Abstract
Background
The predisposition toward gastrointestinal disease is increasing as a result of dietary changes and overindulgence in food preparation. Ninety percent of stomach cancers are adenocarcinomas, a type of cancer of the gastrointestinal tract that is the leading cause of mortality worldwide.
Purpose
Astilbin, a natural dihydroflavonol substance derived from a range of foods and medicinal herbs, has been shown in the current study to have anticancer potential against gastric cancer in AGS cell lines.
Materials and Methods
The half maximal inhibitory concentration value was determined after astilbin was given to AGS cell lines at various doses. To verify apoptosis, the astilbin-treated cell line was subsequently stained with the dual dye acridine orange/ethidium bromide. AGS cell lines, the enzyme-linked immunosorbent assay (ELISA) approach was also used to investigate the inflammatory cytokines. Using ELISA, apoptotic marker expression including Bcl-2-associated X-protein, caspase-9 proteins, and B-cell lymphoma 2 was assessed in gastric cancer cells treated with astilbin.
Results
The study’s findings reveal that astilbin is a prime candidate for gastric cancer therapy since it causes apoptosis in AGS cells and has a stronger effect on the major inflammatory markers of the disease.
Conclusion
The study provides comprehensive methodologies and tools for predicting absorption, distribution, metabolism, excretion, and toxicity properties, facilitating informed decision-making in the development and optimization of astilbin as a gastric cancer therapy.
Introduction
Cancer, with its ever-increasing incidence and death, endangers human health and quality of life globally. By 2030, it is predicted that there will be 20.3 million additional cases and 13.2 million deaths from cancer (She et al., 2015). Gastric cancer is a severe health concern as it is the fourth most prevalent cancer and the second most common cause of fatalities due to cancer globally. Anatomically, there are two types of stomach cancer: gastroesophageal junction adenocarcinomas and true gastric adenocarcinomas, or noncardia gastric malignancies (Ferlay et al., 2013). The multistep process of carcinogenesis consists of three stages: progression, promotion, and initiation. Several molecules that promote inflammation, such as interleukin-1 (IL-1), interleukin-6 (IL-6), and tumor necrosis factor (TNF), trigger transcription factors, such as activator protein 1 (AP-1) and nuclear factor-κB (NF-κB), and early response genes by the activation of intracellular kinases, in addition to the initial damage to deoxyribonucleic acid (DNA). This function is comparable to that of the traditional chemical tumor promoters, which stimulate cell signal transduction to increase cell growth and proliferation (Sokolova & Naumann, 2017).
Epstein–Barr virus is another infection linked to gastric cancer. Only the malignant cells, not the normal epithelial cells, contain this pathogen in 80% of gastric carcinomas with lymphoid stroma. Its function in the development of cancer, however, is still unknown (Wang et al., 1999; Wu et al., 2000). The majority of familial cases of gastric cancer in nations and locations with low prevalence are most likely caused by heritable pathogenic genes that raise danger arising from birth (Oliveira et al., 2015). Oncogenesis linked to inflammation may eventually result from modified cell proliferation, programmed cell death, and specific epigenetic modifications to the genes that restrict tumor growth (Wang et al., 2017). Apoptosis is a genetically driven kind of self-regulated cell death that is profoundly associated with the proliferation of cancer cells and has been demonstrated to be a useful target for cancer treatment (Liu et al., 2023). Extracellular and intracellular signals trigger cell apoptosis through two primary mechanisms: death receptor-mediated pathways and mitochondrial apoptosis (Sun et al., 2018). Activation of intrinsic or extrinsic apoptotic pathways is one method by which anticancer medications might destroy cells (Teng et al., 2016).
Numerous studies conducted recently have demonstrated that anticancer drugs or chemopreventive therapies for cancer work by inducing apoptosis to stop the growth and spread of tumors as well as the emergence of cellular responses to inflammation other than necrosis. A common mechanism of many cytotoxic drugs used in chemotherapy appears to be the induction of apoptosis. Thus, it is anticipated that medicines that induce apoptosis will be the best anticancer medications. Furthermore, by inducing apoptosis, many flavonoids, including apigenin, phloretin, and quercetin, prevent the proliferation of cancer cells. Thus, knowing how apoptosis works has a significant impact on treating and preventing a wide range of illnesses (Shih et al., 2005).
Numerous studies have demonstrated the connection between long-term inflammation and the possibility of gastric cancer. It is thought that a series of cellular pathways leading to malignant epithelial changes can be set off by a chronic inflammatory response. Through its ability to modulate the expression of several target genes, the transcription factor NF-κB commonly referred to as NF-κB has been recognized as an important modulator of immunological and inflammatory responses as well as the apoptotic pathway (Roozbehani et al., 2021). Various protein families govern apoptosis. These include the downstream caspase family, which includes caspase-9 and 3, and the upstream B-cell lymphoma 2 (Bcl-2) family, which includes proapoptotic Bcl-2-associated X-protein (Bax) and antiapoptotic Bcl-2 (Günes-Bayir et al., 2017). Bcl-2 is categorized as an oncogene since it is thought to be a significant antiapoptotic protein. By blocking Bcl-2’s inhibitory effects on apoptosis, medicines can target malignant cells and cause them to undergo apoptosis. Utilizing the caspase enzyme system, several factors that modulate cell apoptosis work (Sokolova & Naumann, 2017).
Surgical excision combined with further adjuvant chemotherapy, radiotherapy, or both have been proposed as ways to extend the survival time of gastric cancer-affected patients by up to 5 years. Chemotherapy, however, has the potential to cause severe adverse effects, increased medication resistance, and treatment failure (Pei et al., 2021). Cisplatin, known as cisplatinum or cis-diamminedichloroplatinum (II), is a renowned chemotherapy drug. It is effective against sarcomas, germ cell tumors, carcinomas, and lymphomas. Its mechanism of action has been linked to its ability to bind with purine bases on DNA, which damages DNA, obstructs repair mechanisms of DNA, and ultimately triggers cancer cells to undergo apoptosis. However, due to drug resistance and several undesirable side effects, including severe kidney problems, decreased immunity to infections, gastrointestinal disorders, and hearing loss, especially in younger patients caused by the consumption of cisplatin, other platinum-containing anticancer medications, such as carboplatin, have also been used. Moreover, cisplatin combination treatments with other medications have drawn a lot of interest as a novel approach to combating medication resistance and minimizing toxicity (Dasari & Tchounwou, 2014).
Combining S-1 and cisplatin as first- or second-line treatment options as chemotherapy for severe and recurrent gastric cancer has shown promise in certain trials, and it is also well tolerated. However, the effectiveness of cisplatin and patient reaction to cisplatin-containing anticancer drugs vary, which can have an impact on treatment results. As a result, there is considerable interest in research on molecules and factors that can predict resistance and patient responses to anticancer medication (Tanida et al., 2012). Thus, in the fight against cancer, both therapeutic and preventative approaches are essential. As a result, there has been a recent surge of interest in certain compounds present naturally, especially in natural food items and phytochemicals for gastric cancer therapy, owing to their low toxicity and better tolerance rate (Pei et al., 2021).
Plant-based bioactive phytochemicals have the potential to treat gastric cancer patients clinically due to their high efficiency and low toxicity, which provides hope for the development of new anticancer medications. As a result, the current study investigated whether astilbin may have any effect on gastric cancer cells and explored its anticarcinogenic and antiproliferative properties to provide light on the molecular mechanisms involved. A dihydroflavonol rhamnoside is astilbin present in many different plants, including Hypericum perforatum, genus Smilax, Drimys brasiliensis, grapevine, and Desmos chinensis. Astilbin-containing plants and herbs have long been used in China and some other Asian nations to treat a range of illnesses, including immunosuppression, hyperuricemia, and chronic renal failure (Sharma et al., 2020). Astilbin has been shown to have antitumor capabilities, although little is known about its proapoptotic and anticancer effects on gastric cancer.
Gastric cancer is the fifth most common cancer worldwide and the third leading cause of cancer-related deaths. Factors contributing to its incidence include age, gender, ethnicity, dietary habits, smoking, and genetic predisposition (Rawla & Barsouk, 2019). Gastric cancer has a high mortality rate due to late diagnosis and limited treatment options for advanced stages. The 5-year survival rate for gastric cancer is relatively low compared to other cancers, especially when diagnosed at advanced stages (Shin et al., 2023).
Apoptosis, or programmed cell death, plays a crucial role in cancer development and treatment resistance. Bcl-2 family proteins are key regulators of apoptosis, with antiapoptotic proteins like Bcl-2 promoting cancer cell survival and resistance to therapy (Qian et al., 2022). Current therapies targeting Bcl-2 and other apoptotic pathways are being developed to overcome treatment resistance and improve patient outcomes. Examples include venetoclax, a Bcl-2 inhibitor approved for certain hematologic malignancies, and other targeted therapies like HER2-targeted therapies and immune checkpoint inhibitors.
Adverse effects of targeted therapies may include gastrointestinal disturbances, hematological toxicities, fatigue, and potential cardiotoxicity (Du et al., 2021). Natural compounds derived from plants, herbs, and other sources have gained attention for their potential anticancer properties, including anti-inflammatory, antioxidant, and proapoptotic effects (Isabel et al., 2022). Natural compounds offer the advantage of being generally well-tolerated with fewer adverse effects compared to conventional therapies, complementing existing treatment modalities, and being cost-effective adjuncts to standard cancer treatment regimens (Banerjee et al., 2023; Dehelean et al., 2021).
Human gastric epithelial adenocarcinoma cell line AGS has been extensively utilized as an exemplary system to assess apoptosis in cancer cells and has been demonstrated to be a good model system for other research concerning the diverse responses to anticancer medicines (Shih et al., 2005). The current study’s major goal is to investigate the anticancer activity of the flavonoid compound astilbin against AGS cell lines. Astilbin’s impact on cell viability as measured by the assay using water-soluble tetrazolium salt (WST-1) reagent, acridine orange (AO)/ethidium bromide (EtBr) staining for assessing apoptosis-inducing potential, TNF-alpha, NF-κB, and IL-6 expression assessments, and the apoptotic protein levels of caspase-9, Bcl-2, and Bax as measured by the enzyme-linked immunosorbent assay (ELISA) technique have all been studied.
Materials and Methods
Materials
A sample of astilbin was purchased from Sigma-Aldrich (USA). The investigations employed only analytical-grade chemicals and reagents. A human cell line for gastric cancer called AGS cells was cultivated and kept in their optimal conditions.
Application of the WST-1 Assay to Assess Cell Proliferation
AGS cells were added equally in 96-well plates using the culture medium for cells and incubated for 24 hours. Further, the cells were subjected to increasing concentrations of astilbin 1.25–200 µg/ml in triplicates. After 24 hours of adding 10 µl of WST-1 reagent with medium to each well, the plate was incubated at 37°C and 5% carbon dioxide (CO2) for 2–3 hours. At 450 nm, the absorbance of every well was measured using the Microplate Reader (Saamarthy et al., 2024).
Apoptosis Identification Through Dual AO/EtBr Staining
The AO/EtBr staining method was used to examine and compare astilbin’s potential to trigger apoptosis in AGS cells compared to that of the control. The cells received astilbin at a half maximal inhibitory concentration (IC50) dose, cisplatin and control incubated for 24 hours. Following incubation, the treated cells were stained with EtBr and AO in equal parts and incubated in the dark for 5 minutes. Following a phosphate-buffered saline (PBS) wash, the fluorescence microscope was used to examine the cells. The experiment was repeated three times (Wang et al., 2023).
Study of Astilbin’s Impact on Inflammatory Cytokines, Such As TNF-α, NF-κB, and IL-6, Using AGS Cell Lines
Evaluate the secretion of TNF-α, IL-6, and NF-κB from AGS cells. Cell lysates from control, positive control, and astilbin-treated cells were prepared, and they were centrifuged at a predetermined speed in a predetermined environment. The resulting supernatant was used to measure the levels of TNF-α, IL-6, and NF-κB according to the directions provided by the manufacturer for the ELISA kit. Triplicate runs of the experiment were carried out (Wang et al., 2023).
Assessment of Apoptotic Markers, Such As Bcl-2, Bax, and Caspase-9 Activities
Astilbin was introduced to AGS cells, and they were cultured for 24 hours and compared with cisplatin (positive control) and control. Following cell harvesting, the protein levels were estimated using an ELISA kit purchased from a commercial source, following the manufacturer’s instructions. Triplicates of the experiment were conducted (Shen et al., 2023).
Statistical Analysis
Software called GraphPad Prism was utilized to conduct statistical analysis on the obtained data from triplicate runs of all experimental analyses. The findings were computed as three separate tests’ mean ± standard deviation (SD), with a significant level established at *p < 0.05.
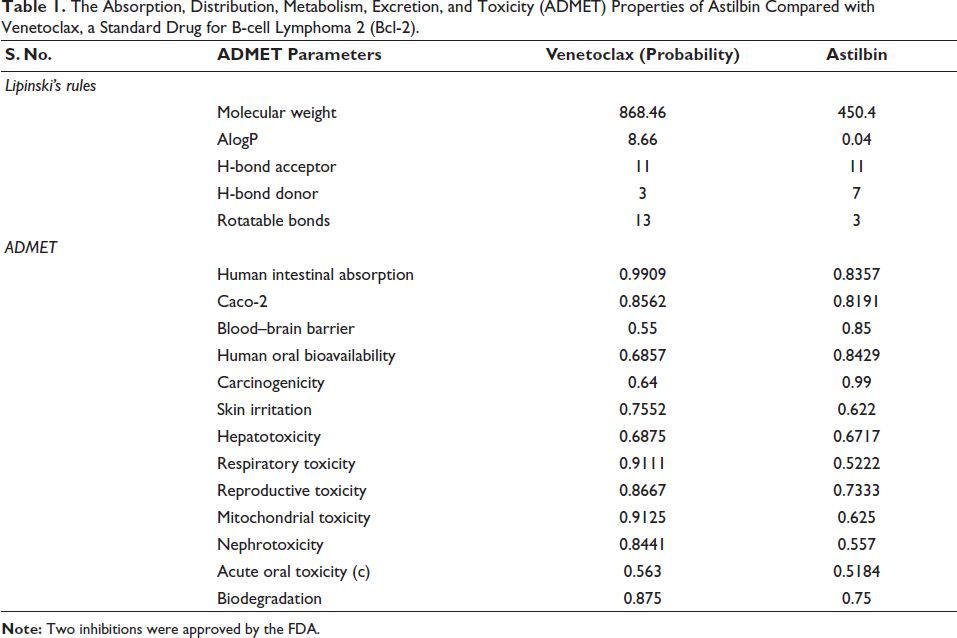
Chemical Absorption, Distribution, Metabolism, Excretion, and Toxicity (ADMET) Properties of Astilbin
The ADMET properties of Astilbin were evaluated using admetSAR v 2.0 and compared with venetoclax defined as a standard inhibitor for Bcl-2 (John & Konopleva, 2023; Yang et al., 2019).
Results
The Effectiveness of Astilbin on Cell Proliferation
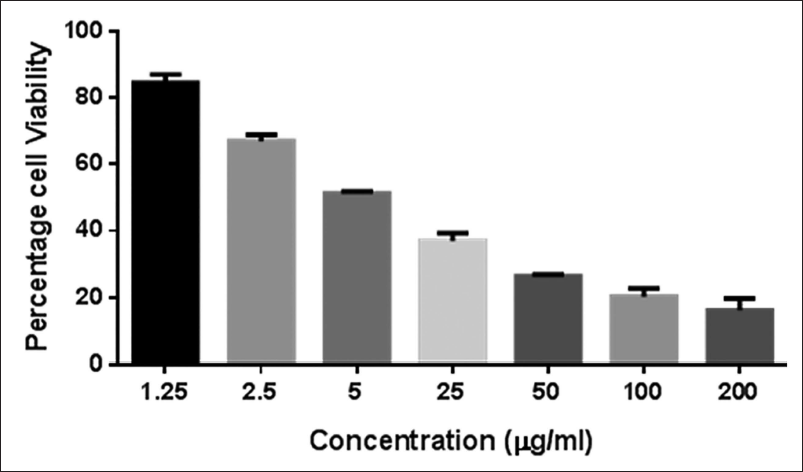
Cell proliferation was assessed. In Figure 1, the dose that results in a 50% inhibition of growth is depicted in the representative data, and the estimated IC50 value is 5 µg/ml. An approximate 80% inhibition of cell growth was observed at the maximum value of 200 µg/ml. Astilbin can suppress the growth of gastric cancer cells, as evidenced by the considerable decrease in the activity observed. The growth rates of cells treated with varying increasing concentrations of the substance.
Evaluation of AGS Cell Viability at Different Astilbin Doses (1.25–200 µg/ml). Samples were Divided into Three Groups to Get the Half Maximal Inhibitory Concentration (IC50) Value. The Representative Result Indicates a 50% Growth Inhibition Dosage.
Importance of Astilbin in Apoptosis
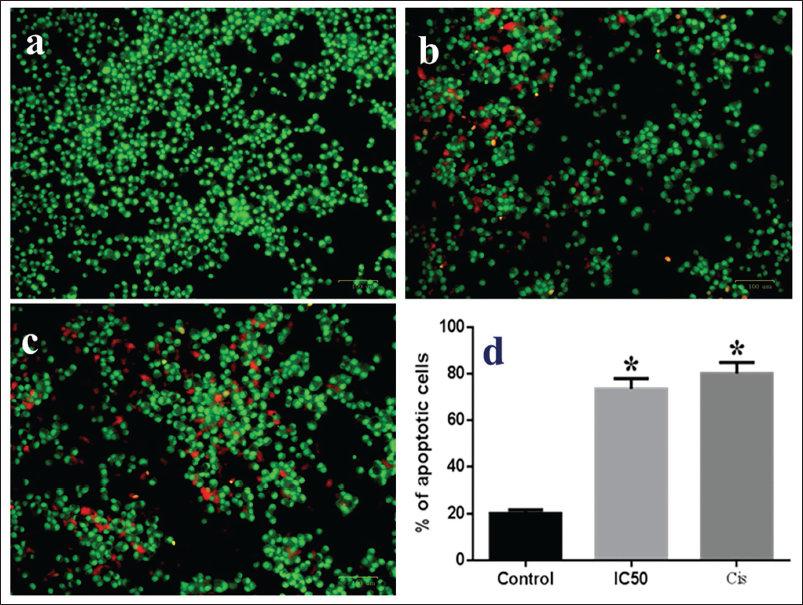
The morphological alterations caused by the astilbin treatment of AGS cells are evaluated by AO/EtBr staining. The microscopic pictures of the untreated control, the 5 µg/ml astilbin-treated cell, and the cisplatin-treated positive control cells are shown in Figure 2. When the three groups were compared, the untreated control cells showed consistently stained green live cells with minute orange cells that represented negligible apoptosis in the control group (Figure 2a). On examination of astilbin-administered AGS cells, it indicated dull green with orange and red stained cells exhibiting apoptotic and early phase apoptotic cells (Figure 2b). But in contrast to the control group and astilbin-treated cells, the orange and red fluorescence is more intense in the cisplatin-treated cells (Figure 2c). The percentage of apoptotic cells in AO/EtBr is shown by a bar graph in Figure 2d. The mean ± SD of three experiments is used to express the values.
The Impact of Astilbin-induced Apoptosis on the AGS Cell Line Using the Acridine Orange/Ethidium Bromide (AO/EtBr) Staining Method. When Astilbin at Half Maximal Inhibitory Concentration (IC50) is Administered, the Fluorescent Microscopic Images of AGS Cells Cultured for 24 Hours Show Dull Green with Orange and Red-stained Cells, Whereas Control Cells Show Bright Green (a). On the Other Hand, the Treated and Cisplatin Group has Cells That are Vivid Orange in Color (b, c). The Percentage of Apoptotic Cells in Each of the Three Groups is Shown in the Bar Graph (d). The Values are Expressed as the Mean ± Standard Deviation (SD) of Three Experiments. *p < 0.05, Compared with the Control Group.
The Effect of Astilbin on Inflammatory Cytokines Like TNF-α, IL-6, and NF-κB in AGS Cells
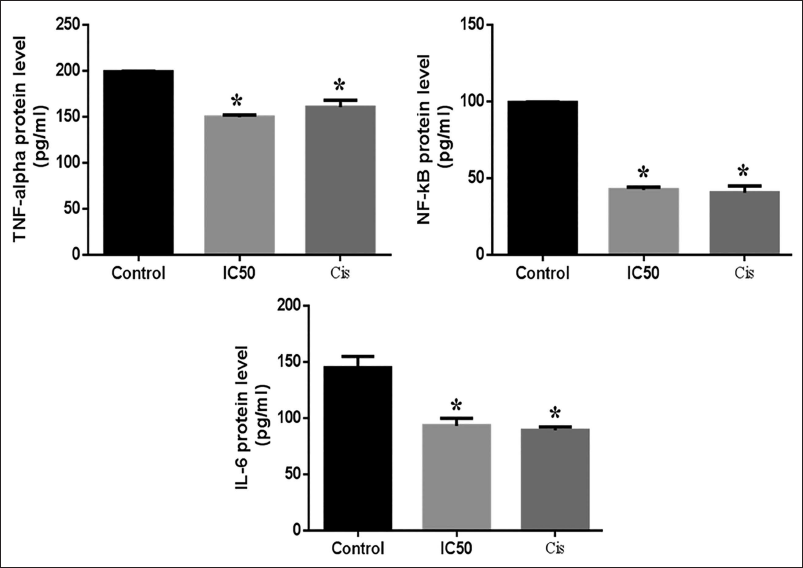
Using the ELISA approach, it was possible to ascertain the role played by inflammatory cytokines and how they are expressed in AGS cells treated with astilbin and cisplatin as well as in cells that were left untreated. In contrast to the untreated control cells that continued to release these inflammatory cytokines at high levels, it was observed that the cells treated with 5 µg/ml astilbin had significantly lowered TNF-α, IL-6, and NF-κB expression (Figure 3). The results show the mean ± SD of triplicate data from three different trials, with a *p < 0.05 difference from the control group.
The Inhibitory Effect of Astilbin on Tumor Necrosis Factor (TNF)-alpha, Nuclear Factor (NF)-κB, and Interleukin (IL)-6 in AGS Cell Lines. The AGS Cells were Given Astilbin, and the Protein Levels were Assessed Using the Enzyme-linked Immunosorbent Assay (ELISA) Technique. The Results Show the Triplicates’ Mean ± Standard Deviation (SD) from Three Different Trials. *p < 0.05, Compared with the Control Group.
The Effect of Astilbin on the Levels of Bax, Bcl-2, and Caspase-9 Proteins in AGS Cells
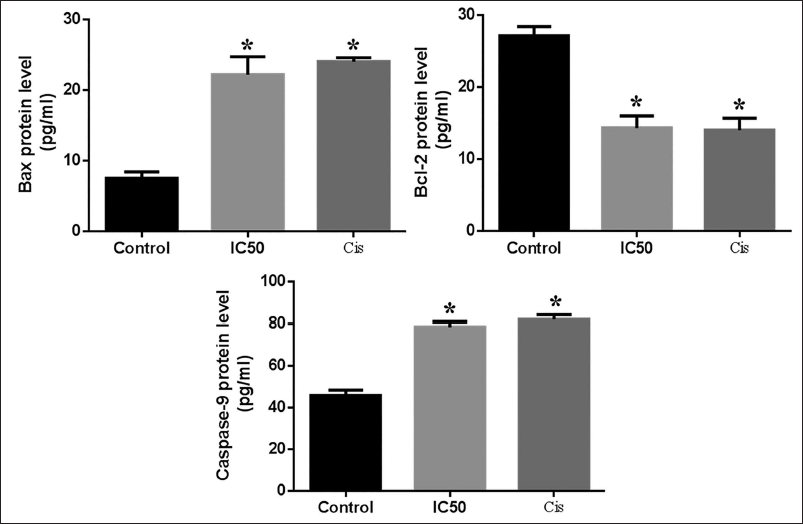
The apoptotic marker repression or stimulation, such as caspase-9, Bax, and Bcl-2 in astilbin-induced AGS cells. Figure 4 shows changes in the expression of Bcl-2, caspase-9, and Bax proteins in treated cultures when compared to control and cisplatin. After incubating cells with astilbin, there was a notable rise in the immunoreactivity for Bax and caspase-9 and a considerable reduction in the level of Bcl-2, suggesting that astilbin had anticarcinogenic potential. It exhibited an essentially identical effect to positive control. The results show the mean ± SD of three different experiments, with a *p < 0.05 difference from the control.
The Resultant Impact of Astilbin on Bcl-2-associated X-protein (Bax), B-cell Lymphoma 2 (Bcl-2), and Caspase-9 Activity in AGS Cell Lines. After a 24-hour Astilbin Treatment, the Cells were Collected. The Enzyme-linked Immunosorbent Assay (ELISA) Technique was Used to Measure the Protein Levels. The Mean ± Standard Deviation (SD) of Three Experiments is Used to Express the Values. *p < 0.05, Compared with the Control Group.
The ADMET Properties of Astilbin are a Significant Factor in its Effectiveness
The ADMET properties of astilbin were compared with the standard drug venetoclax approved by the FDA to inhibit the Bcl-2 expression to treat chronic lymphocytic leukemia as a second-line therapy (John & Konopleva, 2023). The results described that the compound astilbin obeyed Lipinski’s rule of five than venetoclax which has higher molecular weight. Likely, the astilbin intestinal absorption rate was approximately similar to the venetoclax. The estimated probability (p) for carcinogenicity was reduced in the astilbin (p = 0.99) than in the venetoclax (p = 0.64) (Table 1). However, astilbin and venetoclax are estimated for the hepatic, respiratory, reproductive, and mitochondrial toxicity. The results stated that the astilbin and venetoclax both showed high-level toxicity. Also, both the molecules are nonreadily biodegradable. Altogether, the ADMET properties of astilbin were similar to venetoclax with a lesser molecular weight.
The Absorption, Distribution, Metabolism, Excretion, and Toxicity (ADMET) Properties of Astilbin Compared with Venetoclax, a Standard Drug for B-cell Lymphoma 2 (Bcl-2).
Discussion
Throughout the world, one of the most common cancers is gastric cancer. While there has been advancement in adjuvant chemotherapy in the treatment of gastric cancer, the current chemotherapeutic agents have numerous unfavorable side effects in addition to poor curative outcomes. Therefore, it is imperative to create novel active ingredients with lower toxicity or fewer adverse effects, and to discover medications derived from natural sources (Teng et al., 2016). There are many flavonoids in the traditional medical system. Only a few numbers of them demonstrated their effectiveness in preclinical and clinical testing. Among the substances classified as dihydroflavonol rhamnoside that are present in a variety of plants and wines is astilbin (Sharma et al., 2020).
Thus, the goal of the current study aimed to provide a deeper understanding of the oncogenesis of gastric cancer and to aid in the development of therapeutic strategies for the medical condition by examining the role of astilbin in gastric cancer. Here, we demonstrated how astilbin inhibits the growth of AGS cells by inducing apoptosis and antiproliferative effects. The current study’s objective was to look at astilbin’s anticancer and antiproliferative molecular action, specifically how it induces apoptosis in AGS cells. Tumor growth is crucially indicated by cell proliferation. Therefore, the main goal in preventing the progression of a tumor is to limit malignant development by inducing apoptosis in cancer cells. Astilbin’s carcinogenicity has been studied using several cancer cell lines obtained from humans. Astilbin exhibited a dose-dependent reduction in the viability of cancer cells, including Hep G2, HT-29, A549, and MDA-MB-231. Astilbin showed no influence on the growth of Hep G2, A549, MDA-MB-231 cells, etc., at doses less than 200 µM. Astilbin, however, has the potential to kill more cancer cells in a dose-dependent manner at concentrations of more than 200 µM, especially HT-29, A549, and MDA-MB-231 cancer cells (Lv et al., 2019). According to the results of our current study, at 200 µg/ml, astilbin’s effect on AGS cell proliferation drastically decreased, reaching about 20%. These findings point to astilbin’s potential anticancer properties.
A natural biological process called apoptosis aids in preserving cellular equilibrium. Nevertheless, apoptotic pathways frequently experience dysregulation, which can result in a variety of pathogenic traits that promote the development of cancer. As a result, many cytotoxic medications’ antitumor cell inhibitory methods entail promoting cancerous cell death, which is essential for cancer therapy (Shen et al., 2023). In this research, after incubating cells for 24 hours, astilbin exposure was used to examine cytotoxicity and apoptosis at the cellular level. The technique known as AO/EtBr fluorescence labeling allows for the differentiation of viable, necrotic, and apoptotic cells. The current study’s findings demonstrated that astilbin has dose-dependent apoptotic effects on human AGS cells. The impact of the tumor’s internal environment on the onset of cancer is widely acknowledged. Inflammation is known to occur and to be triggered by inflammatory mediators like NF-κB, IL-6, etc. There is mounting evidence that the development of cancer is significantly correlated with elevated expression of inflammatory cytokines (Wang et al., 2023). Astilbin was discovered to have a significant impact on the immune system in earlier research by acting on a variety of immunomodulators and inflammatory modulators as a result. Because astilbin comes from a natural source, it can be a very useful immunosuppressive drug with relatively few adverse effects. Astilbin is also said to have a wide range of additional characteristics, including hepatoprotective, antioxidative, anti-inflammatory, and antibacterial effects (Sharma et al., 2020).
To shed more light on this, the AGS cells in this study that received astilbin treatment displayed a significant decrease in the expression of pro-inflammatory cytokines in comparison to the other cells. As a result, suppressing these factors causes apoptosis and growth inhibition (Sokolova & Naumann, 2017). The two main caspases in the family are caspase-9, an initiator of mitochondria-mediated apoptosis, and caspase-3, an executioner (Wang et al., 2023). Cleaved caspase-9, an active form of caspase-9, is an originator caspase that can activate caspase-3 in the cell death signaling cascade. The caspase-3 enzyme is cleaved to form the caspase-3 enzyme in activated form during apoptosis. This enzyme causes DNA fragmentation, morphological changes, and the breakdown of many cellular proteins. The Bcl-2 family of proteins controls the intrinsic apoptotic process (Shen et al., 2023). Bax belongs to the Bcl-2 family which promotes apoptosis and is activated by multiple apoptotic impulses. Apoptotic signals cause Bax, a proapoptotic protein belonging to the Bcl-2 family, to become activated. The movement of Bax to the mitochondria initiates apoptosis by decreasing mitochondrial membrane potential (MMP), stimulating cytochrome c release and caspase activation (Wang et al., 2023). In many cancers, Bcl-2, an antiapoptotic protein, is significantly higher than in normal, healthy cells. Specifically, Bcl-2/Bax regulates the cytochrome c emission from mitochondria into the cytosol, which triggers the regulation of caspase and ultimately results in apoptosis. By initiating elevated Bcl-2 levels or suppressing Bax protein levels, tumor cells can become resistant to apoptosis (Shen et al., 2023). Using an ELISA technique, the investigation evaluated the levels of caspase-9 and other key apoptotic proteins. The results demonstrated that the AGS cells administered with astilbin exhibited increased production of caspase-9, indicating the activation of death in the cells. It also demonstrated that astilbin-administered cells had downregulated Bcl-2 levels while upregulating Bax expression.
Combining these findings, the research showed that a Bax/Bcl-2 and caspase-dependent mechanism mediates the apoptosis triggered by astilbin. These findings offer significant new understandings of the potential molecular pathways by which astilbin functions as the most promising candidate in the generation of medications to combat gastric cancer drugs (Teng et al., 2016). The study suggests that astilbin may be a promising treatment for gastric cancer, but its safety and efficacy need to be assessed before clinical use. In silico ADMET analysis is recommended to predict astilbin’s pharmacokinetic and pharmacodynamic properties, drug-likeness, and potential adverse effects. This analysis will provide insights into bioavailability, metabolic stability, blood–brain barrier permeability, and potential drug–drug interactions. The article provides comprehensive methodologies and tools for predicting ADMET properties, facilitating informed decision-making in the development and optimization of astilbin as a gastric cancer therapy. Incorporating ADMET analysis will enhance the understanding of astilbin’s pharmacological profile and mitigate safety concerns, ultimately advancing its therapeutic potential.
Conclusion
Astilbin was shown to significantly reduce cell growth using the WTS-1 assay and AO/EtBr staining of the AGS cell line indicated that the molecule had a major impact in inducing apoptosis. Fluorescence microscopy analysis verified the triggering of apoptosis by chromatin condensation. Additionally, the investigation assessed the cytokine expression that causes inflammation and the levels of apoptotic proteins concerning astilbin-treated cells. Comparing treated cells to untreated ones, it dramatically doubled the expression of BAX and caspase-9. In contrast, there was a notable decrease in Bcl-2 expression. In AGS cell lines, the substance has inhibited the generation of cytokines such as TNF-alpha, NF-κB, and IL-6 that promote inflammation. These results imply that, with more research, astilbin may prove to be a highly successful anticancer medication for the efficient treatment of gastric cancer.
Summary
Astilbin notably inhibited the growth of cells. In AGS cell lines, it induces apoptosis and targets important inflammatory markers such as TNF-alpha, NF-κB, and IL-6, demonstrating a significant reduction in their expression. Being able to downregulate the Bcl-2 protein and upregulate the Bax and caspases proteins made them a potentially useful medication for gastric cancer therapy.
Abbreviations
IC50: Half maximal inhibitory concentration; AO: Acridine orange; EtBr: Ethidium bromide; ELISA: Enzyme-linked immunosorbent assay; IL-1: Interleukin-1; TNF: Tumor necrosis factor; IL-6: Interleukin-6; NF: Nuclear factor; Bcl-2: B-cell lymphoma 2; Bax: Bcl-2-associated X-protein; DNA: Deoxyribonucleic acid; WST-1: Water-soluble tetrazolium salt; CO2: Carbon dioxide; PBS: Phosphate-buffered saline; SD: Standard deviation; MMP: Mitochondrial membrane potential.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study strictly adhered to all relevant legal and ethical standards, ensuring rigorous scientific conduct and integrity.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
