Abstract
Background
Renal cell carcinoma, the most prominent kind of adult kidney cancer, lacks early warning symptoms, leading to metastases at the time of diagnosis. Renal cell carcinoma begins with N-diethylnitrosamine (DEN) and is accelerated by ferric nitrilotriacetate (Fe-NTA), which may be a good research model for renal cell carcinoma.
Purpose
This study was designed to unveil the protective potential of punicalagin against renal cell tumors induced by Fe-NTA in male mice.
Materials and Methods
Renal cancer initiation was achieved through a single intraperitoneal injection of DEN (200 mg/kg body weight (b.wt.)), followed by promotion using Fe-NTA (9 mg Fe/kg b.wt. intraperitoneally) administered twice weekly over 16 weeks. Simultaneously, mice were subjected to punicalagin (10 mg/kg b.wt.) for an uninterrupted 16-week duration. The chemopreventive efficacy of punicalagin was assessed by evaluating antioxidant activities, oxidative stress markers, renal function parameters, histopathological examinations, and immunohistochemical analyses.
Results
Results showed a significant reduction in DEN and Fe-NTA-mediated lipid peroxidation, concurrent with the amelioration of renal function, as reflected by diminished levels of blood urea nitrogen, creatinine, and kidney injury molecule-1 in punicalagin-treated mice. Furthermore, punicalagin restored the renal antioxidant parameters like superoxide dismutase, catalase, glutathione peroxidase, glutathione reductase, glutathione in DEN and Fe-NTA-treated mice. Further, it was observed that punicalagin reduces the expression of 8-hydroxy-2′-deoxyguanosine (levels and 4-hydroxy-2-nonenal-modified protein adducts within the kidney tissue.
Conclusion
These findings were further substantiated by histological examinations. Our current findings indicate that punicalagin might be a potential contender for preventing renal cancer, likely due to its capacity to reduce oxidative stress in laboratory animals.
Introduction
Renal cell carcinoma represents the most prevalent form of cancer in the kidney, originating in the renal proximal tubule lining and linked to numerous hazards, including exposure to environmental toxic chemicals. In our quest to curtail cancer incidence, a highly promising strategy involves the intervention of chemical compounds through the integration of antioxidants in food (Padala et al., 2020). This approach, known as chemoprevention, entails the continuous supplementation of naturally occurring compounds with the overarching goal of preventing, reducing, or reversing the initiation, promotion, or progression of tumorigenesis resulting from exposure to cancer-inducing agents (Chen & Kong, 2005). Chemoprevention also extends its purview to address the management of precancerous lesions, variably referred to as preinvasive neoplasia, intraepithelial neoplasia or dysplasia depending on the organ involved (Suvarna et al., 2020).
N-Diethylnitrosamine (DEN) is an established potent toxin and carcinogen that affects the liver (Mansour et al., 2019). It serves as a substrate for the enzyme CYP2E1, actively participating in the generation of reactive oxygen species (ROS) (Tolba et al., 2015). DEN instigates the formation of a variety of mutagenic DNA adducts with varying degrees of potency (Schulien & Hasselblatt, 2021). Due to the robust hepatic alterations induced by DEN, its application in rodent models has emerged as an appealing experimental platform for studying carcinogenesis, unraveling the intricate pathogenetic mechanisms underpinning cancer formation (Kang et al., 2007). Epidemiological investigations have underscored the correlation between heightened body iron storage and an elevated cancer incidence, coupled with premature mortality (Thompson et al., 2021).
Nitrilotriacetate (NTA) stands as a significant toxicant frequently encountered in the atmosphere, primarily as a water pollutant (Puliyel et al., 2015). It is also employed as a substitute for polyphosphates in soaps. The tendency of certain metal ions, particularly Fe3+, to form complexes with NTA, eventually giving ferric nitrilotriacetate (Fe-NTA)—an agent renowned for its nephrotoxic effects, is of special concern (Al-Naimi et al., 2019). The administration of Fe-NTA was documented to instigate renal proximal tubular necrosis at acute and subacute levels, primarily through the production of ROS (Nishida et al., 2007). These ROS are responsible for initiating lipid peroxidation and inflicting DNA damage, culminating in renal cell carcinoma upon repeated exposure to Fe-NTA (Rimbach et al., 2000). It is important to mention that literature has indicated a notable occurrence of Fe-NTA-mediated renal adenocarcinoma in experimental animals (Okazaki, 2022).
Studies have further demonstrated that excessive iron administration causes carcinogenesis, while iron insufficiency impedes the generation of preneoplastic hepatocellular injuries, coinciding with the production of lipid peroxides in experimental animals (Kew, 2014). This solidifies iron’s pivotal role in carcinogenesis, primarily mediated through the generation of reactive oxygen radicals (Ying et al., 2021). Research performed by Greene et al. depicted the deleterious effects of the iron complexes NTA (Fe-NTA), inducing renal proximal tubular necrosis and eventually culminating in the progress of renal cancer in rodents (Greene et al., 2022). These pathogenic events were closely tied to the generation of lipid peroxides and DNA injury caused by reactive oxygen radicals. Notably, this induction of oxidative stress and tumorigenic potential extended to both hepatic and renal tissues (Jelic et al., 2021).
In the pursuit of counteracting the carcinogenesis and harmfulness induced by Fe-NTA, a host of established antioxidants have demonstrated their mettle. These include rutin, quercetin, astaxanthin, vitamin C, and naringin, all of which have been lauded for their chemoprotective effects against Fe-NTA-induced cancer (Tolba et al., 2015). Punicalagin, a polyphenolic tannin extracted from pomegranate, has garnered considerable attention in previous research for its multifaceted biological attributes (Xu et al., 2021). These encompass antiviral, anti-inflammatory, anticancer, and antioxidant properties. Notably, punicalagin has demonstrated its potential to curtail cell division and trigger apoptosis or autophagy in diverse cancer cell lines, including those associated with breast cancer, lung cancer, colon cancer, and cervical cancer (Adaramoye et al., 2017). Moreover, punicalagin performs a key role in the regulation of the immune system and antiviral mechanisms. Prior investigations have revealed its capacity to mitigate inflammation in murine macrophages and exert immunosuppressive effects (Berdowska et al., 2021).
In the current investigation, we have undertaken a comprehensive exploration into the protective attributes of punicalagin, a bioactive compound abundantly found in pomegranate, against the perils of renal carcinogenesis, toxicity, and oxidative stress caused by the renal carcinogen Fe-NTA in a murine model.
Materials and Methods
Chemicals
N-Nitrosodiethylamine (DEN), oxidized and reduced glutathione, ferric nitrate, nitrilotriacetic acid (NTA), and all other chemicals utilized in this investigation were procured from Sigma.
Antibodies
Antibodies targeting 4-hydroxy-2-nonenal (HNE)-protein adducts and 8-hydroxy-2′-deoxyguanosine (8-OHdG) were used in immunohistochemistry examination. These antibodies were purchased from Santa Cruz Biotechnology, CA, USA.
Fe-NTA Solution
The Fe-NTA solution was made in accordance with the method followed by Awai et al.’s procedure. A solution was made by combining ferric nitrate (5 mL, 0.16 mM) with disodium salt of NTA (5 mL, 0.64 mM), and the pH was brought to 7.4 through the addition of sodium bicarbonate. The reagent was freshly prepared just prior to the intraperitoneal administration of Fe-NTA to the mice.
Animals
Mice weighing between 30 g and aged 6–8 weeks were utilized for this experiment. The rats were accommodated in an animal care unit with room temperature set at 25 ± 1°C, following a 12-hour light/dark cycle. They were provided unrestricted exposure to a regular diet as well as drinking water. One week was allotted for the animals to acclimatize before the commencement of treatments. The animals were subjected to humane care as per the regulations and necessary authorization was secured from the Institutional Animal Ethics Committee.
Experimental Design
The dosage chosen to investigate the potential chemopreventive effects of punicalagin against renal cancer was designed based on previous investigations conducted elsewhere (Atrahimovich et al., 2018). Renal carcinogenesis, induced by DEN and encouraged by Fe-NTA, was developed following the methods outlined by Ahmad et al. (2011) and Rehman et al. (2013). To assess the impact of punicalagin on various parameters like biochemical, immunohistochemical, and histopathological, the mice were categorized into four groups: Group I, got distilled water via oral administration for 16 weeks and served as the control. Group II got daily oral administration of distilled water for 16 weeks. Additionally, DEN (200 mg/kg body weight (b.wt.)) was administered at intraperitoneal cavity on the first day of the study, followed by Fe-NTA (9 mg Fe/kg b.wt. i.p.) injections biweekly for 16 weeks, beginning 10 days after the initial injection. Group III received punicalagin through oral gavage at a dosage of 20 mg/kg b.wt. for 16 weeks. Similar to Group II, they were also subjected to DEN and Fe-NTA injections. Group IV received oral gavage of punicalagin at 20 mg/kg b.wt. for 16 weeks. At the completion of the experiment, all animals were euthanized. Kidney tissue samples were prepared for subsequent biochemical, histopathological, and immunohistochemical examinations. Blood samples were gathered and processed for further analysis.
Biochemical Analysis
Superoxide dismutase (SOD), glutathione (GSH), and glutathione peroxidase (GPx) activities were assayed in renal tissues by utilizing a kit from Biotech Inc. In addition, the caspase 3 level was also assessed in experimental tissue by utilizing a kit (Cayman Chemical Company, Ann Arbor, MI, USA). The method described by Buege and Aust was utilized to assess lipid peroxidation in renal tissues. This involved observing the formation of thiobarbituric acid conjugates resulting from the reaction with malondialdehyde (MDA), measured at 535 nm. Protein quantification was carried out utilizing a protein assay kit, specifically the bicinchoninic acid kit from Pierce, Rockford, IL, for all samples.
Histology
The kidneys were extracted, washed with saline solution, and subsequently submerged in 10% neutral buffered formalin for a minimum duration of 24 hours to ensure proper fixation. Subsequent to fixation, the specimens underwent a dehydration process employing ascending concentrations of ethanol, were clarified using benzene, and ultimately embedded in paraffin wax. Renal tissue blocks were prepared, and 5 µm thin sections were precisely sliced from them. The paraffin-embedded tissue sections underwent deparaffinization using a solution of xylene and ethanol. Following this, the slides were thoroughly washed with phosphate-buffered saline and then treated with a permeabilization solution consisting of 0.1 M citrate and 0.1% Triton X-100 to facilitate permeation. Subsequently, these slides were subjected to staining with hematoxylin and eosin and were examined under a light microscope to explore the architectural details of the renal tissue.
Immunohistochemistry
The tumor specimens underwent fixation with formalin, subsequent dehydration, and embedding within paraffin blocks. Thin sections were then precisely cut and mounted onto glass slides. To counteract the endogenous peroxidase activity, the sections were treated with a solution containing 3% H2O2 in methanol for a duration of 10 minutes. Antigen retrieval was carried out by immersing the sections in an ethylenediaminetetraacetic acid buffer at 100°C for 10 minutes, followed by cooling to ambient temperature to uncover the antigenic epitope. Subsequently, the slides were blocked with 5% bovine serum albumin (BSA) at 37°C for 30 minutes, and they were then exposed to specific primary and secondary antibodies for 30 minutes at the same temperature. Additional steps involved incubation with 3,3′-diaminobenzidine and streptavidin–biotin complex followed by counterstaining with hematoxylin. The final stage encompassed subjecting the sections to analyses involving immunohistochemistry and imaging.
Statistical Analysis
Variation between the experimental groups was assessed through analysis of variance (ANOVA), followed by Tukey’s multiple comparisons investigation. The provided data displays the mean ± standard deviation of the mean (SD) for the treatment groups, and significance was deemed present at a minimum threshold of p < 0.05 for all comparisons.
Results
Effects of Punicalagin on Renal Tumorigenesis
Table 1 presents the attenuating outcomes of administering punicalagin in Fe-NTA-induced renal tumorigenesis among mice initiated with DEN and subsequently encouraged by Fe-NTA. The administration of Fe-NTA to DEN-initiated mice led to a 75% increase in the occurrence of renal cell tumors among the studied animals. However, in the cohort of animals receiving punicalagin at a dosage of 20 mg/kg b.wt., the incidence of tumors notably decreased to approximately 43%. Notably, pre-treatment with punicalagin at the same dosage did not exhibit any tumor incidence.
Effects of Punicalagin Administration on Renal Tumorigenesis in N-Diethylnitrosamine (DEN)-initiated and Ferric Nitrilotriacetate (Fe-NTA)-treated Mice.
Effects of Punicalagin on Renal Toxicity Markers
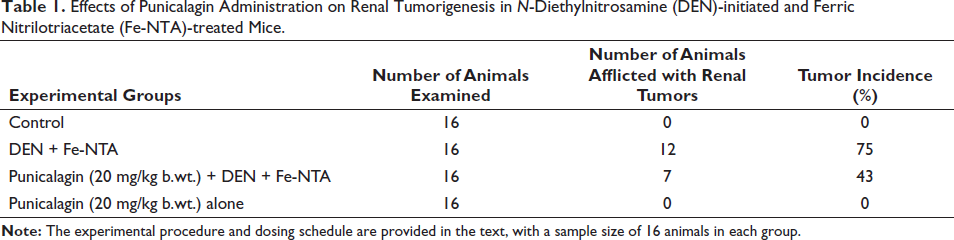
Significant increases (p < 0.05) in the level of blood urea nitrogen (BUN) and creatinine were observed in the mice group treated with DEN and Fe-NTA, as compared to the control mice. Conversely, treatment of punicalagin at the dosage of 20 mg/kg b.wt. resulted in a notable decrease in both BUN (p < 0.05) and creatinine (p < 0.05) levels. Notably, no noteworthy differences in the level of BUN and creatinine were detected between the control and the punicalagin-alone group. Elevated kidney injury molecule-1 (KIM-1) levels were significantly evident (p < 0.05) in mice initiated with DEN and further promoted by Fe-NTA. However, pretreatment with punicalagin led to a reduction in serum KIM-1 levels (p < 0.05). Comparatively, no statistical differences were noted between the punicalagin-alone group and the control one (Figure 1).
Effect of Punicalagin on Oxidative Stress Markers
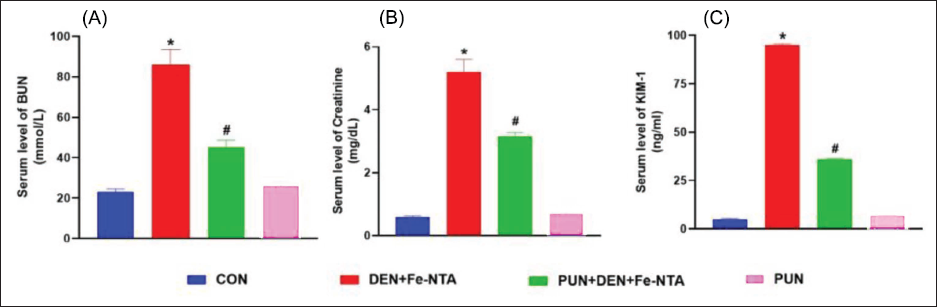
The administration of DEN and Fe-NTA resulted in a noteworthy reduction (p < 0.05) in renal GSH levels and a decrease in the activities of glutathione reductase (GR) (p < 0.05), GPx (p < 0.05, SOD (p < 0.05), and catalase (p < 0.05) when compared to the respective control group. However, when punicalagin was given orally at a dosage of 20 mg/kg b.wt., it led to a significant improvement in GSH levels (p < 0.05), as well as a substantial increase in GPx (p< 0.05), GR (p < 0.05), SOD (p < 0.05), and catalase (p < 0.05) activities. Notably, no statistical variation was observed between the group treated solely with punicalagin and the control (Figure 2A–E). In mice subjected to DEN and Fe-NTA treatment, there was a significant increase (p < 0.05) in MDA levels compared to the control mice. However, punicalagin treatment significantly lowers MDA levels (p < 0.05), thereby mitigating membrane destruction in the renal cells. No significant differences were found in the group administered solely with punicalagin when compared to the control (Figure 2F).
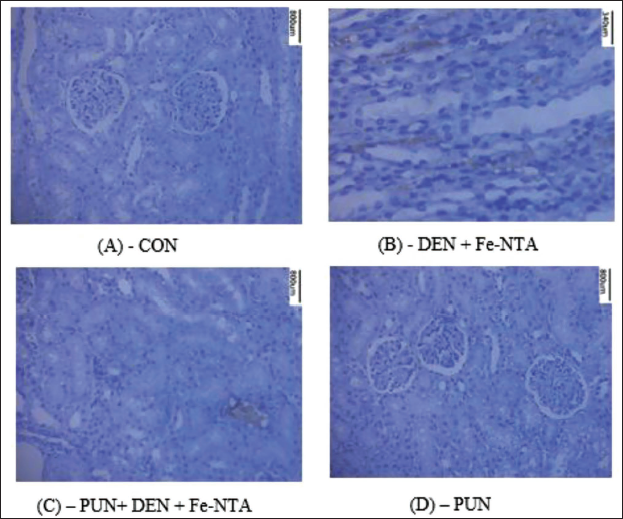
Effect of Punicalagin Administration on Renal Histology on DEN and Fe-NTA-treated Experimental Animals
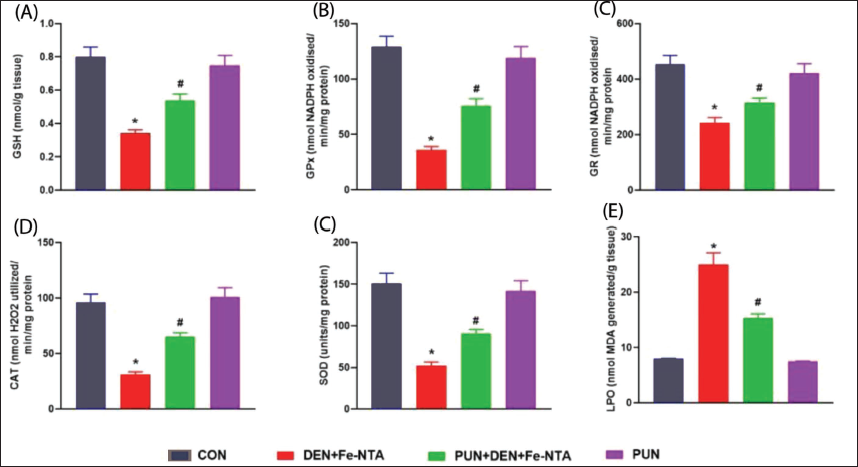
The kidney morphology in control mice appeared normal. However, in the group subjected to DEN initiation and Fe-NTA treatment, there were noticeable distortions in renal structure, including congestion in both tubules and glomeruli, atrophic glomeruli, increased hyperchromatism, and infiltration of inflammatory cells. When punicalagin was administered at a dosage of 20 mg/kg b.wt., some improvements were observed, including a reduction in tubular necrosis, decreased infiltration of inflammatory cells, and decreased hyperchromatism. These findings indicate that punicalagin treatment significantly ameliorated the renal histoarchitecture damage induced by DEN and Fe-NTA. In contrast, the renal sections from the punicalagin-only treated group displayed a regular renal architecture, similar to that of the control mice (Figure 3).
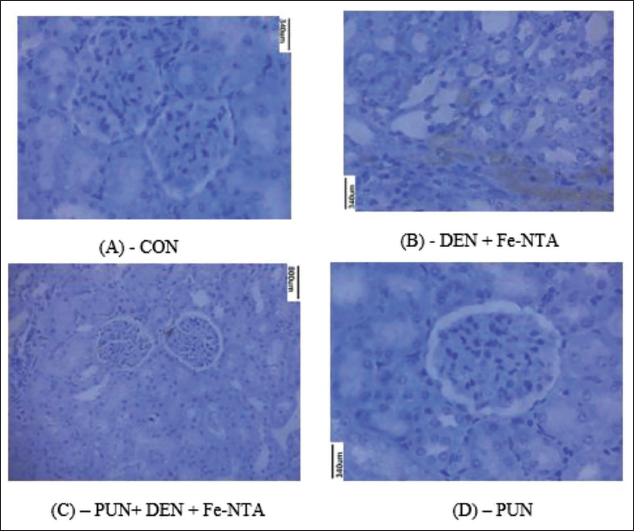
Effect of Punicalagin on DEN and Fe-NTA-induced 4-HNE-modified Protein Adducts and 8-OHdG Expression in Kidney
In Figures 4 and 5, the immunostaining of 4-HNE-protein adducts in experimental mice. Introduction of Fe-NTA to mice resulted in the degeneration of proximal tubular cells and an increase in the generation of 4-HNE-protein adducts, as depicted in Figure 4B. However, in the group of mice pretreated with punicalagin and subsequently given with Fe-NTA, a notable reduction in 4-HNE-modified protein adduct formation was evident, as shown in Figure 4C. No 4-HNE-modified protein adduct was observed in the control group, as illustrated in Figure 4A. Similarly, in animals pretreated with punicalagin-alone, HNE-modified protein adduct formation was also absent. The immunohistochemical assessment of 8-OHdG revealed moderate staining in the renal tissue of control mice. In contrast, mice subjected to DEN initiation and Fe-NTA treatment displayed heightened 8-OHdG immunostaining in their renal cells. However, the administration of a punicalagin resulted in a reduction in 8-OHdG immunostaining within the renal cells. Notably, there was no discernible difference in the immunoreactivity of 8-OHdG between the renal cells of the punicalagin-alone group and those of the control group.
Discussion
In recent days, considerable attention has been created on the creation of chemopreventive agents aimed at preventing cancer at its initial stages, recognizing the challenges of treating cancer once it is diagnosed (Xie et al., 2022). Chemoprevention has emerged as a crucial area of cancer investigations, offering a systematic approach to identifying potential cancer mitigators and investigating the mechanisms of carcinogenesis (Steward & Brown, 2013). It is well-established that various external determinants, like lifestyle choices and encounter to environmental pollutants, play a pivotal role in the development of various cancers in mankind (Parsa, 2012). Consequently, modifying daily dietary intake to incorporate chemopreventive agents with demonstrated potential in tumor bioassay systems becomes essential, offering a promising avenue for cancer chemoprevention (An et al., 2020; Singh et al., 2019).
This study aimed to give substantial mechanistic insights into the chemopreventive properties of punicalagin, known for its diverse biological properties. We employed a robust animal model involving Fe-NTA-induced renal tumorigenesis in mice. Our research demonstrates the effective mitigation of Fe-NTA-induced tumorigenesis by punicalagin, which acts by attenuating kidney injury and oxidative stress in experimental animals. These findings contribute significantly to our understanding of cancer chemoprevention and the potential therapeutic applications of punicalagin in this context (Adaramoye et al., 2017; Berdowska et al., 2021).
The kidney plays a key role in eliminating various xenobiotics, rendering it particularly susceptible to impairment due to its heightened sensitivity to numerous toxins and chemicals (Tolba et al., 2015). Fe-NTA is a known environmental pollutant and a strong nephrotoxic agent and when Fe-NTA is administered to animals, it can be absorbed into the bloodstream and filtered through the kidneys, where it can cause oxidative stress and inflammation. This can lead to a range of toxic manifestations in the renal system, including acute and subacute kidney injury, and the progression of renal cell carcinoma in high incidence (Athar & Iqbal, 1998; El-Maraghy et al., 2009; Iqbal et al., 2007; Mizuno et al., 2006). In our study, we identified elevated levels of creatinine and BUN, well-established markers for impaired kidney function, in mice subjected to DEN and Fe-NTA treatment. However, the administration of punicalagin led to a noteworthy restoration of renal function, as evidenced by a significant reduction in circulating BUN and creatinine levels. Our findings align with the research conducted by An et al. in 2020, documenting the effectiveness of punicalagin in restoring kidney function. KIM-1, a type 1 transmembrane protein, serves as a sensitive marker for kidney injury within the proximal tubule epithelium. In our study, KIM-1 levels were noticed to be increased in the DEN and Fe-NTA-treated group. However, the treatment of punicalagin demonstrated a notable reduction in KIM-1 levels, indicative of its protective effects on kidney tubular cells and its capacity to prevent renal injury.
DEN and Fe-NTA induce various nephrotoxic effects by instigating peroxidative harm to the kidneys through heightened oxidative stress, thereby disrupting the renal redox cycle (Al-Naimi et al., 2019). Oxidative stress denotes the disproportionality between the generation of ROS and the protective mechanisms of antioxidants in the kidneys, which can lead to cellular damage and dysfunction. Fe-NTA can cause oxidative stress in the kidneys by generating ROS through various mechanisms, including the level of ROS. ROS are extremely reactive molecules capable of causing harm to cellular constituents like lipids, proteins, and DNA, consequently inducing oxidative injury and inflammation. In the context of Fe-NTA exposure, ROS can react with lipids in the kidneys, leading to the generation of MDA and other lipid peroxidation products. The antioxidant defense system in the kidneys includes enzymes such as GPx and GR, as well as nonenzymatic antioxidants like GSH. Fe-NTA exposure can deplete the levels of these antioxidants, leading to a further elevation in ROS levels and oxidative stress (Jahangir & Sultana, 2006; Kaur et al., 2010). In this investigation, we observed that DEN and Fe-NTA elevated the levels of MDA while concurrently diminishing the concentrations of antioxidants such as GPx, GSH, and GR, in line with prior reports. GSH, a nonenzymatic antioxidant, plays a crucial role in detoxification by reacting with toxic substances, including ROS. Consequently, it serves a key role in fighting against oxidative stress and inflammation (He et al., 2017). The treatment of punicalagin effectively mitigated lipid peroxidation and augmented the levels of GSH, GR, and GPx. SOD is an antioxidant enzyme responsible for quenching superoxide into hydrogen peroxide, while CAT, a peroxisomal heme protein, catalyzes the removal of hydrogen peroxide generated during the reaction catalyzed by SOD. This enzymatic interplay provides a protective defense against ROS (He et al., 2017). Notably, the activities of SOD and CAT were diminished in mice subjected to DEN and Fe-NTA treatment. However, punicalagin treatment resulted in the elevation of these enzyme activities, indicative of its antioxidant properties, consistent with previous findings reported by Zhang et al. (2022).
One of the main signs of oxidative stress is increased lipid peroxidation, which was shown in the mice treated with Fe-NTA (Al-Naimi et al., 2019). According to one study, Fe-NTA increases the reactive thiobarbituric acid compounds in kidney tissue, which increases lipid peroxidation (Deiana, et al., 2007). Punicalagin reduces the lipid peroxidation brought on by Fe-NTA in the current study’s experimental animals. Malonaldehyde and HNE-modified protein adducts, two lipid peroxidation byproducts that damage DNA and cause renal dysfunction, are cytotoxic and can cause cancer and mutagenesis (Nara et al., 2016). Our study also showed that the pretreatment of rats with punicalagin reduced the production of 8-OHdG and HNE-modified protein adducts brought on by Fe-NTA in the kidney. Punicalagin may therefore have antioxidant components that contribute to the chemoprotection it offers against Fe-NTA-induced renal injury as found in current studies.
Lipid peroxidation, a prominent indicator of oxidative stress, showed an increase in Fe-NTA-treated mice (Ayala et al., 2014). In kidney tissue, Fe-NTA is known to augment lipid peroxidation by elevating the levels of thiobarbituric acid reactive substances (Rimbach et al., 2000). In our present investigation, punicalagin was found to mitigate the lipid peroxidation induced by Fe-NTA through the reduction in the level of MDA in experimental animals. The byproducts of lipid peroxidation, including malonaldehyde and HNE-modified protein adducts, possess cytotoxic properties and can inflict DNA damage, ultimately resulting in the formation of 8-OHdG, a substance known for its carcinogenic and mutagenic potential, leading to renal dysfunction (Barrera, 2012; Jomova et al., 2023). Our data also revealed that pretreatment with punicalagin in animals alleviated the production of HNE-protein adducts and 8-OHdG induced by Fe-NTA in the kidney. Hence, the observed chemoprotection conferred by punicalagin against Fe-NTA-induced renal impairment, as demonstrated in our current study, may be attributed to its antioxidative constituents.
Conclusion
In summary, our current study provides evidence that punicalagin effectively mitigates renal carcinogenesis initiated by DEN and promoted by Fe-NTA in mice. These results indicate that punicalagin holds promise as a chemopreventive agent against renal carcinoma by alleviating oxidative stress. Nevertheless, additional research is necessary to comprehensively elucidate the exact mechanisms through which punicalagin acts to prevent renal cancer.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The animals were subjected to humane care as per the regulations and necessary authorization was secured from the Institutional Animal Ethics Committee of Qingdao University, Qing Dao, China.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
