Abstract
Background
A green and efficient process, free from toxic solvents, was employed to prepare an α-mangostin-rich extract. Conventional techniques like maceration and heat reflux extraction are recognized for their time-intensive nature, as well as the requirement for significant quantities of organic solvents. This innovative process not only reduces energy consumption but also streamlines production steps, providing a more sustainable alternative in herbal medicine preparation.
Purpose
The purpose of the study was to compare the antibacterial activities of alcohol disinfectants and α-mangostin extract-containing disinfectants against tested microorganisms.
Materials and Methods
The α-mangostin-rich extract was obtained from dried powders of Garcinia mangostana pericarps utilizing the microwave-assisted extraction method with polyethylene glycol 400 (PEG 400). The α-mangostin content in the resultant extract was determined through the high-performance liquid chromatography (HPLC) method. Additionally, a broth microdilution method was utilized to investigate the minimum inhibitory concentrations (MICs) and minimum bactericidal concentrations (MBCs) of each compound against methicillin-resistant Staphylococcus aureus (MRSA) and Staphylococcus pseudintermedius.
Results
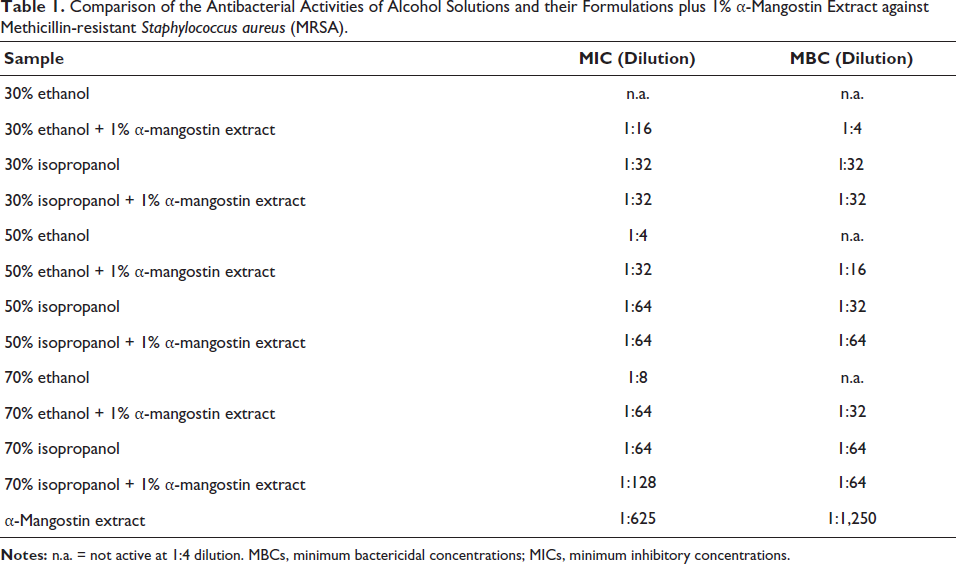
According to a microdilution method, the disinfectant comprising isopropanol (50% and 70%) with 1% α-mangostin extract demonstrated antibacterial effectiveness against both MRSA and S. pseudintermedius. The MICs and MBCs were observed within dilutions ranging from 1:64 to 1:512 and 1:64 to 1:256, respectively. Ethanol (50% and 70%) plus 1% α-mangostin extract disinfectant exhibited antibacterial activity with MICs and MBCs at dilutions of 1:32–1:512 and 1:16–1:128, respectively. Moreover, isopropanol and ethanol (30%) with 1% α-mangostin extract disinfectant demonstrated antibacterial activity with MICs and MBCs at dilutions of 1:16–1:256 and 1:4–1:128, respectively. However, isopropanol (50% and 70%) and ethanol (50% and 70%) disinfectants showed weaker antibacterial activity than α-mangostin extract-containing alcohol solutions. Although isopropanol (30%) disinfectant demonstrated antibacterial activity (MICs and MBCs at dilutions of 1:16–1:32), the 30% ethanol solution did not show any activity against either bacterium. In addition, α-mangostin extract exhibited stronger antibacterial activity (MICs and MBCs at dilutions of 1:625–1:5,000) than all alcohol solutions.
Conclusion
The combination of isopropanol and ethanol disinfectants with α-mangostin-rich extract exhibited significant antibacterial effectiveness against MRSA and S. pseudintermedius. The enhancement of efficacy by α-mangostin-rich extract suggests promising opportunities for enhancing disinfection approaches.
Introduction
Methicillin-resistant Staphylococcus aureus (MRSA) transmission within hospitals often occurs through the transient colonization of staff hands, as reported by Ben-David et al. (2008). To interrupt this transmission chain, barrier precautions and hand disinfection are the primary methods employed (Lee et al., 2020). The Centers for Disease Control and Prevention has underscored proper and regular handwashing as a pivotal infection control measure (Landers et al., 2012). Enhanced compliance with handwashing significantly diminishes the carriage of potential pathogens on healthcare workers’ hands and has notably curtailed nosocomial infections, as evidenced by studies such as Mathur (2011). However, handwashing compliance rates remain below 50% in both European and American hospitals (Landers et al., 2012). Healthcare workers cite reasons such as skin irritation and time constraints for noncompliance (Mouajou et al., 2022). To address these challenges, the use of alcohol-based sanitizers and emollients has been advocated. Increasing effectiveness and efficiency, products other than water and soap have been discovered to streamline procedures and save time, additionally reducing infections (Massa, 2010; Siegel et al., 2007). Various antiseptics and disinfectants, such as alcohols, aldehydes, phenols, peroxygens, biguanides, and cresols, have been used to combat nosocomial infections in shared facilities (Gordin et al., 2005; Noel, 2023). The antimicrobial effects of hand hygiene agents, such as carbohydrate fatty acid derivatives and several antiseptics, against MRSA have been analyzed by various groups (Aboualizadeh et al., 2017; Nobmann et al., 2010). Furthermore, ethyl alcohol and isopropyl alcohol are antiseptically most effective against spores (Thaddeus et al., 2018). Bacterial rupture induced by antiseptics and antibacterial agents affects normal skin cells. An extract of the pericarp of mangosteen is useful for human health. It has been reported to have pharmacological properties, including antibacterial, antifungal, antioxidant, anti-inflammatory, and antiallergy activities (Obolskiy et al., 2009; Pedraza-Chaverri et al., 2008). α-Mangostin, the significant extracted derivative, has antibacterial activities against Gram-positive bacteria, including MRSA and Staphylococcus pseudintermedius (Larsuprom et al., 2019; Meah et al., 2020). It has been reported that incorporating a natural compound in the production of an herbal product is impractical due to its expensive production involving numerous steps (Amalraj et al., 2017). Our focus lies in devising a method to prepare α-mangostin-rich extract from Garcinia mangostana, utilizing environmentally friendly extraction techniques like green extraction (Meah & Panichayupakaranant, 2020). This is prompted by the drawbacks associated with traditional methods, such as heat reflux and maceration extraction, which require significant amounts of organic solvents and consume considerable time, pose challenges due to their toxicity, volatility, and flammability (Azwanida, 2015; Zhou et al., 2011). Hence, there is an urgent need for an eco-friendly and efficient extraction method that minimizes energy consumption and production steps while eliminating reliance on harmful solvents. This approach is particularly crucial for the preparation of herbal extracts enriched with active constituents, which are extensively utilized in pharmaceutical applications (Michel et al., 2011). The purpose of this study was to compare the antibacterial efficacy of alcohol disinfectants when combined with α-mangostin extract.
Materials and Methods
Chemicals and Plant Materials
G. mangostana pericarp powder was sourced from Charoensuk Pharma Supply (Nakhon Pathom, Thailand). Sigma-Aldrich (Steinheim, Germany) provided Diaion® HP 20 resin. Becton, Dickinson, and Company (NJ, USA) supplied brain heart infusion and agar. RCI Labscan (Bangkok, Thailand) provided polyethylene glycol 400 (PEG 400), acetonitrile, and methanol of high-performance liquid chromatography (HPLC) grades. High Science Ltd. (Hat-Yai, Thailand) supplied ethanol of commercial grade. Water was purified using a Milli Q system from Millipore (Bedford, MA, USA).
Bacterial Isolates
The MRSA standard isolate (DMST 20654) was acquired from the Department of Medical Sciences, Ministry of Public Health, Thailand, while S. pseudintermedius was sourced from the Veterinary Teaching Hospital, Prince of Songkla University, Hat-Yai, Thailand.
Preparation of an Enriched α-Mangostin Extract
Dried powders of G. mangostana pericarps (1.5 kg) were subjected to microwave-assisted extraction using a microwave frequency of 1,450 MHz and an electric power of 900 W. The extraction process comprised three cycles, each consisting of 8 minutes of power-on followed by 1 minute of power-off, conducted at 90°C. During the extraction, PEG 400 (3 L) was utilized as the solvent.
Quantitative Analysis using HPLC
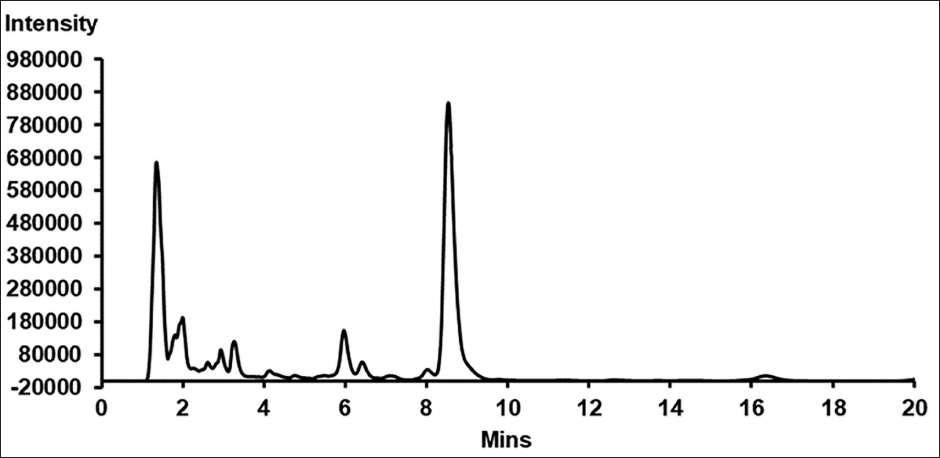
For the quantitative analysis of α-mangostin content in the extract, we adapted the HPLC method outlined by Bundeesomchok et al. (2016) with modifications. Our method utilized a binary HPLC pump (Shimadzu 33236, Tokyo, Japan), coupled with a photodiode array detector (Shimadzu 12623, Tokyo, Japan) and an autosampler (Shimadzu 03224, Tokyo, Japan). Furthermore, we employed a 150 × 4.6 mm ACE 5 C18-PFP column (Advanced Chromatography Technologies, Aberdeen, Scotland). Standardization of the sample injection volume was set at 20 µL, and the column was subjected to isocratic elution using a blend of acetonitrile and 0.2% formic acid (70:30, v/v) at a flow rate of 1 mL/min. The concentration of α-mangostin was determined at a wavelength of 240 nm, utilizing a calibration curve constructed from five α-mangostin concentrations (ranging from 12.5 to 200 µg/mL), with their respective peak areas plotted against concentrations. Each calibration concentration underwent triplicate analysis to establish the linear equation Y = 93,150X – 32,893 (R2 = 1). The analysis revealed that the α-mangostin extract contained 0.04% (w/v) α-mangostin (Figure 1).
High-performance Liquid Chromatography (HPLC) Chromatogram of 40 mg/mL α-Mangostin Extract in Polyethylene Glycol 400 (PEG 400).
Investigation of Minimum Inhibitory Concentrations (MICs) and Minimum Bactericidal Concentrations (MBCs)
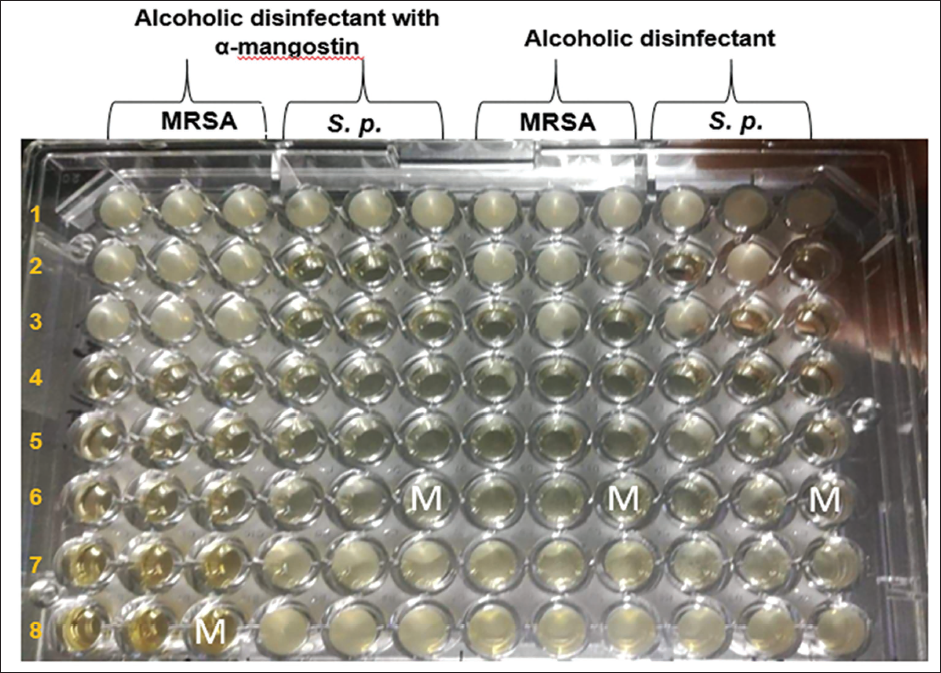
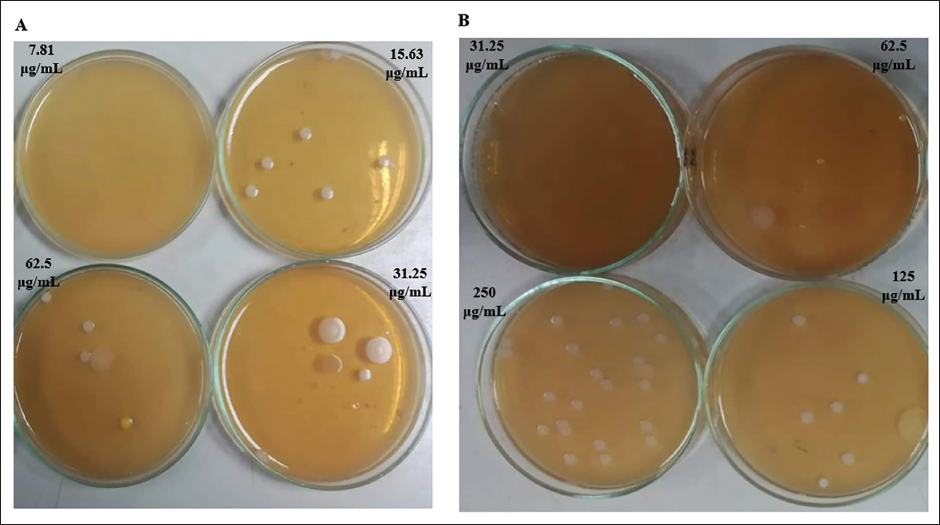
A broth microdilution method, following NCCLS guidelines from 2008 (NCCLS, 2008), was carried out to investigate MICs and MBCs against MRSA and S. pseudintermedius. Initially, the compounds were dissolved in dimethyl sulfoxide and then diluted with brain heart infusion broth to attain a concentration of 1,000 µg/mL. Subsequently, two-fold dilutions were prepared in a 96-well plate. Concurrently, a bacterial suspension was prepared in 0.85% normal saline, with turbidity adjusted to 0.5 McFarland standards (equivalent to 1×108 CFU/mL). This suspension underwent further dilution with normal saline solution to achieve a concentration of 1 × 106 CFU/mL, which was then added to each well of the plate. Ultimately, the final cell concentration was adjusted to 5 × 105 CFU/mL, and the plates were incubated at 37°C for 24 hours. Finally, MICs (Figure 2) and MBCs (Figure 3) were determined as the lowest concentration of the compound that inhibited visible growth.
The M = Minimum Inhibitory Concentrations (MIC) of the Alcoholic Disinfectant containing α-Mangostin was Determined, Yielding a Range of 7.81–31.25 µg/mL. Additionally, the MIC of the Alcoholic Disinfectant Alone was found to be 31.25 µg/mL against both Methicillin-resistant Staphylococcus aureus (MRSA) and S. p. = Staphylococcus pseudintermedius.
The Minimum Bactericidal Concentrations (MBC) of the Alcoholic Disinfectant containing α-Mangostin was Assessed. It was found to be 7.81 µg/mL for the Alcoholic Disinfectant containing α-Mangostin (A), while the MBC for the Alcoholic Disinfectant alone was 31.25 µg/mL (B) against both Bacterial Strains.
Results
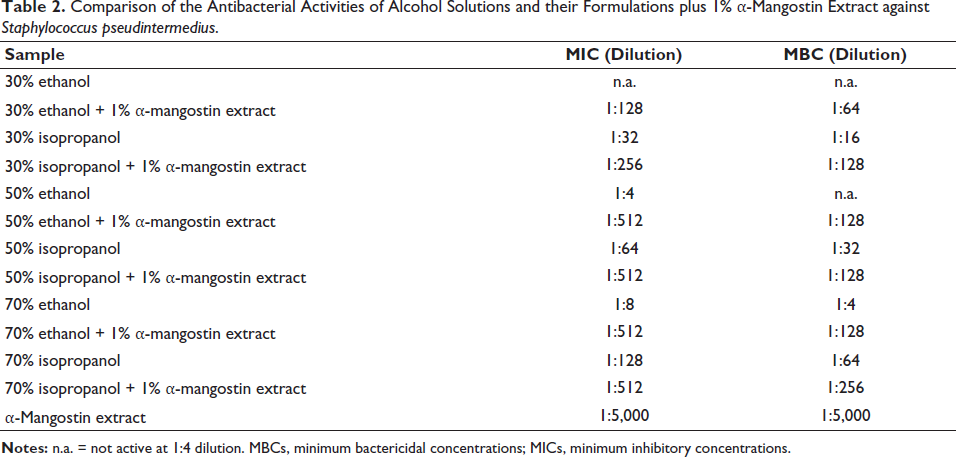
Isopropanol (50% and 70%) disinfectant exhibited antibacterial activity against MRSA and S. pseudintermedius with MICs and MBCs at dilutions of 1:32–1:128 (Tables 1 and 2). Isopropanol (50% and 70%) with 1% α-mangostin extract demonstrated antibacterial activity with MICs and MBCs at dilutions of 1:64–1:512 and 1:64–1:256, respectively. However, ethanol (50% and 70%) disinfectant exhibited weak antibacterial activity against both bacteria with MICs and MBCs at dilutions of 1:4–1:8. Ethanol (50% and 70%) with 1% α-mangostin extract demonstrated antibacterial activity with MICs and MBCs at dilutions of 1:32–1:512 and 1:16–1:128, respectively. Moreover, 30% isopropanol disinfectant demonstrated antibacterial activity with MICs and MBCs at dilutions of 1:32 and 1:16–1:32, respectively. Isopropanol (30%) with 1% α-mangostin extract showed stronger antibacterial activity with MICs and MBCs at dilutions of 1:32–1:256. Furthermore, 30% ethanol elicited no antibacterial activity against either bacterium. However, 30% ethanol plus 1% α-mangostin extract exhibited antibacterial activity with MICs and MBCs at dilutions of 1:16–1:128 and 1:4–1:64, respectively. In addition, α-mangostin extract exhibited the strongest antibacterial activity with MICs and MBCs at dilutions of 1:625–1:5,000.
Comparison of the Antibacterial Activities of Alcohol Solutions and their Formulations plus 1% α-Mangostin Extract against Methicillin-resistant Staphylococcus aureus (MRSA).
Comparison of the Antibacterial Activities of Alcohol Solutions and their Formulations plus 1% α-Mangostin Extract against Staphylococcus pseudintermedius.
Discussion
Based on a microdilution assay, it was observed that both ethanol (50% and 70%) and isopropanol (50% and 70%) disinfectants with α-mangostin extract exhibited strong antibacterial activity against MRSA and S. pseudintermedius. In contrast, alcohol disinfectants without α-mangostin extract showed weak antibacterial activity. MRSA is a leading cause of medical clinic-acquired infections, which are becoming progressively hard to treat due to increasing resistance to all present antibiotic classes. The mecA gene acquisition, its ability to overexpress efflux pumps, and its capacity to produce a β-lactamase enzyme are fundamental factors contributing to MRSA resistance. Numerous countries have reported a significant prevalence of MRSA nosocomial infections (Zuo et al., 2014). Typically, MRSA infections manifest as mild skin issues such as bruises, blisters, or abscesses. Research indicates that the maximum antimicrobial activity is achieved when alcohol is diluted with water, with formulations containing 60–90% alcohol proving to be the most effective (Lim et al., 2023). α-Mangostin’s ability to disrupt cell membranes and enhance permeability has been extensively documented, facilitating the passage of large molecules across the membrane and leading to the release of intracellular components (Koh et al., 2013; Phitaktim et al., 2016). The isoprenyl group within α-mangostin plays a crucial role in modifying the lipid bilayer of the MRSA membrane, thereby causing membrane disruption and increased permeability (Koh et al., 2013). Additionally, Edmonds et al. (2012) demonstrated that 70% ethanol is more effective in reducing bacterial levels compared to 62% ethanol. Among 26 reviewed studies, six (23.08%) specifically investigated the microbicidal efficacy of various concentrations, types, and forms of alcohol-based solutions for hand hygiene. While traditional handwashing with soap or antimicrobial detergent remains prevalent in most Thai hospitals, recent research indicates that greater utilization of alcohol-based hand rubs can enhance hand hygiene practices among healthcare workers (Picheansathian, 2004). Ochwoto et al. (2017) reported that a 99.9% reduction in bacterial colonies on hands following the application of an alcohol-based solution. Hand rubbing with alcohol-based hand rub has been shown to effectively remove microorganisms, requiring less time and causing less irritation to the hands compared to handwashing with soap or other antiseptic agents and water. Moreover, the availability of bedside alcohol-based solutions has been associated with increased compliance with hand hygiene protocols among healthcare workers, as highlighted by Picheansathian (2004). Loveday et al. (2014) showed that rubbing hands with an alcohol-based solution is notably more effective in reducing MRSA compared to washing hands with nonmedicated soap. In the current study, alcohol disinfectants with 1% α-mangostin extract showed greater activity against the bacterial strains than ethanol and isopropanol solutions. In conclusion, a natural product, e.g., α-mangostin extract disinfectant with an alcohol solution, might be useful to treat bacterial infections.
Conclusion
The incorporation of α-mangostin extract significantly bolstered the antibacterial potency of alcohol-based disinfectants, underscoring its potential as a valuable adjunct in combating microbial resistance. These findings underscore with MICs and MBCs at dilutions of 1:625–1:5,000 is the promise of α-mangostin extract in enhancing the effectiveness of disinfectants against challenging bacterial strains like MRSA and S. pseudintermedius.
Footnotes
Abbreviations
MICs: Minimum inhibitory concentrations; MBCs: Minimum bactericidal concentrations; MRSA: Methicillin-resistant Staphylococcus aureus; PEG 400: Polyethylene glycol 400; HPLC: High-performance liquid chromatography.
Acknowledgments
The authors are thankful to the Phytomedicine and Pharmaceutical Biotechnology Excellence Center, Faculty of Pharmaceutical Sciences, Prince of Songkla University, Thailand, for providing all laboratory facilities.
Authors’ Contributions
All authors contributed to the conception and design of the study. M.S. Meah and P. Panichayupakaranant conducted the experiments. M. G. Kim, K. J. Silvano, and R. A. Ramazon Ugli contributed to critically revising the manuscript. All authors approved the final submitted version.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
For this work, ethical approval is not required as it does not involve animal testing. All authors have provided informed consent for this work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Thailand Research Fund (Grant No. DBG6180031) and the National Research Foundation of Korea (Grant No. NRF-2020R1I1A2073610).
