invasion of neutrophil granulocytes and LPS-induced necrosis.
Sanggenol L (SL) Reduces Histopathological Variations of Myocardial Tissue in the Lipopolysaccharide (LPS)-stimulated Septic Rat Model. Scale Bar = 50 µm.
SL Attenuates Inducible Nitric Oxide Synthase (iNOS), Cyclooxygenase-2 (COX-2), and NF-κB p65 Protein Expression in Cardiac Tissues
To examine the possible mechanism of SL on LPS-stimulated myocardial damage, we inspected the protein levels of COX-2, iNOS, and NF-κB p65 in cardiac tissues (Figure 5). LPS-exposed rats unveil elevated protein levels of COX-2, iNOS, p-NF-κB p65, and NF-κB p65. However, SL intensely suppressed the level of iNOS, COX-2, and NF-κB.
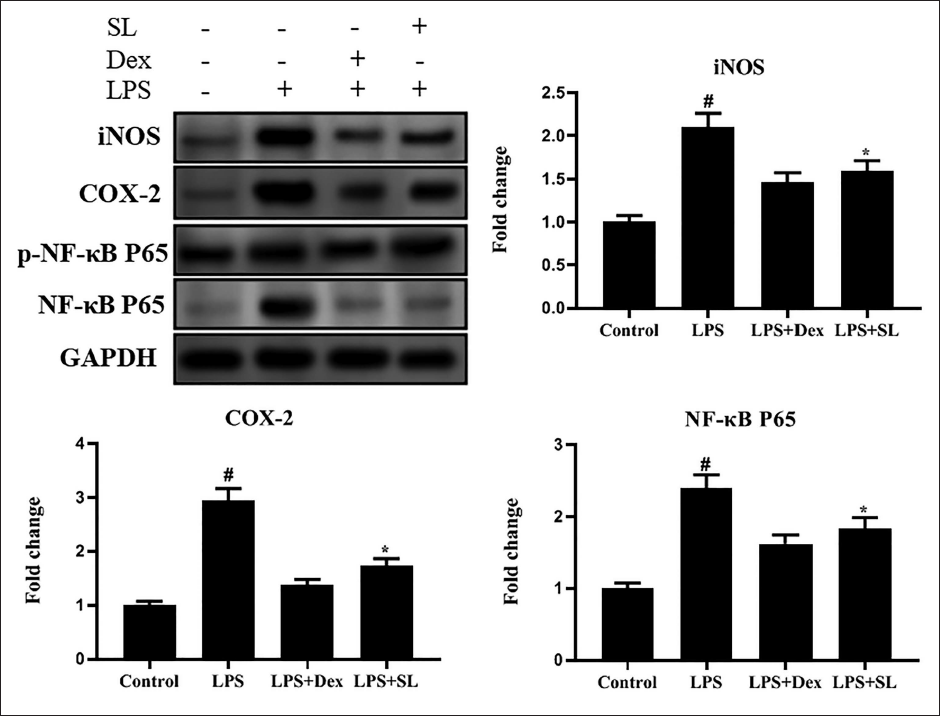
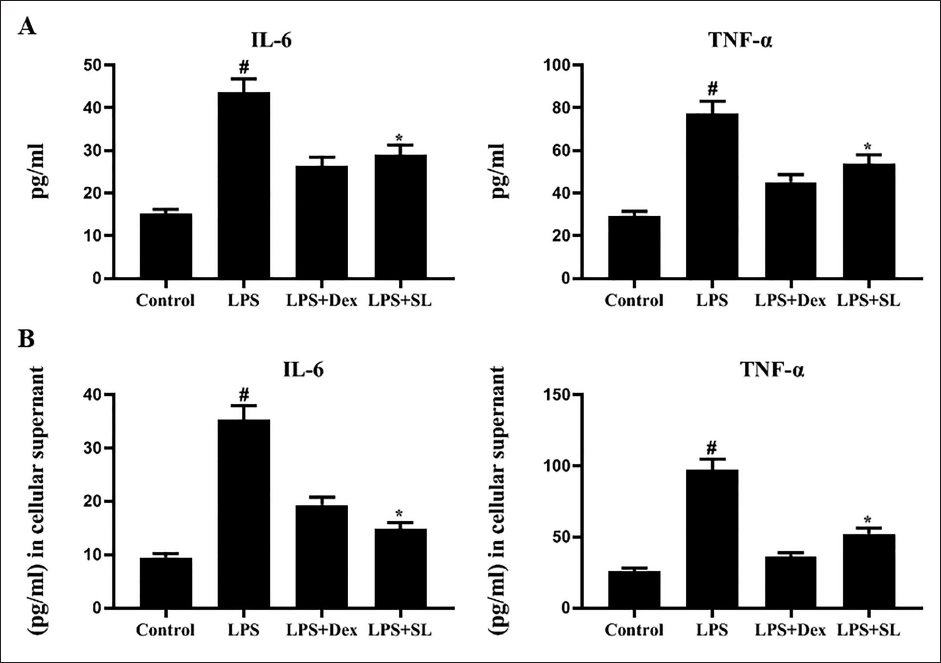
SL Mitigates Inflammatory Cytokines in Serum and Cardiocytes Isolates
LPS-stimulated rats exposed the elevated levels of cytokines such as IL-6 and TNF-α in serum and cardiocyte extracts against control (Figure 6). Treatment with SL substantially repressed these cytokines levels in both serum and extracts. This established that SL could reduce LPS-induced inflammation in the heart.
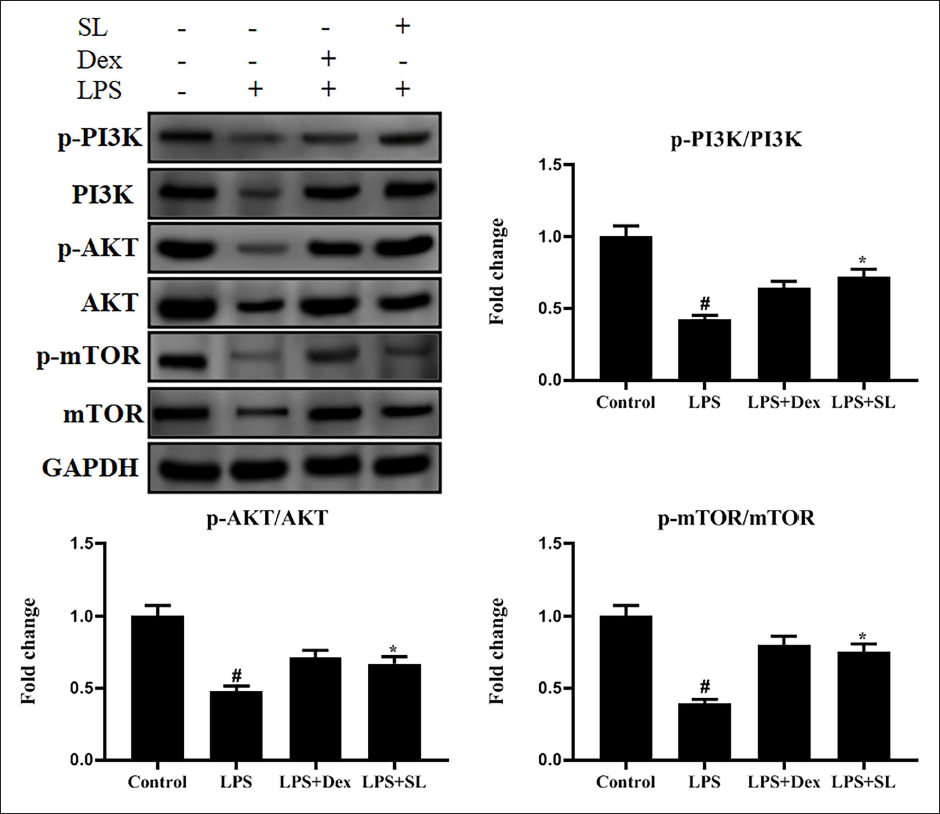
SL Activates PI3K/AKT/mTOR Signaling
Western blot examination exposed that phosphorylated p-PI3K, PI3K, p-AKT, Akt, p-mTOR, and mTOR protein expression levels were inhibited in the sepsis-prompted rats (Figure 7). Conversely, SL administration stimulated the level of these protein expressions.
Discussion
Sepsis is produced by a defective response to host contamination, producing life-hostile organ dysfunction (Liang et al., 2021). In sepsis, numerous factors comprising eicosanoids, cytokines, ROS, and nitrogen species enhance the progression of the symptoms that affect SIRS, which eventually forms multiple organ failure (Chang et al., 2021; Sun et al., 2018; Zhao et al., 2020). Reports suggest that the most susceptible target organ in sepsis is the heart (Jiang et al., 2016; Sun et al., 2018). Former clinical works have established that early sepsis is related to a great occurrence of organic impairment in the myocardium, and these patients frequently have hypotension, heart failure, and arrhythmia (Antonucci et al., 2024; Song et al., 2021). Simultaneously, myocardial inhibition is among the chief signs of a deprived diagnosis in sepsis patients. At present, there is no precise cure for SIMD. In the current exploration, we establish the anti-septic activity of SL, which is a renowned flavonoid isolated from M. alba. Previously, SL has been recognized for its anti-inflammatory (Qin et al., 2015), anticancer (Nam et al., 2016), and neuroprotective (Jung et al., 2015) properties. This study may be the first document to verify that SL could moderate inflammation, cardiotoxicity markers, OS, and apoptosis in the LPS-induced septic model.
Sepsis is a severe clinical issue, and its meager scenario is owing to a SIRS, which affects cardiac dysfunction (Liang et al., 2021; Song et al., 2021). As an endotoxin in gram-negative bacteria, LPS-triggered secretion of pro-inflammatory elements, OS, and cardiac apoptosis. Thus, LPS has been extensively used in the establishment of sepsis in vivo models of induced cardiomyopathy (Jiang et al., 2016; L’Heureux et al., 2020). Outcomes in these experiments also displayed that LPS-prompted cardiac damage with augmentation of cardiotoxic biomarkers, containing CK in serum and LDH in serum as well as cardiocyte isolates. Furthermore, OS, inflammation, and cell apoptosis in cardiac tissues were also promoted by LPS. In this existing research, supplementation of SL attenuated OS through elevated antioxidant enzyme activity comprising SOD, CAT, GSH, and GSH-px, while diminished inflammation and apoptosis both in the serum and heart tissue of LPS-prompted rats. In addition, SL reduced the enzymatic action of CK, LDH, inflammatory enzymes, and cytokines comprising IL-6 and TNF-α in the LPS model, representing a protective influence against organ harm by inducing caspases and suppressing NF-κB, SL promotes antiproliferative and apoptosis regulation in ovarian cancer cells (Nam et al., 2016). Previously, it has been reported that the protective effect of SL inhibits inflammation in DMBA-prompted buccal pouch carcinogenesis in the hamster model (Fu et al., 2023). These results suggest that mechanisms accountable for the organ-defensive deeds of SL may implicate the alleviation of OS, inflammation, and apoptosis.
Inflammation is a central tool of myocardial hurt in sepsis that may facilitate OS and apoptotic cell death. OS and inflammation are interdependent, which is reflected as a key aspect of CVD pathogenesis (Wang & Zhou, 2020; Zhu et al., 2021). ROS and NOS stimulate microvascular coronary injury, which originates anomalous apoptosis and inflammation (Mittal et al., 2021; Zhu et al., 2021). Several reports documented that naturally originated antioxidants guard endothelial and myocardial cells against OS (Chang et al., 2020; Chang et al., 2021). In this investigation, we tested the expression of inflammatory enzymes and it unveiled an enhanced expression of COX-2, iNOS, and NF-κB in the LPS-alone group; however, SL administration averted these modifications. A prior study presented that Puerarin, as a natural antioxidant, could control immune reactions and OS harm prompted by sepsis (Chang et al., 2021). These analytical upshots emphasized that the cardioprotective action of SL might be ascribed to its antioxidative capability. COX-2 and iNOS are fundamental enzymes involved in the development of inflammatory and oxidative dysfunction (You et al., 2013). A crucial transcriptional factor NF-κB moderates inflammatory intermediaries and influences iNOS and COX-2 expressions (Lou et al., 2015). Emerging data have signified that the COX-2 and iNOS which are controlled by NF-κB are closely allied with the LPS-stimulated SIMD (31). Our investigational findings mentioned that SL efficiently subdued COX-2, iNOS, and p-NF-κB deeds in LPS-prompted myocardial harm. Formerly, it has been renowned that SL has diminished inflammation and ankle joint injury in type-II collagen-stimulated arthritis in investigational rats by the repression of the inflammatory cytokines and inflammatory enzymes including iNOS, COX-2, and NF-κB p65 (Sun et al., 2023). Throughout sepsis, elevated intensities of endotoxins trigger immune cells, which in sequence, stimulate the generation of inflammation intermediaries and cytokines (Zhu et al., 2021). IL-6 and TNF-α cytokines are expressed in sepsis, which are accountable for cardiac impairment (Zhao et al., 2020). These findings suggested the anti-inflammatory and antiapoptotic influence of SL on SIMD.
Apoptosis, a kind of involuntary cell demise, shows a crucial action in sepsis-stimulated multiorgan disease (Jiang et al., 2013). Numerous documents established that suppression of apoptosis in the myocardium is correlated to the upgrading of cardiac tasks in sepsis experimental models (Franceschelli et al., 2017). Here, SL exerts antiapoptotic action which suppresses the protein expression of PI3K/AKT/mTOR signaling. On the contrary, reports available previously on cancer cells that SL stimulates apoptosis that inhibits inflammatory and cell proliferative signaling pathways in DMBA-prompted hamster buccal pouch carcinogenesis (Fu et al., 2023). An earlier work identified that SL has declined inflammation and ankle joint damage in type-II collagen-prompted arthritis in trial rats by the subdual of the PI3K/AKT signaling (Sun et al., 2023). Recently, SL caused cell cycle block and apoptosis by the instigation of p53 and repression of PI3K/AKT/mTOR pathways in prostate cancer (Won and Seo, 2020b). In the present study, induction of LPS used to form an SIMD model, which assesses the influence of SL on the PI3K/AKT/mTOR and NF-κB networks in rat models. Outcomes revealed that SL enhanced cardiac function and antioxidant enzyme status, while alleviated cardiotoxicity enzymes, inflammatory enzymes, cytokines, and apoptosis. Our data suggest that SL effectively attenuated LPS-induced acute myocardial injury by stimulating the PI3K/AKT/mTOR singling and averting the NF-κB inflammatory pathway.
Conclusion
Collectively, SL mitigated LPS-triggered cardiac injury, inflammation, cytokines, OS, and apoptosis through its antioxidant activity by triggering the PI3K/AKT/mTOR signaling and repressing the inflammatory NF-κB pathway. Therefore, SL might be a possible anti-septic agent for the treatment of SIMD. Further clinical studies are required.
Summary
SL inhibited toxicity markers, OS, inflammation, apoptosis, and histological changes in experimental rats.
SL effectively attenuated LPS-induced acute myocardial injury rats.
Abbreviations
SL: Sanggenol L; SIMD: Sepsis-induced myocardial dysfunction; LPS: Lipopolysaccharide; Dex: Dexamethasone; NF-κB: Nuclear factor kappa B; SIRS: Systemic inflammatory response syndrome; CVD: Cardiovascular disease; TNF-α: Tumor necrosis factor-α; ROS: Reactive oxygen species; OS: Oxidative stress; DMBA: 7,12-Dimethylbenz[a]anthracene; CK: Creatinine kinase; CAT: Catalase; GSH: Glutathione; SOD: Superoxide dismutase; GSH-Px: Glutathione peroxidase; ANOVA: Analysis of variance; COX-2: Cyclooxygenase-2.
Data Availability
All data generated or analyzed during this study were included in this manuscript. Further inquiries can be directed to the corresponding author.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The animals were maintained as per the rules of the Medical Ethics Committee of Gongli Hospital in Pudong New Area, Shanghai (No.: JK2020040).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
