Abstract
Free radicals and oxidative stress play a central role in gastric injuries caused by ethanol (EtOH). Antioxidant strategies to counteract EtOH toxicity are highly desirable. Norbixin (NBIX) is a carotenoid with antioxidant potential largely used in the food industry. This study evaluated the NBIX effects in a model of gastric ulcer induced by EtOH in rats. Male Wistar rats received NBIX doses of 0, 10, and 25 mg/kg by gavage 1 h after EtOH administration (0 or 75% solution, 1 mL/200 g of animal). The animals were euthanized 1 h after the NBIX administration, and their stomachs were removed for macroscopic and histopathological analyses, quantification of nonprotein sulfhydryl (NPSH) groups, lipid peroxidation (LPO) levels, and catalase (CAT) activity determination. NBIX increased LPO in gastric mucosa and caused CAT inhibition and NPSH depletion in EtOH-treated animals. Results showed that NBIX did not protect gastric tissue against EtOH damage, and this could be associated to a prooxidant effect.
Introduction
In recent decades, the incidence of peptic ulcer disease has declined in the Western world. However, it still ranges from 2 to 10/100,000, remaining a public health problem in modern society. 1 Epidemiological studies relate the occurrence of this gastric disorder to predisposing factors, such as caffeine, tobacco, low-fiber diet, an indiscriminate consumption of alcohol, drugs, nonsteroidal anti-inflammatory and Helicobacter pylori. 2 –4 Besides, physiological abnormalities such as hydrochloric acid hypersecretion, hypersensitivity of the parietal cell to gastrin and the deficiency of mucosal and submucosal blood flow can also be involved. 5,6
Among gastric offenders, there is strong evidence that mucosal defensive actions are exhausted by ethanol (EtOH), 4,7 a xenobiotic that leads to the generation of reactive oxygen species (ROS). 8 ROS are prooxidants, i.e., they have the capacity to oxidize target molecules. The term ROS includes all reactive oxygen forms that participate in the initiation and progression of chain reactions, such as superoxide radical and hydroxyl radical. 9
In view of the damage caused by the excess of ROS, cells have a variety of antioxidant defenses. The principal defense systems against ROS are the enzymes superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), glutathione reductase, catalase (CAT), and nonenzymatic antioxidant nutrients. 10 The mechanisms of action of antioxidant compounds include radical scavenging and suppressing excited states. Besides, catalytic systems neutralize or eliminate ROS, as well as bind metal ions to proteins, making them unavailable for the production of oxidant species. 9
When there is an impairment in the antioxidant system, or there is an overabundance of ROS production, an imbalance between antioxidants and oxidants occurs, and oxidative stress takes place. 11 Previous research shows that EtOH depletes the enzymes SOD, CAT and GSH-Px, and induces lipid peroxidation (LPO), resulting in oxidative stress. 7,12 LPO can be assessed by measuring thiobarbituric acid reactive substances (TBARS), which are an index of oxidative stress. Moreover, superoxide anion and hydroxyl radical production are also increased by EtOH in gastric mucosal along with increased membrane microviscosity, a tissue damage marker. 4 Due to the convincing evidence of the involvement of ROS in EtOH toxicity, it would be worthwhile to find new antioxidant substances to counteract injuries caused by EtOH. 13
Carotenoids are natural substances that present in their chemical structure conjugated double bonds, which are responsible for their color and some biological functions. They are considered powerful antioxidants 14 and in vivo can attenuate the LPO. 15 The seeds of annatto (Bixa orellana L.) are rich in carotenoids, mainly bixin and norbixin (NBIX). Annato preparations are widely used in the food industry and are the most consumed colorant in Brazilian industries (90% of the natural colors) and worldwide (70% of natural colors). 16
NBIX is a water-soluble carotenoid obtained through the hydrolytic removal of a methyl group in the molecule of bixin. 17 Despite the high antioxidant potential of NBIX, especially in lipid membranes, 18 there is not much information available about this carotenoid in animals and humans.
NBIX has been shown to protect rats from DNA damage and oxidative modifications induced by methylmercury. 19 Furthermore, NBIX protects Escherichia coli cells against DNA damage induced by ultraviolet radiation, hydrogen peroxide (H2O2), and superoxide anions. 20 In vitro tests showed that low concentrations of NBIX (up to 50 µM) are effective in protecting the DNA of fibroblasts from the damaging effects caused by H2O2 and metal ions, while higher concentrations concomitantly augmented the extent of DNA damage. 21
To the best of our knowledge (data retrieval), no studies have been conducted on the protective effect of NBIX against gastric ulcer models until this moment. Thus, the objective of this investigation is to clarify the effect of NBIX (10 and 25 mg/kg) against oxidative stress and gastric tissue damage induced by EtOH administration in rats by evaluating macroscopical and histopathological damage along with markers of oxidative stress.
Methods
Chemicals
TBA and trichloroacetic acid (TCA) were purchased from Acros Organics (Morris Plains, New Jersey, USA). H2O2 (Perhidrol®) was acquired from Merck (Darmstadt, Germany), while DTNB (Ellman’s reagent) was purchased from Sigma Chemical Co. (St Louis, Missouri, USA). The other chemicals used were of analytical grade. NBIX (10% m/v) was obtained from Chr. Hansen (Hørsholm, Denmark).
Animals
Seven-weeks-old male Wistar rats (200–250 g), obtained from the General Animal House of the Federal University of Santa Maria, were kept on a separate animal room, in a 12-h light/12-h dark cycle at room temperature with free access to water before the experiment. All animals were used according to the guidelines of the Committee on Care and Use of Experimental Animal Resources from the Federal University of Santa Maria, Brazil (registration number: 030/2012-2).
Experimental design
After a 1-week adaptation period, the animals were divided into six groups (n = 5 per group) and received distilled water as vehicle or 75% aqueous solution of EtOH (v/v) by oral gavage (1 mL/200 g of animal). After 1 h of EtOH administration, NBIX was administered to the ones that received vehicle or EtOH by gavage at doses of 0, 10, and 25 mg/kg of body weight. The chosen model of gastric damage induced by EtOH has already been described. 22 The NBIX doses were chosen based on previous in vivo studies with this carotenoid. 23,24
One hour after NBIX administration, the rats were euthanized by deep anesthesia with thiopental (60 mg/kg intraperitoneally) for stomach removal. The stomach was washed with saline solution (sodium chloride (NaCl 0.9%), and the glandular portion was separated for macroscopic evaluation. Afterward, a portion of gastric tissue was collected for histopathological analysis, and the remaining tissue was homogenized in nine volumes of potassium phosphate buffer (0.1 M, pH 7.4) using a Polytron mixer (Kinematica AG, Switzerland). A part of homogenate was used to assay levels of nonprotein thiol groups (NPSH), an indicator of nonenzymatic defenses. The remaining homogenate was centrifuged at 3000g at 4°C for 10 min to yield a low-speed supernatant that was used to measure the levels of TBARS and the activity of the antioxidant enzyme CAT.
Macroscopical analysis
The stomachs were opened along the greater curvature, washed with 0.9% NaCl, and examined by a blinded pathologist for macroscopic lesions in the glandular part under a dissecting microscope. The severity of gastric mucosal lesions was estimated using an index, where normal stomachs received a score of 0 and discolored stomachs received a score of 1, as previously reported. 22,25 For hemorrhage, petechia, edema, and mucus, the stomachs with minor injury received a score of +1, while those with moderate and severe injuries were given a score of +2 and +3, respectively. 22,25
Histopathological analysis
For histological studies, a portion of stomach from each experimental group was fixed in 10% formalin and embedded in paraffin. Sections of 5 µm were obtained with a standard microtome and were stained with hematoxylin and eosin. The sections were examined by a pathologist without knowledge of the experimental groups for the presence of any negative features such as edema, erosion, ulceration, and necrosis at a microscopic level. The severity of histopathological changes was quantified according to the arbitrary scale described before, with some modifications. Gastric tissue with no negative features was given a score of 0. Gastric tissue with mild histopathological damage was given a score of +1. Those with moderate and severe negative features were given a score of +2 and +3, respectively. Results were expressed as a histopathological score. 26
LPO measurement
The stomach tissue LPO estimation was performed using the TBARS assay, as described before, 27 which quantifies the colorimetric reaction of the LPO product malondialdehyde with TBA. The reaction produces a colored compound that absorbs maximally at 532 nm. One gram of stomach tissue was homogenized in 9 mL of potassium phosphate buffer (0.1 M, pH 7.4) using a Polytron mixer (Kinematica AG, Switzerland). After heating at 80°C for reacting with TBA, tubes were cooled and centrifuged at 2000g. The organic layer was collected and the absorbance was read at 532 nm using a spectrophotometer (Shimadzu, Japan)
NPSH levels
The homogenate fraction was mixed with 10% TCA (1:1 v/v) and distilled water, followed by centrifugation. NPSH content was immediately determined after reaction with 5,5′-dithio-bis(2-nitrobenzoic) acid, as described earlier. 28
CAT activity
CAT activity was determined by measuring the decrease of H2O2 absorption at 25°C. The method is based on the removal of H2O2 by CAT and loss of absorbance at 240 nm, as previously described. 29
Protein quantification
LPO and NPSH levels, as well as CAT activity, were normalized to the amount of stomach protein content. The quantification of the protein was performed following the Lowry method, in which the maximum absorbance for the solution of Folin–ciocalteu, due to its interaction with Bovine serum albumin protein, occurs at 625 nm. 30
Statistical analysis
The data were analyzed using Statistica 7.0 software (StatSoft Inc., Tulsa, Oklahoma, USA), through a two-way analysis of variance (ANOVA; 2 EtOH doses × 3 NBIX doses) and Duncan post hoc when necessary. The data were expressed in means ± standard error of means (SEM), and differences were considered statistically significant when p < 0.05.
Results
Macroscopical analysis
Two-way ANOVA revealed a significant effect of EtOH on gastric tissue at macroscopical level (p < 0.05). Post hoc comparisons demonstrated that 1 h exposure to EtOH is able to cause injury to gastric tissue, characterized by macroscopic features such as discoloration (Figure 1(a)), edema (Figure 1(b)), and mucus loss (Figure 1(c)). Treatment with NBIX at all evaluated doses did not change observed features in color and edema. Besides, all NBIX doses in the presence of EtOH increased mucus loss when compared to EtOH-NBIX 0 group (Figure 1(c)). NBIX per se did not show effects on any macroscopical features described above (Figures 1 –3).

Macroscopical features of stomach from rats treated with EtOH and/or NBIX. (a) Color index (b), edema index, and (c) mucus loss index. Data are means ± SEM (n = 5). *p < 0.05: different from all control groups; # p < 0.05: different from EtOH-NBIX 0 group.EtOH: ethanol; NBIX: norbixin; SEM: standard error of means.

Representative histology (magnification of ×40; a–f) of gastric tissue from animals treated with EtOH and/or NBIX. (a) EtOH 0-NBIX 0 group (control); (b) EtOH-NBIX 0 group; (c) EtOH-NBIX 10 group; (d) EtOH-NBIX 25 group; (e) EtOH 0-NBIX 10 control group; (f) EtOH 0-NBIX 25 control group; Observe the preserved architecture of mucosal cells (a, c, and e). In the EtOH-NBIX 0-treated animals (b), there is edema and mucosal congestion, as well as, inflammatory process characterized by neutrophils infiltration. NBIX 25 mg/kg in the presence or absence of EtOH (d and f) presented negative features such as edema, congestion, and inflammatory infiltration. EtOH: ethanol; NBIX: norbixin.

Histopathological score of gastric tissue from rats treated with EtOH and/or NBIX. Data are means ± SEM (n = 5). *p < 0.05: different from EtOH 0-NBIX 0 control group. # p < 0.05: different from EtOH-NBIX 0 group. The score 0 indicates the absence of negative features (edema, congestion, and inflammation), while score 3 indicates severe negative features. EtOH: ethanol; NBIX: norbixin; SEM: standard error of means.
Histopathological analysis
The acute exposure of rats to EtOH caused mucosal edema and congestion along with an inflammatory process, as demonstrated by histopathological analysis (Figures 2 and 3). These results confirm that an acute exposure to EtOH causes gastric damage also at a microscopic level. Posttreatment with NBIX at 10 mg/kg ameliorates microscopic injuries caused by EtOH, whereas 25 mg/kg did not show any protective effect (Figure 3). Moreover, this NBIX dose caused histopathological changes (Figure 3).
LPO measurement
ANOVA revealed a significant increase in the TBARS levels after EtOH exposure (p < 0.05; Figure 4). This result confirms the ability of EtOH in inducing LPO in gastric tissue even 1 h after its administration. Posttreatment with NBIX exacerbated EtOH-induced LPO in rat stomach at all evaluated doses when compared to the EtOH-NBIX 0 group. Moreover, this effect of NBIX on LPO was dose-related, that is, higher dose produced greater tissue damage. NBIX did not cause LPO per se at any evaluated dose (Figure 4).
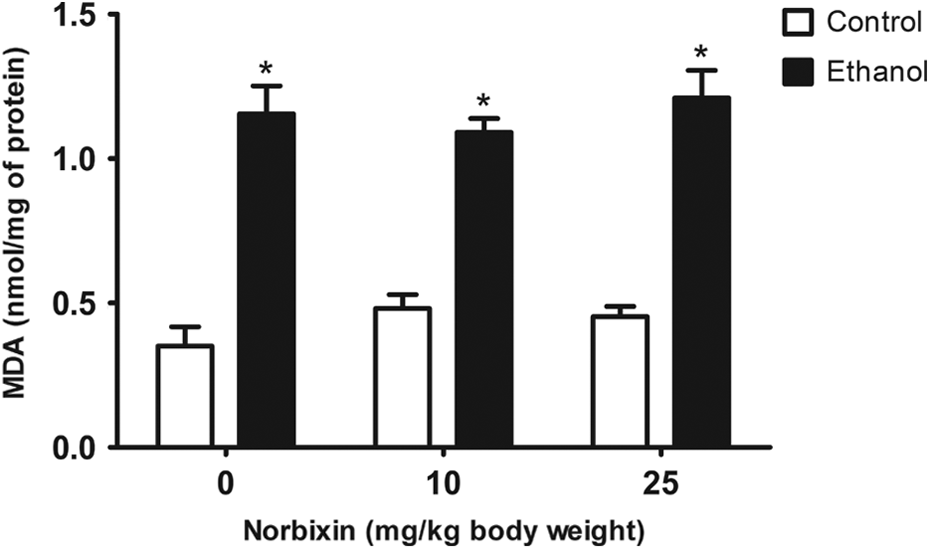
TBARS levels of gastric tissue from rats treated with EtOH and/or NBIX. Data are means ± SEM (n = 5). *p < 0.05: different from all control groups. TBARS: thiobarbituric acid reactive substance; EtOH: ethanol; NBIX: norbixin; SEM: standard error of means.
Antioxidant defenses
Concerning nonenzymatic defenses, 1 h exposure to EtOH alone did not affect NPSH (Figure 5(a)). However, posttreatment with NBIX (10 and 25 mg/kg) induced NPSH depletion when compared to EtOH-NBIX 0 group. Furthermore, 10 mg/kg of NBIX alone caused an increase of NPSH (p < 0.05, Figure 5(a)), while NBIX at 25 mg/kg did not affect NPSH content per se.
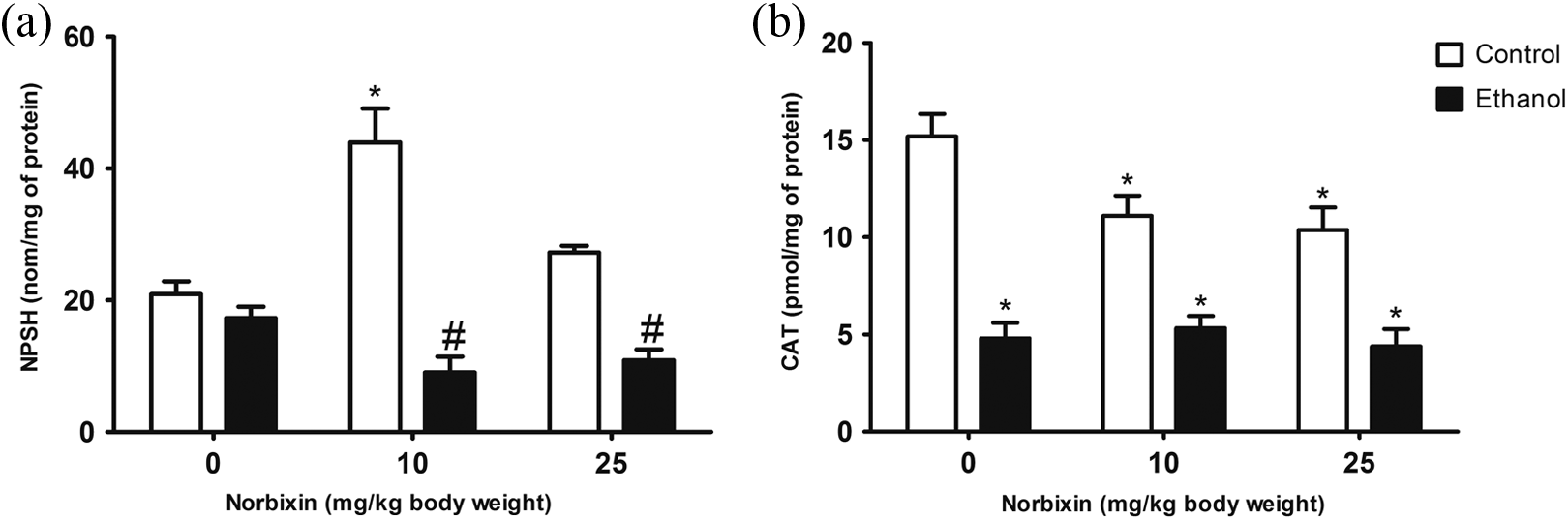
NPSH levels (a) and CAT (b) activity of gastric tissue from rats treated with EtOH and/or NBIX. Data are means ± SEM (n = 5). *p < 0.05: different from EtOH 0-NBIX 0 control group. # p < 0.05: different from EtOH-NBIX 0 group. NPSH: nonprotein sulfhydryl groups; CAT: catalase; EtOH: ethanol; NBIX: norbixin; SEM: standard error of means.
EtOH administration significantly decreased CAT activity in the stomach of the animals in the experimental conditions used in this protocol. No doses of NBIX were able to attenuate the decrease in the activity of this antioxidant enzyme. Moreover, NBIX affected CAT activity per se at all evaluated doses (Figure 5(b)).
Discussion
EtOH consumption and its health complications have become a serious problem around the world. 31 EtOH is known to penetrate the gastric mucosa, causing membrane damage, erosion of gastric cells, impairment in proton (H+) pumping into the gastric lumen, and hence gastric ulceration. 13 Accordingly, EtOH administration in rats caused macroscopic lesions to gastric tissue, such as loss of mucus, discoloration, and edema. Besides, the harmful effect of EtOH on gastric tissue and its ability to cause hemorrhage and mucus loss has already been described. 32 Besides macroscopic lesions, 1-hour exposure to EtOH also caused histopathological damage to gastric tissue; this injury is characterized by edema and inflammatory process characterized by neutrophils infiltration, as described before. 32
Oxidative stress has been considered a crucial step in EtOH-induced mucosal damage and is one of the features most likely to precede cell death in gastric mucosal cells. 33 Here, we observed that EtOH exposure induced a decrease in CAT activity along with an increase in LPO in the gastric tissue of rats. CAT inhibition has already been described after EtOH exposure 12 and may have led to the LPO observed in EtOH-treated group. Besides, LPO in gastric tissue plays a significant role in the pathogenesis of EtOH-induced gastric lessions. 11,34 Thus, we can expeculate that the generation of free radicals by EtOH overwhelmed the antioxidant capacity of gastric tissue, inducing LPO and tissue damage. Interestingly, EtOH exposure per se did not affect NPSH content in gastric tissue in the present study. The principal NPSH is reduced glutathione (GSH), comprising 75–90% of total intracellular NPSH. 35 GSH plays an important role in antioxidant defense, because it possess not only direct radical scavenging properties but is also essential for the antioxidant activity of some enzymes. 36 Previous reports described the depletion of NPSH content after 1-h EtOH exposure. 12,32
Concerning the involvement of oxidative stress in gastric damage induced by EtOH, the search for antioxidant substances is valuable, and many reports are available. 37,38 Despite these evidences of antioxidant uses, there are no reports about the effect of the NBIX on gastric ulcer model induced by EtOH. Here, we described for the first time the behavior of this carotenoid in gastric tissue per se and in the presence of EtOH.
In the present study, we observed that NBIX did not show any protective effects on macroscopic features. Besides, all evaluated doses of the carotenoid exacerbated mucus loss. Mucus production is one of the major mechanisms of local gastric mucosal defense. 39 These tissue damages contrast with the absence of oral toxicity of an extract containing 91.6% of NBIX in rats. 23 Moreover, the same authors described a no observed adverse effect level of NBIX as being 63 mg/kg/day. However, in the present study, we observed the effect of NBIX on gastric tissue even at the dose of 10 mg/kg. Posttreatment with all NBIX doses induced NPSH depletion in EtOH-treated animals. NPSH plays an important role in protecting gastric mucosa from the noxious effect of ROS formation due to EtOH administration, mainly by controlling the production and nature of the mucus. 40 Thus, it seems likely that NPSH depletion induced by NBIX in EtOH-treated rats could be involved in the mucus loss observed in our study.
Besides the absence of protection on NPSH groups, no NBIX dose was able to prevent changes in CAT activity and LPO. These results contrast with the antioxidant effects described for this carotenoid, mainly against H2O2, superoxide anion, as well as transition metals. 20,21 Additionally, NBIX was also able to prevent LPO in liver of rats fed with a high-fat diet. 41
Interestingly, in the present study, we observed prooxidant effects of NBIX, since posttreatment with this carotenoid magnified LPO, CAT inhibition, and NPSH depletion in EtOH-treated animals. Moreover, NBIX affected CAT activity and NPSH groups per se. A prooxidant may be defined as a molecule that can react with ROS to form species capable of exerting toxicological changes to a cell or an organ, greater than the effect of the oxidizing species alone. 42
Carotenoids can exhibit prooxidant effects in situations of changes in certain factors, such as partial pressure of oxygen (O2), interaction with other antioxidant molecules, and the concentration of carotenoid in biological environment. 43 It is the case with the β-carotene, which inhibits the oxidation at low O2 partial pressure (such as those found in most tissues under physiological conditions), but at high oxygen tension (760 mmHg), the initial antioxidant activity is replaced by a prooxidant action. 42 Consistent with these data, it has been proposed that changes in the carotenoid’s concentration or O2 partial pressure would lead to carotenoid auto-oxidation or formation of carotenoid peroxyl radicals. 44 These features alter the properties of biological membranes and may alter their permeability to toxins (such as EtOH) or free radicals. 42
However, since this is greatly influenced by carotenoid structures, different compounds will be incorporated quite differently into membranes. 44 An in vitro study showed that, while low concentrations of NBIX (up to 50 µM) protected DNA against strand breaks, higher concentrations of NBIX augmented the extent of DNA damage. 21 Similar antioxidative–oxidative activities have been described for lycopene and β-carotene on adenocarcinoma cells, 45 and for β-carotene on hepatoma cells. 46 Additionally, the esterification state of carotenoids also seems to be related with their antioxidant/prooxidative status, as described for astaxanthin. 13 Esterified astaxanthin shows comparatively better stability than free astaxanthin, and hence it may exhibit more health beneficial effects. Thus, carotenoids in the nonesterified form (such as NBIX) seem to be more prone to prooxidative states than esterified forms. 13 NBIX could also be exerted its negative effects on LPO levels by interfering in plasma membranes, as described before by its hyperglycemic effect. 47 It is well documented that polar carotenoids, such as NBIX, have the ability to interact with lipids in lipid bilayer membranes in a way that affects their biological properties, as demonstrated in plants 48,49 and photosynthetic bacteria. 50 Changes in the physicochemical properties of membranes interfere with their phase transition, fluidity, and permeability and, therefore, the regulation of membrane-embedded receptors, as well as signal transduction pathways and nutrient transport, might be affected to some degree. 51 Thus, we can speculate that NBIX exerted its negative effects by both mechanisms: a prooxidant effect and interference on membrane properties.
Despite the obvious prooxidant effects exerted by NBIX in the present study, posttreatment with 10 mg/kg of NBIX restored gastric tissue from histopathological damage caused by EtOH. These findings indicate that NBIX could exert some protective action by other mechanism than its antioxidants properties. 19 –21 In fact, the polar carotenoid astaxanthin presented protective effects in gastric ulcer models by controlling acid secretion, showing maximum H+, K+-ATPase-inhibitory activity. 13 Thus, we cannot rule out the possibility that NBIX may present some protective effect other than oxidative stress-related effect that would be implicated in this result. However, we speculate that the histopathological protection offered by NBIX was insufficient to result in macroscopical improvement, since we observed macroscopical damage in all NBIX-EtOH-treated animals.
Our results showed that the carotenoid NBIX was not effective in protecting gastric tissue against EtOH toxicity. Moreover, this carotenoid exacerbated oxidative damage caused by EtOH, besides causing injuries per se, (especially at a dose of 25 mg/kg). To the best of our knowledge, this is the first report on the effect of NBIX on gastric tissue. Since annatto (NBIX source) is extensively used as a natural dye in the food industry, it is important to find information about this carotenoid. Thus, more studies are necessary to clarify the effects and possible mechanisms of the negative effects of NBIX.
Footnotes
Acknowledgements
The authors thank the Department of Physiology and Pharmacology for the support to carry out this study. The authors declare that there is no conflict of interest that would prejudice the impartiality of this scientific work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
