Abstract
Background and Purpose
Non-alcoholic fatty liver disease (NAFLD) is a worldwide health problem with high prevalence and morbidity associated with obesity, insulin resistance, type 2 diabetes mellitus (T2DM), and dyslipidemia. Luteolin is a natural flavonoid with various activities. We aimed to investigate whether luteolin can alleviate NAFLD and its possible mechanism.
Materials and Methods
A mouse model with NAFLD was induced by Western diet (WD) and carbon tetrachloride (CCl4) injection. Twenty-four mice were distributed into three groups randomly: Normal diet group (CON, n = 8), NAFLD group (NAF, n = 8), and luteolin group (LUT, n = 8). NAFLD model was induced by feeding mice in NAF and LUT groups with WD and injecting CCl4. CON group were fed with a normal diet in the same period. The LUT group was administered orally with luteolin (20 mg/kg) every day, starting from the 1st day of the 5th week for 8 weeks, while the CON group was treated with a vehicle. Throughout the experiment, body weight, lipid profile, glucose tolerance test, and the hepatic expressions of CD36/FABP1/LC3B/Bcl-2 were measured beside the histopathological examination.
Results
Luteolin treatment effectively prevents the body from weight gain in mice with NAFLD (NAF vs. LUT: 36.0 g vs. 32.4 g, p < 0.05). Serum total cholesterol and triglyceride levels are both significantly increased in the NAF group (p < 0.01) and reduced to some extent in the LUT group (p < 0.05). Oral glucose tolerance test (OGTT) test results show that luteolin could improve the impaired glucose tolerance and downregulate the elevated fasting blood glucose in mice with NAFLD (p < 0.05). Histopathological results demonstrate luteolin reduces hepatic steatosis and hepatic fibrosis in NAFLD mice. The possible mechanisms may include that luteolin decreases hepatic CD36 and FABP1 expression, and increases levels of autophagy marker LC3B and antiapoptotic protein Bcl-2.
Conclusion
Luteolin could reduce hepatic steatosis and fibrosis, hyperglycemia, body weight, and serum lipid levels in NAFLD mice, which exhibits huge potential in the treatment of metabolic disorders related to NAFLD.
Keywords
Introduction
Non-alcoholic fatty liver disease (NAFLD) is a public health concern all over the world, correlated with obesity, insulin resistance, type 2 diabetes mellitus, hyperlipidemia, and hypertension (Loomba & Sanyal, 2013). It is characterized by hepatic steatosis except secondary reason for hepatic lipid accumulation (e.g., overuse of alcohol). NAFLD may progress to non-alcoholic steatohepatitis (NASH)—the more severe condition—and may further progress to fibrosis and cirrhosis (Pouwels et al., 2022). Recently, a meta-analysis reported that the prevalence of NAFLD and NASH in overweight people was 69.99%, 42.49%, and 33.50%, respectively. Similarly, the prevalence estimates for NAFLD and NASH in obese people were 75.27%, 43.05%, and 33.67%, respectively (Quek et al., 2023). As one can see, with such a high volume of patients and possible commodities, NAFLD is making an enormous impact on life quality and has also caused a huge economic burden (Younossi et al., 2018). Currently, there are no available treatments for NAFLD, although a lot of progress has been made in its pathological mechanisms. Therefore, it is of great significance to find drugs that can prevent and treat this disease.
Natural Compounds Obtained from Natural Plants Play an Important Role for Development of Drugs
Some natural compounds have been discovered to improve NAFLD, demonstrating a huge therapeutic potential, such as cinnamic acid (Wu et al., 2021), alisol B (Zhao et al., 2022), and magnolol (Kuo et al., 2020), which are proven to be CD36 inhibitors. Different from traditional drugs, natural compounds are derived from natural plants, are easily obtainable, and are the ideal option for new drugs (Feng et al., 2022).
Luteolin (3′,4′,5,7-tetrahydroxyflavone) is a type of natural flavonoid found in natural herbs, vegetables, and fruits. Previous studies showed that luteolin exhibits anti-inflammation, antioxidant, and antitumor activity. Besides, it also regulates immune activity (Jeon et al., 2014; Kasala et al., 2016; Xia et al., 2016). It was reported to have antiobesity effects and has been showed to significantly improve NAFLD and obesity (Xu et al., 2014; Yin et al., 2017). Kwon and Choi found that luteolin improved diet-induced hepatic adipocyte fibrosis and ameliorate insulin resistance in mice with obesity through toll-like receptor signaling pathways (Kwon & Choi, 2018). Yin et al. showed that luteolin alleviates NAFLD by targeting liver X receptor and decreasing expression of sterol regulatory element-binding protein 1c (Yin et al., 2017).
The Western diet (WD)/carbon tetrachloride (CCl4) model we used in this study was thought as an ideal mouse model for human NAFLD (Castro & Diehl, 2018), because this model not only has the manifestation of hepatic steatosis but also presents advanced fibrosis, hepatic cirrhosis, and even hepatocellular carcinoma, resembling pathological features of human NAFLD.
Here in the present study, we discussed the effect of luteolin on the WD/CCl4 mouse model of NAFLD and explored the possible mechanisms involved regarding lipid uptake, apoptosis, and autophagy in hepatic tissues.
Materials and Methods
Animals
A total of 24 C57BL/6 male mice (6-week-old) were obtained from Zhejiang Weitong Lihua Laboratory Animal Technology Co., Ltd, Jiaxing, China. All animals were housed in a standard environment at 20 ± 2°C. The humidity was controlled at 55 ± 10% in a 12 h light/12 h dark cycle. All the mice were subjected to 1-week adaptive living and were randomly distributed into three groups: Normal diet group (CON, n = 8), NAFLD group (NAF, n = 8), and luteolin group (LUT, n = 8). NAFLD model was induced by feeding mice with a WD and injecting CCl4 in NAF and LUT groups. The WD contained 21.1% fat, 41% sucrose, and 1.25% cholesterol, and the drinking water was supplemented with 23.1 g/L
Glucose Tolerance Test (GTT)
Following 8 weeks of luteolin or vehicle treatment, a GTT was conducted. Mice were in fasting status for 12 h and given by gavage with glucose (2 g/kg). Blood glucose levels from tail blood samples were measured at 0, 15, 30, 60, 90, and 120 min after injection.
Sample Preparation
Following the last administration of luteolin or vehicle and fasting for 12 h, all the mice were anesthetized, and blood samples were collected from the orbital vein. After the samples were centrifuged at 3,000 × g for 5 min, serum biomarkers were measured. The livers were removed, rinsed with normal saline, and stored in a –80°C refrigerator. Paraffin sections of the liver tissues were made for further pathological evaluation.
Biochemical Analysis
Total cholesterol (TC), triglyceride (TG), and fasting plasma glucose (FPG) were analyzed by using Automatic Biochemical Analyzer (Beckman Coulter, Indianapolis, US). All procedures were conducted in accordance with the manufacturer’s instructions.
Histopathological Examination
Paraffin-embedded liver was sectioned into 3 µm and fixed in 4% paraformaldehyde for hematoxylin–eosin (H&E) and Masson’s trichrome staining, which were processed routinely with commercial kits (Boster & Yeasen, China). The stained sections were observed and photographed under an optical microscope (Nikon, Tokyo, Japan) with a ×200 magnitude.
Real-time Polymerase Chain Reaction (PCR)
Total ribonucleic acid (RNA) was extracted from hepatic tissues using TRIzol reagent and reverse-transcribed into complementary deoxyribonucleic acid (cDNA) using PrimeScript™ RT kit (Roche, Basel, Switzerland). According to the instructions of the SYBR Premix Ex Taq™ kit (Takara, Tokyo, Japan), reagents were added in turn, and gene amplification was performed by PCR. The specific primer series are listed in Table 1.
The Specific Primer Series in Real-time Polymerase Chain Reaction (PCR).

Statistical Analysis
All data were expressed as mean ± standard error of the mean (SEM). Data of body weight from week 4 to week 12 were analyzed using a factorial repeated measures analysis of variance (ANOVA) with the between-subjects factor being groups and the within-subjects factor being test weeks (CON group versus NAF group and NAF group versus LUT group, respectively). Statistical significance was calculated using two-tailed Student’s t-tests for comparisons between two groups. For all analyses, p ≤ 0.05 was considered statistically significant, and all data were analyzed using SPSS version 23.
Results
Luteolin Decreases Body Weight and Improves Insulin Resistance in Mice with NAFLD
We adopted the WD/CCl4 model to induce NAFLD in mice, which resembles human NAFLD the most (Castro & Diehl, 2018). Figure 2a shows that body weight in NAF group increased rapidly throughout the study period, and luteolin could prevent the body from weight gain (NAF vs. LUT: 36.0 g vs. 32.4 g, p < 0.05), demonstrating the antiobesity effect of luteolin in mice with NAFLD.
(a) Luteolin Prevented the Body from Weight Gain in Mice with Non-alcoholic Fatty Liver Disease (NAFLD). Body Weight of Mice Fed with Western Diet (WD) and Carbon Tetrachloride (CCl4) Injection (Red and Blue) or Normal Diet and Vehicle Injection (Green) Over 12 Weeks were Presented. (B) Luteolin Could Reduce Serum Total Cholesterol and Triglyceride Levels in Mice with NAFLD. (c) Luteolin Could Improve Glucose Metabolism in Mice with NAFLD, Including Amelioration of Impaired Glucose Tolerance (Left) and Decrease of Fasting Blood Glucose (Right). All Data are Presented as Means ± Standard Error of the Mean (SEM) (in Comparison with NAF, *p < 0.05, **p < 0.01, in Comparison with CON, #p < 0.01).
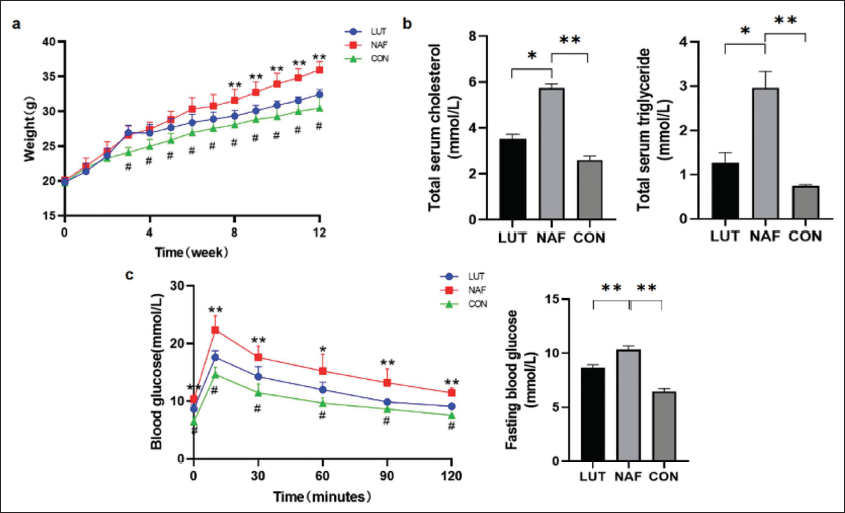
Luteolin Downregulates Serum Lipids in Mice with WD/CCl4-induced NAFLD Mice
Figure 2b shows that serum TC and TG levels both significantly increase in the NAF group (p < 0.01) and are reduced to some extent in LUT group (p < 0.05), suggesting a lipid-lowering effect of luteolin.
Luteolin Ameliorate Glucose Metabolism in WD/CCl4-induced NAFLD Mice
We also conducted OGTT test at the end of the study. The result demonstrates that 100% mice in NAF group showed fasting blood glucose >7 mmol/L. Luteolin could improve the impaired glucose tolerance and downregulate the elevated fasting blood glucose in mice with NAFLD (p < 0.01) (Figure 2c).
Luteolin Reduces Hepatic Steatosis and Prevents Hepatic Fibrosis in NAFLD Mice
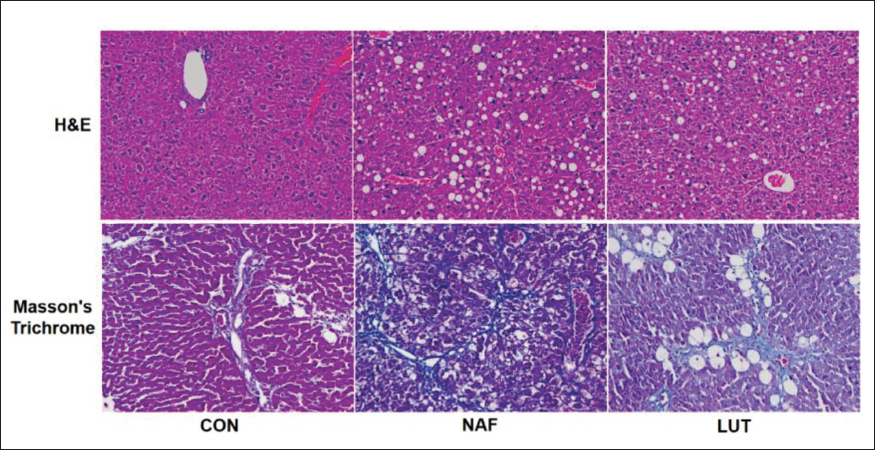
The H&E staining of the liver tissue demonstrates hepatic steatosis in NAF group, characterized by extensive microvacuolar steatosis (Figure 3). Luteolin reduced the pathological changes. Masson’s trichrome staining of hepatic tissues in mice with NAFLD showed more collagen fiber staining in NAF mice compared with CON group, and luteolin alleviated the amount of collagen fiber in hepatic tissues, implying an antifibrosis effect of luteolin in NAFLD.
Luteolin Improved Hepatic Steatosis and Hepatic Fibrosis in Non-alcoholic Fatty Liver Disease (NAFLD) Mice. 4 µm Thick Liver Sections from 12-Week-Old Male Mice Treated with Luteolin were Stained with Hematoxylin & Eosin (H&E) (Upper) (×100 Magnification) and Masson’s Trichrome (Lower) (×400 Magnification).
Luteolin Improves WD/CCl4-induced NAFLD in Mice by Inhibiting CD36 and FABP1 mRNA Expression in Hepatic Cells
As shown in Figure 4, hepatic lipid uptake-related proteins CD36 and FABP1 are elevated in NAF group. Luteolin shows the tendency of alleviating increased CD36 and FABP1 mRNA levels, implicating that luteolin may ameliorate NAFLD by inhibiting hepatic lipid uptake through CD36 and FABP1 pathways.
Luteolin Alleviated CD36 and FABP1 Messenger Ribonucleic Acid (mRNA) Expression (a) and Increased Hepatic Autophagy Protein LC3B and Apoptosis Protein Bcl-2 mRNA Expression (b) in Non-alcoholic Fatty Liver Disease (NAFLD) Mice. All Data are Presented as Means ± Standard Error of the Mean (SEM) (Compared with NAF, *p < 0.05, **p < 0.01).
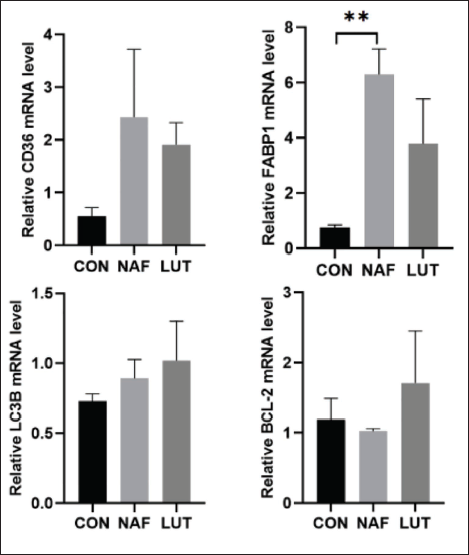
Luteolin Improves WD/CCl4-induced NAFLD in mice by increasing LC3B and Bcl-2 mRNA expression in hepatic cells
In the present study, we have also observed increased hepatic autophagy protein LC3B and antiapoptosis protein Bcl-2 mRNA expression after luteolin intervention, demonstrating that luteolin may also plays roles in promoting hepatic autophagy and inhibiting hepatic apoptosis (Figure 4).
Discussion
The present study, for the first time, demonstrates that luteolin administration could ameliorate body weight gain, hyperlipidemia, glucose metabolic disorders, hepatic steatosis, and fibrosis in the WD/CCl4 mouse model of NAFLD. The possible mechanisms may be through inhibition of the lipid uptake pathways, promotion of autophagy, and decrease of apoptosis in hepatocytes.
CD36, a multiligand receptor and membrane glycoprotein in hepatocytes, enterocytes, adipocytes, etc., plays a role in the hepatic uptake of lipids (Glatz & Luiken, 2017). CD36 in the liver is positively correlated with TG contents in the liver of patients with NAFLD, and this transporter plays a potential role in this disease and is considered a crucial treatment target (Greco et al., 2008; Rada et al., 2020; Samovski & Abumrad, 2019). In CD36 gene knockout hepatocytes, upregulation of β-oxidation and reduction of steatosis were observed as a result of lipid phagocytosis (Li et al., 2019). Mice with hepatocytes deficient in CD36 are protected from diet-induced hepatic steatosis and insulin resistance (Wilson et al., 2016). In obese rats, CD36 mRNA expression is positively related to liver steatosis severity (Buqué et al., 2010). FABP1 is one of the cytosolic lipid-binding proteins expressed in hepatic cells and facilitates intracellular transport of FFAs. Mice with FABP1-deficient hepatocytes are protected from diet-induced obesity (Newberry et al., 2003). FABP1 knockdown in the liver reduced liver weight and TG accumulation in the liver (Mukai et al., 2017), suggesting that inhibition of FABP1 in hepatocytes is an effective way to improve NAFLD.
Our results in the present study show that CD36 and FABP1 mRNA levels were significantly downregulated as predicted, indicating that luteolin may improve NAFLD by inhibiting the lipid uptake pathways in hepatocytes.
We also analyzed mRNA expression of LC3B in hepatic cells to find the role of luteolin in autophagy. We found that luteolin could, to some extent, increase LC3B levels, revealing that luteolin may induce autophagy of hepatic cells in NAFLD mice. BCL-2 protein family is a key regulator of physiological and pathological apoptosis. The mRNA levels of antiapoptotic protein Bcl-2 increased after luteolin administration, demonstrating that luteolin may also counteract liver steatosis by targeting apoptosis.
Moreover, a study shows that exercise ameliorates hepatic steatosis and lipid disorders in NAFLD through the autophagy-lysosomal machinery mediated by FABP1 inhibition (Pi et al., 2019), demonstrating the correlation between FABP1 and autophagy in hepatic cells. However, the underlying mechanisms involved are still unknown.
Conclusion
In the present study, we find luteolin treatment for 8 weeks could, to some extent, prevent obesity, lipid and glucose disorders, hepatic steatosis, and fibrosis in the WD/CCl4 mouse model of NAFLD. The possible mechanisms may be through inhibiting hepatic lipid uptake proteins CD36 and FABP1, promoting hepatic autophagy, and alleviating apoptotic processes. However, the exact mechanisms are still unknown due to the limitations of our experimental conditions. Further research is urgently needed to discuss the more precise molecular mechanisms of luteolin in NAFLD prevention and treatment.
Abbreviations
NAFLD: Non-alcoholic fatty liver disease; T2DM: Type 2 diabetes mellitus; WD: Western diet; NASH: Non-alcoholic steatohepatitis; TC: Total cholesterol; TG: Triglyceride; FPG: Fasting plasma glucose; H&E: Hematoxylin–eosin; FABP-1: Fatty acid-binding protein 1; GTT: Glucose tolerance test.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
All methods used in this study were conducted in accordance with relevant guidelines and regulations and approved by the Research Ethics Committee of Shanghai General Hospital (Approval No. 2024AWS245).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by the Jiading Branch of Shanghai General Hospital.
